Metabolic Activity of Human Embryos after Thawing Differs in Atmosphere with Different Oxygen Concentrations
Abstract
1. Introduction
2. Experimental Section
2.1. In Vitro Fertilization and Embryo Culture
2.2. Cryoembryotransfer and Sample Collection
2.3. Amino Acids Analyses
2.4. Statistics
3. Results
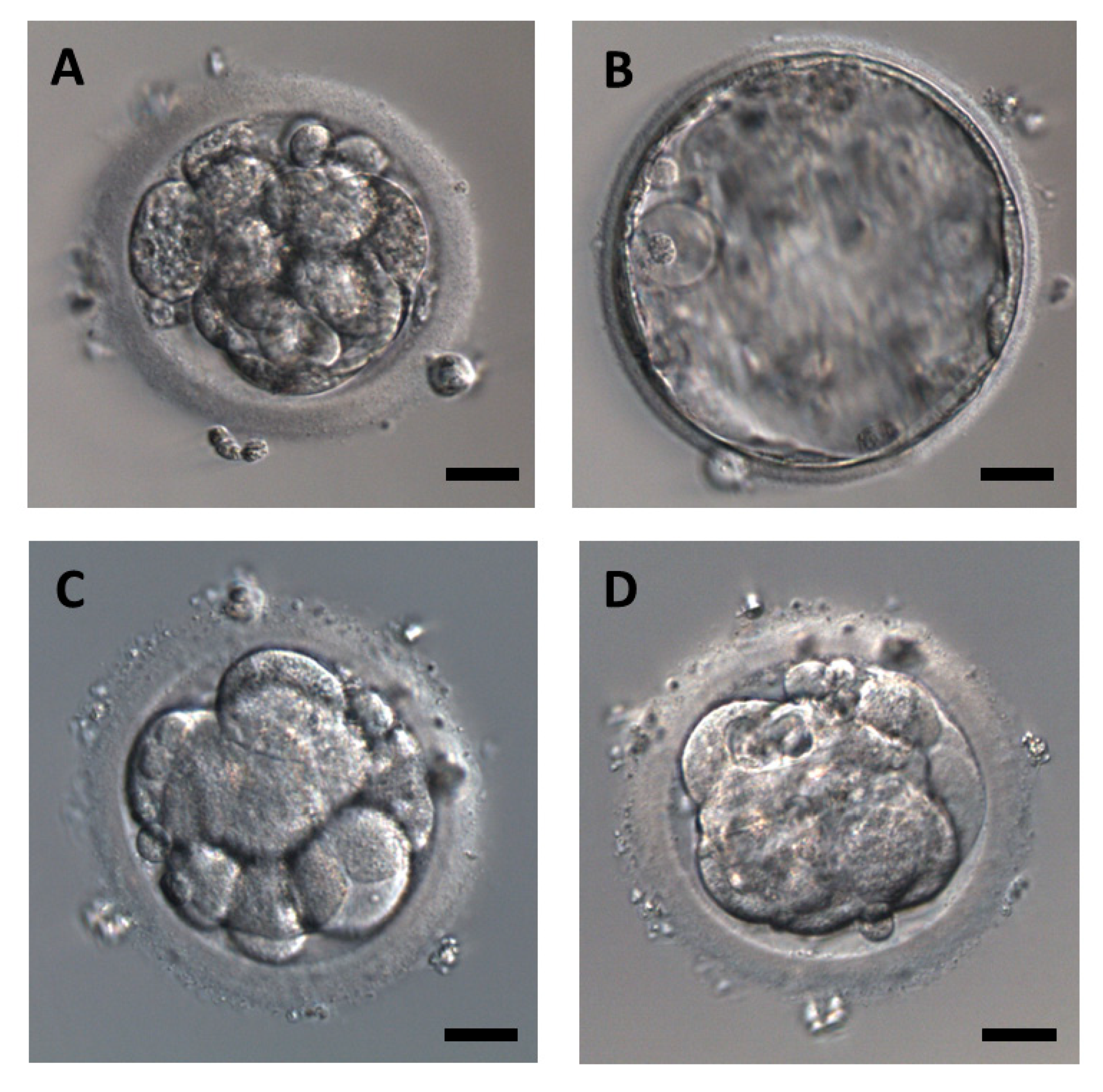
3.1. Development of Embryos after Thawing
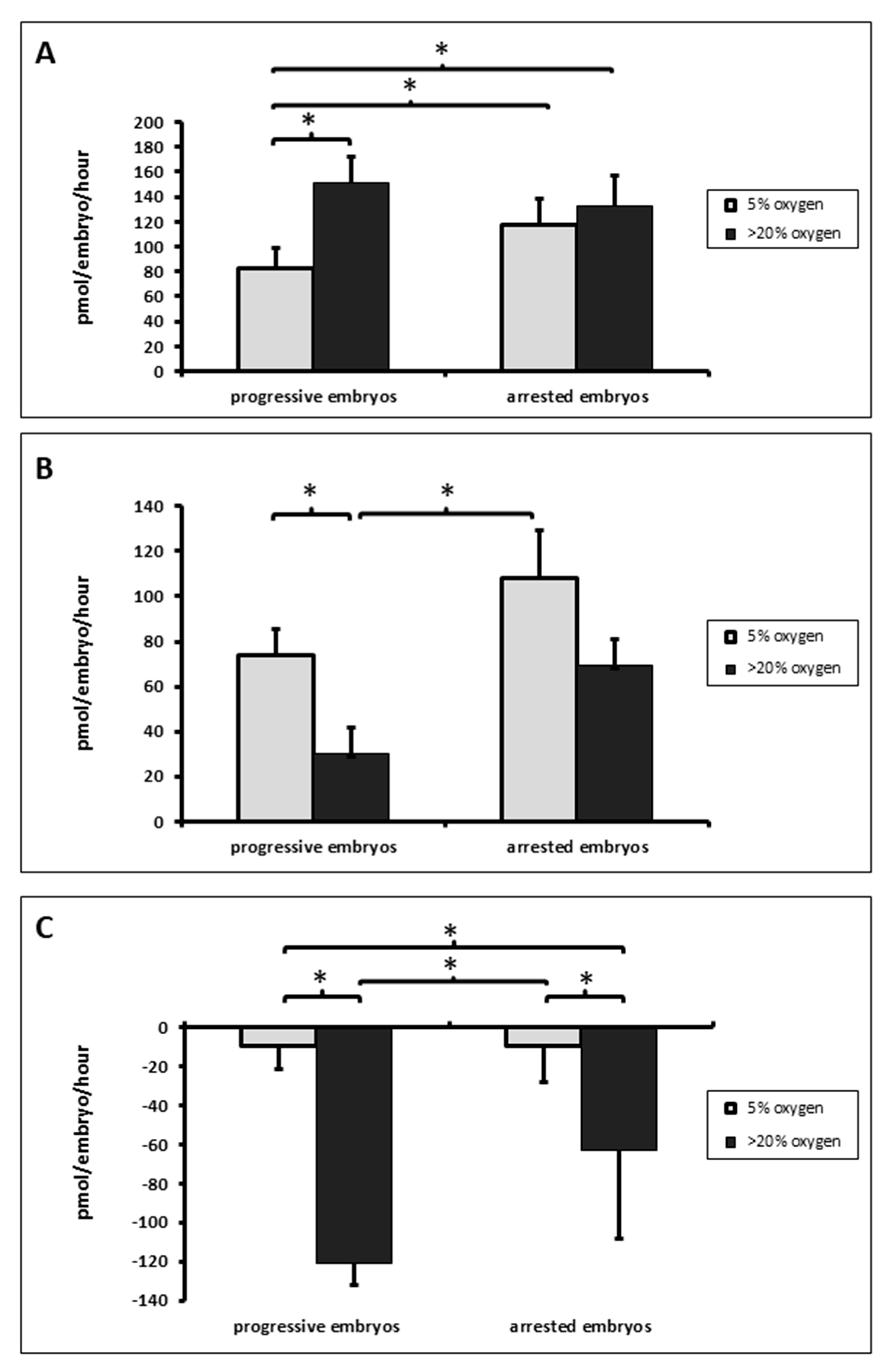
3.2. Total Amino Acid Consumption, Production and Turnover
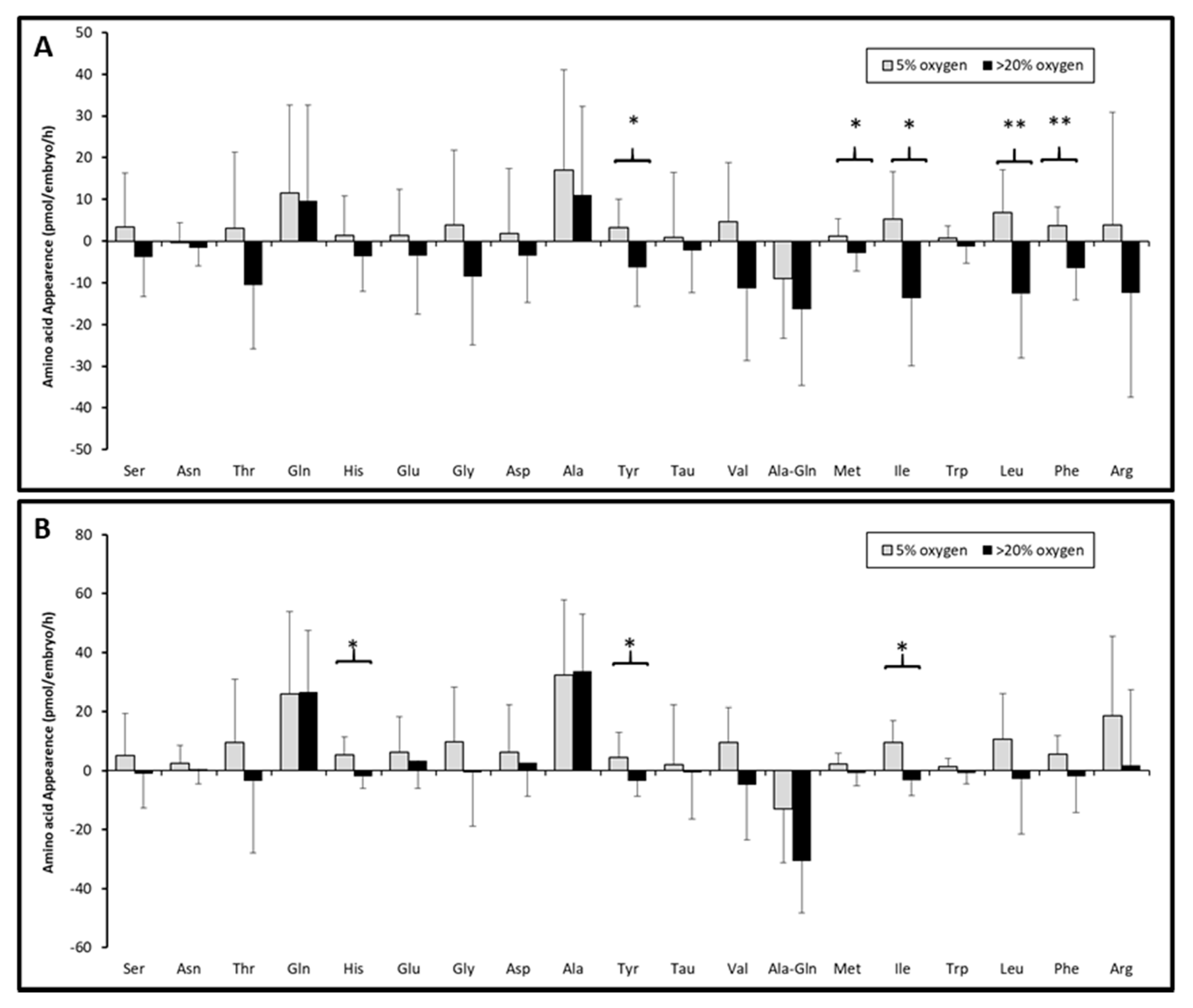
3.3. Changes of Individual Amino Acids
3.4. Development after Transfer
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Liebermann, J.; Nawroth, F.; Isachenko, V.; Isachenko, E.; Rahimi, G.; Tucker, M. Potential importance of vitrification in reproductive medicine. Biol. Reprod. 2002, 67, 1671–1680. [Google Scholar] [CrossRef] [PubMed]
- Pal, L.; Kovacs, P.; Witt, B.; Jindal, S.; Santoro, N.; Barad, D. Postthaw blastomere survival is predictive of the success of frozen-thawed embryo transfer cycles. Fertil. Steril. 2004, 82, 821–826. [Google Scholar] [CrossRef] [PubMed]
- Lane, M.; Maybach, J.; Gardner, D. Addition of ascorbate during cryopreservation stimulates subsequent embryo development. Hum. Reprod. 2002, 17, 2686–2693. [Google Scholar] [CrossRef]
- Kitagawa, Y.; Suzuki, K.; Yoneda, A.; Watanabe, T. Effects of oxygen concentration and antioxidants on the in vitro developmental ability, production of reactive oxygen species (ROS), and DNA fragmentation in porcine embryos. Theriogenology 2004, 62, 1186–1197. [Google Scholar] [CrossRef] [PubMed]
- Hosseini, S.; Forouzanfar, M.; Hajian, M.; Asgari, V.; Abedi, P.; Hosseini, L.; Ostadhosseini, S.; Moulavi, F.; Langrroodi, M.; Sadeghi, H.; et al. Antioxidant supplementation of culture medium during embryo development and/or after vitrification-warming; which is the most important? J. Assist. Reprod. Genet. 2009, 26, 355–364. [Google Scholar] [CrossRef] [PubMed][Green Version]
- VanBlerkom, J.; Antczak, M.; Schrader, R. The developmental potential of the human oocyte is related to the dissolved oxygen content of follicular fluid: Association with vascular endothelial growth factor levels and perifollicular blood flow characteristics. Hum. Reprod. 1997, 12, 1047–1055. [Google Scholar] [CrossRef]
- Fischer, B.; Bavister, B. Oxygen-tension in the oviduct and uterus of rhesus-monkeys, hamsters and rabbits. J. Reprod. Fertil. 1993, 99, 673–679. [Google Scholar] [CrossRef]
- Dumoulin, J.; Meijers, C.; Bras, M.; Coonen, E.; Geraedts, J.; Evers, J. Effect of oxygen concentration on human in-vitro fertilization and embryo culture. Hum. Reprod. 1999, 14, 465–469. [Google Scholar] [CrossRef]
- Waldenstrom, U.; Engstrom, A.; Hellberg, D.; Nilsson, S. Low-oxygen compared with high-oxygen atmosphere in blastocyst culture, a prospective randomized study. Fertil. Steril. 2009, 91, 2461–2465. [Google Scholar] [CrossRef]
- Bontekoe, S.; Mantikou, E.; van Wely, M.; Seshadri, S.; Repping, S.; Mastenbroek, S. Low oxygen concentrations for embryo culture in assisted reproductive technologies. In Cochrane Database of Systematic Reviews; John Wiley & Sons: Hoboken, NJ, USA, 2012. [Google Scholar] [CrossRef]
- Meintjes, M.; Chantilis, S.; Douglas, J.; Rodriguez, A.; Guerami, A.; Bookout, D.; Barnett, B.; Madden, J. A controlled randomized trial evaluating the effect of lowered incubator oxygen tension on live births in a predominantly blastocyst transfer program dagger. Hum. Reprod. 2009, 24, 300–307. [Google Scholar] [CrossRef]
- Guo, N.; Li, Y.; Ai, J.; Gu, L.; Chen, W.; Liu, Q. Two different concentrations of oxygen for culturing precompaction stage embryos on human embryo development competence: A prospective randomized sibling-oocyte study. Int. J. Clin. Exp. Pathol. 2014, 7, 6191–6198. [Google Scholar] [PubMed]
- Madr, A.; Glatz, Z.; Crha, I. ‘Omics’ Techniques: Genomics, Transcriptomics, Proteomics and Metabolomics in Embryo Developmental Capacity Assessment. Chem. Listy 2017, 111, 551–558. [Google Scholar]
- Baumann, C.; Morris, D.; Sreenan, J.; Leese, H. The quiet embryo hypothesis: Molecular characteristics favoring viability. Mol. Reprod. Dev. 2007, 74, 1345–1353. [Google Scholar] [CrossRef] [PubMed]
- Houghton, F.; Hawkhead, J.; Humpherson, P.; Hogg, J.; Balen, A.; Rutherford, A.; Leese, H. Non-invasive amino acid turnover predicts human embryo developmental capacity. Hum. Reprod. 2002, 17, 999–1005. [Google Scholar] [CrossRef]
- Leese, H.; Guerif, F.; Allgar, V.; Brison, D.; Lundin, K.; Sturmey, R. Biological Optimization, the Goldilocks Principle, and How Much Is Lagom in the Preimplantation Embryo. Mol. Reprod. Dev. 2016, 83, 748–754. [Google Scholar] [CrossRef]
- Stokes, P.; Hawkhead, J.; Fawthrop, R.; Picton, H.; Sharma, V.; Leese, H.; Houghton, F. Metabolism of human embryos following cryopreservation: Implications for the safety and selection of embryos for transfer in clinical IVF. Hum. Reprod. 2007, 22, 829–835. [Google Scholar] [CrossRef]
- Brison, D.; Houghton, F.; Falconer, D.; Roberts, S.; Hawkhead, J.; Humpherson, P.; Lieberman, B.; Leese, H. Identification of viable embryos in IVF by non-invasive measurement of amino acid turnover. Hum. Reprod. 2004, 19, 2319–2324. [Google Scholar] [CrossRef]
- Picton, H.; Elder, K.; Houghton, F.; Hawkhead, J.; Rutherford, A.; Hogg, J.; Leese, H.; Harris, S. Association between amino acid turnover and chromosome aneuploidy during human preimplantation embryo development in vitro. Mol. Hum. Reprod. 2010, 16, 557–569. [Google Scholar] [CrossRef]
- Cela, A.; Madr, A.; Jeseta, M.; Zakova, J.; Crha, I.; Glatz, Z. Study of metabolic activity of human embryos focused on amino acids by capillary electrophoresis with light-emitting diode-induced fluorescence detection. Electrophoresis 2018, 39, 3040–3048. [Google Scholar] [CrossRef]
- Gardner, K.; Schoolcraft, W. A randomized trial of blastocyst culture and transfer in in-vitro fertilization. Hum. Reprod. 1999, 14, 1663. [Google Scholar] [CrossRef]
- Leese, H. Quiet please, do not disturb: A hypothesis of embryo metabolism and viability. Bioessays 2002, 24, 845–849. [Google Scholar] [CrossRef] [PubMed]
- Swain, J.; Carrell, D.; Cobo, A.; Meseguer, M.; Rubio, C.; Smith, G. Optimizing the culture environment and embryo manipulation to help maintain embryo developmental potential. Fertil. Steril. 2016, 105, 571–587. [Google Scholar] [CrossRef] [PubMed]
- Wale, P.; Gardner, D. Oxygen Regulates Amino Acid Turnover and Carbohydrate Uptake During the Preimplantation Period of Mouse Embryo Development. Biol. Reprod. 2012, 87. [Google Scholar] [CrossRef] [PubMed]
- Kirkegaard, K.; Hindkjaer, J.; Ingerslev, H. Effect of oxygen concentration on human embryo development evaluated by time-lapse monitoring. Fertil. Steril. 2013, 99, 738. [Google Scholar] [CrossRef]
- Yu, Y.; Xu, Y.; Ding, C.; Khoudja, R.Y.; Lin, M.; Awoniyi, A.O.; Dai, J.; Puscheck, E.E.; Rappolee, D.A.; Zhou, C.E. Comparison of 2, 5, and 20% O2 on the development of post-thaw human embryos. J. Assist. Reprod. Genet. 2016, 33, 919–927. [Google Scholar]
- Lane, M.; Gardner, D. Ammonium induces aberrant blastocyst differentiation, metabolism, pH regulation, gene expression and subsequently alters fetal development in the mouse. Biol. Reprod. 2003, 69, 1109–1117. [Google Scholar] [CrossRef]
- Karagenc, L.; Sertkaya, Z.; Ciray, N.; Ulug, U.; Bahceci, M. Impact of oxygen concentration on embryonic development of mouse zygotes. Reprod. Biomed. Online 2004, 9, 409–417. [Google Scholar] [CrossRef]
- Houghton, F.; Leese, H. Metabolism and developmental competence of the preimplantation embryo. Eur. J. Obstet. Gynecol. Reprod. Biol. 2004, 115, S92–S96. [Google Scholar] [CrossRef]
- Hemmings, K.; Maruthini, D.; Vyjayanthi, S.; Hogg, J.; Balen, A.; Campbell, B.; Leese, H.; Picton, H. Amino acid turnover by human oocytes is influenced by gamete developmental competence, patient characteristics and gonadotrophin treatment. Hum. Reprod. 2013, 28, 1031–1044. [Google Scholar] [CrossRef]
- Gardner, D.; Lane, M. Amino-acids and ammonium regulate mouse embryo development in culture. Biol. Reprod. 1993, 48, 377–385. [Google Scholar] [CrossRef]
- Wale, P.; Gardner, D. Time-lapse analysis of mouse embryo development in oxygen gradients. Reprod. Biomed. Online 2010, 21, 402–410. [Google Scholar] [CrossRef] [PubMed]
- Edwards, L.; Williams, D.; Gardner, D. Intracellular pH of the mouse preimplantation embryo: Amino acids act as buffers of intracellular pH. Hum. Reprod. 1998, 13, 3441–3448. [Google Scholar] [CrossRef]
- Mandal, N.; Mangroo, D.; Dalluge, J.; McCloskey, J.; Rajbhandary, U. Role of the three consecutive G:C base pairs conserved in the anticodon stem of initiator tRNAs in initiation of protein synthesis in Escherichia coli. RNA Publ. RNA Soc. 1996, 2, 473–482. [Google Scholar]
- Bin, P.; Huang, R.; Zhou, X. Oxidation resistance of the sulfur amino acides: Methionine and Cysteine. Biomed Res. Int. 2017. [Google Scholar] [CrossRef]
- Kimball, S.; Shantz, L.; Horetsky, R.; Jefferson, L. Leucine regulates translation of specific mRNAs in L6 myoblasts through mTOR-mediated changes in availability of eIF4E and phosphorylation of ribosomal protein S6. J. Biol. Chem. 1999, 274, 11647–11652. [Google Scholar] [CrossRef] [PubMed]
- Anthony, J.; Yoshizawa, F.; Anthony, T.; Vary, T.; Jefferson, L.; Kimball, S. Leucine stimulates translation initiation in skeletal muscle of postabsorptive rats via a rapamycin-sensitive pathway. J. Nutr. 2000, 130, 2413–2419. [Google Scholar] [CrossRef]
- Lynch, C.; Fox, H.; Vary, T.; Jefferson, L.; Kimball, S. Regulation of amino acid-sensitive TOR signaling by leucine analogues in adipocytes. J. Cell. Biochem. 2000, 77, 234–251. [Google Scholar] [CrossRef]
- Meucci, E.; Mele, M. Amino acids and plasma antioxidant capacity. Amino Acids 1997, 12, 373–377. [Google Scholar] [CrossRef]
- Devreker, F.; Winston, R.; Hardy, K. Glutamine improves human preimplantation development in vitro. Fertil. Steril. 1998, 69, 293–299. [Google Scholar] [CrossRef]
- Orsi, N.; Leese, H. Ammonium exposure and pyruvate affect the amino acid metabolism of bovine blastocysts in vitro. Reproduction 2004, 127, 131–140. [Google Scholar] [CrossRef]
- Hansen, J.; Harris, C. Glutathione during embryonic development. Biochim. Biophys. Acta Gen. Subj. 2015, 1850, 1527–1542. [Google Scholar] [CrossRef] [PubMed]
- Guerin, P.; El Mouatassim, S.; Menezo, Y. Oxidative stress and protection against reactive oxygen species in the pre-implantation embryo and its surroundings. Hum. Reprod. Update 2001, 7, 175–189. [Google Scholar] [CrossRef] [PubMed]
- Wale, P.; Gardner, D. The effects of chemical and physical factors on mammalian embryo culture and their importance for the practice of assisted human reproduction. Hum. Reprod. Update 2016, 22, 2–22. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ješeta, M.; Celá, A.; Žáková, J.; Mádr, A.; Crha, I.; Glatz, Z.; Kempisty, B.; Ventruba, P. Metabolic Activity of Human Embryos after Thawing Differs in Atmosphere with Different Oxygen Concentrations. J. Clin. Med. 2020, 9, 2609. https://doi.org/10.3390/jcm9082609
Ješeta M, Celá A, Žáková J, Mádr A, Crha I, Glatz Z, Kempisty B, Ventruba P. Metabolic Activity of Human Embryos after Thawing Differs in Atmosphere with Different Oxygen Concentrations. Journal of Clinical Medicine. 2020; 9(8):2609. https://doi.org/10.3390/jcm9082609
Chicago/Turabian StyleJešeta, Michal, Andrea Celá, Jana Žáková, Aleš Mádr, Igor Crha, Zdeněk Glatz, Bartosz Kempisty, and Pavel Ventruba. 2020. "Metabolic Activity of Human Embryos after Thawing Differs in Atmosphere with Different Oxygen Concentrations" Journal of Clinical Medicine 9, no. 8: 2609. https://doi.org/10.3390/jcm9082609
APA StyleJešeta, M., Celá, A., Žáková, J., Mádr, A., Crha, I., Glatz, Z., Kempisty, B., & Ventruba, P. (2020). Metabolic Activity of Human Embryos after Thawing Differs in Atmosphere with Different Oxygen Concentrations. Journal of Clinical Medicine, 9(8), 2609. https://doi.org/10.3390/jcm9082609






