Cardiovascular Manifestations and Complications of Pheochromocytomas and Paragangliomas
Abstract
1. Introduction
2. PPGLs and Hypertension
3. PPGL- and Hypertensive-Crisis
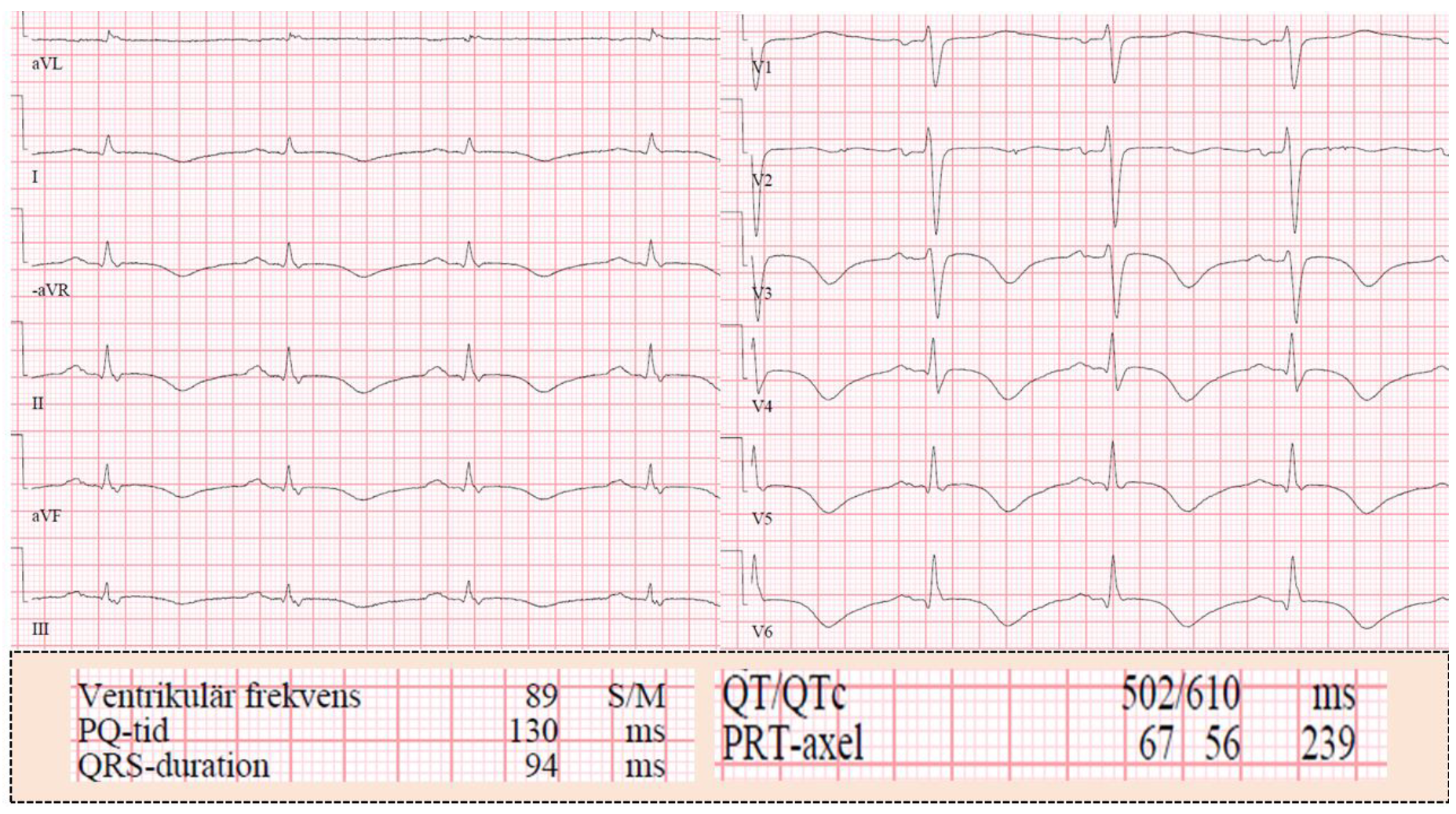
4. PPGLs and Electrocardiogram Changes
5. PPGLs and Arrhythmias
6. PPGLs and Acute Coronary Syndrome
7. PPGLs and Dilated Cardiomyopathy
8. PPGLs and Myocarditis
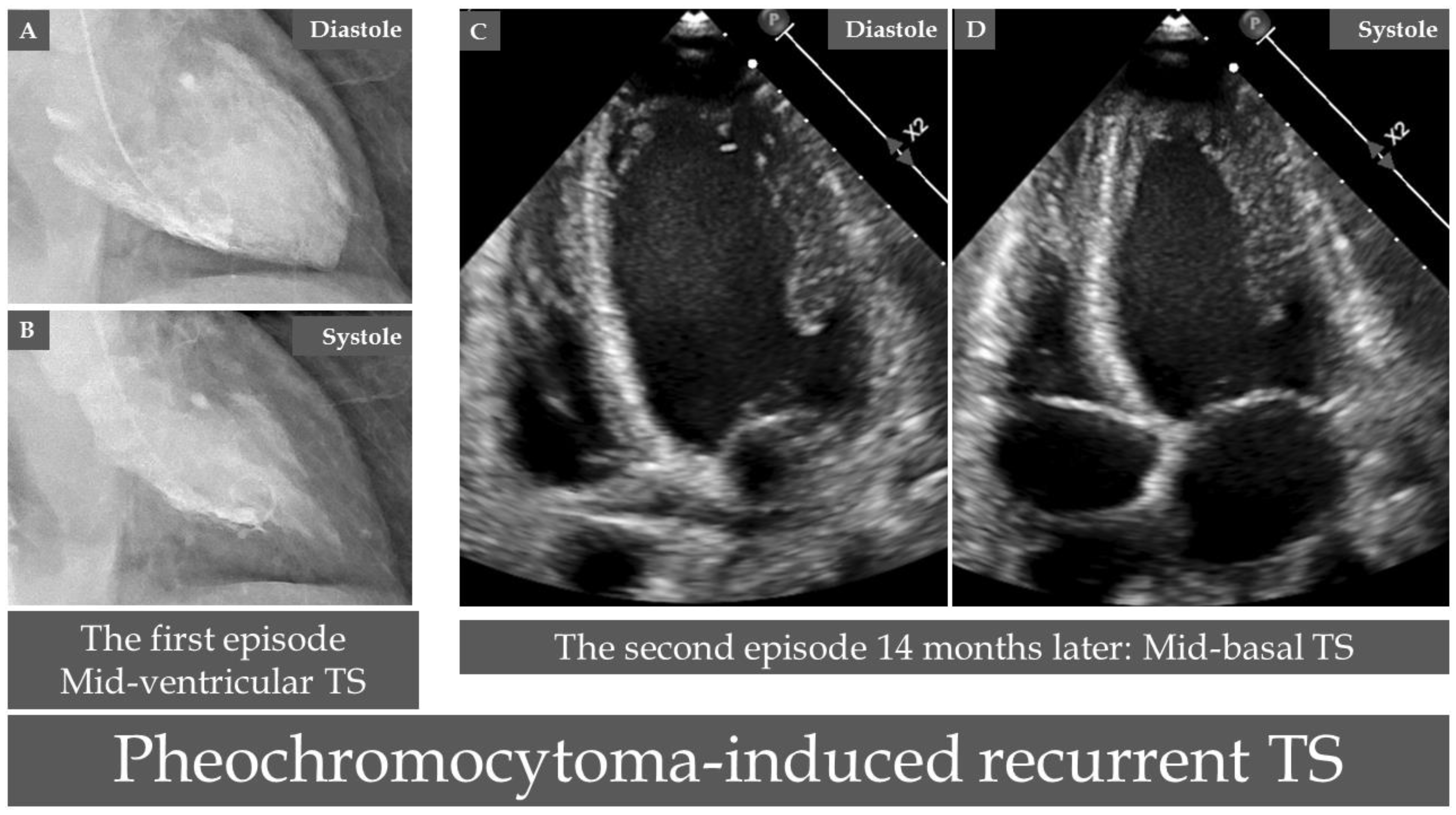
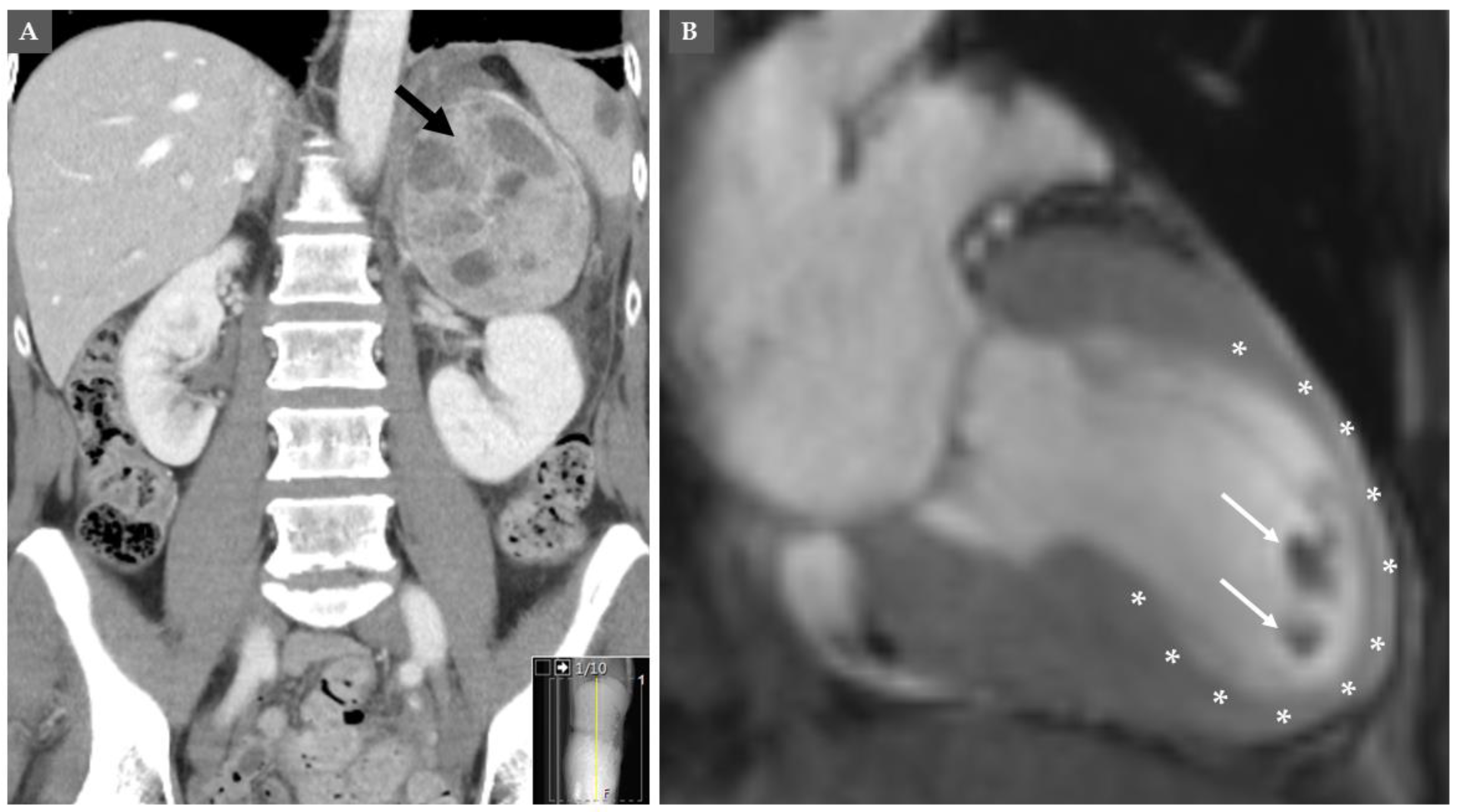
9. PPGLs and Takotsubo Syndrome
10. PPGLs and Thrombo-Embolism
11. PPGLs in Children and Cardiovascular Complications
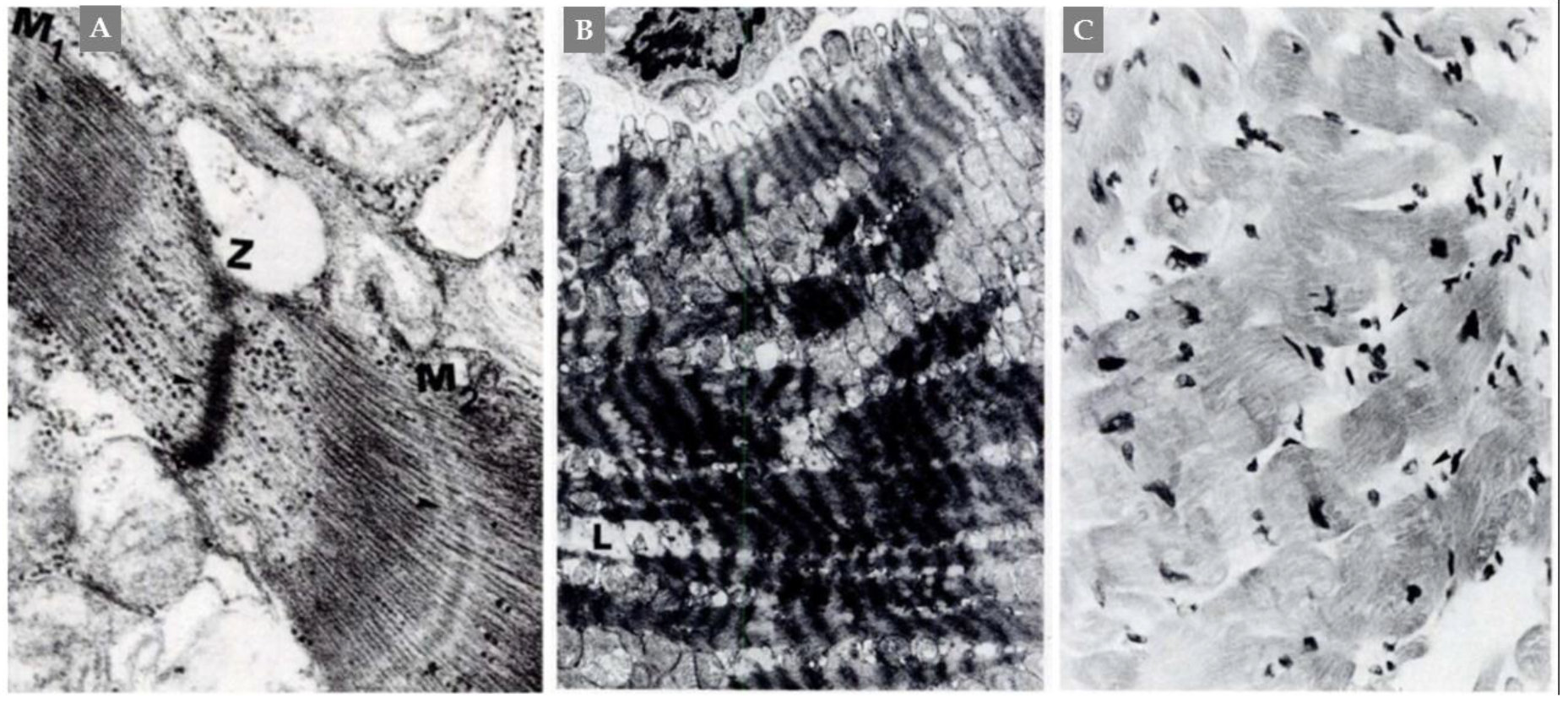
12. Histopathology of Myocardium in PPGLs-Induced Cardiac Complications
13. Is There a Common Thread Tethering the Cardiac Manifestation Induced by PPGLs?
14. PPGL Management
15. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Lenders, J.W.; Duh, Q.Y.; Eisenhofer, G.; Gimenez-Roqueplo, A.P.; Grebe, S.K.; Murad, M.H.; Naruse, M.; Pacak, K.; Young, W.F., Jr.; Endocrine, S. Pheochromocytoma and paraganglioma: An endocrine society clinical practice guideline. J. Clin. Endocrinol. Metab. 2014, 99, 1915–1942. [Google Scholar] [CrossRef] [PubMed]
- Neumann, H.P.H.; Young, W.F., Jr.; Eng, C. Pheochromocytoma and Paraganglioma. N. Engl. J. Med. 2019, 381, 552–565. [Google Scholar] [CrossRef]
- Stratakis, C.A. Pheochromocytomas: Fabulous, Fascinating, and First (in everything)! Horm. Metab. Res. 2019, 51, 401–402. [Google Scholar] [CrossRef] [PubMed]
- Santos, J.R.U.; Brofferio, A.; Viana, B.; Pacak, K. Catecholamine-Induced Cardiomyopathy in Pheochromocytoma: How to Manage a Rare Complication in a Rare Disease? Horm. Metab. Res. 2019, 51, 458–469. [Google Scholar] [CrossRef] [PubMed]
- Pappachan, J.M.; Tun, N.N.; Arunagirinathan, G.; Sodi, R.; Hanna, F.W.F. Pheochromocytomas and Hypertension. Curr. Hypertens. Rep. 2018, 20, 3. [Google Scholar] [CrossRef]
- Falhammar, H.; Kjellman, M.; Calissendorff, J. Initial clinical presentation and spectrum of pheochromocytoma: A study of 94 cases from a single center. Endocr. Connect. 2018, 7, 186–192. [Google Scholar] [CrossRef]
- Yeomans, H.; Calissendorff, J.; Volpe, C.; Falhammar, H.; Mannheimer, B. Limited value of long-term biochemical follow-up in patients with adrenal incidentalomas-a retrospective cohort study. BMC Endocr. Disord. 2015, 15, 6. [Google Scholar] [CrossRef]
- Patrova, J.; Jarocka, I.; Wahrenberg, H.; Falhammar, H. Clinical Outcomes in Adrenal Incidentaloma: Experience from One Center. Endocr. Pract. Off. J. Am. Coll. Endocrinol. Am. Assoc. Clin. Endocrinol. 2015, 21, 870–877. [Google Scholar] [CrossRef]
- Mantero, F.; Terzolo, M.; Arnaldi, G.; Osella, G.; Masini, A.M.; Ali, A.; Giovagnetti, M.; Opocher, G.; Angeli, A. A survey on adrenal incidentaloma in Italy. Study Group on Adrenal Tumors of the Italian Society of Endocrinology. J. Clin. Endocrinol. Metab. 2000, 85, 637–644. [Google Scholar] [CrossRef]
- Amar, L.; Servais, A.; Gimenez-Roqueplo, A.P.; Zinzindohoue, F.; Chatellier, G.; Plouin, P.F. Year of diagnosis, features at presentation, and risk of recurrence in patients with pheochromocytoma or secreting paraganglioma. J. Clin. Endocrinol. Metab. 2005, 90, 2110–2116. [Google Scholar] [CrossRef]
- Gruber, L.M.; Hartman, R.P.; Thompson, G.B.; McKenzie, T.J.; Lyden, M.L.; Dy, B.M.; Young, W.F.; Bancos, I. Pheochromocytoma Characteristics and Behavior Differ Depending on Method of Discovery. J. Clin. Endocrinol. Metab. 2019, 104, 1386–1393. [Google Scholar] [CrossRef] [PubMed]
- Hamidi, O.; Young, W.F., Jr.; Iniguez-Ariza, N.M.; Kittah, N.E.; Gruber, L.; Bancos, C.; Tamhane, S.; Bancos, I. Malignant Pheochromocytoma and Paraganglioma: 272 Patients Over 55 Years. J. Clin. Endocrinol. Metab. 2017, 102, 3296–3305. [Google Scholar] [CrossRef] [PubMed]
- Y-Hassan, S.; Falhammar, H. Clinical features, complications, and outcomes of exogenous and endogenous catecholamine-triggered Takotsubo syndrome: A systematic review and meta-analysis of 156 published cases. Clin. Cardiol. 2020, 43, 459–467. [Google Scholar] [CrossRef] [PubMed]
- Falhammar, H.; Stenman, A.; Calissendorff, J.; Juhlin, C.C. Presentation, Treatment, Histology, and Outcomes in Adrenal Medullary Hyperplasia Compared With Pheochromocytoma. J. Endocr. Soc. 2019, 3, 1518–1530. [Google Scholar] [CrossRef] [PubMed]
- Zelinka, T.; Petrak, O.; Turkova, H.; Holaj, R.; Strauch, B.; Krsek, M.; Vrankova, A.B.; Musil, Z.; Duskova, J.; Kubinyi, J.; et al. High incidence of cardiovascular complications in pheochromocytoma. Horm. Metab. Res. 2012, 44, 379–384. [Google Scholar] [CrossRef]
- Zhang, R.; Gupta, D.; Albert, S.G. Pheochromocytoma as a reversible cause of cardiomyopathy: Analysis and review of the literature. Int. J. Cardiol. 2017, 249, 319–323. [Google Scholar] [CrossRef]
- Pacak, K.; Linehan, W.M.; Eisenhofer, G.; Walther, M.M.; Goldstein, D.S. Recent advances in genetics, diagnosis, localization, and treatment of pheochromocytoma. Ann. Intern. Med. 2001, 134, 315–329. [Google Scholar] [CrossRef]
- Greenleaf, C.E.; Griffin, L.A.; Shake, J.G.; Orr, W.S. Hypertensive crisis secondary to pheochromocytoma. Bayl. Univ. Med. Cent. Proc. 2017, 30, 314–315. [Google Scholar] [CrossRef][Green Version]
- Yeoh, C.J.; Ng, S.Y.; Goh, B.K. Pheochromocytoma Multisystem Crisis Triggered by Glucocorticoid Administration and Aggravated by Citrate Dialysis. A A Case Rep. 2017, 8, 58–60. [Google Scholar] [CrossRef]
- Huddle, K.R.; Kalliatakis, B.; Skoularigis, J. Pheochromocytoma associated with clinical and echocardiographic features simulating hypertrophic obstructive cardiomyopathy. Chest 1996, 109, 1394–1397. [Google Scholar] [CrossRef]
- Jacob, J.L.; da Silveira, L.C.; de Freitas, C.G.; Centola, C.A.; Nicolau, J.C.; Lorga, A.M. Pheochromocytoma with echocardiographic features of obstructive hypertrophic cardiomyopathy. A case report. Angiology 1994, 45, 985–989. [Google Scholar] [CrossRef] [PubMed]
- Shub, C.; Williamson, M.D.; Tajik, A.J.; Eubanks, D.R. Dynamic left ventricular outflow tract obstruction associated with pheochromocytoma. Am. Heart J. 1981, 102, 286–290. [Google Scholar] [CrossRef]
- Schuiki, E.R.; Jenni, R.; Amann, F.W.; Ziegler, W.H. A reversible form of apical left ventricular hypertrophy associated with pheochromocytoma. J. Am. Soc. Echocardiogr. 1993, 6, 327–331. [Google Scholar] [CrossRef]
- Falhammar, H.; Kjellman, M.; Calissendorff, J. Treatment and outcomes in pheochromocytomas and paragangliomas: A study of 110 cases from a single center. Endocrine 2018, 62, 566–575. [Google Scholar] [CrossRef]
- Wiesner, T.D.; Bluher, M.; Windgassen, M.; Paschke, R. Improvement of insulin sensitivity after adrenalectomy in patients with pheochromocytoma. J. Clin. Endocrinol. Metab. 2003, 88, 3632–3636. [Google Scholar] [CrossRef]
- Juszczak, K.; Drewa, T. Adrenergic crisis due to pheochromocytoma—practical aspects. A short review. Cent. Eur. J. Urol. 2014, 67, 153–155. [Google Scholar] [CrossRef]
- Y-Hassan, S.; Falhammar, H. Pheochromocytoma-and paraganglioma-triggered Takotsubo syndrome. Endocrine 2019, 65, 483–493. [Google Scholar] [CrossRef]
- Y-Hassan, S.; Tornvall, P. Epidemiology, pathogenesis, and management of takotsubo syndrome. Clin. Auton. Res. 2018, 28, 53–65. [Google Scholar] [CrossRef]
- Riester, A.; Weismann, D.; Quinkler, M.; Lichtenauer, U.D.; Sommerey, S.; Halbritter, R.; Penning, R.; Spitzweg, C.; Schopohl, J.; Beuschlein, F.; et al. Life-threatening events in patients with pheochromocytoma. Eur. J. Endocrinol. Eur. Fed. Endocr. Soc. 2015, 173, 757–764. [Google Scholar] [CrossRef]
- Pekic, S.; Jovanovic, V.; Tasic, G.; Paunovic, I.; Tatic, S.; Dundjerovic, D.; Doknic, M.; Miljic, D.; Stojanovic, M.; Nikolic Djurovic, M.; et al. Intracerebral hemorrhage as a first sign of pheochromocytoma: Case report and review of the literature. Endokrynol. Pol. 2019, 70, 298–303. [Google Scholar] [CrossRef]
- Sayer, W.J.; Moser, M.; Mattingly, T.W. Pheochromocytoma and the abnormal electrocardiogram. Am. Heart J. 1954, 48, 42–53. [Google Scholar] [CrossRef]
- Haas, G.J.; Tzagournis, M.; Boudoulas, H. Pheochromocytoma: Catecholamine-mediated electrocardiographic changes mimicking ischemia. Am. Heart J. 1988, 116, 1363–1365. [Google Scholar] [CrossRef]
- Hirata, M.; Takeda, K.; Sasaki, S.; Oguro, M.; Kawasaki, S.; Hayashi, J.; Nakata, T.; Itoh, H.; Fushiki, S.; Nakagawa, M. A case of pheochromocytoma with an AMI-like ECG change corrected by an alpha-blocking agent. Nihon Naibunpi Gakkai Zasshi 1990, 66, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Cheng, T.O.; Bashour, T.T. Striking electrocardiographic changes associated with pheochromocytoma. Masquerading as ischemic heart disease. Chest 1976, 70, 397–399. [Google Scholar] [CrossRef]
- Trevethan, S.; Castilla, R.; Medrano, G.; de Michelli, A. Giant T waves simulating apical hypertrophic myocardiopathy that disappear with sodium nitroprusside administration. Case report of pheochromocytoma. J. Electrocardiol. 1991, 24, 267–275. [Google Scholar] [CrossRef]
- Y-Hassan, S. The pathogenesis of reversible T-wave inversions or large upright peaked T-waves: Sympathetic T-waves. Int. J. Cardiol. 2015, 191, 237–243. [Google Scholar] [CrossRef]
- Y-Hassan, S. Clinical Features and Outcome of Pheochromocytoma-Induced Takotsubo Syndrome: Analysis of 80 Published Cases. Am. J. Cardiol. 2016, 117, 1836–1844. [Google Scholar] [CrossRef]
- Lee, M.; Langsjeon, D.; Devabhaktuni, S.; Olsovsky, G. Pheochromocytoma and sinus node dysfunction. Bayl. Univ. Med. Cent. Proc. 2019, 32, 119–120. [Google Scholar] [CrossRef]
- Rostoff, P.; Nessler, B.; Pikul, P.; Golinska-Grzybala, K.; Miszalski-Jamka, T.; Nessler, J. Fulminant adrenergic myocarditis complicated by pulmonary edema, cardiogenic shock and cardiac arrest. Am. J. Emerg. Med. 2018, 36, 344.e1–344.e4. [Google Scholar] [CrossRef]
- Gould, J.; Mundal, A.L. Multifocal ventricular tachycardia induced by etamon in a case of pheochromocytoma. Am. Heart J. 1951, 42, 460–466. [Google Scholar] [CrossRef]
- Buist, R.J. Phaeochromocytoma presenting as an acute abdomen. Br. Med. J. 1985, 291, 1724–1725. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Boldt, M.H.; Flexner, M.; Ortner, A.B. Pheochromocytoma associated with painless myocardial infarction. Ann. Intern. Med. 1957, 46, 1165–1177. [Google Scholar] [CrossRef] [PubMed]
- Daubert, J.C.; Rouxel, P.; Langevin, P.; Lardy, B.; Pony, J.C.; Gouffault, J. Coronary insufficiency in pheochromocytoma. Arch. Mal. Coeur Vaiss 1976, 69, 49–58. [Google Scholar] [PubMed]
- Priest, W.M. Phaeochromocytoma with fatal myocardial infarction in a man aged 22. Br. Med. J. 1952, 2, 860–862. [Google Scholar] [CrossRef][Green Version]
- Chen, F.; Zheng, M.; Li, X.; Peng, Y.; Chen, M. ST-Segment Elevation Myocardial Infarction Related to Potential Spontaneous Coronary Thrombosis in Pheochromocytoma Crisis. Front. Endocrinol. 2020, 11, 140. [Google Scholar] [CrossRef]
- Short, I.A.; Padfield, P.L. Malignant phaeochromocytoma with severe constipation and myocardial necrosis. Br. Med. J. 1976, 2, 793–794. [Google Scholar] [CrossRef]
- Mattman, P.E. Successful removal of a pheochromocytoma four weeks after acute myocardial infarction. Am. J. Cardiol. 1961, 8, 426–430. [Google Scholar] [CrossRef]
- Bourke, S.J.; Wilkes, G.; Maloney, D.L.; Terry, G. Phaeochromocytoma-induced myocardial necrosis. Scott. Med. J. 1991, 36, 184. [Google Scholar] [CrossRef]
- Melson, E.; Amir, S.; Shepherd, L.; Kauser, S.; Freestone, B.; Kempegowda, P. Myocardial Infarction with non-obstructed coronaries—Atypical presentation of pheochromocytoma. Endocrinol. Diabetes Metab. Case Rep. 2019. [Google Scholar] [CrossRef]
- McGonigle, P.; Webb, S.W.; Adgey, A.A. Phaeochromocytoma: An unusual cause of chest pain. Br. Med. J. (Clin. Res. Ed.) 1983, 286, 1477–1478. [Google Scholar] [CrossRef][Green Version]
- Boulmier, D.; Bazin, P. Myocardial pseudo-infarction: “Stress”-associated catecholamine-induced acute cardiomyopathy or coronary spasm? Ann. Cardiol. Angeiol. 2000, 49, 449–454. [Google Scholar]
- Mauser, M.; Billmann, P.; Fleischmann, D. Acute myocardial infarct in pheochromocytoma crisis. Early coronary angiography findings and echocardiography follow-up. Z. Kardiol. 2001, 90, 297–303. [Google Scholar] [CrossRef] [PubMed]
- Jessurun, C.R.; Adam, K.; Moise, K.J., Jr.; Wilansky, S. Pheochromocytoma-induced myocardial infarction in pregnancy. A case report and literature review. Tex. Heart Inst. J. 1993, 20, 120–122. [Google Scholar] [PubMed]
- Shaw, T.R.; Rafferty, P.; Tait, G.W. Transient shock and myocardial impairment caused by phaeochromocytoma crisis. Br. Heart J. 1987, 57, 194–198. [Google Scholar] [CrossRef]
- Case records of the Massachusetts General Hospital. Weekly clinicopathological exercises. Case 15–1988. A 26-year-old woman with cardiomyopathy, multiple strokes, and an adrenal mass. N. Engl. J. Med. 1988, 318, 970–981. [CrossRef] [PubMed]
- Iga, K.; Gen, H.; Tomonaga, G.; Matsumura, T.; Hori, K. Reversible left ventricular wall motion impairment caused by pheochromocytoma—A case report. Jpn. Circ. J. 1989, 53, 813–818. [Google Scholar] [CrossRef]
- Elian, D.; Harpaz, D.; Sucher, E.; Kaplinsky, E.; Motro, M.; Vered, Z. Reversible catecholamine-induced cardiomyopathy presenting as acute pulmonary edema in a patient with pheochromocytoma. Cardiology 1993, 83, 118–120. [Google Scholar] [CrossRef]
- Dinckal, M.H.; Davutoglu, V.; Soydinc, S.; Kirilmaz, A. Phaeochromocytoma-induced myocarditis mimicking acute myocardial infarction. Int. J. Clin. Pract. 2003, 57, 842–843. [Google Scholar] [PubMed]
- Roubille, F.; Tournant, G.; Sportouch-Dukhan, C.; Davy, J.M.; Piot, C. Recurrent severe acute apical-sparing left ventricular dysfunction in a young woman: Don’t forget pheochromocytoma. Ann. Cardiol. Angeiol. 2010, 59, 52–53. [Google Scholar] [CrossRef] [PubMed]
- Miura, M.; Kawano, H.; Yoshida, T.; Yamagata, Y.; Nakata, T.; Koga, S.; Ikeda, S.; Kageyama, K.; Abe, K.; Maemura, K. The Histological Features of a Myocardial Biopsy Specimen in a Patient in the Acute Phase of Reversible Catecholamine-induced Cardiomyopathy due to Pheochromocytoma. Intern. Med. 2017, 56, 665–671. [Google Scholar] [CrossRef] [PubMed]
- Khattak, S.; Sim, I.; Dancy, L.; Whitelaw, B.; Sado, D. Phaeochromocytoma found on cardiovascular magnetic resonance in a patient presenting with acute myocarditis: An unusual association. BMJ Case Rep. 2018. [Google Scholar] [CrossRef] [PubMed]
- Stowers, S.A.; Gilmore, P.; Stirling, M.; Morantz, J.M.; Miller, A.B.; Meyer, L.J.; Glazer, G.; Behrendt, D. Cardiac pheochromocytoma involving the left main coronary artery presenting with exertional angina. Am. Heart J. 1987, 114, 423–427. [Google Scholar] [CrossRef]
- Goldbaum, T.S.; Henochowicz, S.; Mustafa, M.; Blunda, M.; Lindsay, J., Jr. Pheochromocytoma presenting with Prinzmetal’s angina. Am. J. Med. 1986, 81, 921–922. [Google Scholar] [PubMed]
- Baker, G.; Zeller, N.H.; Weitzner, S.; Leach, J.K. Pheochromocytoma without hypertension presenting as cardiomyopathy. Am. Heart J. 1972, 83, 688–693. [Google Scholar] [CrossRef]
- Garcia, R.; Jennings, J.M. Pheochromocytoma masquerading as a cardiomyopathy. Am. J. Cardiol. 1972, 29, 568–571. [Google Scholar] [CrossRef]
- Giavarini, A.; Chedid, A.; Bobrie, G.; Plouin, P.F.; Hagege, A.; Amar, L. Acute catecholamine cardiomyopathy in patients with phaeochromocytoma or functional paraganglioma. Heart 2013, 99, 1438–1444. [Google Scholar] [CrossRef]
- Gatzoulis, K.A.; Tolis, G.; Theopistou, A.; Gialafos, J.H.; Toutouzas, P.K. Cardiomyopathy due to a pheochromocytoma. A reversible entity. Acta Cardiol. 1998, 53, 227–229. [Google Scholar]
- Wilkenfeld, C.; Cohen, M.; Lansman, S.L.; Courtney, M.; Dische, M.R.; Pertsemlidis, D.; Krakoff, L.R. Heart transplantation for end-stage cardiomyopathy caused by an occult pheochromocytoma. J. Heart Lung. Transpl. 1992, 11, 363–366. [Google Scholar]
- Vilcant, V. Pheochromocytoma-Induced Cardiomyopathy Mimicking Acute Coronary Syndrome. J. Am. Osteopath. Assoc. 2017, 117, 537–540. [Google Scholar] [CrossRef][Green Version]
- Sardesai, S.H.; Mourant, A.J.; Sivathandon, Y.; Farrow, R.; Gibbons, D.O. Phaeochromocytoma and catecholamine induced cardiomyopathy presenting as heart failure. Br. Heart J. 1990, 63, 234–237. [Google Scholar] [CrossRef]
- Van Vliet, P.D.; Burchell, H.B.; Titus, J.L. Focal myocarditis associated with pheochromocytoma. N. Engl. J. Med. 1966, 274, 1102–1108. [Google Scholar] [CrossRef]
- Jepson, R.P.; Slavotinek, A.H.; Gilligan, J.E. Myocarditis and phaeochromocytomata. Aust. N. Z. J. Surg. 1969, 38, 324–328. [Google Scholar] [CrossRef] [PubMed]
- Fauvre, F.M.; John, D.R.; Watkins, L.E. Cardiomyopathy secondary to pheochromocytoma. Calif. Med. 1972, 117, 58–60. [Google Scholar] [PubMed]
- Baratella, M.C.; Menti, L.; Angelini, A.; Daliento, L. An unusual case of myocarditis. Int. J. Cardiol. 1998, 65, 305–310. [Google Scholar] [CrossRef]
- Karch, S.B.; Billingham, M.E. Myocardial contraction bands revisited. Hum. Pathol. 1986, 17, 9–13. [Google Scholar] [CrossRef]
- Silver, M.D. Myocardial lesions in pheochromocytoma. CMAJ 1990, 142, 99–100. [Google Scholar]
- Ferreira, V.M.; Marcelino, M.; Piechnik, S.K.; Marini, C.; Karamitsos, T.D.; Ntusi, N.A.B.; Francis, J.M.; Robson, M.D.; Arnold, J.R.; Mihai, R.; et al. Pheochromocytoma Is Characterized by Catecholamine-Mediated Myocarditis, Focal and Diffuse Myocardial Fibrosis, and Myocardial Dysfunction. J. Am. Coll. Cardiol. 2016, 67, 2364–2374. [Google Scholar] [CrossRef]
- Y-Hassan, S. Myocarditis is an essential feature rather than an exclusion criterion for takotsubo syndrome: Case report. Int. J. Cardiol. 2015, 187, 304–306. [Google Scholar] [CrossRef]
- Y-Hassan, S. Myocarditis changes in the stunned myocardial segments in takotsubo syndrome: The role of the pattern of ventricular wall motion abnormality. Int. J. Cardiol. 2015, 191, 267–269. [Google Scholar] [CrossRef]
- Rolf, A.; Nef, H.M.; Mollmann, H.; Troidl, C.; Voss, S.; Conradi, G.; Rixe, J.; Steiger, H.; Beiring, K.; Hamm, C.W.; et al. Immunohistological basis of the late gadolinium enhancement phenomenon in tako-tsubo cardiomyopathy. Eur. Heart J. 2009, 30, 1635–1642. [Google Scholar] [CrossRef]
- Y-Hassan, S.; De Palma, R. Contemporary review on the pathogenesis of takotsubo syndrome: The heart shedding tears: Norepinephrine churn and foam at the cardiac sympathetic nerve terminals. Int. J. Cardiol. 2017, 228, 528–536. [Google Scholar] [CrossRef] [PubMed]
- Ghadri, J.R.; Wittstein, I.S.; Prasad, A.; Sharkey, S.; Dote, K.; Akashi, Y.J.; Cammann, V.L.; Crea, F.; Galiuto, L.; Desmet, W.; et al. International Expert Consensus Document on Takotsubo Syndrome (Part I): Clinical Characteristics, Diagnostic Criteria, and Pathophysiology. Eur. Heart J. 2018, 39, 2032–2046. [Google Scholar] [CrossRef] [PubMed]
- Y-Hassan, S.; Yamasaki, K. History of takotsubo syndrome: Is the syndrome really described as a disease entity first in 1990? Some inaccuracies. Int. J. Cardiol. 2013, 166, 736–737. [Google Scholar] [CrossRef] [PubMed]
- Sato, H.; Tateishi, H.; Uchida, T.; Dote, K.; Ishihara, M.; Sasaki, K. Stunned myocardium with specific (tsubo-type) left ventriculographic configuration due to multivessel spasm. In Clinical Aspect of Myocardial Injury: From Ischemia to Heart Failure; Kodama, K., Haze, K., Hori, M., Eds.; Kagakuhyouronsha Publ. Co.: Tokyo, Japan, 1990; pp. 56–64. [Google Scholar]
- Dote, K.; Sato, H.; Tateishi, H.; Uchida, T.; Ishihara, M. Myocardial stunning due to simultaneous multivessel coronary spasms: A review of 5 cases. J. Cardiol. 1991, 21, 203–214. [Google Scholar] [PubMed]
- Gagnon, N.; Mansour, S.; Bitton, Y.; Bourdeau, I. Takotsubo-Like Cardiomyopathy in a Large Cohort of Patients with Pheochromocytoma and Paraganglioma. Endocr. Pract. 2017, 23, 1178–1192. [Google Scholar] [CrossRef]
- Y-Hassan, S.; Falhammar, H. Stumbling broke the spleen and unveiled pheochromocytoma, which in turn broke the heart. Endocrine 2020, 67, 727–728. [Google Scholar] [CrossRef]
- Dagartzikas, M.I.; Sprague, K.; Carter, G.; Tobias, J.D. Cerebrovascular event, dilated cardiomyopathy, and pheochromocytoma. Pediatr. Emerg. Care 2002, 18, 33–35. [Google Scholar] [CrossRef]
- Y-Hassan, S. Recurrent takotsubo syndrome triggered by undiagnosed pheochromocytoma. Int. J. Cardiol. 2015, 187, 369–371. [Google Scholar] [CrossRef]
- Schmidt, K.H.; Herholz, T.; Rodeck, J.; Abegunewardene, N.; Kreitner, K.F.; Munzel, T. Pheochromocytoma triggers takotsubo syndrome complicated by cerebral and peripheral embolic events. Eur. Heart J. 2017, 38, 1522–1523. [Google Scholar] [CrossRef]
- Waguespack, S.G.; Rich, T.; Grubbs, E.; Ying, A.K.; Perrier, N.D.; Ayala-Ramirez, M.; Jimenez, C. A current review of the etiology, diagnosis, and treatment of pediatric pheochromocytoma and paraganglioma. J. Clin. Endocrinol. Metab. 2010, 95, 2023–2037. [Google Scholar] [CrossRef]
- Virgone, C.; Andreetta, M.; Avanzini, S.; Chiaravalli, S.; De Pasquale, D.; Crocoli, A.; Inserra, A.; D’Angelo, P.; Alaggio, R.; Opocher, G.; et al. Pheochromocytomas and paragangliomas in children: Data from the Italian Cooperative Study (TREP). Pediatr. Blood Cancer 2020, 67, e28332. [Google Scholar] [CrossRef] [PubMed]
- Nirgiotis, J.G.; Andrassy, R.J. Pheochromocytoma and acute myocardial infarction. South. Med. J. 1990, 83, 1478–1480. [Google Scholar] [CrossRef] [PubMed]
- Imperato-McGinley, J.; Gautier, T.; Ehlers, K.; Zullo, M.A.; Goldstein, D.S.; Vaughan, E.D., Jr. Reversibility of catecholamine-induced dilated cardiomyopathy in a child with a pheochromocytoma. N. Engl. J. Med. 1987, 316, 793–797. [Google Scholar] [CrossRef]
- Frustaci, A.; Loperfido, F.; Gentiloni, N.; Caldarulo, M.; Morgante, E.; Russo, M.A. Catecholamine-induced cardiomyopathy in multiple endocrine neoplasia. A histologic, ultrastructural, and biochemical study. Chest 1991, 99, 382–385. [Google Scholar] [CrossRef]
- Baroldi, G.; Mittleman, R.E.; Parolini, M.; Silver, M.D.; Fineschi, V. Myocardial contraction bands. Definition, quantification and significance in forensic pathology. Int. J. Leg. Med. 2001, 115, 142–151. [Google Scholar] [CrossRef]
- Haft, J.I. Cardiovascular injury induced by sympathetic catecholamines. Prog. Cardiovasc. Dis. 1974, 17, 73–86. [Google Scholar] [CrossRef]
- Sato, K.; Masuda, T.; Izumi, T. Subarachnoid hemorrhage and myocardial damage clinical and experimental studies. Jpn. Heart J. 1999, 40, 683–701. [Google Scholar] [CrossRef]
- Nef, H.M.; Mollmann, H.; Kostin, S.; Troidl, C.; Voss, S.; Weber, M.; Dill, T.; Rolf, A.; Brandt, R.; Hamm, C.W.; et al. Tako-Tsubo cardiomyopathy: Intraindividual structural analysis in the acute phase and after functional recovery. Eur. Heart J. 2007, 28, 2456–2464. [Google Scholar] [CrossRef]
- Burch, G.E.; Sun, S.C.; Colcolough, H.L.; DePasquale, N.P.; Sohal, R.S. Acute myocardial lesions; following experimentally-induced intracranial hemorrhage in mice: A histological and histochemical study. Arch. Pathol. 1967, 84, 517–521. [Google Scholar]
- Thiene, G.; Valente, M.; Cecchetto, A.; Giordano, R.; Pennelli, N. Myocellular necrosis by cathecolamines in pheochromocytoma (author’s transl). G. Ital. Cardiol. 1975, 5, 779–783. [Google Scholar]
- Y-Hassan, S. Insights into the pathogenesis of takotsubo syndrome, which with persuasive reasons should be regarded as an acute cardiac sympathetic disease entity. Isrn. Cardiol. 2012, 2012, 593735. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Samuels, M.A. The brain-heart connection. Circulation 2007, 116, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Wood, R.; Commerford, P.J.; Rose, A.G.; Tooke, A. Reversible catecholamine-induced cardiomyopathy. Am. Heart J. 1991, 121, 610–613. [Google Scholar] [CrossRef]
- Yamanaka, O.; Yasumasa, F.; Nakamura, T.; Ohno, A.; Endo, Y.; Yoshimi, K.; Miura, K.; Yamaguchi, H. “Myocardial stunning”-like phenomenon during a crisis of pheochromocytoma. Jpn. Circ. J. 1994, 58, 737–742. [Google Scholar] [CrossRef]
- Batisse-Lignier, M.; Pereira, B.; Motreff, P.; Pierrard, R.; Burnot, C.; Vorilhon, C.; Maqdasy, S.; Roche, B.; Desbiez, F.; Clerfond, G.; et al. Acute and Chronic Pheochromocytoma-Induced Cardiomyopathies: Different Prognoses?: A Systematic Analytical Review. Medicine 2015, 94, e2198. [Google Scholar] [CrossRef]
- Lyon, A.R.; Rees, P.S.; Prasad, S.; Poole-Wilson, P.A.; Harding, S.E. Stress (Takotsubo) cardiomyopathy—A novel pathophysiological hypothesis to explain catecholamine-induced acute myocardial stunning. Nat. Clin. Pract. Cardiovasc. Med. 2008, 5, 22–29. [Google Scholar] [CrossRef]
- Y-Hassan, S. Plasma Epinephrine Level and its Causal Link to Takotsubo Syndrome Revisited: Critical Review with a Diverse Conclusion. Cardiovasc. Revasc. Med. 2019, 20, 907–914. [Google Scholar] [CrossRef]
- Y-Hassan, S. Tight coronary artery stenosis and takotsubo syndrome triggered each other: Well-illustrated in a case. Cardiovasc. Revasc. Med. 2018, 19, 2–4. [Google Scholar] [CrossRef]
- Nakajo, M.; Shapiro, B.; Glowniak, J.; Sisson, J.C.; Beierwaltes, W.H. Inverse relationship between cardiac accumulation of meta-[131I]iodobenzylguanidine (I-131 MIBG) and circulating catecholamines in suspected pheochromocytoma. J. Nucl. Med. 1983, 24, 1127–1134. [Google Scholar]
- Nakajo, M.; Shimabukuro, K.; Miyaji, N.; Shimada, J.; Shirono, K.; Sakata, H.; Yoshimura, H.; Yonekura, R.; Shinohara, S. Rapid clearance of iodine-131 MIBG from the heart and liver of patients with adrenergic dysfunction and pheochromocytoma. J. Nucl. Med. 1985, 26, 357–365. [Google Scholar]
- Matsumoto, K.; Sugihara, H.; Ito, K.; Terada, K.; Taniguchi, Y.; Ohtsuki, K.; Nakata, T.; Ushijima, Y.; Maeda, T.; Nakagawa, M. 123I-metaiodobenzylguanidine myocardial scintigraphy in a case of pheochromocytoma before and after resection of it. Kaku Igaku 1995, 32, 1023–1028. [Google Scholar] [PubMed]
- Agostini, D.; Darlas, Y.; Filmont, J.E.; Dronne, F.; Babatasi, G.; Grollier, G.; Potier, J.C.; Bouvard, G. The reversibility of cardiac neuronal function after removal of a pheochromocytoma: An I-123 MIBG Scintigraphic Study. Clin. Nucl. Med. 1999, 24, 514–518. [Google Scholar] [CrossRef] [PubMed]
- Samuels, M.A. Neurogenic heart disease: A unifying hypothesis. Am. J. Cardiol. 1987, 60, 15J–19J. [Google Scholar] [CrossRef]
- Samuels, M.A. Neurally induced cardiac damage. Definition of the problem. Neurol. Clin. 1993, 11, 273–292. [Google Scholar] [CrossRef]
- Y-Hassan, S. Acute cardiac sympathetic disruption in the pathogenesis of the takotsubo syndrome: A systematic review of the literature to date. Cardiovasc. Revasc. Med. 2014, 15, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Y-Hassan, S. Pathophysiology of takotsubo syndrome: Acute cardiac sympathetic disruption (ACSD) syndrome. Cardiovasc. Revasc. Med. 2014, 15, 311–312. [Google Scholar] [CrossRef]
- Lenders, J.W.M.; Kerstens, M.N.; Amar, L.; Prejbisz, A.; Robledo, M.; Taieb, D.; Pacak, K.; Crona, J.; Zelinka, T.; Mannelli, M.; et al. Genetics, diagnosis, management and future directions of research of phaeochromocytoma and paraganglioma: A position statement and consensus of the Working Group on endocrine hypertension of the European society of hypertension. J. Hypertens. 2020. [Google Scholar] [CrossRef]
- Buitenwerf, E.; Osinga, T.E.; Timmers, H.; Lenders, J.W.M.; Feelders, R.A.; Eekhoff, E.M.W.; Haak, H.R.; Corssmit, E.P.M.; Bisschop, P.; Valk, G.D.; et al. Efficacy of alpha-Blockers on Hemodynamic Control during Pheochromocytoma Resection: A Randomized Controlled Trial. J. Clin. Endocrinol. Metab. 2020, 105. [Google Scholar] [CrossRef]
- Suzuki, K.; Miyake, T.; Okada, H.; Yamaji, F.; Kitagawa, Y.; Fukuta, T.; Yasuda, R.; Tanaka, Y.; Okamoto, H.; Nachi, S.; et al. Thyrotoxic and pheochromocytoma multisystem crisis: A case report. J. Med. Case Rep. 2017, 11, 173. [Google Scholar] [CrossRef]
- Ghadri, J.R.; Wittstein, I.S.; Prasad, A.; Sharkey, S.; Dote, K.; Akashi, Y.J.; Cammann, V.L.; Crea, F.; Galiuto, L.; Desmet, W.; et al. International Expert Consensus Document on Takotsubo Syndrome (Part II): Diagnostic Workup, Outcome, and Management. Eur. Heart J. 2018, 39, 2047–2062. [Google Scholar] [CrossRef]
| Authors and the Year of Publication | Age Gender | Published with Diagnosis in Addition to PPGL | Important other Findings and Complications | Findings Consistent with Takotsubo Syndrome |
|---|---|---|---|---|
| McGonigle et al., 1983 [50] | 38/F | Recurrent myocardial infarction | Marked reversible ST-elevation and “left ventricular aneurysm” complicated by left ventricular thrombus | Recurrent TS |
| Shaw et al., 1987 [54] | 41/M | Transient shock and myocardial impairment | A left ventricular angiogram showed hyperkinesis of the basal segments and the remaining areas of myocardium were akinetic | Mid-apical TS pattern |
| Case records of the MGH (case 15-1988) [55] | 26/F | Embolic stroke of cardiac origin, cardiomyopathy | Echo: the left ventricle was markedly hypokinetic at the base and the midventricular level, and function at the apex was hyperkinetic Endomyocardial biopsy: vacuolization of the myocytes, myocyte necrosis, and scattered myocytes were hypereosinophilic with focal disruption of their sarcolemmal membrane The left ventricular dysfunction resolved completely | Mid-basal TS (inverted TS) complicated with embolic stroke |
| Iga et al., 1989 [56] | 44/F | Reversible left ventricular wall motion impairment | A figure in the article shows an echo finding typical for mid-apical ballooning | Mid-apical TS |
| Elian et al., 1993 [57] | 41/M | Reversible cardiomyopathy with acute pulmonary edema | Left ventricular thrombus and heart failure | Mid-apical TS |
| Boulmier et al., 2000 [51] | 56/F | Myocardial pseudo-infarction Acute cardiomyopathy or coronary spasm | Diagnosis of myocardial necrosis, acute myocarditis | Mid-apical TS |
| Mauser et al., 2001 [52] | 41/F | A large anterolateral-apical-diaphragmal myocardial infarction | Complicated by acute pulmonary edema and cardiogenic shock, the left ventricular dysfunction resolved after 4 weeks | Mid-apical TS |
| Dinckal et al., 2003 [58] | 44/F | Myocarditis mimicking myocardial infarction | Echo showed basal and midventricular hypokinesia. The ECG and echo changes normalized within few days | Typical mid basal (inverted) TS |
| Roubille et al., 2010 [59] | 35/F | Recurrent severe acute apical sparing left ventricular dysfunction | Echo reveals typical severe basal and midventricular left ventricular dysfunction but preserved apical contractility | Recurrent inverted TS |
| Miura et al., 2017 [60] | 63/F | Reversible cardiomyopathy | Defects of MIBG uptake in the mid-apical segments corresponding to the akinetic segments with normal MIBG uptake in the normo-kinetic basal segments, and histopathological findings consistent with TS | Biventricular mid-apical ballooning pattern consistent with TS |
| Khattak et al., 2018 [61] | 25/M | Acute myocarditis | ECG findings of wide-spread ST-depression besides echo and C-MRI findings of hypokinesis at the basal segments argue strongly for basal TS pattern (inverted TS) with late gadolinium enhancement at the basal segments | Mid-basal (inverted TS) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Y-Hassan, S.; Falhammar, H. Cardiovascular Manifestations and Complications of Pheochromocytomas and Paragangliomas. J. Clin. Med. 2020, 9, 2435. https://doi.org/10.3390/jcm9082435
Y-Hassan S, Falhammar H. Cardiovascular Manifestations and Complications of Pheochromocytomas and Paragangliomas. Journal of Clinical Medicine. 2020; 9(8):2435. https://doi.org/10.3390/jcm9082435
Chicago/Turabian StyleY-Hassan, Shams, and Henrik Falhammar. 2020. "Cardiovascular Manifestations and Complications of Pheochromocytomas and Paragangliomas" Journal of Clinical Medicine 9, no. 8: 2435. https://doi.org/10.3390/jcm9082435
APA StyleY-Hassan, S., & Falhammar, H. (2020). Cardiovascular Manifestations and Complications of Pheochromocytomas and Paragangliomas. Journal of Clinical Medicine, 9(8), 2435. https://doi.org/10.3390/jcm9082435






