Effect of an Enhanced Self-Care Protocol on Lymphedema Status among People Affected by Moderate to Severe Lower-Limb Lymphedema in Bangladesh, a Cluster Randomized Controlled Trial
Abstract
1. Introduction
2. Materials and Methods
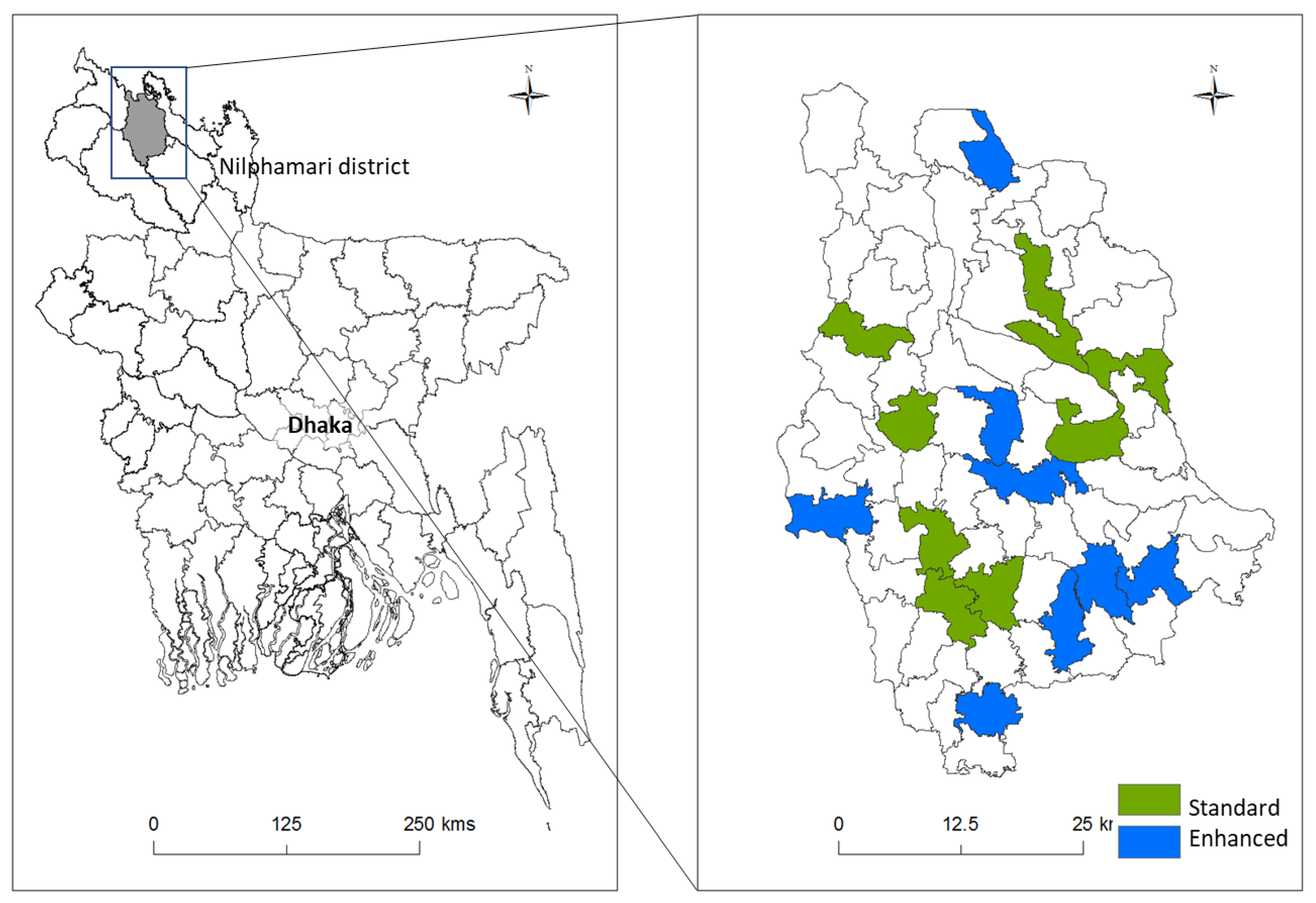
2.1. Study Design, Research Personnel, and Recruitment
2.2. Outcomes Measures and Data Collection
2.3. Intervention
2.4. Analysis
3. Results
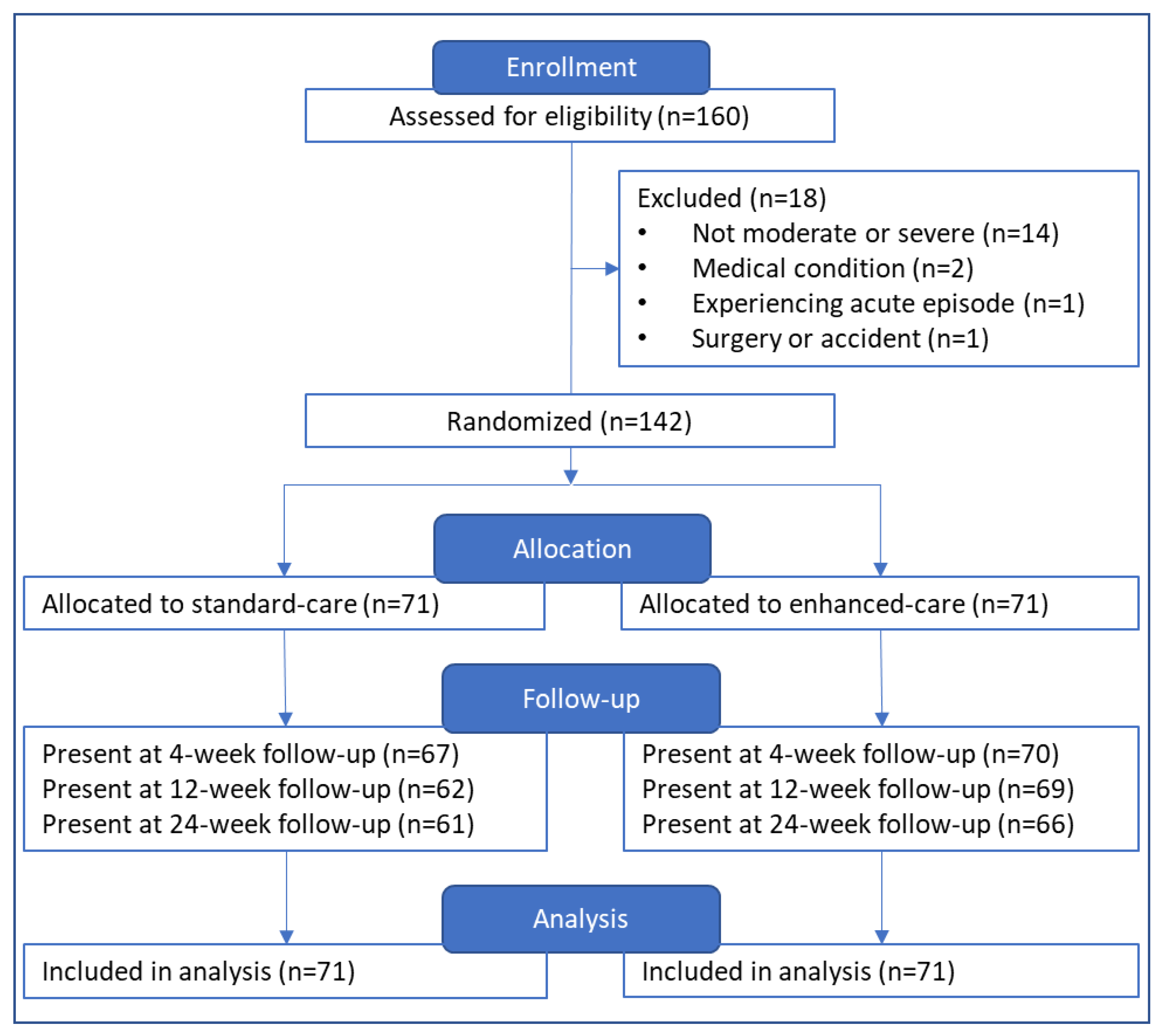
3.1. Participants
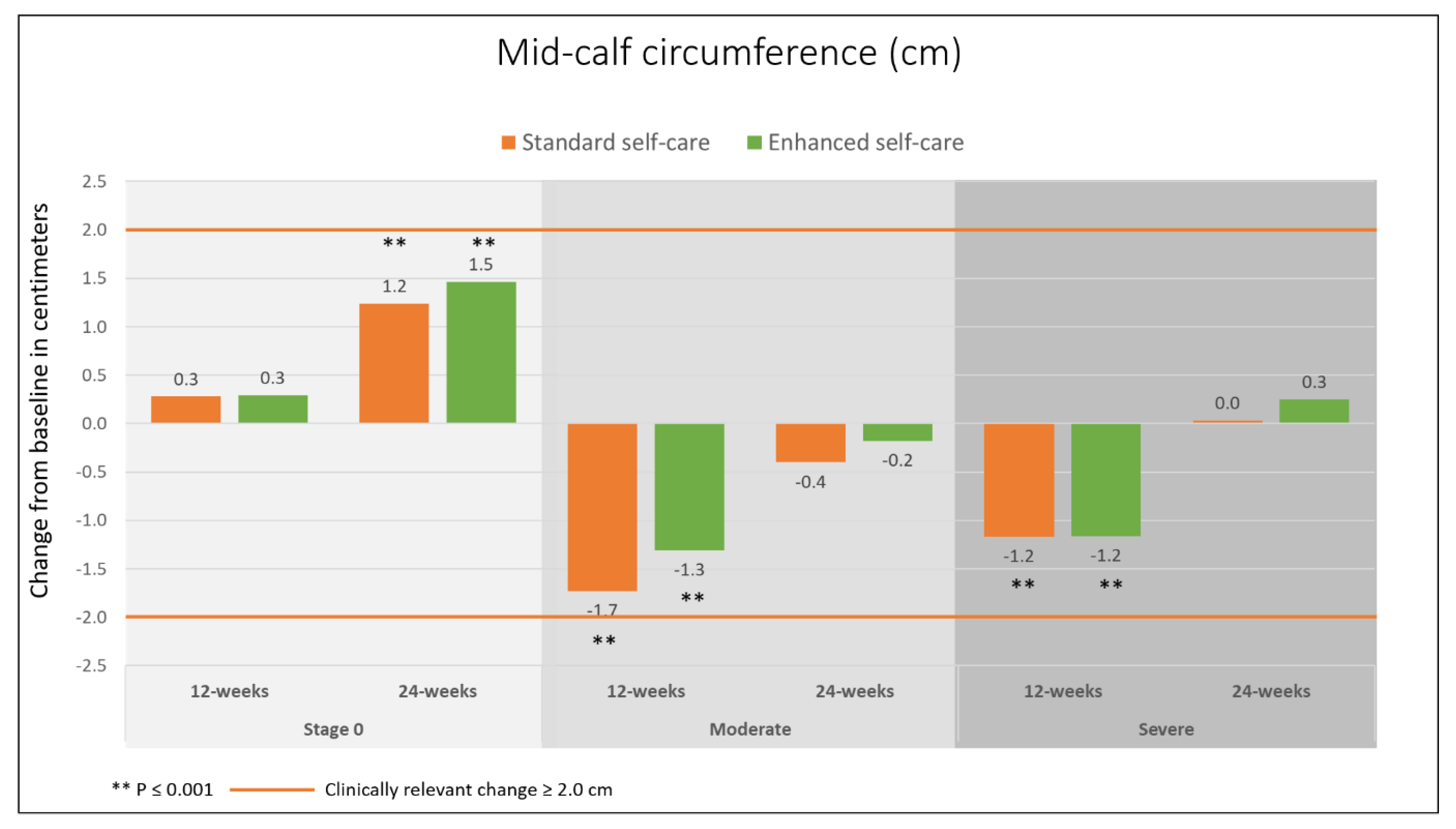
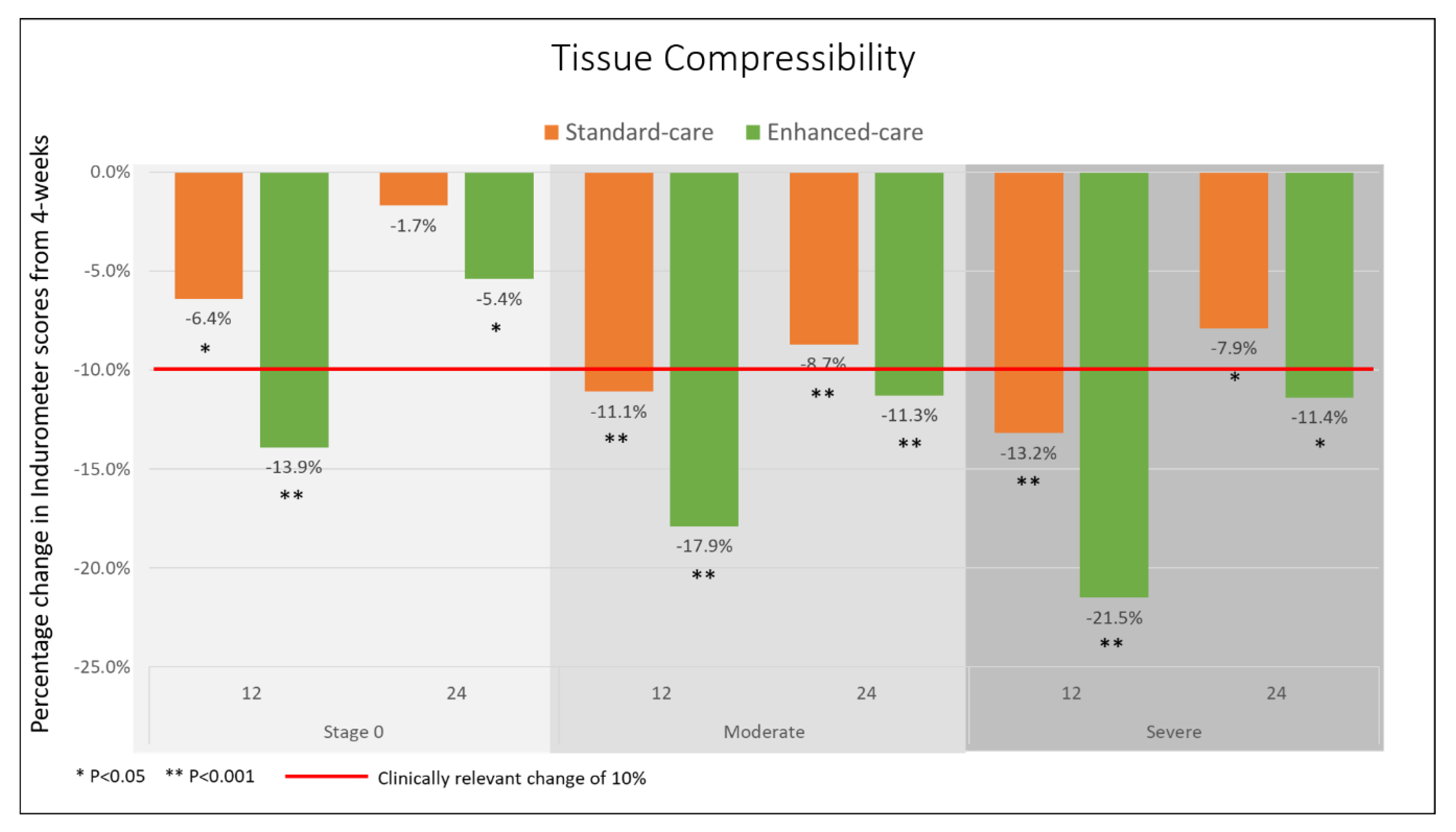
3.2. Primary Outcomes
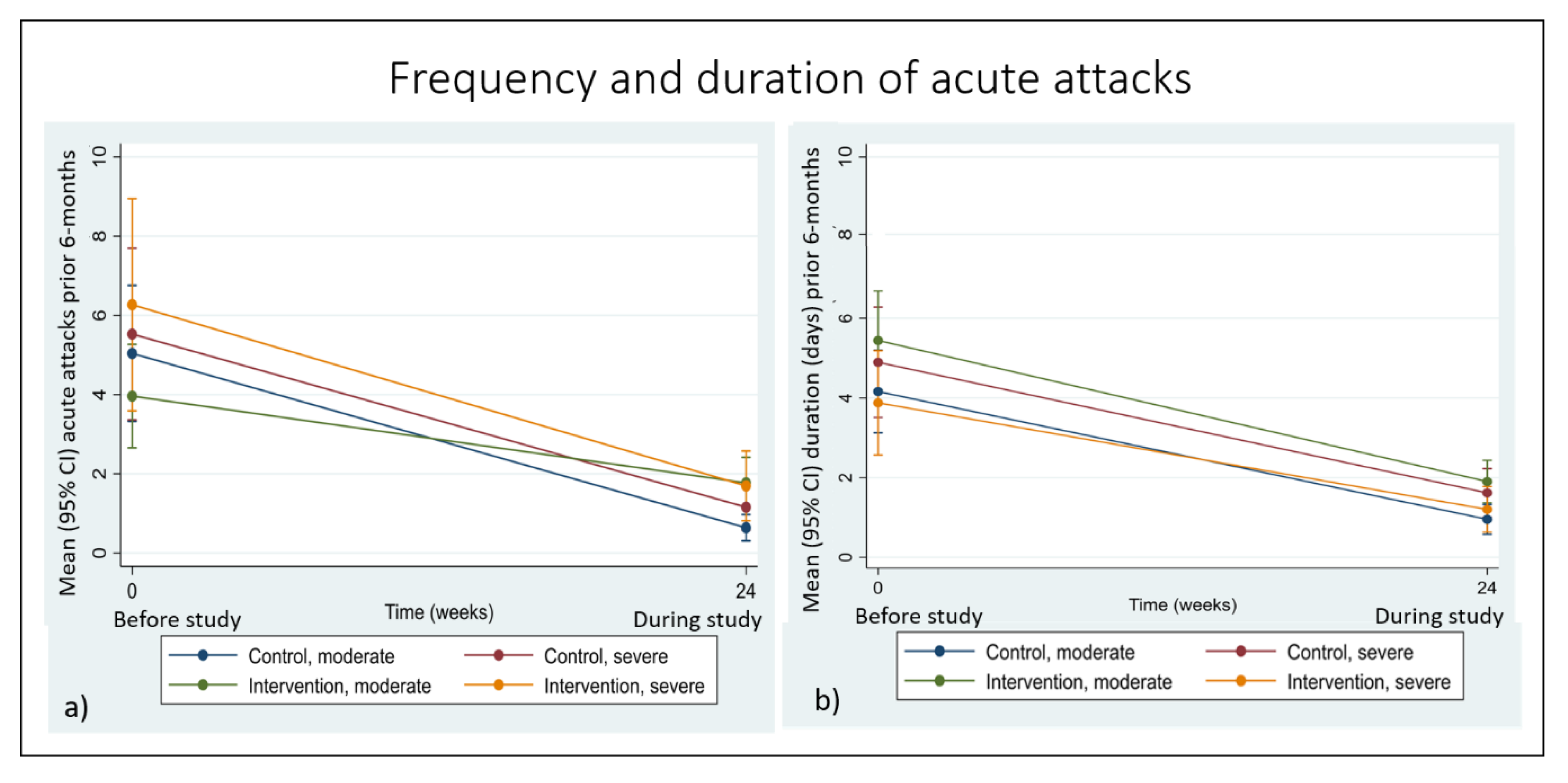
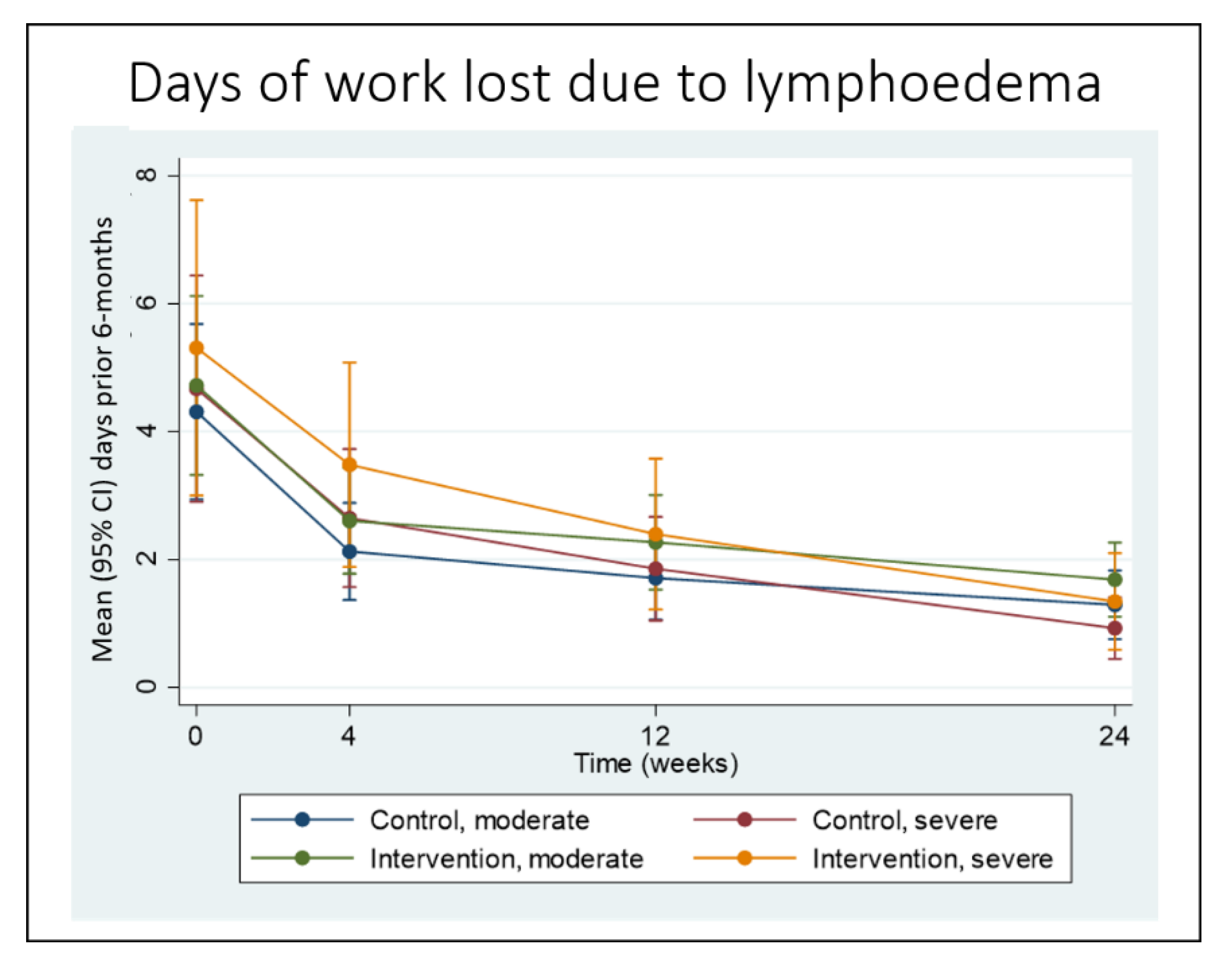
3.3. Secondary Outcomes
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- International Society of Lymphology. The diagnosis and treatment of peripheral lymphedema: 2016 consensus document of the international society of lymphology. Lymphology 2016, 49, 170–184. [Google Scholar]
- Nutman, T.B. Insights into the pathogenesis of disease in human lymphatic filariasis. Lymphat. Res. Biol. 2013, 11, 144–148. [Google Scholar] [CrossRef] [PubMed]
- Szuba, A.; Rockson, S.G. Lymphedema: Anatomy, physiology and pathogenesis. Vasc Med 1997, 2, 321–326. [Google Scholar] [CrossRef]
- Addiss, D.G.; Brady, M.A. Morbidity management in the Global Programme to Eliminate Lymphatic Filariasis: A review of the scientific literature. Filaria J. 2007, 6, 2. [Google Scholar] [CrossRef] [PubMed]
- WHO. Progress report 2000–2009 and strategic plan 2010–2020 of the global programme to eliminate lymphatic filariasis: Halfway towards eliminating lymphatic filariasis. In WHO Library Catalogue; World Health Organization: Geneva, Switzerland, 2010. [Google Scholar]
- Betts, H.; Martindale, S.; Chiphwanya, J.; Mkwanda, S.Z.; Matipula, D.E.; Ndhlovu, P.; Mackenzie, C.; Taylor, M.J.; Kelly-Hope, L.A. Significant improvement in quality of life following surgery for hydrocoele caused by lymphatic filariasis in Malawi: A prospective cohort study. PLoS Negl. Trop. Dis. 2020, 14, e0008314. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Lymphatic Filariasis: Managing Morbidity and Preventing Disability: An Aide-Memoire for national Programme Managers; World Health Organization: Geneva, Switzerland, 2013. [Google Scholar]
- Shamsuzzaman, A.K.M.; Haq, R.; Karim, M.J.; Azad, M.B.; Mahmood, A.S.M.S.; Khair, A.; Rahman, M.M.; Hafiz, I.; Ramaiah, K.D.; Mackenzie, C.D.; et al. The significant scale up and success of Transmission Assessment Surveys ’TAS’ for endgame surveillance of lymphatic filariasis in Bangladesh: One step closer to the elimination goal of 2020. PLoS Negl. Trop. Dis. 2017, 11, e0005340. [Google Scholar] [CrossRef]
- Karim, M.J.; Haq, R.; Mableson, H.E.; Mahmood, A.S.M.; Rahman, M.; Chowdhury, S.M.; Rahman, A.; Hafiz, I.; Betts, H.; Mackenzie, C.; et al. Developing the first national database and map of lymphatic filariasis clinical cases in Bangladesh: Another step closer to the elimination goals. PLoS Negl. Trop. Dis. 2019, 13, e0007542. [Google Scholar] [CrossRef]
- Hafiz, I.; Graves, P.; Haq, R.; Flora, M.S.; Kelly-Hope, L. Clinical case estimates of lymphatic filariasis in an endemic district of Bangladesh after a decade of mass drug administration. Trans. R. Soc. Trop. Med. Hyg. 2015, 109, 700–709. [Google Scholar] [CrossRef]
- Suma, T.K.; Shenoy, R.K.; Kumaraswami, V. Efficacy and sustainability of a footcare programme in preventing acute attacks of adenolymphangitis in Brugian filariasis. Trop. Med. Int. Health 2002, 7, 763–766. [Google Scholar] [CrossRef]
- Addiss, D.G.; Michel, M.C.; Michelus, A.; Radday, J.; Billhimer, W.; Louis-Charles, J.; Roberts, J.M.; Kramp, K.; Dahl, B.A.; Keswick, B. Evaluation of antibacterial soap in the management of lymphoedema in Leogane, Haiti. Trans. R. Soc. Trop. Med. Hyg. 2011, 105, 58–60. [Google Scholar] [CrossRef]
- Douglass, J.; Graves, P.; Gordon, S. Self-Care for Management of Secondary Lymphedema: A Systematic Review. PLoS Negl. Trop. Dis. 2016, 10, e0004740. [Google Scholar] [CrossRef] [PubMed]
- Stocks, M.E.; Freeman, M.C.; Addiss, D.G. The Effect of hygiene-based lymphedema management in lymphatic filariasis-endemic areas: A systematic review and meta-analysis. PLoS Negl. Trop. Dis. 2015, 9, e0004171. [Google Scholar] [CrossRef] [PubMed]
- Douglass, J.; Mableson, H.E.; Martindale, S.; Kelly-Hope, L.A. An enhanced self-care protocol for people affected by moderate to severe lymphedema. Methods Protoc. 2019, 2, 77. [Google Scholar] [CrossRef] [PubMed]
- Douglass, J.; Kelly-Hope, L. Comparison of staging systems to assess lymphedema caused by cancer therapies, lymphatic filariasis, and podoconiosis. Lymphat. Res. Biol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Soran, A.; Ozmen, T.; McGuire, K.P.; Diego, E.J.; McAuliffe, P.F.; Bonaventura, M.; Ahrendt, G.M.; Degore, L.; Johnson, R. The importance of detection of subclinical lymphedema for the prevention of breast cancer-related clinical lymphedema after axillary lymph node dissection; A prospective observational study. Lymphat. Res. Biol. 2014, 12, 289–294. [Google Scholar] [CrossRef]
- Wilson, S.F.; Guarner, J.; Valme, A.L.; Louis-Charles, J.; Jones, T.L.; Addiss, D.G. Histopathologic improvement with lymphedema management, Leogane, Haiti. Emerg. Infect. Dis. 2004, 10, 1938–1946. [Google Scholar] [CrossRef]
- Gordon, S.; Melrose, W.; Warner, J.; Buttner, P.; Ward, L. Lymphatic filariasis: A method to identify subclinical lower limb change in PNG adolescents. PLoS Negl. Trop. Dis. 2011, 5, e1242. [Google Scholar] [CrossRef]
- Kar, S.K.; Kar, P.K.; Mania, J. Tissue tonometry: A useful tool for assessing filarial lymphedema. Lymphology 1992, 25, 55–61. [Google Scholar]
- Moseley, A.L.; Piller, N.B. Reliability of bioimpedance spectroscopy and tonometry after breast conserving cancer treatment. Lymphat. Res. Biol. 2008, 6, 85–87. [Google Scholar] [CrossRef]
- Bagheri, S.; Ohlin, K.; Olsson, G.; Brorson, H. Tissue tonometry before and after liposuction of arm lymphedema following breast cancer. Lymphat. Res. Biol. 2005, 3, 66–80. [Google Scholar] [CrossRef]
- Douglass, J.; Mableson, H.; Martindale, S.; Karim, J.; Mahmood, A.S.; Hailekiros, F.; Kelly-Hope, L. Intra-rater reliability and agreement of the indurometer when used to assess mid-calf tissue compressibility among people affected by moderate to severe lymphedema in bangladesh and ethiopia. Lymphat. Res. Biol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Douglass, J.; Graves, P.; Gordon, S. Intrarater reliability of tonometry and bioimpedance spectroscopy to measure tissue compressibility and extracellular fluid in the legs of healthy young people in australia and myanmar. Lymphat. Res. Biol. 2017, 15, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Vanderstelt, S.; Pallotta, O.J.; McEwen, M.; Ullah, S.; Burrow, L.; Piller, N. Indurometer vs. tonometer: Is the indurometer currently able to replace and improve upon the tonometer? Lymphat. Res. Biol. 2015, 13, 131–136. [Google Scholar] [CrossRef] [PubMed]
- Dreyer, G.; Addiss, D.; Dreyer, P.; Noroes, J. Basic Lymphoedema Management, Treatment and Prevention Problems Associated with Lymphatic Filariasis; Hollis Publishing Company: Hollis, NH, USA, 2002. [Google Scholar]
- Addiss, D.G.; Louis-Charles, J.; Roberts, J.; Leconte, F.; Wendt, J.M.; Milord, M.D.; Lammie, P.J.; Dreyer, G. Feasibility and effectiveness of basic lymphedema management in Leogane, Haiti, an area endemic for bancroftian filariasis. PLoS Negl. Trop. Dis. 2010, 4, e668. [Google Scholar] [CrossRef]
- Hartung, C.; Lerer, A.; Anokwa, Y.; Tseng, C.; Brunette, W.; Borriello, G. Open data kit: Tools to build information services for developing regions. In Proceedings of the 4th ACM/IEEE International Conference on Information and Communication Technologies and Development, London, UK, 13–16 December 2010; pp. 1–12. [Google Scholar]
- World Medical Association. World Medical Association Declaration of Helsinki: Ethical principles for medical research involving human subjects. JAMA 2013, 310, 2191–2194. [Google Scholar] [CrossRef]
- Douglass, J.; Graves, P.; Lindsay, D.; Becker, L.; Roineau, M.; Masson, J.; Aye, N.N.; Win, S.S.; Wai, T.; Win, Y.Y.; et al. Lymphatic filariasis increases tissue compressibility and extracellular fluid in lower limbs of asymptomatic young people in central myanmar. Trop. Med. Infect. Dis. 2017, 2, 50. [Google Scholar] [CrossRef]
- Lawenda, B.D.; Mondry, T.E.; Johnstone, P.A.S. Lymphedema: A primer on the identification and management of a chronic condition in oncologic treatment. Ca Cancer J. Clin. 2009, 59, 8–24. [Google Scholar] [CrossRef]
- Kerketta, A.S.; Babu, B.V.; Rath, K.; Jangid, P.K.; Nayak, A.N.; Kar, S.K. A randomized clinical trial to compare the efficacy of three treatment regimens along with footcare in the morbidity management of filarial lymphoedema. Trop. Med. Int. Health 2005, 10, 698–705. [Google Scholar] [CrossRef]
- Martindale, S.; Mackenzie, C.; Mkwanda, S.; Smith, E.; Stanton, M.; Molyneux, D.; Kelly-Hope, L. “Unseen” caregivers: The disproportionate gender balance and role of females in the home- based care of lymphatic filariasis patients in Malawi. Front. Women’s Health 2017, 2, 1–3. [Google Scholar] [CrossRef][Green Version]
- Caprioli, T.; Martindale, S.; Mengiste, A.; Assefa, D.; Hailekiros, F.; Tamiru, M.; Negussu, N.; Taylor, M.; Betts, H.; Kelly-Hope, L.A. Quantifying the socio-economic impact of leg lymphoedema on patient caregivers in a lymphatic filariasis and podoconiosis co-endemic district of Ethiopia. PLoS Negl. Trop. Dis. 2020, 14, e0008058. [Google Scholar] [CrossRef]
- Stillwaggon, E.; Sawers, L.; Rout, J.; Addiss, D.; Fox, L. Economic costs and benefits of a community-based lymphedema management program for lymphatic filariasis in Odisha State, India. Am. J. Trop. Med. Hyg. 2016, 95, 877–884. [Google Scholar] [CrossRef] [PubMed]
- Martindale, S.; Mkwanda, S.Z.; Smith, E.; Molyneux, D.; Stanton, M.C.; Kelly-Hope, L.A. Quantifying the physical and socio-economic burden of filarial lymphoedema in Chikwawa District, Malawi. Trans. R. Soc. Trop. Med. Hyg. 2014, 108, 759–767. [Google Scholar] [CrossRef] [PubMed]
- Stout, N.L.; Pfalzer, L.A.; Springer, B.; Levy, E.; McGarvey, C.L.; Danoff, J.V.; Gerber, L.H.; Soballe, P.W. Breast cancer–related lymphedema: Comparing direct costs of a prospective surveillance model and a traditional model of care. Phys. Ther. 2012, 92, 152–163. [Google Scholar] [CrossRef] [PubMed]
- Shenoy, R.K.; Kumaraswami, V.; Suma, T.K.; Rajan, K.; Radhakuttyamma, G. A double-blind, placebo-controlled study of the efficacy of oral penicillin, diethylcarbamazine or local treatment of the affected limb in preventing acute adenolymphangitis in lymphoedema caused by brugian filariasis. Ann. Trop Med. Parasitol. 1999, 93, 367–377. [Google Scholar] [CrossRef]
- Yahathugoda, T.C.; Weerasooriya, M.V.; Samarawickrema, W.A.; Kimura, E.; Itoh, M. Impact of two follow-up schemes on morbidity management and disability prevention (MMDP) programme for filarial lymphedema in Matara, Sri Lanka. Parasitol. Int. 2018, 67, 176–183. [Google Scholar] [CrossRef]
- Narahari, S.R.; Aggithaya, M.G.; Prasanna, K.S.; Bose, K.S. An integrative treatment for lower limb lymphedema (elephantiasis). J. Altern Complement. Med. 2010, 16, 145–149. [Google Scholar] [CrossRef]
- Bernhard, L.; Bernhard, P.; Magnussen, P. Management of patients with lymphoedema caused by filariasis in north-eastern Tanzania. Physiotherapy 2003, 89, 743–749. [Google Scholar] [CrossRef]
- Moseley, A.L.; Piller, N.B.; Carati, C.J. The effect of gentle arm exercise and deep breathing on secondary arm lymphedema. Lymphology 2005, 38, 136–145. [Google Scholar]
- Gautam, A.P.; Maiya, A.G.; Vidyasagar, M.S. Effect of home-based exercise program on lymphedema and quality of life in female postmastectomy patients: Pre-post intervention study. J. Rehabil. Res. Dev. 2011, 48, 1261–1268. [Google Scholar] [CrossRef]
- Barclay, J.; Vestey, J.; Lambert, A.; Balmer, C. Reducing the symptoms of lymphoedema: Is there a role for aromatherapy? Eur J. Oncol. Nurs. 2006, 10, 140–149. [Google Scholar] [CrossRef]
- Aggithaya, M.G.; Narahari, S.R.; Vayalil, S.; Shefuvan, M.; Jacob, N.K.; Sushma, K.V. Self care integrative treatment demonstrated in rural community setting improves health related quality of life of lymphatic filariasis patients in endemic villages. Acta Trop. 2013, 126, 198–204. [Google Scholar] [CrossRef] [PubMed]
- Lehman, L.F.; Geyer, M.J.; Bolton, L. Ten Steps. A Guide for Health Promotion and Empowerment of People Affected by Neglected Tropical Diseases. Available online: https://leprosy.org/ten-steps/ (accessed on 26 June 2019).
| Standard Self-Care | Enhanced Self-Care | |||
|---|---|---|---|---|
| (Controls) | (Intervention) | p = | ||
| Participants | n = 71 | n = 71 | ||
| Age, median (IQR) | 55 (48, 65) | 52 (45, 60) | 0.16 | |
| Gender = female | 55 (77%) | 52 (73%) | 0.56 | |
| Marital status = married | 50 (71%) | 59 (82%) | 0.17 | |
| Highest education = illiterate | 35 (50%) | 48 (67%) | 0.22 | |
| Maximum stage of lymphedema | 0.22 | |||
| = moderate | n (%) | 43 (61%) | 50 (70%) | |
| = severe | n (%) | 18 (39%) | 21 (30%) | |
| Acute attacks—last 1 month | Median (IQR) | 1 (0, 1) | 1 (0, 1) | 0.86 |
| Workdays lost—last 1 month | Median (IQR) | 3 (0, 6) | 4 (0, 7) | 0.48 |
| Acute attacks—last 6 months | Median (IQR) | 3.5 (1.5, 6) | 3 (2, 6) | 0.74 |
| Workdays lost—last 6 months | Median (IQR) | 18 (8, 30) | 18 (7, 30) | 0.96 |
| Circumference—left leg (cm) | Median (IQR) Mean (SD | 29.2 (24.6, 33.8) 29.4 (5.91) | 27.8 (24.2, 32.4) 28.6 (6.80) | 0.48 0.41 |
| Circumference—right leg (cm) | Median (IQR) Mean (SD) | 30.5 (25.8, 34.1) 29.9 (5.75) | 27.9 (24.9, 32.4) 29.2 (5.77) | 0.25 0.40 |
| Indurometer score—left leg * | Median (IQR) Mean (SD) | 3.20 (2.71, 3.62) 3.17 (0.78) | 3.45 (2.95, 4.02) 3.48 (0.82) | 0.03 0.03 |
| Indurometer score—right leg* | Median (IQR) Mean (SD) | 3.51 (3, 3.86) 3.44 (0.76) | 3.53 (3.18, 3.94) 3.56 (0.64) | 0.40 0.34 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Douglass, J.; Mableson, H.; Martindale, S.; Jhara, S.T.; Karim, M.J.; Rahman, M.M.; Kawsar, A.A.; Khair, A.; Mahmood, A.S.; Rahman, A.F.; et al. Effect of an Enhanced Self-Care Protocol on Lymphedema Status among People Affected by Moderate to Severe Lower-Limb Lymphedema in Bangladesh, a Cluster Randomized Controlled Trial. J. Clin. Med. 2020, 9, 2444. https://doi.org/10.3390/jcm9082444
Douglass J, Mableson H, Martindale S, Jhara ST, Karim MJ, Rahman MM, Kawsar AA, Khair A, Mahmood AS, Rahman AF, et al. Effect of an Enhanced Self-Care Protocol on Lymphedema Status among People Affected by Moderate to Severe Lower-Limb Lymphedema in Bangladesh, a Cluster Randomized Controlled Trial. Journal of Clinical Medicine. 2020; 9(8):2444. https://doi.org/10.3390/jcm9082444
Chicago/Turabian StyleDouglass, Janet, Hayley Mableson, Sarah Martindale, Sanya Tahmina Jhara, Mohammad Jahirul Karim, Muhammad Mujibur Rahman, Abdullah Al Kawsar, Abul Khair, ASM Sultan Mahmood, AKM Fazlur Rahman, and et al. 2020. "Effect of an Enhanced Self-Care Protocol on Lymphedema Status among People Affected by Moderate to Severe Lower-Limb Lymphedema in Bangladesh, a Cluster Randomized Controlled Trial" Journal of Clinical Medicine 9, no. 8: 2444. https://doi.org/10.3390/jcm9082444
APA StyleDouglass, J., Mableson, H., Martindale, S., Jhara, S. T., Karim, M. J., Rahman, M. M., Kawsar, A. A., Khair, A., Mahmood, A. S., Rahman, A. F., Chowdhury, S. M., Kim, S., Betts, H., Taylor, M., & Kelly-Hope, L. (2020). Effect of an Enhanced Self-Care Protocol on Lymphedema Status among People Affected by Moderate to Severe Lower-Limb Lymphedema in Bangladesh, a Cluster Randomized Controlled Trial. Journal of Clinical Medicine, 9(8), 2444. https://doi.org/10.3390/jcm9082444





