GlycA Levels during the Earliest Stages of Rheumatoid Arthritis: Potential Use as a Biomarker of Subclinical Cardiovascular Disease
Abstract
1. Introduction
2. Experimental Section
2.1. Study Participants
2.2. Carotid Ultrasound Imaging
2.3. Glycoprotein Profiling
2.4. Lipoprotein Characterization
2.5. Serum Glycosyltransferase Activity
2.6. Statistical Analyses
3. Results
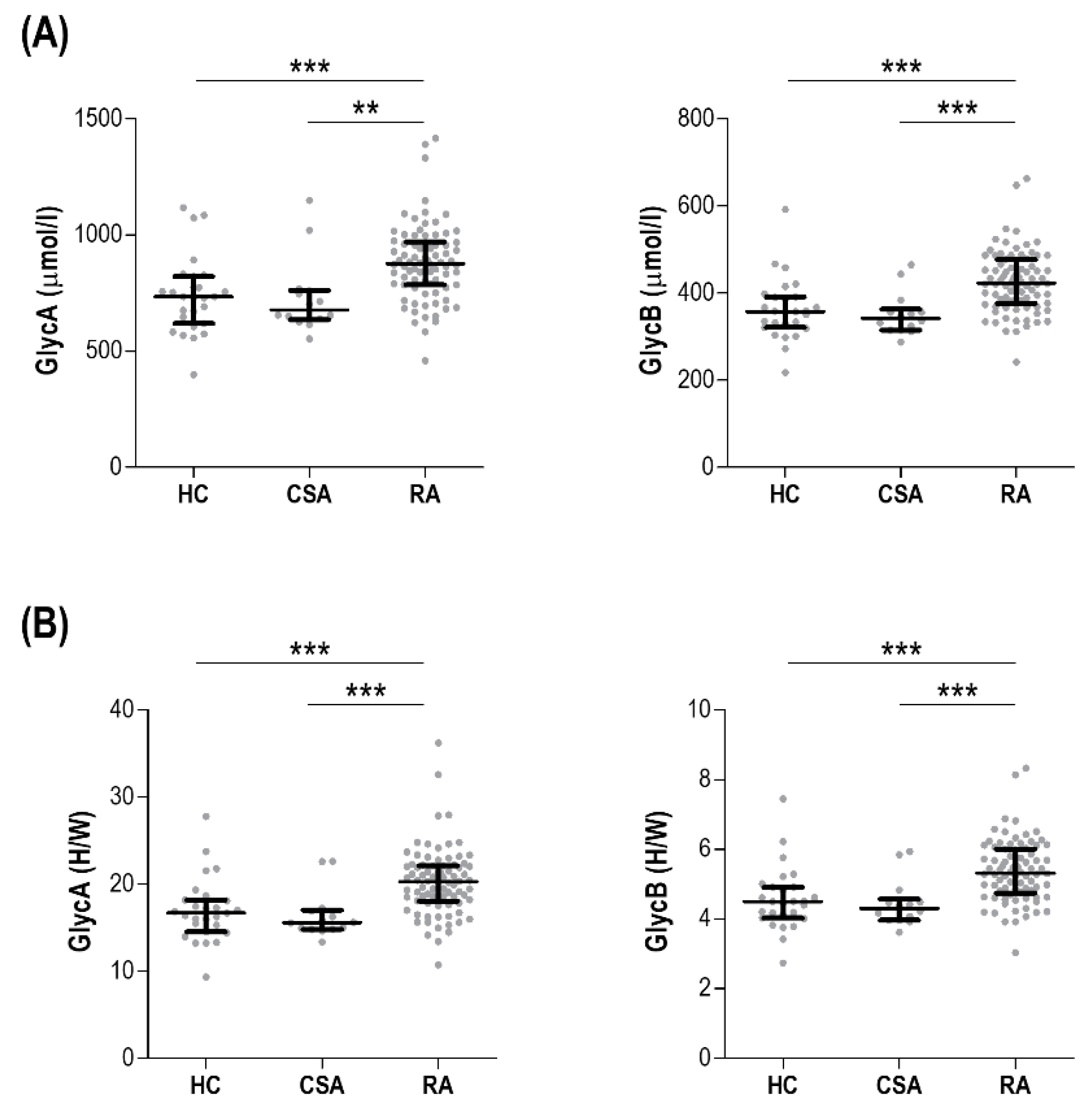
3.1. GlycA and GlycB Are Increased in Early RA Patients
3.2. GlycA, Traditional CV Risk Factors, and Glucose Homeostasis in Early RA
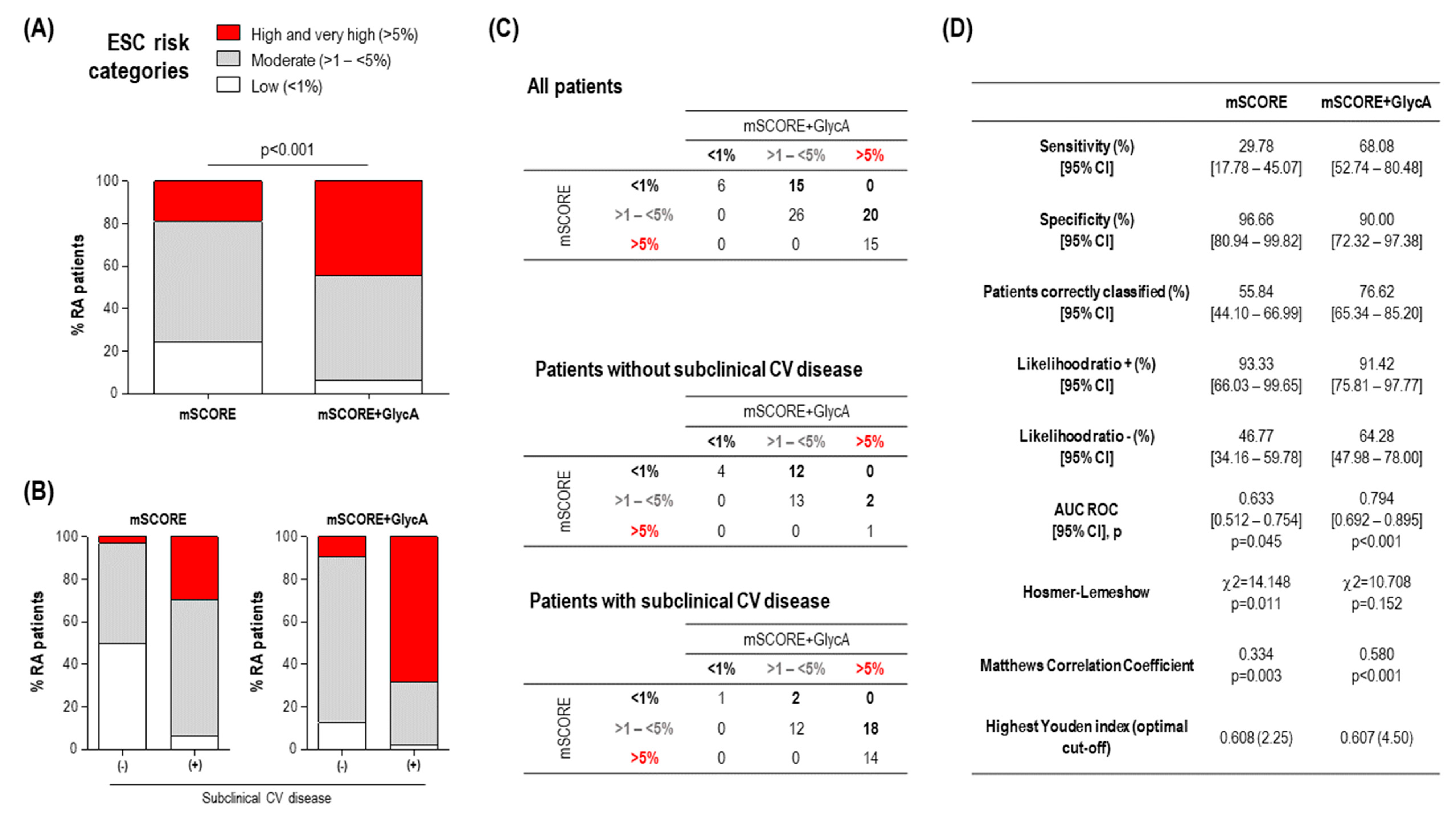
3.3. GlycA and Subclinical CV Disease in Early RA
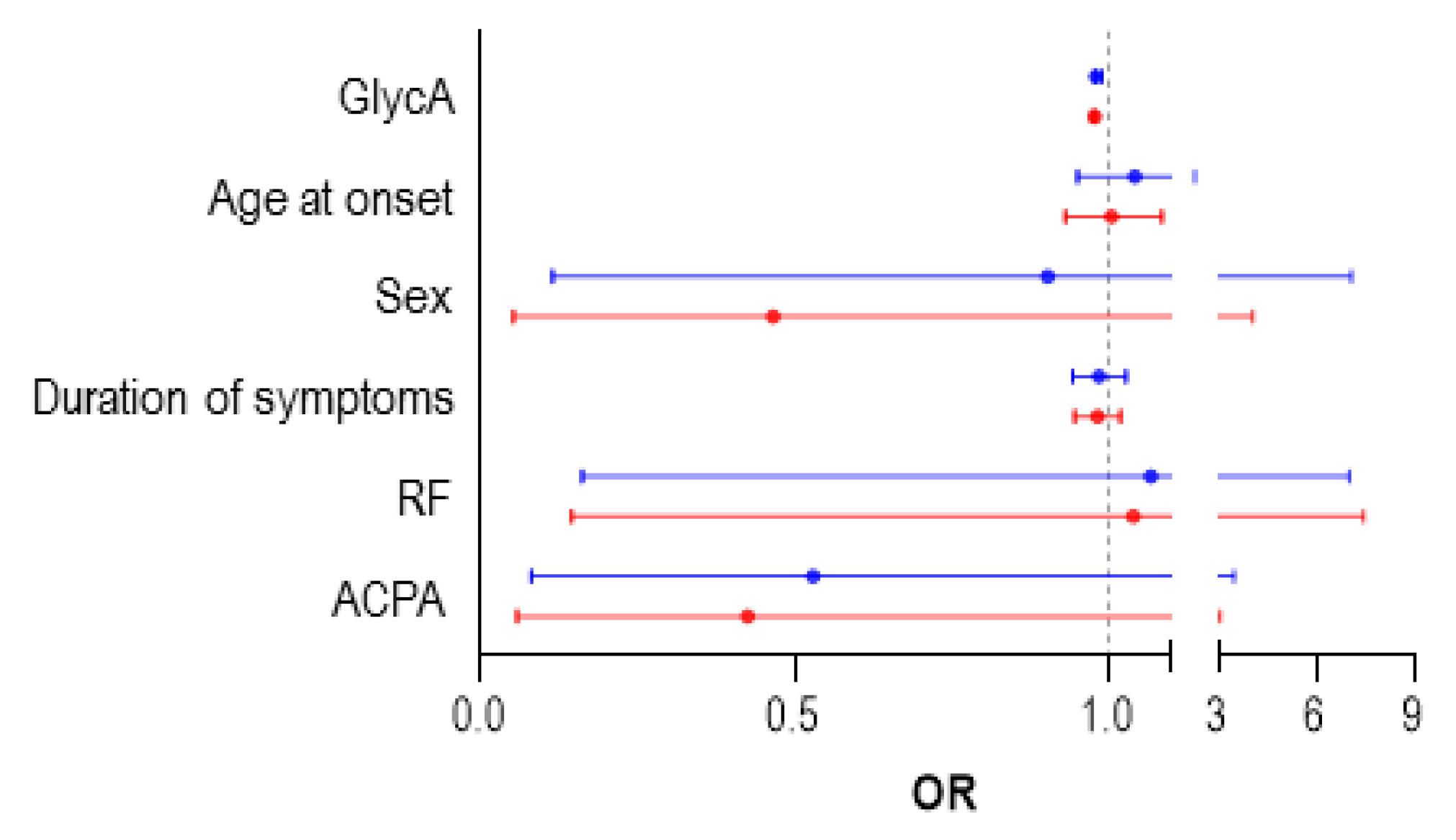
3.4. GlycA as a Biomarker of Treatment Outcome in Early RA Patients
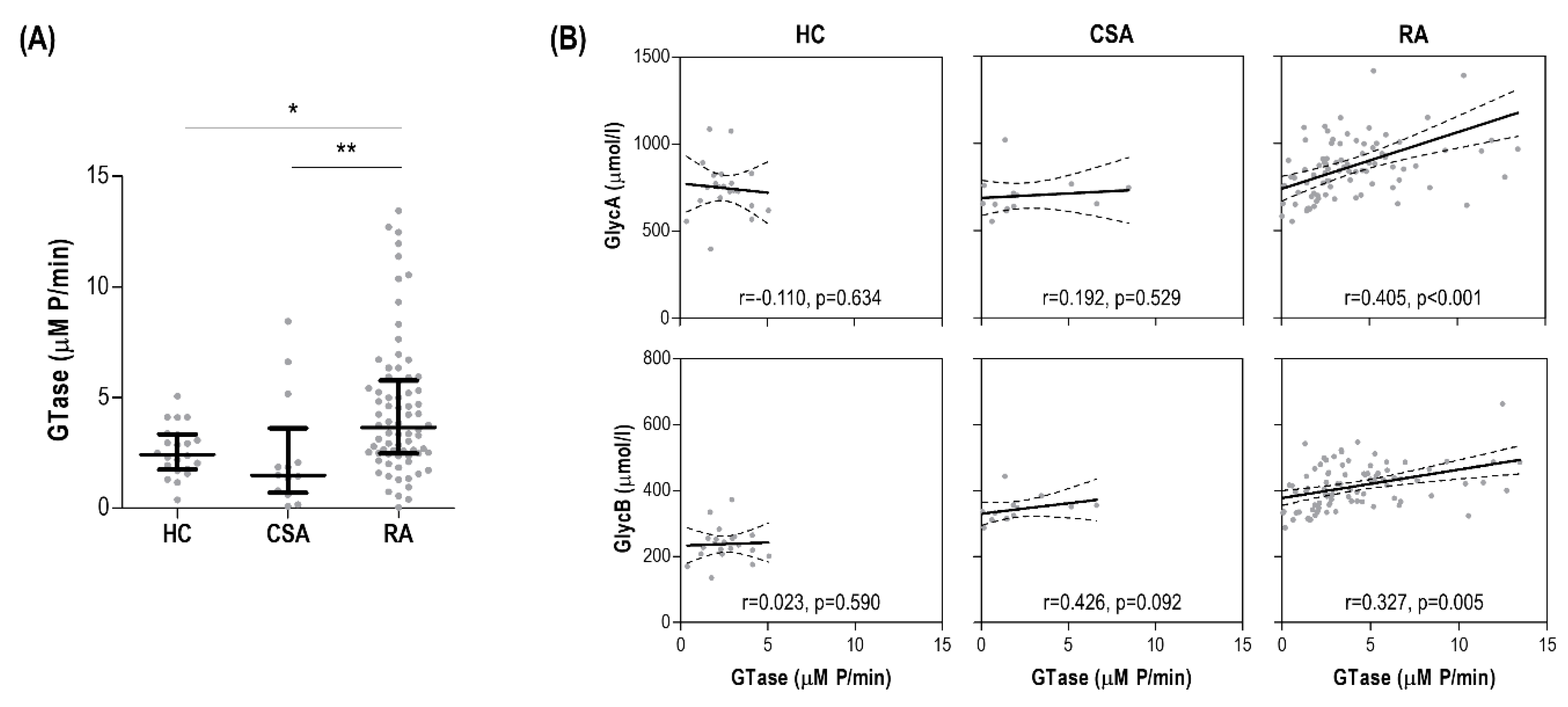
3.5. GlycA Levels Are Associated with Serum Glycosyltransferase Activity
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Carubbi, F.; Alunno, A.; Gerli, R.; Giacomelli, R. Post-Translational Modifications of Proteins: Novel Insights in the Autoimmune Response in Rheumatoid Arthritis. Cells 2019, 8, 657. [Google Scholar] [CrossRef] [PubMed]
- Marth, J.D.; Grewal, P.K. Mammalian glycosylation in immunity. Nat. Rev. Immunol. 2008, 8, 874–887. [Google Scholar] [CrossRef] [PubMed]
- Schachter, H.; Freeze, H.H. Glycosylation diseases: Quo vadis? Biochim. Biophys. Acta Mol. Basis Dis. 2009, 1792, 925–930. [Google Scholar] [CrossRef] [PubMed]
- Ohtsubo, K.; Marth, J.D. Glycosylation in Cellular Mechanisms of Health and Disease. Cell 2006, 126, 855–867. [Google Scholar] [CrossRef] [PubMed]
- Ceciliani, F.; Pocacqua, V. The Acute Phase Protein α1-Acid Glycoprotein: A Model for Altered Glycosylation During Diseases. Curr. Protein Pept. Sci. 2007, 8, 91–108. [Google Scholar] [CrossRef] [PubMed]
- Otvos, J.D.; Shalaurova, I.; Wolak-Dinsmore, J.; Connelly, M.A.; Mackey, R.H.; Stein, J.H.; Tracy, R.P. GlycA: A Composite Nuclear Magnetic Resonance Biomarker of Systemic Inflammation. Clin. Chem. 2015, 61, 714–723. [Google Scholar] [CrossRef]
- Bartlett, D.B.; Connelly, M.A.; AbouAssi, H.; Bateman, L.A.; Tune, K.N.; Huebner, J.L.; Kraus, V.B.; Winegar, D.A.; Otvos, J.D.; Kraus, W.E.; et al. A novel inflammatory biomarker, GlycA, associates with disease activity in rheumatoid arthritis and cardio-metabolic risk in BMI-matched controls. Arthritis Res. Ther. 2016, 18, 86. [Google Scholar] [CrossRef]
- Joshi, A.A.; Lerman, J.B.; Aberra, T.M.; Afshar, M.; Teague, H.L.; Rodante, J.A.; Krishnamoorthy, P.; Ng, Q.; Aridi, T.Z.; Salahuddin, T.; et al. GlycA Is a Novel Biomarker of Inflammation and Subclinical Cardiovascular Disease in Psoriasis. Circ. Res. 2016, 119, 1242–1253. [Google Scholar] [CrossRef]
- Chung, C.P.; Ormseth, M.J.; Connelly, M.A.; Oeser, A.; Solus, J.F.; Otvos, J.D.; Raggi, P.; Stein, C.M. GlycA, a novel marker of inflammation, is elevated in systemic lupus erythematosus. Lupus 2016, 25, 296–300. [Google Scholar] [CrossRef]
- Fuertes-Martín, R.; Taverner, D.; Vallvé, J.-C.; Paredes, S.; Masana, L.; Correig Blanchar, X.; Amigó Grau, N. Characterization of 1 H NMR Plasma Glycoproteins as a New Strategy To Identify Inflammatory Patterns in Rheumatoid Arthritis. J. Proteome Res. 2018, 17, 3730–3739. [Google Scholar] [CrossRef]
- Purmalek, M.M.; Carlucci, P.M.; Dey, A.K.; Sampson, M.; Temesgen-Oyelakin, Y.; Sakhardande, S.; Lerman, J.B.; Fike, A.; Davis, M.; Chung, J.H.; et al. Association of lipoprotein subfractions and glycoprotein acetylation with coronary plaque burden in SLE. Lupus Sci. Med. 2019, 6, e000332. [Google Scholar] [CrossRef] [PubMed]
- Huizinga, T.W.; Landewé, R.B. Early aggressive therapy in rheumatoid arthritis: A “window of opportunity”? Nat. Clin. Pract. Rheumatol. 2005, 1, 2–3. [Google Scholar] [CrossRef] [PubMed]
- Ballout, R.A.; Remaley, A.T. GlycA: A new biomarker for systemic inflammation and cardiovascular disease (CVD) risk assessment. J. Lab. Precis. Med. 2020, 5, 17. [Google Scholar] [CrossRef] [PubMed]
- Van den Oever, I.A.M.; Sattar, N.; Nurmohamed, M.T. Thromboembolic and cardiovascular risk in rheumatoid arthritis: Role of the haemostatic system. Ann. Rheum. Dis. 2014, 73, 954–957. [Google Scholar] [CrossRef]
- Agca, R.; Heslinga, S.C.; Rollefstad, S.; Heslinga, M.; McInnes, I.B.; Peters, M.J.L.; Kvien, T.K.; Dougados, M.; Radner, H.; Atzeni, F.; et al. EULAR recommendations for cardiovascular disease risk management in patients with rheumatoid arthritis and other forms of inflammatory joint disorders: 2015/2016 update. Ann. Rheum. Dis. 2017, 76, 17–28. [Google Scholar] [CrossRef]
- Aletaha, D.; Neogi, T.; Silman, A.J.; Funovits, J.; Felson, D.T.; Bingham, C.O.; Birnbaum, N.S.; Burmester, G.R.; Bykerk, V.P.; Cohen, M.D.; et al. 2010 rheumatoid arthritis classification criteria: An American College of Rheumatology/European League Against Rheumatism collaborative initiative. Ann. Rheum. Dis. 2010, 69, 1580–1588. [Google Scholar] [CrossRef]
- Smolen, J.S.; Landewé, R.; Bijlsma, J.; Burmester, G.; Chatzidionysiou, K.; Dougados, M.; Nam, J.; Ramiro, S.; Voshaar, M.; van Vollenhoven, R.; et al. EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs: 2016 update. Ann. Rheum. Dis. 2017, 76, 960–977. [Google Scholar] [CrossRef]
- Van Steenbergen, H.W.; Aletaha, D.; Beaart-van de Voorde, L.J.J.; Brouwer, E.; Codreanu, C.; Combe, B.; Fonseca, J.E.; Hetland, M.L.; Humby, F.; Kvien, T.K.; et al. EULAR definition of arthralgia suspicious for progression to rheumatoid arthritis. Ann. Rheum. Dis. 2017, 76, 491–496. [Google Scholar] [CrossRef]
- Piepoli, M.F.; Hoes, A.W.; Agewall, S.; Albus, C.; Brotons, C.; Catapano, A.L.; Cooney, M.T.; Corrà, U.; Cosyns, B.; Deaton, C.; et al. 2016 European Guidelines on cardiovascular disease prevention in clinical practice. Eur. Heart J. 2016, 37, 2315–2381. [Google Scholar] [CrossRef]
- Fuertes-Martín, R.; Moncayo, S.; Insenser, M.; Martínez-García, M.Á.; Luque-Ramírez, M.; Grau, N.A.; Blanchar, X.C.; Escobar-Morreale, H.F. Glycoprotein A and B Height-to-Width Ratios as Obesity-Independent Novel Biomarkers of Low-Grade Chronic Inflammation in Women with Polycystic Ovary Syndrome (PCOS). J. Proteome Res. 2019, 18, 4038–4045. [Google Scholar] [CrossRef]
- Nicholson, J.K.; Foxall, P.J.D.; Spraul, M.; Farrant, R.D.; Lindon, J.C. 750 MHz 1H and 1H-13C NMR Spectroscopy of Human Blood Plasma. Anal. Chem. 1995, 67, 793–811. [Google Scholar] [CrossRef] [PubMed]
- Mallol, R.; Amigó, N.; Rodríguez, M.A.; Heras, M.; Vinaixa, M.; Plana, N.; Rock, E.; Ribalta, J.; Yanes, O.; Masana, L.; et al. Liposcale: A novel advanced lipoprotein test based on 2D diffusion-ordered 1 H NMR spectroscopy. J. Lipid Res. 2015, 56, 737–746. [Google Scholar] [CrossRef] [PubMed]
- Shafiqur Rahman, M.; Qin, W. Glycosyltransferase Activity Assay Using Colorimetric Methods. Methods Mol. Biol. 2019, 1954, 237–243. [Google Scholar] [PubMed]
- Burgers, L.E.; Raza, K.; van der Helm-van Mil, A.H. Window of opportunity in rheumatoid arthritis—Definitions and supporting evidence: From old to new perspectives. RMD Open 2019, 5, e000870. [Google Scholar] [CrossRef]
- Ferraz-Amaro, I.; García-Dopico, J.A.; Medina-Vega, L.; González-Gay, M.A.; Díaz-González, F. Impaired beta cell function is present in nondiabetic rheumatoid arthritis patients. Arthritis Res. Ther. 2013, 15, R17. [Google Scholar] [CrossRef]
- Alavi, A.; Axford, J.; Pool, A. Serum galactosyltransferase isoform changes in rheumatoid arthritis. J. Rheumatol. 2004, 31, 1513–1520. [Google Scholar]
- Feelders, R.A.; Vreugdenhil, G.; de Jong, G.; Swaak, A.J.G.; Van Eijk, H.G. Transferrin microheterogeneity in rheumatoid arthritis. Rheumatol. Int. 1992, 12, 195–199. [Google Scholar] [CrossRef][Green Version]
- Nielen, M.M.J.; Van Schaardenburg, D.; Reesink, H.W.; Twisk, J.W.R.; Van De Stadt, R.J.; Van Der Horst-Bruinsma, I.E.; De Gast, T.; Habibuw, M.R.; Vandenbroucke, J.P.; Dijkmans, B.A.C. Increased levels of C-reactive protein in serum from blood donors before the onset of rheumatoid arthritis. Arthritis Rheum. 2004, 50, 2423–2427. [Google Scholar] [CrossRef]
- Nielen, M.M.J. Simultaneous development of acute phase response and autoantibodies in preclinical rheumatoid arthritis. Ann. Rheum. Dis. 2006, 65, 535–537. [Google Scholar] [CrossRef]
- Van Dijk, W.; Turner, G.A.; Mackiewicz, A. Changes in glycosylation of acute-phase proteins in health and disease: Occurrence, regulation and function. Glycoconj. J. 1994, 1, 5–14. [Google Scholar] [CrossRef]
- Gornik, O.; Lauc, G. Glycosylation of Serum Proteins in Inflammatory Diseases. Dis. Markers 2008, 25, 267–278. [Google Scholar] [CrossRef] [PubMed]
- Maverakis, E.; Kim, K.; Shimoda, M.; Gershwin, M.E.; Patel, F.; Wilken, R.; Raychaudhuri, S.; Ruhaak, L.R.; Lebrilla, C.B. Glycans in the immune system and The Altered Glycan Theory of Autoimmunity: A critical review. J. Autoimmun. 2015, 57, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Wolfert, M.A.; Boons, G.-J. Adaptive immune activation: Glycosylation does matter. Nat. Chem. Biol. 2013, 9, 776–784. [Google Scholar] [CrossRef] [PubMed]
- Malmström, V.; Catrina, A.I.; Klareskog, L. The immunopathogenesis of seropositive rheumatoid arthritis: From triggering to targeting. Nat. Rev. Immunol. 2017, 17, 60–75. [Google Scholar] [CrossRef] [PubMed]
- Connelly, M.A.; Otvos, J.D.; Shalaurova, I.; Playford, M.P.; Mehta, N.N. GlycA, a novel biomarker of systemic inflammation and cardiovascular disease risk. J. Transl. Med. 2017, 15, 219. [Google Scholar] [CrossRef]
- Ritchie, S.C.; Würtz, P.; Nath, A.P.; Abraham, G.; Havulinna, A.S.; Fearnley, L.G.; Sarin, A.-P.; Kangas, A.J.; Soininen, P.; Aalto, K.; et al. The Biomarker GlycA Is Associated with Chronic Inflammation and Predicts Long-Term Risk of Severe Infection. Cell Syst. 2015, 1, 293–301. [Google Scholar] [CrossRef]
- Rodriguez-Carrio, J.; Alperi-Lopez, M.; Lopez, P.; Alonso-Castro, S.; Ballina-Garcia, F.J.; Suarez, A. Red cell distribution width is associated with cardiovascular risk and disease parameters in rheumatoid arthritis. Rheumatology 2015, 54, 641–646. [Google Scholar] [CrossRef]
- Hassan, S.; Antonelli, M.; Ballou, S. Red cell distribution width: A measure of cardiovascular risk in rheumatoid arthritis patients? Clin. Rheumatol. 2015, 34, 1053–1057. [Google Scholar] [CrossRef]
- Orr, C.K.; Najm, A.; Young, F.; McGarry, T.; Biniecka, M.; Fearon, U.; Veale, D.J. The Utility and Limitations of CRP, ESR and DAS28-CRP in Appraising Disease Activity in Rheumatoid Arthritis. Front. Med. 2018, 5, 185. [Google Scholar] [CrossRef]
- Tang, Y.; Liang, P.; Chen, J.; Fu, S.; Liu, B.; Feng, M.; Lin, B.; Lee, B.; Xu, A.; Lan, H.Y. The baseline levels and risk factors for high-sensitive C-reactive protein in Chinese healthy population. Immun. Ageing 2018, 15, 21. [Google Scholar] [CrossRef]
- Lakoski, S.G.; Cushman, M.; Criqui, M.; Rundek, T.; Blumenthal, R.S.; D’Agostino, R.B.; Herrington, D.M. Gender and C-reactive protein: Data from the Multiethnic Study of Atherosclerosis (MESA) cohort. Am. Heart J. 2006, 152, 593–598. [Google Scholar] [CrossRef] [PubMed]
- Pankow, J.S.; Folsom, A.R.; Cushman, M.; Borecki, I.B.; Hopkins, P.N.; Eckfeldt, J.H.; Tracy, R.P. Familial and genetic determinants of systemic markers of inflammation: The NHLBI family heart study. Atherosclerosis 2001, 154, 681–689. [Google Scholar] [CrossRef]
- Greenland, P.; Alpert, J.S.; Beller, G.A.; Benjamin, E.J.; Budoff, M.J.; Fayad, Z.A.; Foster, E.; Hlatky, M.A.; Hodgson, J.M.; Kushner, F.G.; et al. 2010 ACCF/AHA Guideline for Assessment of Cardiovascular Risk in Asymptomatic Adults. J. Am. Coll. Cardiol. 2010, 56, e50–e103. [Google Scholar] [CrossRef] [PubMed]
- Pearson, T.A.; Mensah, G.A.; Alexander, R.W.; Anderson, J.L.; Cannon, R.O.; Criqui, M.; Fadl, Y.Y.; Fortmann, S.P.; Hong, Y.; Myers, G.L.; et al. Markers of Inflammation and Cardiovascular Disease. Circulation 2003, 107, 499–511. [Google Scholar] [CrossRef]
- Scott, D.W.; Vallejo, M.O.; Patel, R.P. Heterogenic Endothelial Responses to Inflammation: Role for Differential N -Glycosylation and Vascular Bed of Origin. J. Am. Heart Assoc. 2013, 2. [Google Scholar] [CrossRef]
- Menni, C.; Gudelj, I.; Macdonald-Dunlop, E.; Mangino, M.; Zierer, J.; Bešić, E.; Joshi, P.K.; Trbojević-Akmačić, I.; Chowienczyk, P.J.; Spector, T.D.; et al. Glycosylation Profile of Immunoglobulin G Is Cross-Sectionally Associated With Cardiovascular Disease Risk Score and Subclinical Atherosclerosis in Two Independent Cohorts. Circ. Res. 2018, 122, 1555–1564. [Google Scholar] [CrossRef]
- Crowson, C.S.; Gabriel, S.E.; Semb, A.G.; van Riel, P.L.C.M.; Karpouzas, G.; Dessein, P.H.; Hitchon, C.; Pascual-Ramos, V.; Kitas, G.D.; Douglas, K.; et al. Rheumatoid arthritis-specific cardiovascular risk scores are not superior to general risk scores: A validation analysis of patients from seven countries. Rheumatology 2017, 56, 1102–1110. [Google Scholar] [CrossRef]
- Alemao, E.; Cawston, H.; Bourhis, F.; Al, M.; Rutten-van Molken, M.; Liao, K.P.; Solomon, D.H. Comparison of cardiovascular risk algorithms in patients with vs. without rheumatoid arthritis and the role of C-reactive protein in predicting cardiovascular outcomes in rheumatoid arthritis. Rheumatology 2017, 56, 777–786. [Google Scholar]
- Crowson, C.S.; Rollefstad, S.; Ikdahl, E.; Kitas, G.D.; van Riel, P.L.C.M.; Gabriel, S.E.; Matteson, E.L.; Kvien, T.K.; Douglas, K.; Sandoo, A.; et al. Impact of risk factors associated with cardiovascular outcomes in patients with rheumatoid arthritis. Ann. Rheum. Dis. 2018, 77, 48–54. [Google Scholar] [CrossRef]
- Van den Boogert, M.A.W.; Larsen, L.E.; Ali, L.; Kuil, S.D.; Chong, P.L.W.; Loregger, A.; Kroon, J.; Schnitzler, J.G.; Schimmel, A.W.M.; Peter, J.; et al. N-Glycosylation Defects in Humans Lower Low-Density Lipoprotein Cholesterol Through Increased Low-Density Lipoprotein Receptor Expression. Circulation 2019, 140, 280–292. [Google Scholar] [CrossRef]
- Choy, E.; Sattar, N. Interpreting lipid levels in the context of high-grade inflammatory states with a focus on rheumatoid arthritis: A challenge to conventional cardiovascular risk actions. Ann. Rheum. Dis. 2009, 68, 460–469. [Google Scholar] [CrossRef] [PubMed]
- Macy, E.M.; Hayes, T.E.; Tracy, R.P. Variability in the measurement of C-reactive protein in healthy subjects: Implications for reference intervals and epidemiological applications. Clin. Chem. 1997, 43, 52–58. [Google Scholar] [CrossRef] [PubMed]
- DeGoma, E.M.; French, B.; Dunbar, R.L.; Allison, M.A.; Mohler, E.R.; Budoff, M.J. Intraindividual variability of C-reactive protein: The Multi-Ethnic Study of Atherosclerosis. Atherosclerosis 2012, 224, 274–279. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Fuertes-Martín, R.; Correig, X.; Vallvé, J.-C.; Amigó, N. Human Serum/Plasma Glycoprotein Analysis by 1H-NMR, an Emerging Method of Inflammatory Assessment. J. Clin. Med. 2020, 9, 354. [Google Scholar] [CrossRef]

| Variables | CSA n = 14 | RA n = 82 |
|---|---|---|
| Age (years), mean ± SD | 49.28 ± 10.53 | 58.51 ± 10.49 |
| Sex (women/men) | 14/0 | 66/16 |
| Clinical features | ||
| Duration of symptoms (weeks) | 24.00 (40.00) | 20.00 (22.00) |
| Morning stiffness (minutes) | 30.00 (50.00) | 60.00 (80.0) |
| Tender joint count | 3.00 (3.00) | 8.00 (7.00) |
| Swollen joint count | 0.00 (1.00) | 6.00 (5.00) |
| ESR (mm/h) | 7.50 (8.84) | 24.00 (27.00) |
| CRP (mg/dL) | 0.15 (0.30) | 0.80 (2.20) |
| Patient global assessment (VAS 0–100) | 30.00 (50.00) | 70.00 (25.00) |
| Pain assessment (VAS 0–10) | 5.00 (5.00) | 7.00 (2.00) |
| DAS28 | 5.40 (1.78) | |
| SDAI | 24.22 (16.90) | |
| HAQ | 0.55 (0.60) | 1.11 (1.00) |
| Fatigue (VAS 0–10) | 4.50 (5.00) | 5.00 (7.00) |
| RF+, n (%) | 8 (66.6) | 57 (69.5) |
| ACPA+, n (%) | 7 (58.3) | 56 (68.2) |
| Traditional CV risk factors | ||
| Hypertension, n (%) | 1 (12.5) | 28 (34.11) |
| Diabetes, n (%) | 0 (0.0) | 9 (10.9) |
| Dyslipidemia, n (%) | 3 (37.5) | 24 (29.2) |
| Smoking, n (%) | 10 (71.4) | 31 (27.8) |
| Obesity, n (%) | 3 (37.5) | 33 (40.2) |
| Waist circumference | 92.00 (16.00) | 101.00 (20.00) |
| Glucose homeostasis features | ||
| Glucose (mg/dL) | 98.00 (19.00) | 92.00 (10.00) |
| Insulin (U/mL) | 7.87 (4.57) | 10.10 (11.80) |
| C-peptide (ng/mL) | 2.10 (1.4) | 2.79 (1.6) |
| HOMA-IR | 1.00 (0.60) | 1.30 (1.38) |
| QUICKI | 0.36 (0.02) | 0.33 (0.05) |
| Subclinical atherosclerosis (n = 92) | n = 13 | n = 79 |
| cIMT (mm) | 0.58 ± 0.15 | 0.67 ± 0.10 |
| Plaque presence, n (%) | 4 (30.7) | 46 (58.2) |
| Plaque number | 0.46 ± 0.87 | 0.96 ± 1.01 |
| Plaque presence or cIMT > 0.90, n (%) | 4 (30.7) | 47 (59.6) |
| High-risk plaque | 0 (0.0) | 20 (25.3) |
| Treatments, n (%) | ||
| None | 14 (100) | 69 (84.1) |
| Glucocorticoids | 0 (0) | 13 (15.8) |
| Methotrexate | 0 (0) | 5 (6.0) |
| Variables | GlycA | GlycB |
|---|---|---|
| Traditional CV Risk Factors | ||
| Hypertension | p = 0.115 | p = 0.374 |
| Diabetes | p = 0.078 | p = 0.090 |
| Dyslipidemia | p = 0.163 | p = 0.536 |
| Smoking | p = 0.977 | p = 0.793 |
| Obesity | p = 0.107 | p = 0.113 |
| Waist circumference | r = 0.145 p = 0.233 | r = 0.078 p = 0.524 |
| Glucose Homeostasis Parameters | ||
| Glucose | r = 0.068 p = 0.542 | r = 0.218 p = 0.050 |
| Insulin | r = 0.087 p = 0.485 | r = −0.001 p = 0.994 |
| C-peptide | r = −0.034 p = 0.786 | r = −0.075 p = 0.545 |
| HOMA-IR | r = 0.068 p = 0.593 | r = 0.002 p = 0.990 |
| QUICKI | r = −0.181 p = 0.147 | r = −0.135 p = 0.279 |
| Models | OR | 95% CI | p-Value |
|---|---|---|---|
| Univariate | |||
| GlycA, per unit | 1.004 | 1.002–1.007 | 0.020 |
| Multivariate | |||
| GlycA, per unit | 1.008 | 1.003–1.012 | 0.023 |
| Age at sampling, per year | 1.091 | 1.017–1.169 | 0.015 |
| Sex, women | 4.609 | 0.635–33.427 | 0.131 |
| Hypertension, yes | 3.811 | 0.752–19.305 | 0.106 |
| Smoking, yes | 2.491 | 0.908–6.833 | 0.076 |
| Dyslipidemia, yes | 1.093 | 0.250–4.776 | 0.906 |
| Diabetes, yes | 3.731 | 0.345–40.371 | 0.278 |
| BMI, per unit | 0.013 | 0.012–22.705 | 0.254 |
| Model | 6 Months | 12 Months | ||||
|---|---|---|---|---|---|---|
| OR | 95% CI | p-Value | OR | 95% CI | p-Value | |
| Univariate | ||||||
| GlycA, per unit | 0.993 | 0.988–0.998 | 0.008 | 0.994 | 0.989–0.999 | 0.015 |
| Multivariate | ||||||
| GlycA, per unit | 0.992 | 0.986–0.998 | 0.015 | 0.993 | 0.986–0.999 | 0.030 |
| Age at onset, per year | 1.005 | 0.931–1.085 | 0.894 | 1.042 | 0.951–1.142 | 0.381 |
| Sex, women | 0.466 | 0.053–4.055 | 0.489 | 0.903 | 0.115–7.079 | 0.903 |
| Duration of symptoms, per week | 0.982 | 0.946–1.019 | 0.342 | 0.985 | 0.943–1.028 | 0.489 |
| RF, + | 0.426 | 0.059–3.055 | 0.396 | 0.530 | 0.081–3.482 | 0.509 |
| ACPA, + | 1.039 | 0.146–7.417 | 0.696 | 1.067 | 0.162–7.024 | 0.946 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rodríguez-Carrio, J.; Alperi-López, M.; López, P.; Pérez-Álvarez, Á.I.; Gil-Serret, M.; Amigó, N.; Ulloa, C.; Benavente, L.; Ballina-García, F.J.; Suárez, A. GlycA Levels during the Earliest Stages of Rheumatoid Arthritis: Potential Use as a Biomarker of Subclinical Cardiovascular Disease. J. Clin. Med. 2020, 9, 2472. https://doi.org/10.3390/jcm9082472
Rodríguez-Carrio J, Alperi-López M, López P, Pérez-Álvarez ÁI, Gil-Serret M, Amigó N, Ulloa C, Benavente L, Ballina-García FJ, Suárez A. GlycA Levels during the Earliest Stages of Rheumatoid Arthritis: Potential Use as a Biomarker of Subclinical Cardiovascular Disease. Journal of Clinical Medicine. 2020; 9(8):2472. https://doi.org/10.3390/jcm9082472
Chicago/Turabian StyleRodríguez-Carrio, Javier, Mercedes Alperi-López, Patricia López, Ángel I. Pérez-Álvarez, Miriam Gil-Serret, Núria Amigó, Catalina Ulloa, Lorena Benavente, Francisco J. Ballina-García, and Ana Suárez. 2020. "GlycA Levels during the Earliest Stages of Rheumatoid Arthritis: Potential Use as a Biomarker of Subclinical Cardiovascular Disease" Journal of Clinical Medicine 9, no. 8: 2472. https://doi.org/10.3390/jcm9082472
APA StyleRodríguez-Carrio, J., Alperi-López, M., López, P., Pérez-Álvarez, Á. I., Gil-Serret, M., Amigó, N., Ulloa, C., Benavente, L., Ballina-García, F. J., & Suárez, A. (2020). GlycA Levels during the Earliest Stages of Rheumatoid Arthritis: Potential Use as a Biomarker of Subclinical Cardiovascular Disease. Journal of Clinical Medicine, 9(8), 2472. https://doi.org/10.3390/jcm9082472





