Prior Routine Use of Non-Steroidal Anti-Inflammatory Drugs (NSAIDs) and Important Outcomes in Hospitalised Patients with COVID-19
Abstract
1. Introduction
2. Methods
2.1. Study Population
2.2. Participants
2.3. Endpoints
2.4. Exposure
2.5. Covariates
2.6. Statistical Analysis
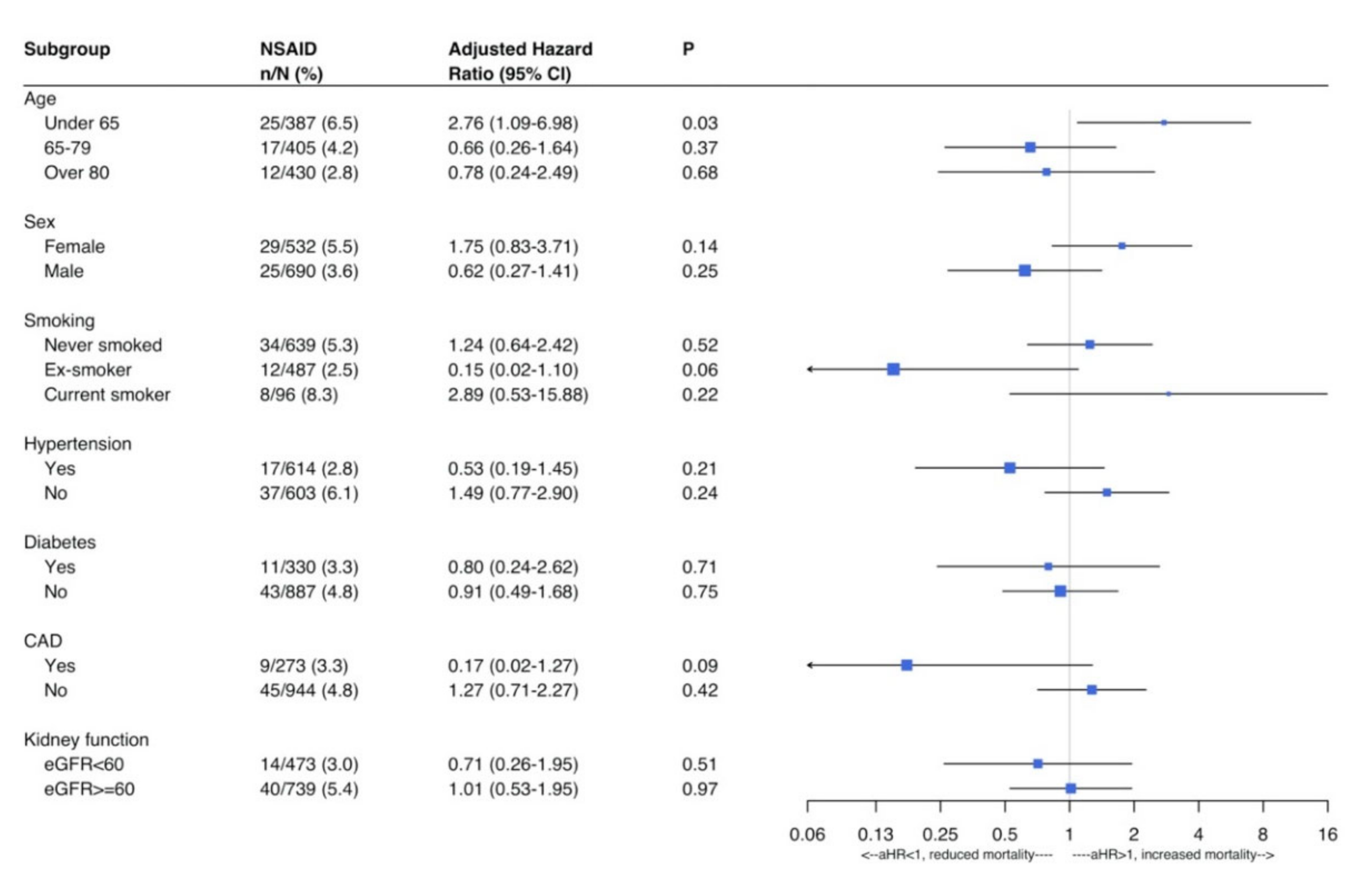
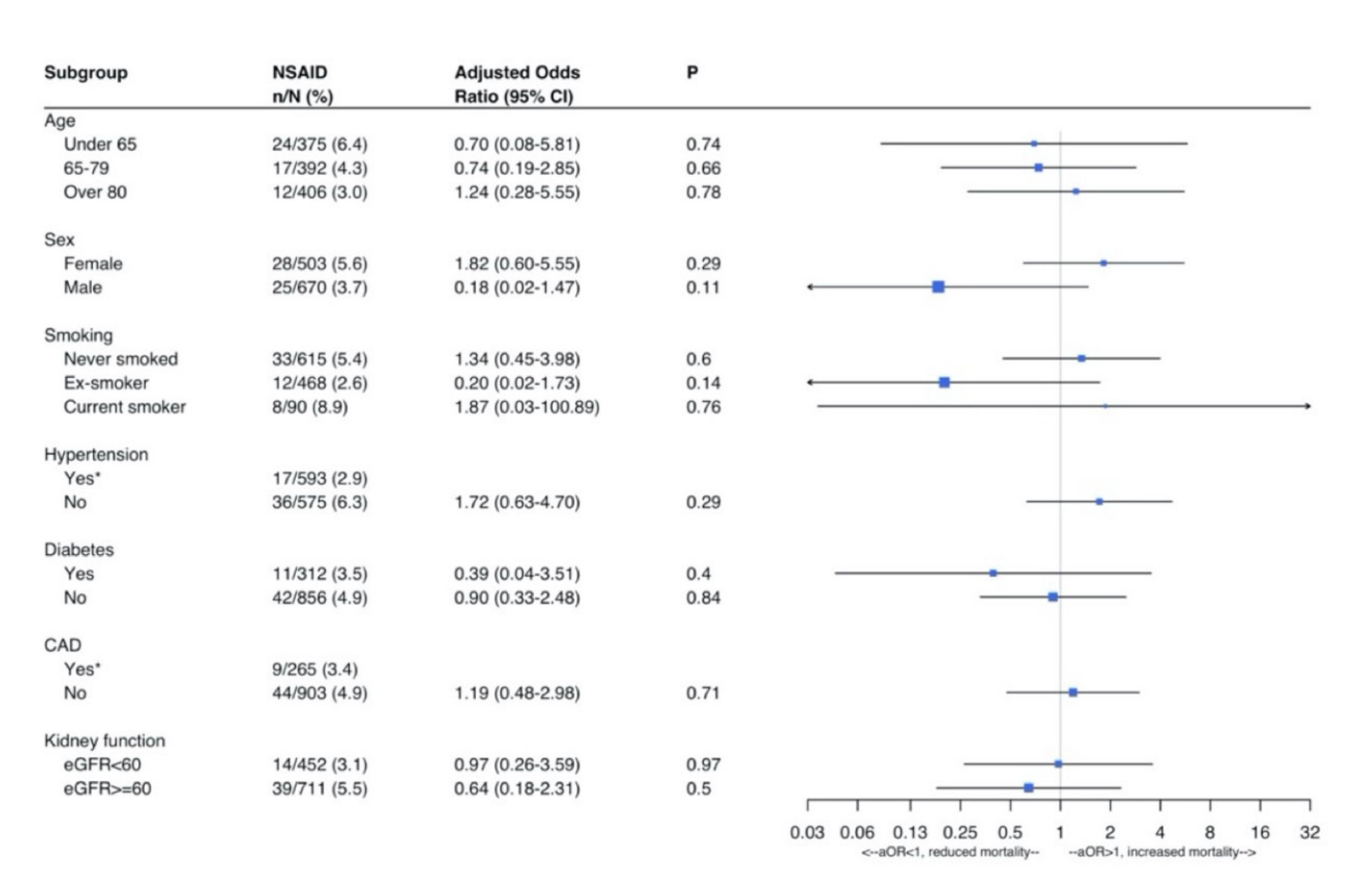
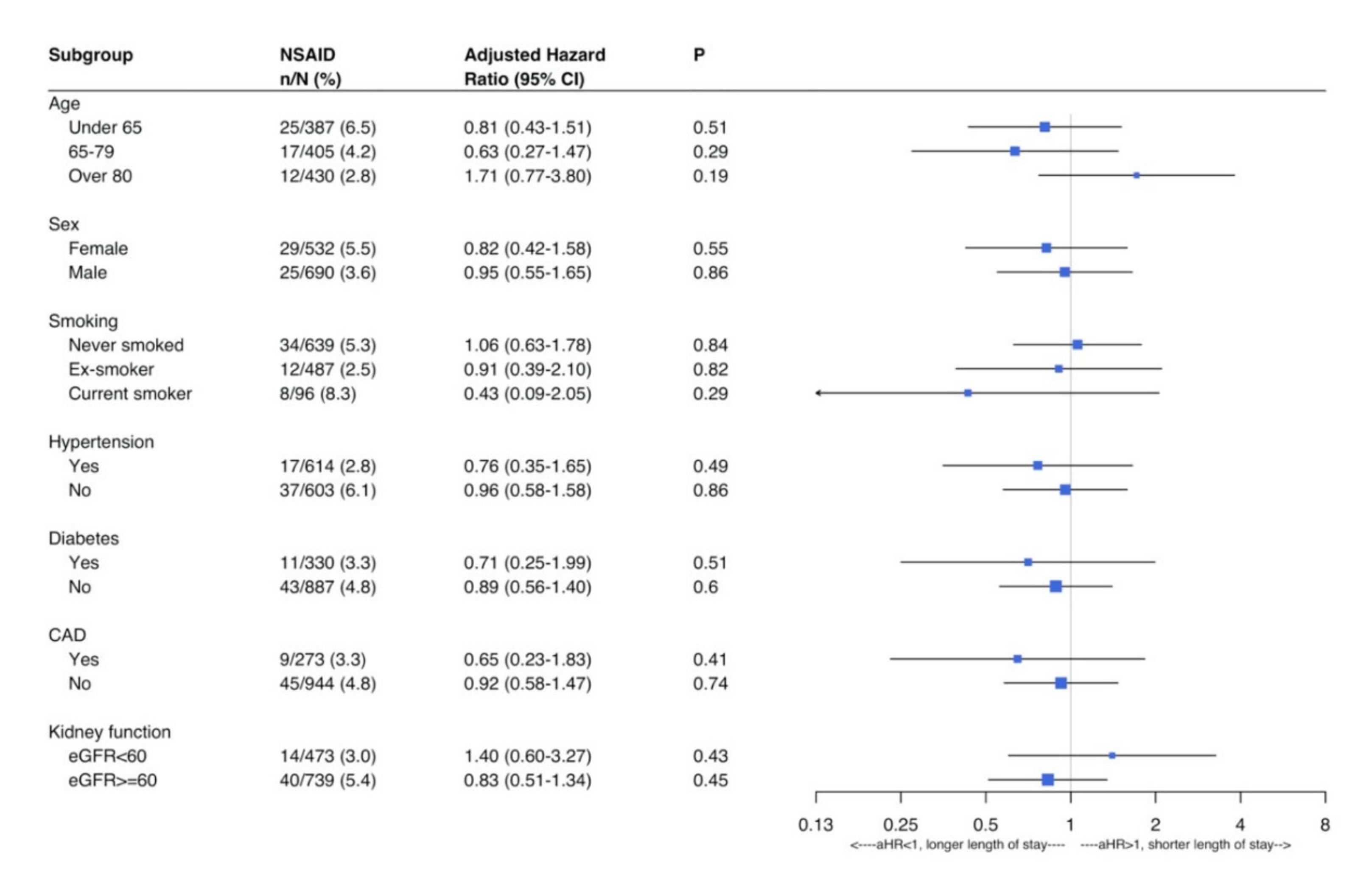
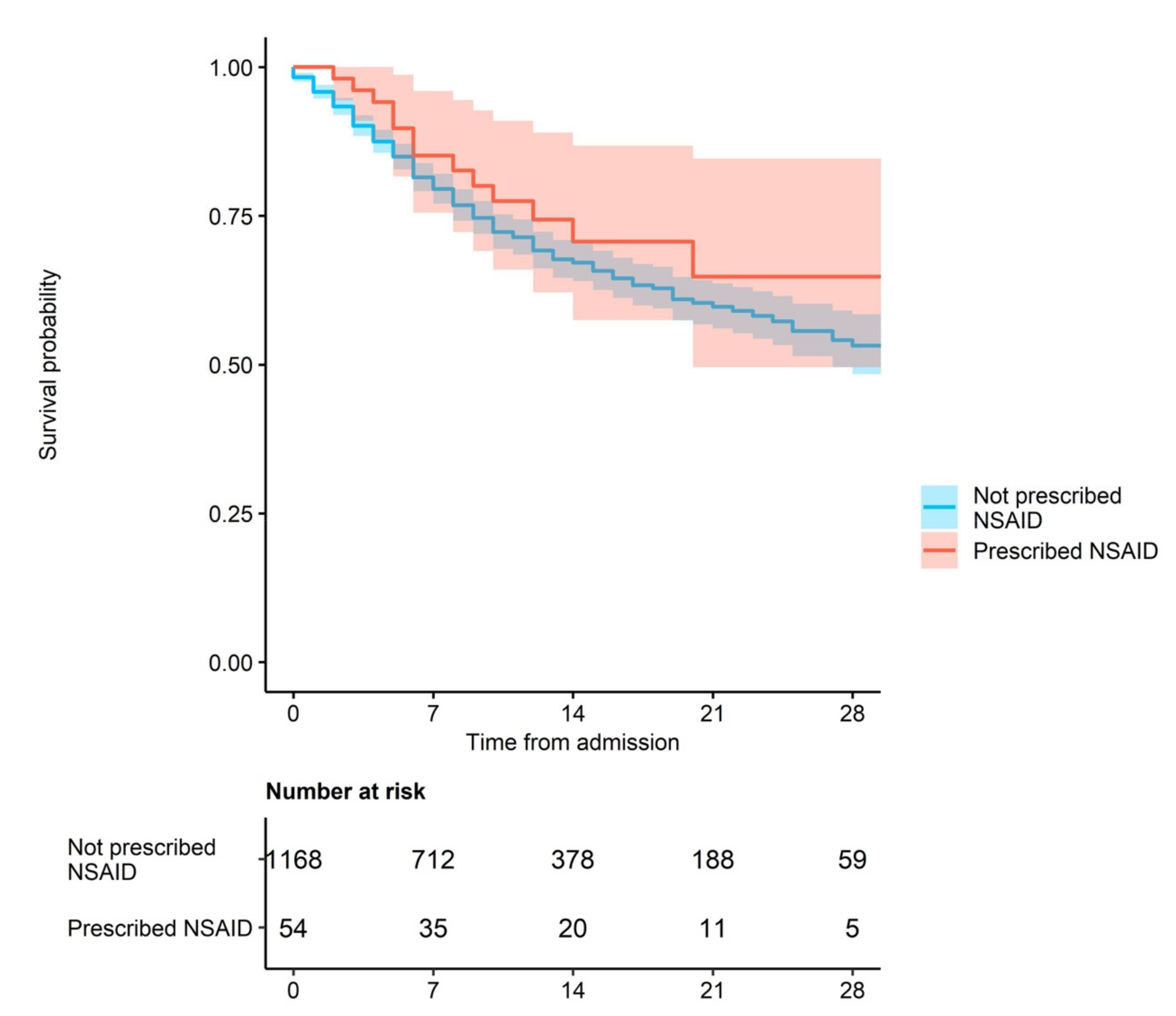
3. Results
Outcome Analysis
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| No | Yes | Total | |
|---|---|---|---|
| (n = 1168) | (n = 54) | ||
| Sites | |||
| Hospital A | 146 (95.4) | 7 (4.6) | 153 (12.5) |
| Hospital B | 35 (81.4) | 8 (18.6) | 43 (3.5) |
| Hospital C | 117 (95.1) | 6 (4.9) | 123 (10.7) |
| Hospital D | 366 (96.3) | 14 (3.7) | 380 (31.1) |
| Hospital E | 109 (97.3) | 3 (3.0) | 112 (9.2) |
| Hospital F | 235 (95.5) | 11 (4.5) | 246 (20.1) |
| Hospital G | 112 (97.4) | 3 (2.6) | 115 (9.4) |
| Hospital H | 48 (96.0) | 2 (4.0) | 50 (4.1) |
| Age | |||
| Under 65 yrs | 362 (93.4) | 25 (6.5) | 387 (31.7) |
| 65 to 79 yrs | 388 (95.8) | 17 (4.2) | 405 (33.1) |
| Over 80 yrs | 418 (97.2) | 12 (2.8) | 430 (35.2) |
| Sex | |||
| Female | 503 (94.6) | 29 (5.5) | 532 (43.5) |
| Male | 665 (96.4) | 25 (3.6) | 690 (56.5) |
| Smoking Status | |||
| Never smokers | 585 (94.5) | 34 (5.5) | 619 (50.7) |
| Ex-smokers | 475 (97.5) | 12 (2.5) | 487 (39.9) |
| Current smokers | 88 (91.7) | 8 (8.3) | 96 (7.9) |
| Missing | 20 | 0 | 20 |
| Diabetes | |||
| No | 844 (95.2) | 43 (4.9) | 887 (72.6) |
| Yes | 319 (96.7) | 11 (3.3) | 330(27.0) |
| Missing | 5 | 0 | 5 |
| Hypertension | |||
| No | 566 (93.9) | 37 (6.1) | 603 (49.4) |
| Yes | 597 (97.2) | 17 (2.8) | 614 (50.3) |
| Missing | 5 | 0 | 5 |
| Coronary Artery disease | |||
| No | 899 (95.2) | 45 (4.8) | 944 (77.3) |
| Yes | 264 (96.7) | 9 (3.3) | 273 (22.3) |
| Missing | 5 | 0 | 5 |
| Elevated CRP (>40) | |||
| No | 324 (97.0) | 10 (3.0) | 334 (27.3) |
| Yes | 825 (94.9) | 44 (5.1) | 888 (71.1) |
| Missing | 19 | 0 | 19 |
| Renal function (eGFR < 60) | |||
| No | 699 (94.6) | 10 (5.4) | 739 (60.5) |
| Yes | 459 (97.0) | 14 (3.0) | 473 (38.7) |
| Missing | 10 | 0 | 10 |
References
- Fu, Y.; Cheng, Y.; Wu, Y. Understanding SARS-CoV-2-mediated inflammatory responses: From mechanisms to potential therapeutic tools. Virol. Sin. 2020, 35, 266–271. [Google Scholar] [CrossRef]
- Russell, B.; Moss, C.; Rigg, A.; Van Hemelrijck, M. COVID-19 and treatment with NSAIDs and corticosteroids: Should we be limiting their use in the clinical setting? Ecancermedicalscience 2020, 14, 1023. [Google Scholar] [CrossRef]
- Wongrakpanich, S.; Wongrakpanich, A.; Melhado, K.; Rangaswami, J. A Comprehensive Review of Non-Steroidal Anti-Inflammatory Drug Use in The Elderly. Aging Dis. 2018, 9, 143–150. [Google Scholar] [CrossRef]
- Coxib and traditional NSAID trialists (CNT) collaboration. Vascular and upper gastrointestinal effects of non-steroidal anti-inflammatory drugs: Meta-analyses of individual participant data from randomised trials. Lancet 2013, 38, 769–779. [Google Scholar]
- Voiriot, G.; Philippot, Q.; Elabbadi, A.; Elbim, C.; Chalumeau, M.; Fartoukh, M. Risks related to the use of non-steroidal anti-inflammatory drugs in community-acquired pneumonia in adult and pediatric patients. J. Clin. Med. 2019, 8, E786. [Google Scholar] [CrossRef]
- Gracia-Ramos, A.E. Is the ACE2 Overexpression a Risk Factor for COVID-19 infection? Arch. Med. Res. 2020. [Google Scholar] [CrossRef]
- Basille, D.; Thomsen, R.W.; Madsen, M.; Duhaut, P.; Andrejak, C.; Jounieaux, V.; Sørensen, H.T. Nonsteroidal anti-inflammatory drug use and clinical outcomes of community- acquired pneumonia. Am. J. Rspir. Crit. Care Med. 2018, 198, 128–131. [Google Scholar] [CrossRef]
- Amici, C.; Di Caro, A.; Ciucci, A.; Chiappa, L.; Castilletti, C.; Martella, V.; Decaro, N.; Buonavoglia, C.; Capobianchi, M.R.; Santoro, M.G. Indomethacin has a potent antiviral activity against SARS coronavirus. Antivir. Ther. 2006, 11, 1021–1030. [Google Scholar]
- Hewitt, J.; Carter, B.; Vilches-Moraga, A.; Quinn, T.J.; Braude, P.; Verduri, A.; Pearce, L.; Stechman, M.; Short, R.; Price, A.; et al. The effect of frailty on survival in patients with COVID-19 (COPE): A multicentre, European, observational cohort study. Lancet Public Health 2020, 5, e444–e451. [Google Scholar] [CrossRef]
- Heyneman, C.A.; Lawless-Liday, C.; Wall, G.C. Oral versus Topical NSAIDs in Rheumatic Diseases. Drugs 2000, 60, 555–574. [Google Scholar] [CrossRef]
- Honvo, G.; Leclercq, V.; Geerinck, A.; Thomas, T.; Veronese, N.; Charles, A.; Rabenda, V.; Beaudart, C.; Cooper, C.; Reginster, J.Y.; et al. Safety of Topical Non-steroidal Anti-Inflammatory Drugs in Osteoarthritis: Outcomes of a Systematic Review and Meta-Analysis. Drugs Aging 2019, 36 (Suppl. 1), 45–64. [Google Scholar] [CrossRef] [PubMed]
- StataCorp. Stata Statistical Software; Release 15; StataCorp LLC.: College Station, TX, USA, 2017. [Google Scholar]
- R Core Team. A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria. 2019. Available online: https://www.R-project.org/ (accessed on 11 June 2020).
- Therneau, T. A Package for Survival Analysis in R. R Package Version 3.1–12, 2020. Available online: https://CRAN.R-project.org/package=survival (accessed on 11 June 2020).
- Kassambara, A.; Kosinski, M.; Biecek, P. Survminer: Drawing Survival Curves Using 'Ggplot2'. R Package Version 0.4.6. Available online: https://CRAN.R-project.org/package=survminer (accessed on 11 June 2020).
- Russell, B.; Moss, C.; George, G.; Santaolalla, A.; Cope, A.; Papa, S.; Van Hemelrijck, M. Associations between immune-suppressive and stimulating drugs and novel COVID-19-a systematic review of current evidence. Ecancermedicalscience 2020, 14, 1022. [Google Scholar] [CrossRef]
- MaassenVanDenBrink, A.; de Vries, T.; Danser, A.H.J. Headache medication and the COVID-19 pandemic. J. Headache Pain 2020, 21, 1–4. [Google Scholar] [CrossRef]
- Pergolizzi, J.V.; Varrassi, G.; Magnusson, P.; LeQuang, J.A.; Paladini, A.; Taylor, R.; Wollmuth, C.; Breve, F.; Christo, P. COVID-19 and NSAIDS: A Narrative Review of Knowns and Unknowns. Pain Ther. 2020, 1–6. [Google Scholar] [CrossRef]
- Yousefifard, M.; Zali, A.; Zarghi, A.; Madani Neishaboori, A.; Hosseini, M.; Safari, S. Non-steroidal anti-inflammatory drugs in management of COVID-19; a systematic review on current evidence. Int. J. Clin. Pract. 2020, e13557. [Google Scholar] [CrossRef]
- Capuano, A.; Scavone, C.; Racagni, G.; Scaglione, F. Italian Society of Pharmacology. NSAIDs in patients with viral infections, including Covid-19: Victims or perpetrators? Pharmacol. Res. 2020, 1. [Google Scholar] [CrossRef]
- Rinott, E.; Kozer, E.; Shapira, Y.; Bar-Haim, A. Youngster, I. Ibuprofen use and clinical outcomes in COVID-19 patients. Clin. Microbiol. Infect. 2020. [Google Scholar] [CrossRef]
- Gómez-Acebo, I.; Dierssen-Sotos, T.; De Pedro, M.; Pérez-Gómez, B.; Castaño-Vinyals, G.; Fernández-Villa, T.; Palazuelos-Calderón, C.; Amiano, P.; Etxeberria, J.; Benavente, Y. Epidemiology of non-steroidal anti-inflammatory drugs consumption in Spain. The MCC-Spain study. BMC Public Health 2018, 18, 1134. [Google Scholar] [CrossRef]
- Zhang, X.; Donnan, P.T.; Bell, S.; Guthrie, B. Non-steroidal anti-inflammatory drug induced acute kidney injury in the community dwelling general population and people with chronic kidney disease: Systematic review and meta-analysis. BMC Nephrol. 2017, 18, 256. [Google Scholar] [CrossRef]
- Pan, Y.; Zhang, L.; Wang, F.; Li, X.; Wang, H. China National Survey of Chronic Kidney Disease Working Group. Status of non-steroidal anti-inflammatory drugs use and its association with chronic kidney disease: A cross-sectional survey in China. Nephrology 2014, 19, 655–660. [Google Scholar] [CrossRef]
- Lanas, A.; Carrera, P.; Arguedas, Y.; Garcia, S.; Bujanda, L.; Calvet, X.; Ponce, J.; Perez-Aisa, M.; Fernández, M.; muñoz, M.; et al. Risk of upper and lower gastrointestinal bleeding in patients taking nonsteroidal anti-inflammatory drugs, antiplatelet agents, or anticoagulants. Clin. Gastroenterol. Hepatol. 2014, 13. [Google Scholar] [CrossRef]
- LIBERATE Trial in COVID-19 (LIBERATE). ClinicalTrials.gov Identifier: NCT04334629. Available online: https://clinicaltrials.gov/ct2/show/study/NCT04334629 (accessed on 11 June 2020).
| Alive | Dead | Total | p-Value & | |
|---|---|---|---|---|
| (n = 864) | (n = 358) | (n = 1222) | ||
| NSAID Prescription | 0.578 | |||
| No | 824 (70.6) | 344 (29.5) | 1168 (95.6) | |
| Yes | 40 (74.1) | 14 (25.9) | 54 (4.4) | |
| Sites | <0.001 | |||
| Hospital A | 119 (77.8) | 34 (22.2) | 153 (12.5) | |
| Hospital B | 33 (76.7) | 10 (23.3) | 43 (3.5) | |
| Hospital C | 108 (87.8) | 15 (12.2) | 123 (10.1) | |
| Hospital D | 254 (66.8) | 126 (33.2) | 380 (31.1) | |
| Hospital E | 76 (67.9) | 36 (32.1) | 112 (9.2) | |
| Hospital F | 138 (56.1) | 108 (43.9) | 246 (20.1) | |
| Hospital G | 100 (87.0) | 15 (13.0) | 115 (9.4) | |
| Hospital H | 36 (72.0) | 14 (28.0) | 50 (4.1) | |
| Age | <0.001 | |||
| Under 65 years | 337 (87.1) | 50 (12.9) | 387 (31.7) | |
| 65 to 79 years | 266 (65.7) | 139 (34.3) | 405 (33.1) | |
| Over 80 years | 261 (60.7) | 169 (39.3) | 430 (35.2) | |
| Sex | 0.625 | |||
| Female | 380 (71.4) | 152 (28.6) | 532 (43.5) | |
| Male | 484 (70.1) | 206 (29.9) | 690 (56.5) | |
| Smoking Status | 0.049 | |||
| Never smokers | 453 (73.2) | 166 (26.8) | 619 (50.7) | |
| Ex-smokers | 325 (66.7) | 162 (33.3) | 487 (39.9) | |
| Current smokers | 74 (77.1) | 22 (22.9) | 96 (7.8) | |
| Missing | 12 | 8 | 20 | |
| Diabetes | 0.100 | |||
| No | 638 (71.9) | 249 (28.1) | 887 (72.6) | |
| Yes | 223 (67.6) | 107 (32.4) | 330 (27.0) | |
| Missing | 3 | 2 | 5 | |
| Hypertension | 0.008 | |||
| No | 448 (74.3) | 155 (25.7) | 603 (49.4) | |
| Yes | 414 (67.4) | 200 (32.6) | 614 (50.3) | |
| Missing | 2 | 3 | 5 | |
| Coronary Artery disease | <0.001 | |||
| No | 696 (73.7) | 248 (26.3) | 944 (77.3) | |
| Yes | 166 (60.8) | 107 (39.2) | 273 (22.3) | |
| Missing | 2 | 3 | 5 | |
| Elevated CRP (>40) | <0.001 | |||
| No | 282 (84.4) | 52 (15.6) | 334 (27.3) | |
| Yes | 571 (65.7) | 298 (34.3) | 869 (72.7) | |
| Missing | 11 | 8 | 19 | |
| Renal function (eGFR < 60) | <0.001 | |||
| No | 568 (76.9) | 171 (23.1) | 739 (60.5) | |
| Yes | 290 (61.3) | 183 (38.7) | 473 (38.7) | |
| Missing | 6 | 4 | 10 |
| Crude Hazard Ratio (HR) | Adjusted Hazard Ratio (aHR) & | |||
|---|---|---|---|---|
| (n = 1181) | (n = 1167) | |||
| HR, (95% CI) | p-Value | aHR, (95% CI) | p-Value | |
| NSAID | 0.82 (0.48–1.40) | 0.46 | 0.89 (0.52–1.53) | 0.67 |
| Age | ||||
| Under 65 | -Ref- | -Ref- | ||
| 65 to 79 | 3.21 (2.29–4.51) | <0.001 | 3.14 (2.20–4.48) | <0.001 |
| Over 80 | 3.94 (2.82–5.50) | <0.001 | 4.00 (2.81–5.71) | <0.001 |
| Sex | ||||
| Female | -Ref- | -Ref- | ||
| Male | 0.88 (0.71–1.10) | 0.25 | 0.88 (0.70–1.11) | 0.28 |
| Smoking status | ||||
| Never | ||||
| Ex-smokers | 1.24 (1.0–1.55) | 0.06 | 1.02 (0.80–1.28) | 0.92 |
| Current smokers | 0.90 (0.56–1.42) | 0.62 | 1.11 (0.68–1.82) | 0.66 |
| Elevated CRP (>40) | 2.24 (1.65–3.05) | <0.001 | 2.75 (2.01–3.76) | <0.001 |
| Patients with diabetes | 1.09 (0.87–1.38) | 0.45 | 1.03 (0.81–1.32) | 0.80 |
| Patients with CAD | 1.47 (1.16–1.87) | 0.001 | 1.09 (0.84–1.40) | 0.53 |
| Patients with hypertension | 1.27 (1.03–1.58) | 0.03 | 0.97 (0.77–1.22) | 0.81 |
| Patients with reduced renal function (eGFR < 60) | 1.80 (1.45–2.24) | <0.001 | 1.40 (1.11–1.76) | 0.004 |
| Adjusted Odds Ratio (OR) | Adjusted Hazard Ratio (aHR) & | |||
|---|---|---|---|---|
| (n = 1158) | (n = 1167) | |||
| aOR, (95% CI) | p-Value | aHR, (95% CI) | p-Value | |
| NSAID | 0.79 (0.32–1.92) | 0.602 | 0.89 (0.59–1.35) | 0.58 |
| Age | ||||
| Under 65 | -Ref- | -Ref- | ||
| 65 to 79 | 3.80 (2.26–6.37) | <0.001 | 0.76 (0.61–0.95) | 0.02 |
| Over 80 | 5.14 (3.04–8.69) | <0.001 | 0.56 (0.44–0.73) | <0.001 |
| Sex | ||||
| Female | -Ref- | -Ref- | ||
| Male | 0.81 (0.57–1.14) | 0.227 | 0.92 (0.76–1.11) | 0.37 |
| Smoking status | ||||
| Never | ||||
| Ex-smokers | 1.17 (0.83–1.67) | 0.372 | 0.95 (0.78–1.15) | 0.61 |
| Current smokers | 1.12 (0.54–2.33) | 0.765 | 1.04 (0.73–1.48) | 0.84 |
| Elevated CRP (>40) | 4.91 (2.99–8.06) | <0.001 | 0.69 (0.57–0.84) | <0.001 |
| Patients with diabetes | 1.04 (0.71–1.52) | 0.838 | 0.84 (0.68–1.04) | 0.12 |
| Patients with CAD | 1.46 (1.00–2.13) | 0.051 | 1.12 (0.88–1.42) | 0.36 |
| Patients with hypertension | 0.78 (0.55–1.10) | 0.157 | 0.95 (0.79–1.16) | 0.63 |
| Patients with reduced renal function (eGFR < 60) | 2.02 (1.42–2.86) | <0.001 | 0.92 (0.75–1.13) | 0.58 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bruce, E.; Barlow-Pay, F.; Short, R.; Vilches-Moraga, A.; Price, A.; McGovern, A.; Braude, P.; Stechman, M.J.; Moug, S.; McCarthy, K.; et al. Prior Routine Use of Non-Steroidal Anti-Inflammatory Drugs (NSAIDs) and Important Outcomes in Hospitalised Patients with COVID-19. J. Clin. Med. 2020, 9, 2586. https://doi.org/10.3390/jcm9082586
Bruce E, Barlow-Pay F, Short R, Vilches-Moraga A, Price A, McGovern A, Braude P, Stechman MJ, Moug S, McCarthy K, et al. Prior Routine Use of Non-Steroidal Anti-Inflammatory Drugs (NSAIDs) and Important Outcomes in Hospitalised Patients with COVID-19. Journal of Clinical Medicine. 2020; 9(8):2586. https://doi.org/10.3390/jcm9082586
Chicago/Turabian StyleBruce, Eilidh, Fenella Barlow-Pay, Roxanna Short, Arturo Vilches-Moraga, Angeline Price, Aine McGovern, Philip Braude, Michael J. Stechman, Susan Moug, Kathryn McCarthy, and et al. 2020. "Prior Routine Use of Non-Steroidal Anti-Inflammatory Drugs (NSAIDs) and Important Outcomes in Hospitalised Patients with COVID-19" Journal of Clinical Medicine 9, no. 8: 2586. https://doi.org/10.3390/jcm9082586
APA StyleBruce, E., Barlow-Pay, F., Short, R., Vilches-Moraga, A., Price, A., McGovern, A., Braude, P., Stechman, M. J., Moug, S., McCarthy, K., Hewitt, J., Carter, B., & Myint, P. K. (2020). Prior Routine Use of Non-Steroidal Anti-Inflammatory Drugs (NSAIDs) and Important Outcomes in Hospitalised Patients with COVID-19. Journal of Clinical Medicine, 9(8), 2586. https://doi.org/10.3390/jcm9082586






