Plasma C-Peptide and Risk of Developing Type 2 Diabetes in the General Population
Abstract
1. Introduction
2. Materials and Methods
2.1. Design and Study Population
2.2. Data Collection
2.3. Laboratory Measurements
2.4. Outcome Definition
2.5. Statistical Analyses
3. Results
3.1. Baseline Characteristics
3.2. Cross-Sectional Associations
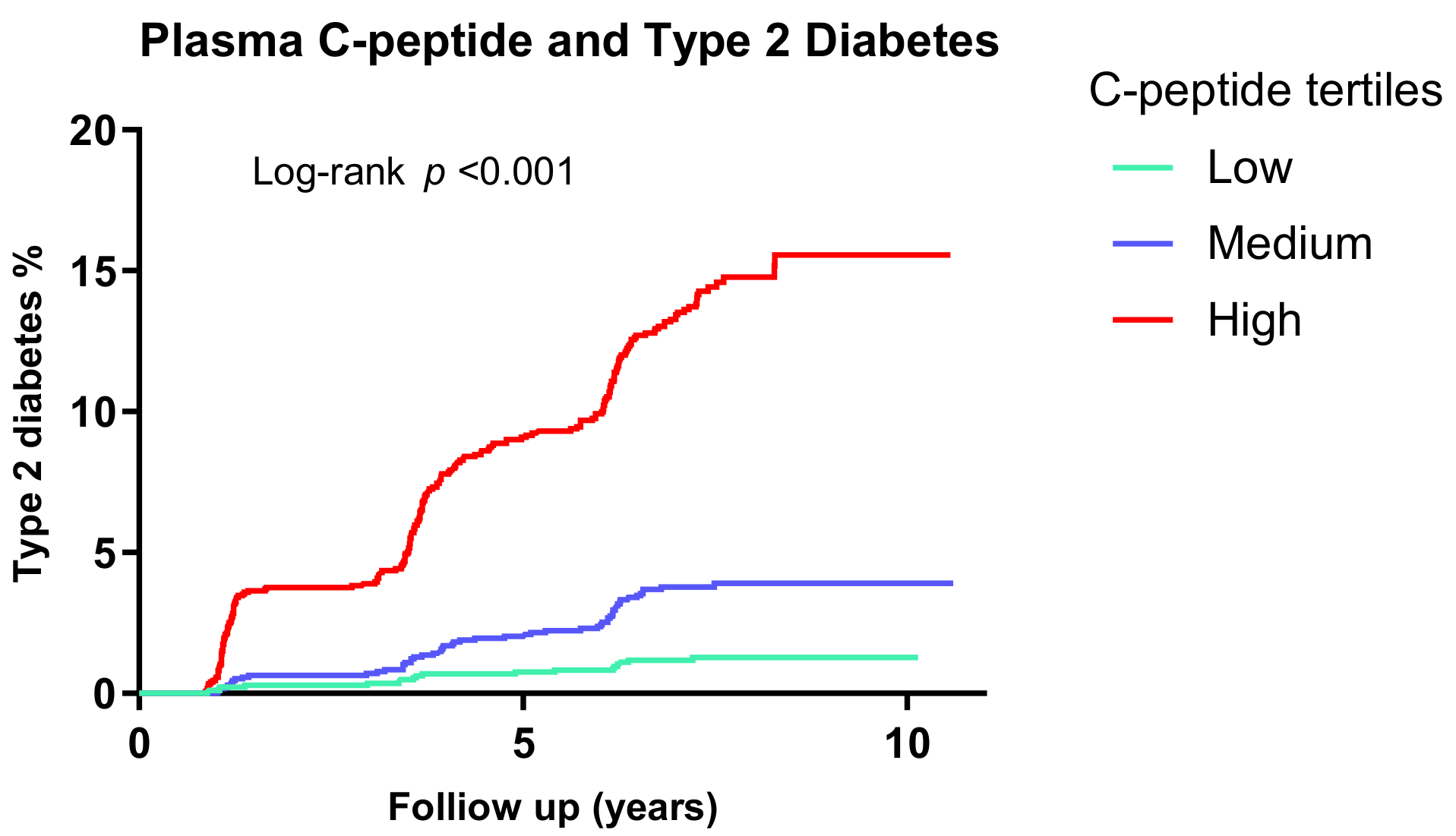
3.3. Plasma C-Peptide and Type 2 Diabetes
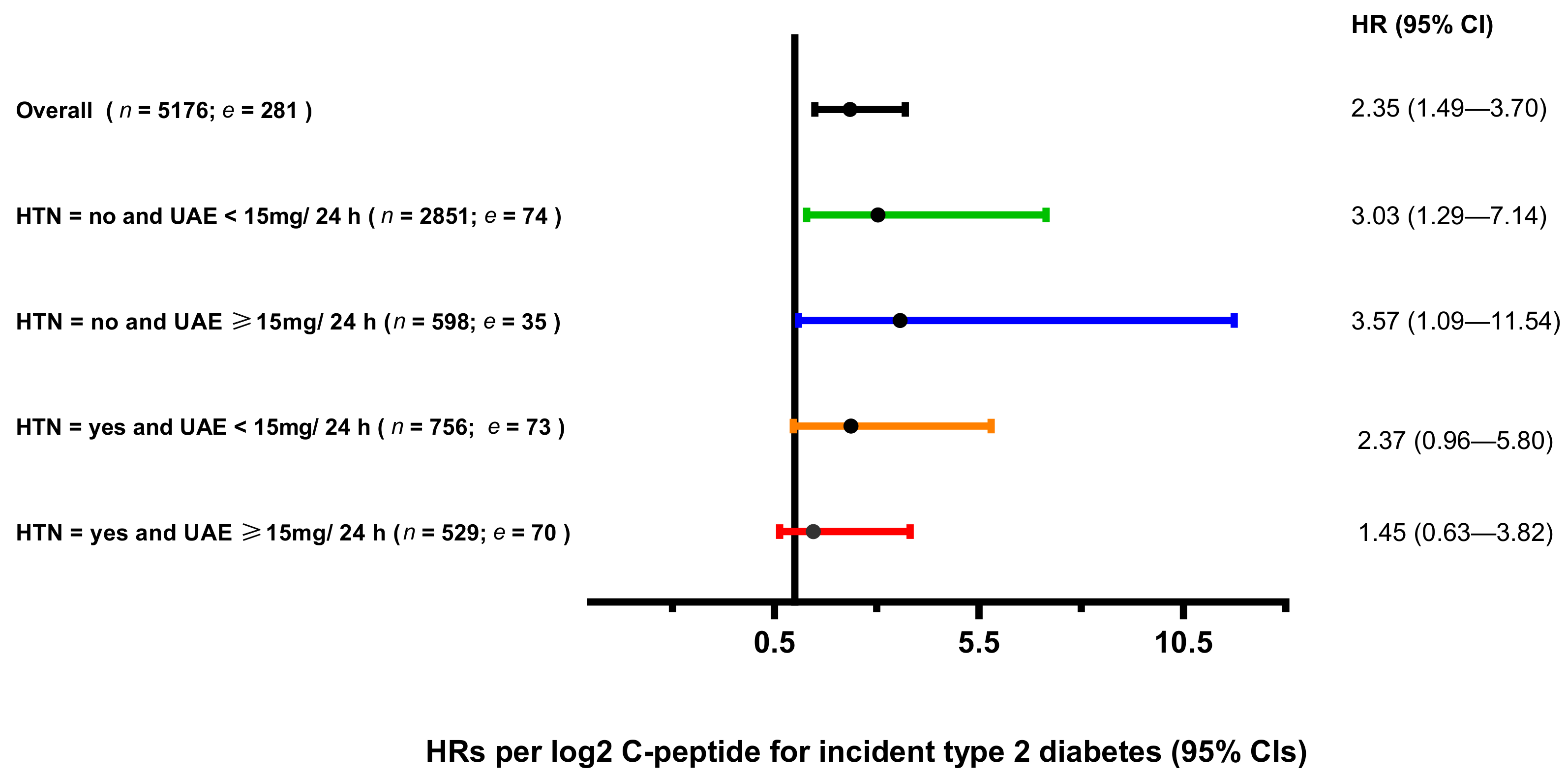
3.4. Secondary Analyses on C-Peptide and Type 2 Diabetes
3.5. Sensitivity Analyses on C-Peptide and Type 2 Diabetes
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Ogurtsova, K.; Fernandes, J.D.R.; Huang, Y.; Linnenkamp, U.; Guariguata, L.; Cho, N.; Cavan, D.; Shaw, J.; Makaroff, L.E. IDF Diabetes Atlas: Global estimates for the prevalence of diabetes for 2015 and 2040. Diabetes Res. Clin. Pract. 2017, 128, 40–50. [Google Scholar] [CrossRef]
- Weyer, C.; Bogardus, C.; Mott, D.M.; Pratley, R.E. The natural history of insulin secretory dysfunction and insulin resistance in the pathogenesis of type 2 diabetes mellitus. J. Clin. Investig. 1999, 104, 787–794. [Google Scholar] [CrossRef]
- Martin, B.; Warram, J.; Krolewski, A.; Soeldner, J.; Kahn, C.; Bergman, R. Role of glucose and insulin resistance in development of type 2 diabetes mellitus: Results of a 25-year follow-up study. Lancet 1992, 340, 925–929. [Google Scholar] [CrossRef]
- Patel, N.; Taveira, T.H.; Choudhary, G.; Whitlatch, H.; Wu, W.-C. Fasting Serum C-Peptide Levels Predict Cardiovascular and Overall Death in Nondiabetic Adults. J. Am. Heart Assoc. 2012, 1, e003152. [Google Scholar] [CrossRef]
- Min, J.-Y.; Min, K.-B. Serum C-peptide levels as an independent predictor of diabetes mellitus mortality in non-diabetic individuals. Eur. J. Epidemiol. 2013, 28, 771–774. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.D.; Kang, S.J.; Lee, M.K.; Park, S.E.; Rhee, E.-J.; Park, C.-Y.; Oh, K.-W.; Park, S.-W.; Lee, W.-Y. C-Peptide-Based Index Is More Related to Incident Type 2 Diabetes in Non-Diabetic Subjects than Insulin-Based Index. Endocrinol. Metab. 2016, 31, 320–327. [Google Scholar] [CrossRef]
- De León, A.C.; García, J.G.O.; Rodríguez, I.M.; González, D.A.; Sánchez, J.J.A.; Díaz, B.B.; Coello, S.D.; Martínez-Sellés, M.; Jaime, A.A.; Pérez, M.D.C.R. C-peptide as a risk factor of coronary artery disease in the general population. Diabetes Vasc. Dis. Res. 2015, 12, 199–207. [Google Scholar] [CrossRef]
- Hsu, C.-N.; Chang, C.-H.; Lin, Y.-S.; Lin, J.-W.; Caffrey, J.L. Association of Serum C-Peptide Concentrations with Cancer Mortality Risk in Pre-Diabetes or Undiagnosed Diabetes. PLoS ONE 2013, 8, e55625. [Google Scholar] [CrossRef]
- Cardellini, M.; Farcomeni, A.; Ballanti, M.; Morelli, M.; Davato, F.; Cardolini, I.; Grappasonni, G.; Porzio, O.; Pecchioli, C.; Rizza, S.; et al. C-peptide: A predictor of cardiovascular mortality in subjects with established atherosclerotic disease. Diabetes Vasc. Dis. Res. 2017, 14, 395–399. [Google Scholar] [CrossRef]
- Pikkemaat, M.; Andersson, T.; Melander, O.; Chalmers, J.P.; Rådholm, K.; Boström, K.B. C-peptide predicts all-cause and cardiovascular death in a cohort of individuals with newly diagnosed type 2 diabetes. The Skaraborg diabetes register. Diabetes Res. Clin. Pract. 2019, 150, 174–183. [Google Scholar] [CrossRef]
- Chen-Huan, C.; Shih-Tzer, T.; Jen-Hsiang, C.; Mau-Song, C.; Shih-Pu, W.; Pesus, C. Population-based study of insulin, C-peptide, and blood pressure in Chinese with normal glucose tolerance. Am. J. Cardiol. 1995, 76, 585–588. [Google Scholar] [CrossRef]
- Kruszynska, Y.T.; Home, P.D.; Hanning, I.; Alberti, K.G.M.M. Basal and 24-h C-peptide and insulin secretion rate in normal man. Diabetologia 1987, 30, 16–21. [Google Scholar] [CrossRef] [PubMed]
- Ashby, J.P.; Frier, B.M. Circulating C-Peptide: Measurement and Clinical Application. Ann. Clin. Biochem. Int. J. Lab. Med. 1981, 18, 125–130. [Google Scholar] [CrossRef] [PubMed]
- Tura, A.; Ludvik, B.; Nolan, J.J.; Pacini, G.; Thomaseth, K. Insulin and C-peptide secretion and kinetics in humans: Direct and model-based measurements during OGTT. Am. J. Physiol. Metab. 2001, 281, E966–E974. [Google Scholar] [CrossRef]
- Steiner, D.F.; Park, S.-Y.; Støy, J.; Philipson, L.H.; Bell, G.I. A brief perspective on insulin production. Diabetes Obes. Metab. 2009, 11, 189–196. [Google Scholar] [CrossRef]
- Heerspink, H.J.L.; Brantsma, A.H.; De Zeeuw, D.; Bakker, S.J.L.; De Jong, P.E.; Gansevoort, R.T.; for The PREVEND Study Group. Albuminuria Assessed From First-Morning-Void Urine Samples Versus 24-Hour Urine Collections as a Predictor of Cardiovascular Morbidity and Mortality. Am. J. Epidemiol. 2008, 168, 897–905. [Google Scholar] [CrossRef]
- Abbasi, A.; Corpeleijn, E.; Postmus, D.; Gansevoort, R.T.; De Jong, P.E.; Gans, R.O.B.; Struck, J.; Hillege, H.L.; Stolk, R.P.; Navis, G.; et al. Plasma procalcitonin and risk of type 2 diabetes in the general population. Diabetologia 2011, 54, 2463–2465. [Google Scholar] [CrossRef]
- Gruppen, E.G.; Bakker, S.J.; James, R.W.; Dullaart, R.P.F. Serum paraoxonase-1 activity is associated with light to moderate alcohol consumption: The PREVEND cohort study. Am. J. Clin. Nutr. 2018, 108, 1283–1290. [Google Scholar] [CrossRef]
- Kunutsor, S.K.; Bakker, S.J.; Kootstra-Ros, J.E.; Blokzijl, H.; Gansevoort, R.T.; Dullaart, R.P. Inverse linear associations between liver aminotransferases and incident cardiovascular disease risk: The PREVEND study. Atherosclerosis 2015, 243, 138–147. [Google Scholar] [CrossRef]
- Inker, L.A.; Schmid, C.H.; Tighiouart, H.; Eckfeldt, J.H.; Feldman, H.I.; Greene, T.; Kusek, J.W.; Manzi, J.; Van Lente, F.; Zhang, Y.L.; et al. Estimating Glomerular Filtration Rate from Serum Creatinine and Cystatin C. N. Engl. J. Med. 2012, 367, 20–29. [Google Scholar] [CrossRef]
- Matthews, D.R.; Hosker, J.P.; Rudenski, A.S.; Naylor, B.A.; Treacher, D.F.; Turner, R.C. Homeostasis model assessment: Insulin resistance and ?-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985, 28, 412–419. [Google Scholar] [CrossRef]
- Wilson, P.W.F.; Meigs, J.B.; Sullivan, L.M.; Fox, C.S.; Nathan, D.M.; D’Agostino, R.B. Prediction of Incident Diabetes Mellitus in Middle-aged Adults. Arch. Intern. Med. 2007, 167, 1068–1074. [Google Scholar] [CrossRef] [PubMed]
- Pencina, M.J.; Sr, R.B.D.; Steyerberg, E.W. Extensions of net reclassification improvement calculations to measure usefulness of new biomarkers. Stat. Med. 2010, 30, 11–21. [Google Scholar] [CrossRef]
- Teitelbaum, J.; Zatorre, J.; Carpenter, S.; Daniel, G.; Evans, A.; Gjedde, A.; Cashman, N. Insulin Resistance and Insulin Secretory Dysfunction as Precursors of Non-Insulin Dependent Diabetes Mellitus. N. Engl. J. Med. 1993, 322, 2012. [Google Scholar]
- Haffner, S.M.; Stern, M.P.; Mitchell, B.D.; Hazuda, H.P.; Patterson, J.K. Incidence of Type II Diabetes in Mexican Americans Predicted by Fasting Insulin and Glucose Levels, Obesity, and Body-Fat Distribution. Diabetes 1990, 39, 283–288. [Google Scholar] [CrossRef]
- Bergstrom, R.W.; Newell-Morris, L.L.; Leonetti, D.L.; Shuman, W.P.; Wahl, P.W.; Fujimoto, W.Y. Association of Elevated Fasting C-Peptide Level and Increased Intra-Abdominal Fat Distribution With Development of NIDDM in Japanese-American Men. Diabetes 1990, 39, 104–111. [Google Scholar] [CrossRef]
- Chailurkit, L.-O.; Jongjaroenprasert, W.; Chanprasertyothin, S.; Ongphiphadhanakul, B. Insulin and C-peptide levels, pancreatic beta cell function, and insulin resistance across glucose tolerance status in Thais. J. Clin. Lab. Anal. 2007, 21, 85–90. [Google Scholar] [CrossRef]
- Cersosimo, E.; Solis-Herrera, C.; Trautmann, M.E.; Malloy, J.; Triplitt, C.L. Assessment of Pancreatic β-Cell Function: Review of Methods and Clinical Applications. Curr. Diabetes Rev. 2014, 10, 2–42. [Google Scholar] [CrossRef]
- Muniyappa, R.; Lee, S.; Chen, H.; Quon, M.J. Current approaches for assessing insulin sensitivity and resistance in vivo: Advantages, limitations, and appropriate usage. Am. J. Physiol. Metab. 2008, 294, E15–E26. [Google Scholar] [CrossRef]
- Laakso, M. How Good a Marker Is Insulin Level for Insulin Resistance? Am. J. Epidemiol. 1993, 137, 959–965. [Google Scholar] [CrossRef]
- Akter, S.; Nanri, A.; Yi, S.; Pham, N.M.; Kurotani, K.; Kimura, Y.; Matsushita, Y.; Mizoue, T. Dietary patterns and C-peptide concentrations in a Japanese working population. Nutrition 2012, 28, e29–e35. [Google Scholar] [CrossRef] [PubMed]
- Meyer, K.A.; Conigrave, K.; Chu, N.-F.; Rifai, N.; Spiegelman, N.; Stampfer, M.J.; Rimm, E.B. Alcohol consumption patterns and HbA1c, C-peptide and insulin concentrations in men. J. Am. Coll. Nutr. 2003, 22, 185–194. [Google Scholar] [CrossRef] [PubMed]
- Saad, M.F.; Rewers, M.; Selby, J.; Howard, G.; Jinagouda, S.; Fahmi, S.; Zaccaro, D.; Bergman, R.N.; Savage, P.J.; Haffner, S.M. Insulin Resistance and Hypertension. Hypertension 2004, 43, 1324–1331. [Google Scholar] [CrossRef] [PubMed]
- Manolio, T.A.; Savage, P.J.; Burke, G.L.; Liu, K.A.; Wagenknecht, L.E.; Sidney, S.; Jacobs, D.R.; Roseman, J.M.; Donahue, R.P.; Oberman, A. Association of fasting insulin with blood pressure and lipids in young adults. The CARDIA study. Arteriosclerosis 1990, 10, 430–436. [Google Scholar] [CrossRef]
- Zhou, M.-S.; Wang, A.; Yu, H. Link between insulin resistance and hypertension: What is the evidence from evolutionary biology? Diabetol. Metab. Syndr. 2014, 6, 12. [Google Scholar] [CrossRef]
- Boller, S.; Joblin, B.A.; Xu, L.; Item, F.; Trüb, T.; Boschetti, N.; Spinas, G.A.; Niessen, M. From signal transduction to signal interpretation: An alternative model for the molecular function of insulin receptor substrates. Arch. Physiol. Biochem. 2012, 118, 148–155. [Google Scholar] [CrossRef]
- Rocchini, A.P.; Katch, V.; Kveselis, D.; Moorehead, C.; Martin, M.; Lampman, R.; Gregory, M. Insulin and renal sodium retention in obese adolescents. Hypertension 1989, 14, 367–374. [Google Scholar] [CrossRef]
- Landsberg, L.; Aronne, L.J.; Beilin, L.J.; Burke, V.; Igel, L.I.; Sowers, J.; Lloyd-Jones, N.; Lloyd-Jones, D.M. Obesity-Related Hypertension: Pathogenesis, Cardiovascular Risk, and Treatment. J. Clin. Hypertens. 2012, 15, 14–33. [Google Scholar] [CrossRef]
- Venkatesh, P.; Jayasingh, K.; Srikanth, K.; Green, S.R. Cross sectional study of microalbuminuria, C-peptide and fundal changes in pre-diabetics. Int. J. Adv. Med. 2018, 5, 271. [Google Scholar] [CrossRef]
- Nosadini, R.; Cipollina, M.; Slonini, A. Close relationship between Microalbuminuria and insulin resistance. J. Chem. Inf. Model. 2019, 53, 1689–1699. [Google Scholar] [CrossRef]
- Lopez-Giacoman, S.; Madero, M. Biomarkers in chronic kidney disease, from kidney function to kidney damage. World J. Nephrol. 2015, 4, 57–73. [Google Scholar] [CrossRef] [PubMed]
- Viazzi, F.; Pontremoli, R. Blood pressure, albuminuria and renal dysfunction: The ’chicken or egg’ dilemma. Nephrol. Dial. Transplant. 2014, 29, 1453–1455. [Google Scholar] [CrossRef] [PubMed]
- Kazi, A.A.; Blonde, L. Classification of Diabetes Mellitus. Clin. Lab. Med. 2019, 23–25. [Google Scholar] [CrossRef]
| Sex-Specific Tertiles of C-Peptide pmol/L | |||||
|---|---|---|---|---|---|
| Male | <642 | 642–890 | >890 | p value for trend * | |
| Female | <592 | 592–803 | >803 | ||
| Participants, n | 1723 | 1725 | 1728 | ||
| Female, (%) | 50.3% | 50% | 50.3% | 0.986 | |
| Age | 48.7 ± 10.6 | 52.3 ± 11.4 | 56.5 ± 11 | <0.001 | |
| Race, white (%) | 99 | 99.1 | 99.5 | 0.123 | |
| The family history of diabetes (%) | 13.8 | 17.6 | 21.8 | <0.001 | |
| Smoking status, | |||||
| Never (%) | 34.3 | 29.6 | 26.1 | <0.001 | |
| Current (%) | 27.0 | 28.4 | 25.3 | ||
| Former (%) | 38.7 | 42.0 | 48.6 | ||
| Alcohol consumption, | |||||
| none (%) | 18.9 | 22.6 | 28.9 | <0.001 | |
| 1–4 units per month (%) | 15.8 | 18.2 | 17.8 | ||
| 2–7 units per week (%) | 35.3 | 32.7 | 29.2 | ||
| 1–3 units per day (%) | 25.2 | 22.9 | 19.8 | ||
| >3 units per day (%) | 4.8 | 3.5 | 4.4 | ||
| Gestational Diabetes (%) | 1.0 | 2.2 | 1.6 | 0.337 | |
| Length (cm) | 174 ± 9 | 173 ± 9 | 171 ± 9 | <0.001 | |
| Weight (kg) | 72.9 ± 11.3 | 78.5 ± 12.3 | 86.7 ± 15.1 | <0.001 | |
| BMI (kg/m2) | 23.9 ± 2.7 | 26.1 ± 3.2 | 29.3 ± 4.3 | <0.001 | |
| Systolic blood pressure (mmHg) | 118.9 ± 15.8 | 124.1 ± 17 | 131.7 ± 18.2 | <0.001 | |
| Diastolic blood pressure (mmHg) | 70.3 ± 8.7 | 73.1 ± 8.6 | 75.8 ± 8.6 | <0.001 | |
| Use of antihypertensive medication (%) | 6.5 | 12.2 | 27.3 | <0.001 | |
| Hypertension (%) | 14.7 | 23.4 | 44.3 | <0.001 | |
| Total Cholesterol (mmol/L) | 5.1 ± 0.9 | 5.4±1 | 5.6±1 | <0.001 | |
| HDL-cholestrerol (mmol/L) | 1.37 ± 0.30 | 1.26 ± 0.26 | 1.15 ± 0.27 | <0.001 | |
| Triglycerides (mmol/L) | 0.85 (0.64–1.11) | 1.08 (0.83–1.49) | 1.47 (1.08–2.03) | <0.001 | |
| Use of lipid-lowering medication (%) | 3 | 6.7 | 14.2 | <0.001 | |
| Glucose (mmol/L) | 4.5 ± 0.5 | 4.8 ± 0.5 | 5 ± 0.6 | <0.001 | |
| Insulin (mU/L) | 5.2 (4.2–6.6) | 7.8 (6.5–9.5) | 13.1 (10.3–17.9) | <0.001 | |
| HOMA-IR ((mU mmol/l2)/22.5) | 1.1 ± 0.5 | 1.8 ± 0.8 | 3.4 ± 1.9 | <0.001 | |
| Plasma ASAT (U/L) | 22 (19–25) | 22 (19–26) | 24 (20–28) | <0.001 | |
| Plasma ALAT (U/L) | 15 (12–20) | 17 (12–23) | 21 (15–29) | <0.001 | |
| Plasma urea (mmol/L) | 4.9 ± 1.2 | 5.1 ± 1.3 | 5.5 ± 1.6 | <0.001 | |
| eGFR (mL/min/1.73 m2) | 99.1 ± 13.7 | 94.1 ± 15.4 | 86.9 ± 17.3 | <0.001 | |
| UAE (mg/24 h) | 7.52 (5.73–11.15) | 8.26 (6.00–13.27) | 10.19 (6.56–20.91) | <0.001 | |
| Sex Specific Tertiles of Plasma C-Peptide, pmol/L | C-Peptide Per Log2 Unit Increase | |||||
|---|---|---|---|---|---|---|
| Male | <642 | 642–890 | >890 | p value | ||
| Female | <592 1 | 592–803 2 | >803 3 | |||
| Cases | 19 | 57 | 213 | 289 | ||
| Person-years | 11,860 | 11,643 | 10,756 | 34,260 | <0.001 | |
| Crude analysis | 1.00 (ref) | 3.05 (1.81–5.13) | 12.45 (7.78–19.91) | 6.09 (5.05–7.36) | <0.001 | |
| Model 1 | 1.00 (ref) | 2.83 (1.68–4.77) | 10.77 (6.69–17.33) | 5.47 (4.48–6.68) | <0.001 | |
| Model 2 | 1.00 (ref) | 2.78 (1.65–4.68) | 10.41 (4.46–16.75) | 5.38 (4.35–6.51) | <0.001 | |
| Model 3 | 1.00 (ref) | 2.16 (1.28–3.66) | 5.21 (3.15–8.61) | 3.47 (2.72–4.43) | <0.001 | |
| Model 4 | 1.00 (ref) | 2.01 (1.16–3.50) | 3.97 (2.30–6.85) | 2.90 (2.20–3.80) | <0.001 | |
| Model 5 | 1.00 (ref) | 1.93 (1.1–3.37) | 3.75 (2.16–6.52) | 3.26 (2.42–4.36) | <0.001 | |
| Model 6 | 1.00 (ref) | 1.65 (0.94–2.89) | 2.40 (1.32–4.36) | 2.35 (1.49–3.70) | <0.001 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sokooti, S.; Kieneker, L.M.; Borst, M.H.d.; Muller Kobold, A.; Kootstra-Ros, J.E.; Gloerich, J.; van Gool, A.J.; Heerspink, H.J.L.; T Gansevoort, R.; Dullaart, R.P.F.; et al. Plasma C-Peptide and Risk of Developing Type 2 Diabetes in the General Population. J. Clin. Med. 2020, 9, 3001. https://doi.org/10.3390/jcm9093001
Sokooti S, Kieneker LM, Borst MHd, Muller Kobold A, Kootstra-Ros JE, Gloerich J, van Gool AJ, Heerspink HJL, T Gansevoort R, Dullaart RPF, et al. Plasma C-Peptide and Risk of Developing Type 2 Diabetes in the General Population. Journal of Clinical Medicine. 2020; 9(9):3001. https://doi.org/10.3390/jcm9093001
Chicago/Turabian StyleSokooti, Sara, Lyanne M. Kieneker, Martin H. de Borst, Anneke Muller Kobold, Jenny E. Kootstra-Ros, Jolein Gloerich, Alain J. van Gool, Hiddo J. Lambers Heerspink, Ron T Gansevoort, Robin P.F. Dullaart, and et al. 2020. "Plasma C-Peptide and Risk of Developing Type 2 Diabetes in the General Population" Journal of Clinical Medicine 9, no. 9: 3001. https://doi.org/10.3390/jcm9093001
APA StyleSokooti, S., Kieneker, L. M., Borst, M. H. d., Muller Kobold, A., Kootstra-Ros, J. E., Gloerich, J., van Gool, A. J., Heerspink, H. J. L., T Gansevoort, R., Dullaart, R. P. F., & Bakker, S. J. L. (2020). Plasma C-Peptide and Risk of Developing Type 2 Diabetes in the General Population. Journal of Clinical Medicine, 9(9), 3001. https://doi.org/10.3390/jcm9093001





