What Is the Effectiveness of Different Duration Interdisciplinary Treatment Programs in Patients with Chronic Pain? A Large-Scale Longitudinal Register Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Design
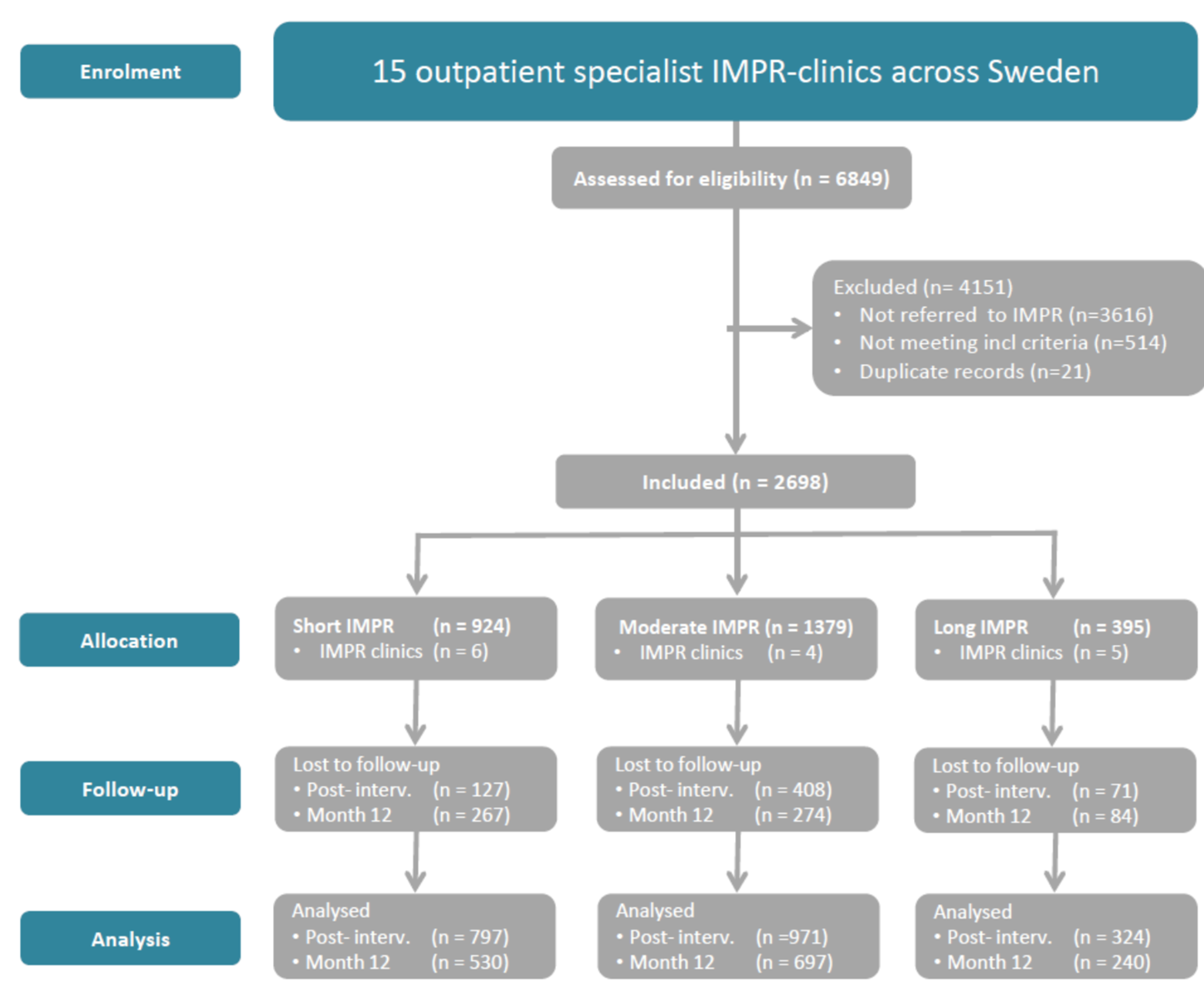
2.2. Participants
2.3. Intervention
2.4. Outcome (Dependent) Measures
2.4.1. Primary Outcome
2.4.2. Secondary Outcomes
2.5. Independent Variable and Statistics
3. Results
3.1. Results of Main Outcomes
3.1.1. Within-Group Analyses
3.1.2. Between-Group Analyses
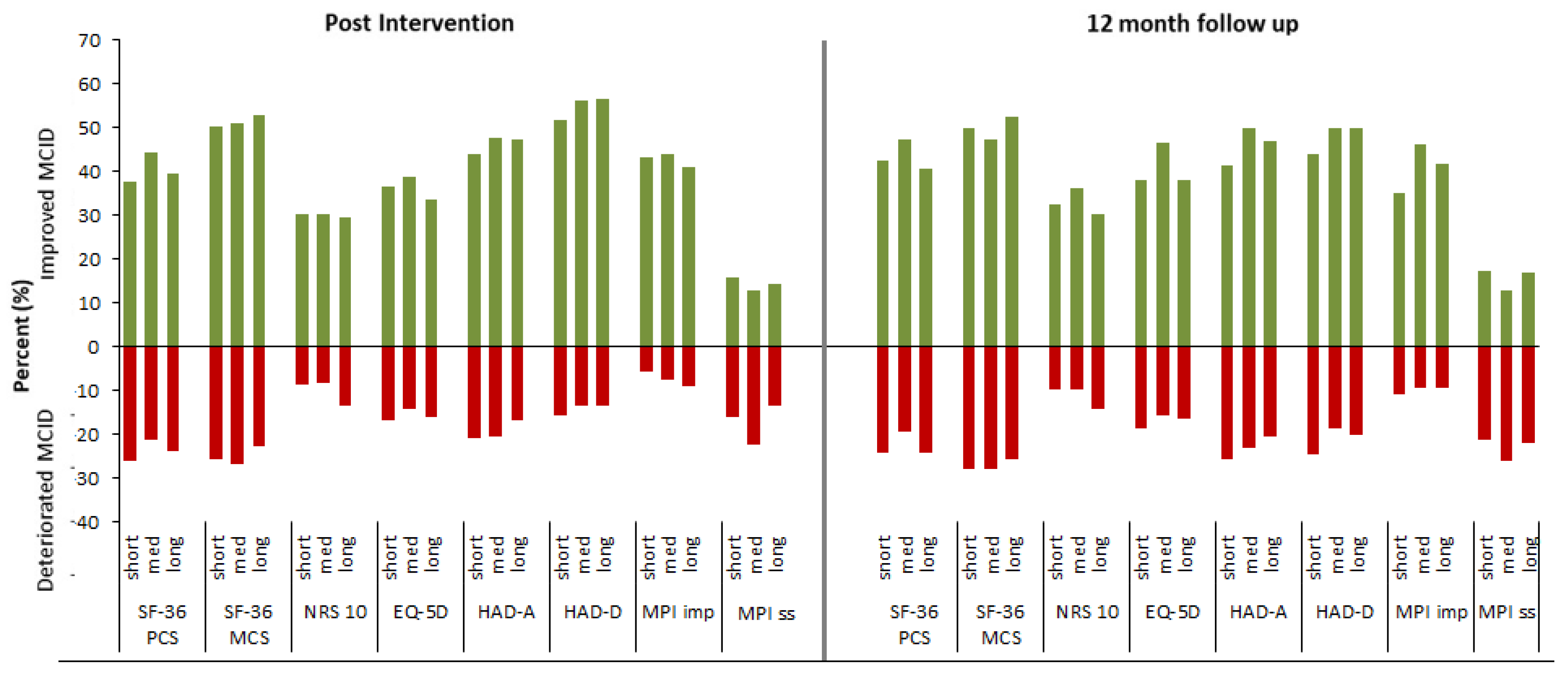
3.2. Results of Secondary Outcomes
3.2.1. Within-Group Analyses
3.2.2. Between-Group Analyses
4. Discussion
4.1. Summary of Main Findings
4.2. Effectiveness of IMPR
4.3. Comparative Effectiveness
4.4. Methodological Discussion
4.5. Implications and Future Directions
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Merskey, H.; Bogduk, N. International Association for the Study of Pain. Task Force on, T. In Classification of Chronic Pain: Descriptions of Chronic Pain Syndromes and Definitions of Pain Terms, 2nd ed.; IASP Press: Seattle, WA, USA, 1994. [Google Scholar]
- Leadley, R.M.; Armstrong, N.; Lee, Y.C.; Allen, A.; Kleijnen, J. Chronic diseases in the European Union: The prevalence and health cost implications of chronic pain. J. Pain Palliat. Care Pharmacother. 2012, 26, 310–325. [Google Scholar] [CrossRef] [PubMed]
- Raffaeli, W.; Arnaudo, E. Pain as a disease: An overview. J. Pain Res. 2017, 10, 2003–2008. [Google Scholar] [CrossRef] [PubMed]
- Turk, D.C.; Dworkin, R.H.; Revicki, D.; Harding, G.; Burke, L.B.; Cella, D.; Cleeland, C.S.; Cowan, P.; Farrar, J.T.; Hertz, S.; et al. Identifying important outcome domains for chronic pain clinical trials: An IMMPACT survey of people with pain. Pain 2008, 137, 276–285. [Google Scholar] [CrossRef] [PubMed]
- Vos, T.; Abajobir, A.A.; Abate, K.H.; Abbafati, C.; Abbas, K.M.; Abd-Allah, F.; Abdulkader, R.S.; Abdulle, A.M.; Abebo, T.A.; Abera, S.F.; et al. Global, regional, and national incidence, prevalence, and years lived with disability for 328 diseases and injuries for 195 countries, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet 2017, 390, 1211–1259. [Google Scholar] [CrossRef]
- IASP. Task Force on Multimodal Pain Treatment Defines Terms for Chronic Pain Care. Available online: https://www.iasp-pain.org/PublicationsNews/NewsDetail.aspx?ItemNumber=6981 (accessed on 14 December 2017).
- SBU. Rehabilitering vid Långvarig Smärta: En Systematisk Litteraturöversikt: Partiell Uppdatering och Fördjupning av SBU-Rapport nr 177/1+2; Statens beredning för medicinsk utvärdering (SBU): Stockholm, Sweden, 2010. [Google Scholar]
- Kamper, S.J.; Apeldoorn, A.T.; Chiarotto, A.; Smeets, R.J.E.M.; Ostelo, R.W.J.G.; Guzman, J.; van Tulder, M.W. Multidisciplinary biopsychosocial rehabilitation for chronic low back pain: Cochrane systematic review and meta-analysis. BMJ 2015, 350. [Google Scholar] [CrossRef]
- Scascighini, L.; Toma, V.; Dober-Spielmann, S.; Sprott, H. Multidisciplinary treatment for chronic pain: A systematic review of interventions and outcomes. Rheumatology (Oxford) 2008, 47, 670–678. [Google Scholar] [CrossRef]
- Van Middelkoop, M.; Rubinstein, S.M.; Kuijpers, T.; Verhagen, A.P.; Ostelo, R.; Koes, B.W.; Van Tulder, M.W. A systematic review on the effectiveness of physical and rehabilitation interventions for chronic non-specific low back pain. Eur. Spine J. 2011, 20, 19–39. [Google Scholar] [CrossRef]
- Gatchel, R.J.; McGeary, D.D.; McGeary, C.A.; Lippe, B. Interdisciplinary chronic pain management: Past, present, and future. Am. Psychol. 2014, 69, 119–130. [Google Scholar] [CrossRef]
- Kaiser, U.; Treede, R.-D.; Sabatowski, R. Multimodal pain therapy in chronic noncancer pain—Gold standard or need for further clarification? PAIN 2017, 158, 1853–1859. [Google Scholar] [CrossRef]
- Waterschoot, F.P.C.; Dijkstra, P.U.; Hollak, N.; de Vries, H.J.; Geertzen, J.H.B.; Reneman, M.F. Dose or content? Effectiveness of pain rehabilitation programs for patients with chronic low back pain: A systematic review. Pain 2014, 155, 179–189. [Google Scholar] [CrossRef]
- Bandura, A. Self-efficacy: Toward a unifying theory of behavioral change. Psychol. Rev. 1977, 84, 191. [Google Scholar] [CrossRef]
- Bovend’Eerdt, T.J.; Botell, R.E.; Wade, D.T. Writing SMART rehabilitation goals and achieving goal attainment scaling: A practical guide. Clin. Rehabil. 2009, 23, 352–361. [Google Scholar] [CrossRef] [PubMed]
- Haugmark, T.; Hagen, K.B.; Provan, S.A.; Baerheim, E.; Zangi, H.A. Effects of a community-based multicomponent rehabilitation programme for patients with fibromyalgia: Protocol for a randomised controlled trial. BMJ Open 2018, 8, e021004. [Google Scholar] [CrossRef]
- Patient-Centered Outcomes Research Institute. Available online: https://www.pcori.org/research-results/research-we-support (accessed on 14 December 2019).
- Whiteneck, G.G.; Gassaway, J. SCIRehab Uses Practice-Based Evidence Methodology to Associate Patient and Treatment Characteristics With Outcomes. Arch. Phys. Med. Rehabil. 2013, 94, S67–S74. [Google Scholar] [CrossRef] [PubMed]
- Turk, D.C.; Dworkin, R.H.; Allen, R.R.; Bellamy, N.; Brandenburg, N.; Carr, D.B.; Cleeland, C.; Dionne, R.; Farrar, J.T.; Galer, B.S.; et al. Core outcome domains for chronic pain clinical trials: IMMPACT recommendations. Pain 2003, 106, 337–345. [Google Scholar] [CrossRef] [PubMed]
- Dworkin, R.H.; Turk, D.C.; Farrar, J.T.; Haythornthwaite, J.A.; Jensen, M.P.; Katz, N.P.; Kerns, R.D.; Stucki, G.; Allen, R.R.; Bellamy, N.; et al. Core outcome measures for chronic pain clinical trials: IMMPACT recommendations. Pain 2005, 113, 9–19. [Google Scholar] [CrossRef]
- Haldorsen, E.M.H.; Grasdal, A.L.; Skouen, J.S.; Risa, A.E.; Kronholm, K.; Ursin, H. Is there a right treatment for a particular patient group? Comparison of ordinary treatment, light multidisciplinary treatment, and extensive multidisciplinary treatment for long-term sick-listed employees with musculoskeletal pain. Pain 2002, 95, 49–63. [Google Scholar] [CrossRef]
- Guzman, J.; Esmail, R.; Karjalainen, K.; Malmivaara, A.; Irvin, E.; Bombardier, C. Multidisciplinary rehabilitation for chronic low back pain: Systematic review. BMJ 2001, 322, 1511–1516. [Google Scholar] [CrossRef]
- Gerdle, B.; Stålnacke, B.-M.; Söderlund, A.; Åsenlöf, P. Indikation för multimodal rehabilitering vid långvarig smärta. Samarbetsprojektet Nationella Med. Indik. 2011, 1–24. [Google Scholar]
- Ware, J.E., Jr. SF-36 health survey update. Spine 2000, 25, 3130–3139. [Google Scholar] [CrossRef]
- Farrar, J.T.; Young, J.P.; LaMoreaux, L.; Werth, J.L.; Poole, R.M. Clinical importance of changes in chronic pain intensity measured on an 11-point numerical pain rating scale. Pain 2001, 94, 149–158. [Google Scholar] [CrossRef]
- van Agt, H.M.; Essink-Bot, M.L.; Krabbe, P.F.; Bonsel, G.J. Test-retest reliability of health state valuations collected with the EuroQol questionnaire. Soc. Sci. Med. 1994, 39, 1537–1544. [Google Scholar] [CrossRef]
- Kerns, R.D.; Turk, D.C.; Rudy, T.E. The West Haven-Yale Multidimensional Pain Inventory (WHYMPI). Pain 1985, 23, 345–356. [Google Scholar] [CrossRef]
- McKillop, J.M.; Nielson, W.R. Improving the usefulness of the Multidimensional Pain Inventory. Pain Res. Manag. 2011, 16, 239–244. [Google Scholar] [CrossRef]
- Zigmond, A.S.; Snaith, R.P. The hospital anxiety and depression scale. Acta Psychiatr. Scand. 1983, 67, 361–370. [Google Scholar] [CrossRef] [PubMed]
- Zetterqvist, J.; Vansteelandt, S.; Pawitan, Y.; Sjölander, A. Doubly robust methods for handling confounding by cluster. Biostatistics 2016, 17, 264–276. [Google Scholar] [CrossRef] [PubMed]
- Sjölander, A. Regression standardization with the R package stdReg. Eur. J. Epidemiol. 2016, 31, 563–574. [Google Scholar] [CrossRef]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences, 2nd ed.; Lawrence Erlbaum Associates: Hillsdale, NJ, USA, 1988. [Google Scholar]
- Jaeschke, R.; Singer, J.; Guyatt, G.H. Measurement of health status: Ascertaining the minimal clinically important difference. Control. Clin. Trials 1989, 10, 407–415. [Google Scholar] [CrossRef]
- Auffinger, B.M.; Lall, R.R.; Dahdaleh, N.S.; Wong, A.P.; Lam, S.K.; Koski, T.; Fessler, R.G.; Smith, Z.A. Measuring surgical outcomes in cervical spondylotic myelopathy patients undergoing anterior cervical discectomy and fusion: Assessment of minimum clinically important difference. PLoS ONE 2013, 8, e67408. [Google Scholar] [CrossRef]
- Lauche, R.; Langhorst, J.; Dobos, G.J.; Cramer, H. Clinically meaningful differences in pain, disability and quality of life for chronic nonspecific neck pain–a reanalysis of 4 randomized controlled trials of cupping therapy. Complementary Ther. Med. 2013, 21, 342–347. [Google Scholar] [CrossRef]
- Coretti, S.; Ruggeri, M.; McNamee, P. The minimum clinically important difference for EQ-5D index: A critical review. Expert Rev. Pharm. Outcomes Res. 2014, 14, 221–233. [Google Scholar] [CrossRef]
- Dworkin, R.H.; Turk, D.C.; Wyrwich, K.W.; Beaton, D.; Cleeland, C.S.; Farrar, J.T.; Haythornthwaite, J.A.; Jensen, M.P.; Kerns, R.D.; Ader, D.N.; et al. Interpreting the Clinical Importance of Treatment Outcomes in Chronic Pain Clinical Trials: IMMPACT Recommendations. J. Pain 2008, 9, 105–121. [Google Scholar] [CrossRef] [PubMed]
- Salaffi, F.; Stancati, A.; Silvestri, C.A.; Ciapetti, A.; Grassi, W. Minimal clinically important changes in chronic musculoskeletal pain intensity measured on a numerical rating scale. Eur. J. Pain 2004, 8, 283–291. [Google Scholar] [CrossRef] [PubMed]
- Puhan, M.A.; Frey, M.; Büchi, S.; Schünemann, H.J. The minimal important difference of the hospital anxiety and depression scale in patients with chronic obstructive pulmonary disease. Health Qual. Life Outcomes 2008, 6, 46. [Google Scholar] [CrossRef]
- Ringqvist, A.; Dragioti, E.; Bjork, M.; Larsson, B.; Gerdle, B. Moderate and Stable Pain Reductions as a Result of Interdisciplinary Pain Rehabilitation-A Cohort Study from the Swedish Quality Registry for Pain Rehabilitation (SQRP). J. Clin. Med. 2019, 8, 905. [Google Scholar] [CrossRef] [PubMed]
- Volker, G.; van Vree, F.; Wolterbeek, R.; van Gestel, M.; Smeets, R.; Köke, A.; Vlieland, T.V. Long-Term Outcomes of Multidisciplinary Rehabilitation for Chronic Musculoskeletal Pain. Musculoskelet. Care 2017, 15, 59–68. [Google Scholar] [CrossRef] [PubMed]
- Dragioti, E.; Evangelou, E.; Larsson, B.; Gerdle, B. Effectiveness of multidisciplinary programmes for clinical pain conditions: An umbrella review. J. Rehabil. Med. 2018, 50, 779–791. [Google Scholar] [CrossRef]
- Vartiainen, P.; Heiskanen, T.; Sintonen, H.; Roine, R.P.; Kalso, E. Health-related quality of life change in patients treated at a multidisciplinary pain clinic. Eur. J. Pain 2019. [Google Scholar] [CrossRef]
- Dragioti, E.; Björk, M.; Larsson, B.; Gerdle, B. A Meta-Epidemiological Appraisal of the Effects of Interdisciplinary Multimodal Pain Therapy Dosing for Chronic Low Back Pain. J. Clin. Med. 2019, 8, 871. [Google Scholar] [CrossRef]
- Stenberg, G.; Pietilä Holmner, E.; Stålnacke, B.-M.; Enthoven, P. Healthcare professional experiences with patients who participate in multimodal pain rehabilitation in primary care–a qualitative study. Disabil. Rehabil. 2016, 38, 2085–2094. [Google Scholar] [CrossRef]
- Holmner, E.P.; Fahlström, M.; Nordström, A. The effects of interdisciplinary team assessment and a rehabilitation program for patients with chronic pain. Am. J. Phys. Med. Rehabil. 2013, 92, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Merrick, D.; Sundelin, G.; Stålnacke, B.M. One-Year Follow-up of Two Different Rehabilitation Strategies for Patients with Chronic Pain. J. Rehabil. Med. 2012, 44, 764–773. [Google Scholar] [CrossRef] [PubMed]

| Short IMPR (n = 924) | Moderate IMPR (n = 1379) | Long IMPR (n = 395) | |
|---|---|---|---|
| Gender (%) females | 78.1 | 81.4 | 84.1 |
| Age mean (SD) | 43.2 (10.5) | 39.7 (10.7) | 42.6 (10.0) |
| Education level (%) | |||
| Elementary | 13.7 | 12.0 | 12.2 |
| Secondary | 56.7 | 56.8 | 53.7 |
| University | 22.9 | 25.0 | 25.3 |
| Other unspecified | 6.4 | 5.1 | 7.8 |
| Country of origin (%) | |||
| Sweden | 86.0 | 77.0 | 84.1 |
| Other European country | 7.5 | 7.2 | 5.6 |
| Outside Europe | 6.4 | 15.7 | 10.1 |
| Pain duration (months) mean (SD) | 110.0 (102.5) | 93.7 (99.1) | 119.8 (108.1) |
| Pain duration (months) median (IQR) | 73.0 (137.4) | 56.6 (122.9) | 87.0 (145.1) |
| Number of pain regions mean (SD) | 16.0 (8.4) | 15.8 (8.6) | 16.1 (8.4) |
| Working (%) | 51.5 | 59.5 | 53.5 |
| Mean Within-Subject Change and Effect Size | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Baseline Mean (SD) | Post-IMPR Mean (SD) | 12-Month FU Mean (SD) | Baseline to PostIMPR * | 95% CI | ES | Baseline to 12 Month FU * | 95% CI | ES | |
| SF-36 PCS (0–100) | 28.5 (7.8) | 30.8 (8.7) | 31.7 (9.6) | 2.0 | 1.7–2.3 | 0.28 | 2.8 | 2.4–3.2 | 0.37 |
| SF-36 MCS (0–100) | 34.6 (12.7) | 39.1 (13.0) | 38.7 (13.3) | 4.3 | 3.8–4.9 | 0.35 | 3.8 | 3.1–4.4 | 0.32 |
| Pain intensity last 7 days (NRS) (0–10) | 7.0 (1.6) | 6.1 (1.9) | 6.0 (2.1) | −0.8 | −0.9–−0.7 | −0.51 | −0.9 | 1.0–−0.8 | −0.54 |
| EQ-5D index (−0.594–1) | 0.24 (0.30) | 0.36 (0.32) | 0.40 (0.34) | 0.11 | 0.10–0.12 | 0.39 | 0.14 | 0.12–0.15 | 0.50 |
| HADS A (0–21) | 9.5 (4.8) | 8.0 (4.5) | 7.9 (4.8) | −1.4 | −1.6–−1.2 | −0.32 | −1.3 | −1.5–−1.1 | −0.33 |
| HADS D (0–21) | 8.8 (4.4) | 6.6 (4.2) | 7.1 (4.6) | −2.2 | −2.3–−2.0 | −0.51 | −1.6 | −1.8–1.4 | −0.38 |
| MPI Impairment (0–6) | 4.0 (0.8) | 3.4 (0.9) | 3.4 (1.1) | −0.5 | −0.6–−0.5 | −0.70 | −0.5 | −0.6–−0.5 | −0.62 |
| MPI Social Support (0–6) | 3.5 (1.0) | 3.4 (1.0) | 3.4 (1.0) | −0.1 | −0.1–0.0 | 0.10 | −0.1 | −0.2–−0.1 | −0.10 |
| Baseline | Post-IMPR | 12-Month Follow-Up | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Mean | 95% CI | Mean | 95% CI | p-Value | ES | Mean | 95% CI | p-Value | ES | |
| SF-36 PCS (0–100) | ||||||||||
| short | 27.9 | 27.4–28.4 | 30.2 | 29.7–30.8 | Ref. | 30.4 | 29.9–31.0 | Ref. | ||
| medium | 28.6 | 28.2–29.1 | 31.6 | 31.1–32.2 | <0.01 | 0.17 | 32.1 | 31.6–32.6 | <0.01 | 0.26 |
| long | 29.3 | 28.5–30.1 | 29.7 | 28.8–30.6 | 0.335 | −0.06 | 30.2 | 29.4–31.0 | 0.563 | −0.03 |
| SF-36 MCS (0–100) | ||||||||||
| short | 36.2 | 35.3–37.0 | 39.6 | 38.8–40.5 | 39.5 | 38.7–40.3 | ||||
| medium | 33.8 | 33.1–34.5 | 38.6 | 37.9–39.4 | 0.084 | −0.08 | 38.5 | 37.8–39.2 | 0.055 | −0.11 |
| long | 33.7 | 32.4–35.0 | 39.5 | 38.1–40.8 | 0.853 | −0.01 | 39.2 | 38.0–40.4 | 0.672 | −0.03 |
| Pain intensity last 7 days (NRS) (0-10) | ||||||||||
| short | 7.0 | 6.9–7.1 | 6.2 | 6.1–6.3 | 6.1 | 6.0–6.3 | ||||
| medium | 7.0 | 6.9–7.1 | 6.1 | 5.96–6.2 | 0.166 | −0.07 | 6.0 | 5.9–6.1 | 0.100 | −0.08 |
| long | 6.8 | 6.7–7.0 | 6.2 | 5.9–6.4 | 0.891 | −0.01 | 6.2 | 6.0–6.4 | 0.769 | 0.03 |
| EQ-5D index (−0.594–1) | ||||||||||
| short | 0.25 | 0.23–0.27 | 0.35 | 0.33–0.38 | 0.36 | 0.34–0.38 | ||||
| medium | 0.22 | 0.20–0.23 | 0.37 | 0.35–0.39 | 0.313 | 0.05 | 0.39 | 0.37–0.41 | 0.052 | 0.11 |
| long | 0.28 | 0.25–0.31 | 0.34 | 0.31–0.38 | 0.632 | −0.03 | 0.36 | 0.33–0.39 | 0.773 | −0.02 |
| HADS A (0–21) | ||||||||||
| short | 8.8 | 8.5–9.1 | 7.9 | 7.6–8.2 | 7.9 | 7.7–8.2 | ||||
| medium | 9.9 | 9.6–10.1 | 8.0 | 7.8–8.3 | 0.528 | 0.03 | 8.0 | 7.7–8.2 | 0.824 | 0.01 |
| long | 9.6 | 9.1–10.1 | 7.9 | 7.5–8.3 | 0.941 | −0.01 | 7.8 | 7.5–8.2 | 0.650 | −0.03 |
| HADS A (0–21) | ||||||||||
| short | 8.5 | 8.2–8.8 | 6.5 | 6.2–6.8 | 6.8 | 6.6–7.1 | ||||
| medium | 9.0 | 8.8–9.2 | 6.6 | 6.4–6.8 | 0.519 | 0.03 | 6.7 | 6.5–6.9 | 0.555 | −0.03 |
| long | 8.9 | 8.5- 9.3 | 6.7 | 6.4–7.1 | 0.293 | 0.07 | 7.0 | 6.6–7.4 | 0.425 | 0.06 |
| MPI Impairment (0–6) | ||||||||||
| short | 3.9 | 3.9–4.0 | 3.4 | 3.4–3.5 | 3.4 | 3.4–3.5 | ||||
| medium | 4.1 | 4.0–4.1 | 3.4 | 3.4–3.5 | 0.547 | 0.02 | 3.4 | 3.3–3.5 | 0.240 | −0.05 |
| long | 3.9 | 3.8–4.0 | 3.5 | 3.4–3.6 | 0.298 | 0.07 | 3.5 | 3.3–3.6 | 0.731 | 0.03 |
| MPI Social Support (0–6) | ||||||||||
| short | 3.5 | 3.5–3.6 | 3.5 | 3.4–3.5 | 3.5 | 3.4–3.5 | ||||
| medium | 3.5 | 3.5–3.6 | 3.4 | 3.3–3.4 | 0.006 | −0.11 | 3.4 | 3.3–3.4 | 0.003 | −0.15 |
| long | 3.4 | 3.3–3.5 | 3.5 | 3.4–3.6 | 0.388 | 0.06 | 3.5 | 3.4–3.6 | 0.879 | 0.02 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tseli, E.; LoMartire, R.; Vixner, L.; Grooten, W.J.A.; Gerdle, B.; Äng, B.O. What Is the Effectiveness of Different Duration Interdisciplinary Treatment Programs in Patients with Chronic Pain? A Large-Scale Longitudinal Register Study. J. Clin. Med. 2020, 9, 2788. https://doi.org/10.3390/jcm9092788
Tseli E, LoMartire R, Vixner L, Grooten WJA, Gerdle B, Äng BO. What Is the Effectiveness of Different Duration Interdisciplinary Treatment Programs in Patients with Chronic Pain? A Large-Scale Longitudinal Register Study. Journal of Clinical Medicine. 2020; 9(9):2788. https://doi.org/10.3390/jcm9092788
Chicago/Turabian StyleTseli, Elena, Riccardo LoMartire, Linda Vixner, Wilhelmus Johannes Andreas Grooten, Björn Gerdle, and Björn O. Äng. 2020. "What Is the Effectiveness of Different Duration Interdisciplinary Treatment Programs in Patients with Chronic Pain? A Large-Scale Longitudinal Register Study" Journal of Clinical Medicine 9, no. 9: 2788. https://doi.org/10.3390/jcm9092788
APA StyleTseli, E., LoMartire, R., Vixner, L., Grooten, W. J. A., Gerdle, B., & Äng, B. O. (2020). What Is the Effectiveness of Different Duration Interdisciplinary Treatment Programs in Patients with Chronic Pain? A Large-Scale Longitudinal Register Study. Journal of Clinical Medicine, 9(9), 2788. https://doi.org/10.3390/jcm9092788





