Effect of Manual Therapy and Splint Therapy in People with Temporomandibular Disorders: A Preliminary Study
Abstract
1. Introduction
2. Methods
2.1. Participants
2.2. Study Design
2.3. Randomization and Blinding
2.4. Interventions
2.5. Outcomes
2.5.1. Primary Outcome
2.5.2. Secondary Outcomes
2.5.3. Statistical Analysis
3. Results
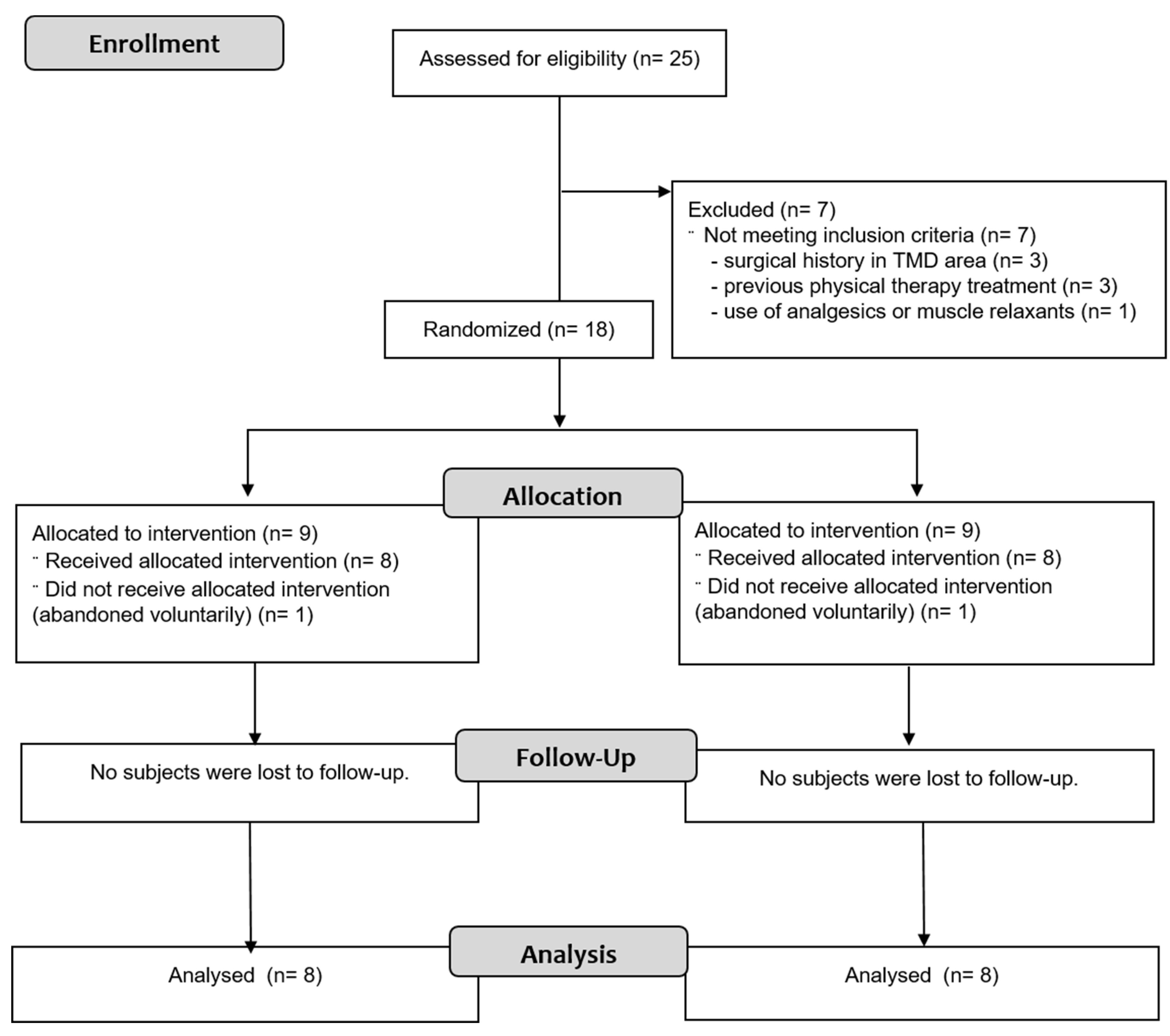
3.1. Participants
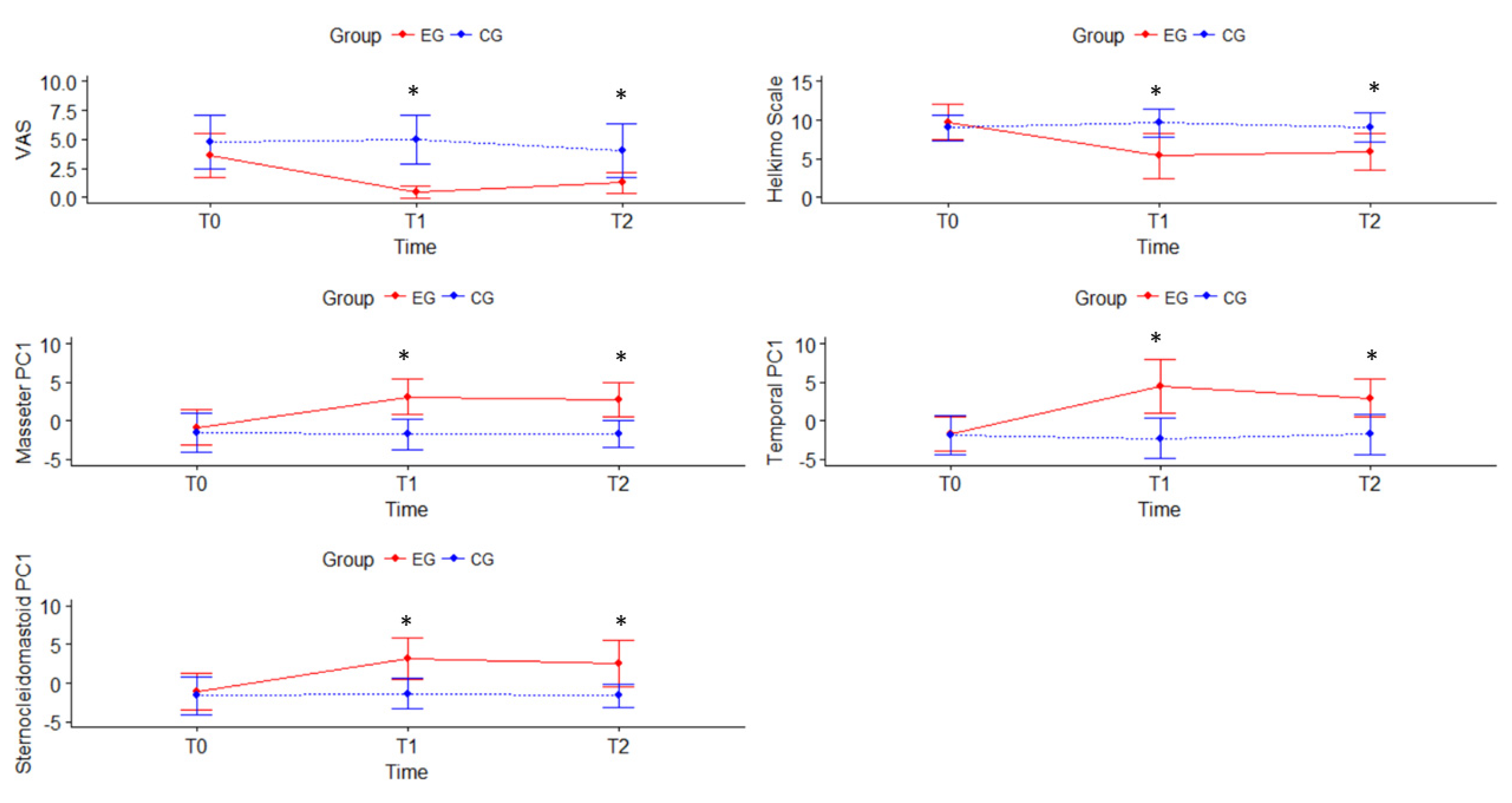
3.2. Effect of the Treatment
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Appendix A
| APPENDIX | ||
| DESCRIPTION OF MANUAL THERAPY INTERVENTIONS | ||
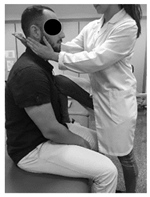 |  |  |
| (1) Neck accessory mobilization technique | (2) Mobilization neck central | (3) Mobilization up neck |
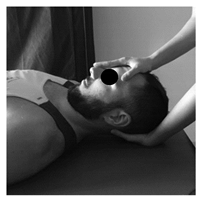
| Patient lies sitting. Non-thrust facet joint distraction mobilization is applied. Clinician left hand separates right lower facet of C7 away from top facet of T1; right hand maintains ventral and medial pressure against lower facet of C7 and compresses it against T1 thus facilitating greater motion on right side of the C7. Manual stabilization of T1 occurs by using the right lower extremity passively, which rotates the T1 segment in opposite direction of the intended mobilization. | Patient lies prone. The posterior-anterior mobilization of the C5 vertebra is carried out. The tips of the clinician’s thumbs are positioned on the spinous process of C5 and an oscillatory pressure is applied in the postero-anterior direction. This mobilization is carried out with a frequency of 2 oscillations per second (2 Hz), with a total of 9 min. The time was distributed in 3 series of 3 min, with 1 min of rest between them. | Patient lies supine. The mobilization is done with one hand in the occipital to perform head traction, and another in the frontal region to apply caudal pressure. This mobilization is carried out at a slow pace of 2 s per oscillation, with a total time of 10 min. |
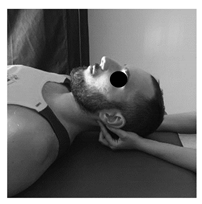 |  |  |
| (4) Suboccipital inhibition technique | (5) Suboccipital accessory mobilization technique | (6) Trigger points technique |
| Patient lies supine. Contact is made with the hands in the occiput. A progressive and deep pressure is applied with the fingertips on the rear arc of the atlas in the direction of the ceiling with slight traction in a cranial direction for 2 min. | Patient lies supine, head turned to the side with slight lateroflexion and neutral flexion-extension. A gentle cephalic decompression is applied, then small circumductions until joint barrier is reached and then a rotation is performed with cranial helical movement. 2 times on each side without passing the joint barrier. | Patient lies supine. Inhibitory pressure is performed on the most sensitive trigger point of the muscles: masseter, temporal and sternocleidomastoid. Gradual pressure is exerted until the onset of pain and maintained 90 s. |
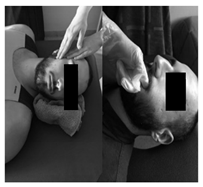 | 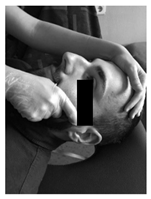 | 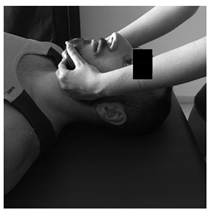 |
| (7) Myofascial technique | (8) Temporomandibular Joint Mobilization technique | (9) Temporomandibular massage |
| Patient lies supine. (a) One hand is placed in the zygomatic arch and the other one makes slow longitudinal movements on the masseter muscle for 5 min. (b) The index finger is introduced in the last molar and pressure is applied in the posterior and cephalic direction in the region of the lateral and medial pterygoid for 5 s in each. | Patient lies supine. 5 mobilizations are made, introducing the thumb in the area of the lower molars applying a caudal traction force taking the mandibular branch with the rest of the fingers. With the opposite hand, the clinician stabilizes the patient’s forehead. | Patient lies supine. Superficial and deep synchronized frictions, compressions and kneading are performed with moderate pressure on the anterior temporal, masseter and sternocleidomastoid muscles, bilaterally for 2 min. |
References
- Pavia, S.; Fischer, R.; Roy, R. Chiropractic treatment of temporomandibular dysfunction: A retrospective case series. J. Chiropr. Med. 2015, 14, 279–284. [Google Scholar] [CrossRef][Green Version]
- Liu, F.; Steinkeler, A. Epidemiology, diagnosis, and treatment of temporomandibular disorders. Dent. Clin. North Am. 2013, 57, 465–479. [Google Scholar] [CrossRef]
- Zhang, C.; Wu, J.-Y.; Deng, D.-L.; He, B.-Y.; Tao, Y.; Niu, Y.-M.; Deng, M.-H. Efficacy of splint therapy for the management of temporomandibular disorders: A meta-analysis. Oncotarget 2016, 7, 84043–84053. [Google Scholar] [CrossRef] [PubMed]
- Chaves, T.C.; Turci, A.M.; Pinheiro, C.F.; Sousa, L.M.; Grossi, D.B. Static body postural misalignment in individuals with temporomandibular disorders: A systematic review. Braz. J. Phys. Ther. 2014, 18, 481–501. [Google Scholar] [CrossRef] [PubMed]
- Armijo-Olivo, S.; Silvestre, R.A.; Fuentes, J.P.; Da Costa, B.R.; Major, P.W.; Warren, S.; Thie, N.M.R.; Magee, D.J. Patients with temporomandibular disorders have increased fatigability of the cervical extensor muscles. Clin. J. Pain 2012, 28, 55–64. [Google Scholar] [CrossRef]
- Wieckiewicz, M.; Boening, K.; Wiland, P.; Shiau, Y.Y.; Paradowska-Stolarz, A. Reported concepts for the treatment modalities and pain management of temporomandibular disorders. J. Headache Pain 2015, 16, 106. [Google Scholar] [CrossRef] [PubMed]
- Schiffman, E.; Ohrbach, R.; Truelove, E.; Look, J.; Anderson, G.; Goulet, J.-P.; List, T.; Svensson, P.; Gonzalez, Y.; Lobbezoo, F.; et al. Diagnostic criteria for temporomandibular disorders (DC/TMD) for clinical and research applications: Recommendations of the international RDC/TMD consortium network* and orofacial pain special interest group. J. Oral. Facial Pain Headache 2014, 28, 6–27. [Google Scholar] [CrossRef]
- Pihut, M.; Szuta, M.; Ferendiuk, E.; Zeńczak-Więckiewicz, D. Differential diagnostics of pain in the course of trigeminal neuralgia and temporomandibular joint dysfunction. Biomed. Res. Int. 2014, 563786. [Google Scholar] [CrossRef]
- Al-Ani, Z.; Gray, R.J.; Davies, S.J.; Sloan, P.; Glenny, A.M. Stabilization splint therapy for the treatment of temporomandibular myofascial pain: A systematic review. J. Dent. Educ. 2005, 69, 1242–1250. [Google Scholar] [CrossRef]
- Dashnyam, K.; Lee, J.H.; Mandakhbayar, N.; Jin, G.Z.; Lee, H.H.; Kim, H.W. Intra-articular biomaterials-assisted delivery to treat temporomandibular joint disorders. J. Tissue Eng. 2018, 9, 204173141877651. [Google Scholar] [CrossRef]
- Pihut, M.; Szuta, M.; Ferendiuk, E.; Zeńczak-Więckiewicz, D. Evaluation of pain regression in patients with temporomandibular dysfunction treated by intra-articular platelet-rich plasma injections: A preliminary report. Biomed. Res. Int. 2014, 132369. [Google Scholar] [CrossRef] [PubMed]
- Conti, P.C.R.; Pinto-Fiamengui, L.M.S.; Cunha, C.O.; de Castro Ferreira Conti, A.C. Orofacial pain and temporomandibular disorders: The impact on oral health and quality of life. Braz. Oral Res. 2012, 26, 120–123. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, M.A.; Tahara, A.K.; Ferreira, I.C.; Intelangelo, L.; Barbosa, A.C. Effects of 8 weeks of masticatory muscles focused endurance exercises on women with oro-facial pain and temporomandibular disorders: A placebo randomised controlled trial. J. Oral Rehabil. 2019, 46, 885–894. [Google Scholar] [CrossRef]
- Goadsby, P.J.; Hoskin, K.L. The distribution of trigeminovascular afferents in the nonhuman primate brain Macaca nemestrina: A c-fos immunocytochemical study. J. Anat. 1997, 190, 367–375. [Google Scholar] [CrossRef] [PubMed]
- La Touche, R.; Fernández-de-las-Peñas, C.; Fernández-Carnero, J.; Escalante, K.; Angulo-Díaz-Parreño, S.; Paris-Alemany, A.; Cleland, J.A. The effects of manual therapy and exercise directed at the cervical spine on pain and pressure pain sensitivity in patients with myofascial temporomandibular disorders. J. Oral Rehabil. 2009, 36, 644–652. [Google Scholar] [CrossRef] [PubMed]
- Creighton, D.; Gruca, M.; Marsh, D.; Murphy, N. A comparison of two non-thrust mobilization techniques applied to the C7 segment in patients with restricted and painful cervical rotation. J. Man. Manip. Ther. 2014, 22, 206–212. [Google Scholar] [CrossRef]
- Oliveira-Campelo, N.M.; Rubens-Rebelatto, J.; Martí, N.-V.F.J.; Alburquerque-Sendí, F.; Fernández-de-Las-Peñas, C. The immediate effects of atlanto-occipital joint manipulation and suboccipital muscle inhibition technique on active mouth opening and pressure pain sensitivity over latent myofascial trigger points in the masticatory muscles. J. Orthop. Sports Phys. Ther. 2010, 40, 310–317. [Google Scholar] [CrossRef]
- Espejo-Antúnez, L.; Castro-Valenzuela, E.; Ribeiro, F.; Albornoz-Cabello, M.; Silva, A.; Rodríguez-Mansilla, J. Immediate effects of hamstring stretching alone or combined with ischemic compression of the masseter muscle on hamstrings extensibility, active mouth opening and pain in athletes with temporomandibular dysfunction. J. Bodyw. Mov. Ther. 2016, 20, 579–587. [Google Scholar] [CrossRef]
- Alajbeg, I.; Gikić, M.; Valentić Peruzović, M. Mandibular range of movement and pain intensity in patients with anterior disc displacement without reduction. Acta Stomatol. Croat. 2015, 49, 119–127. [Google Scholar] [CrossRef]
- Shaffer, S.M.; Brismée, J.-M.; Sizer, P.S.; Courtney, C.A. Temporomandibular disorders. Part 2: Conservative management. J. Man. Manip. Ther. 2014, 22, 13–23. [Google Scholar] [CrossRef]
- Kalamir, A.; Graham, P.L.; Vitiello, A.L.; Bonello, R.; Pollard, H. Intra-oral myofascial therapy versus education and self-care in the treatment of chronic, myogenous temporomandibular disorder: A randomised, clinical trial. Chiropr. Man. Therap. 2013, 21, 17. [Google Scholar] [CrossRef] [PubMed]
- El Hage, Y.; Politti, F.; De Sousa, D.F.M.; Herpich, C.M.; Glória, I.P.D.S.; Gomes, C.A.F.D.P.; Amaral, A.P.; De Melo, N.C.; Da Silva, T.C.; Arruda, E.E.C.; et al. Effect of mandibular mobilization on electromyographic signals in muscles of mastication and static balance in individuals with temporomandibular disorder: Study protocol for a randomized controlled trial. Trials 2013, 14, 316. [Google Scholar] [CrossRef] [PubMed]
- Zwarenstein, M.; Treweek, S.; Gagnier, J.J.; Altman, U.G.; Tunis, S.; Haynes, B.; Oxman, A.D.; Moher, D. Improving the reporting of pragmatic trials: An extension of the CONSORT statement. BMJ 2008, 337, a2390. [Google Scholar] [CrossRef]
- van Grootel, R.J.; Buchner, R.; Wismeijer, D.; van der Glas, H.W. Towards an optimal therapy strategy for myogenous TMD, physiotherapy compared with occlusal splint therapy in an RCT with therapy-and-patient-specific treatment durations. BMC Musculoskelet. Disord. 2017, 18, 76. [Google Scholar] [CrossRef] [PubMed]
- Bijur, P.E.; Silver, W.; Gallagher, E.J. Reliability of the visual analog scale for measurement of acute pain. Acad. Emerg. Med. 2001, 8, 1153–1157. [Google Scholar] [CrossRef]
- Chesterton, L.S.; Sim, J.; Wright, C.C.; Foster, N.E. Interrater reliability of algometry in measuring pressure pain thresholds in healthy humans, using multiple raters. Clin. J. Pain 2007, 23, 760–766. [Google Scholar] [CrossRef]
- Gomes, M.B.; Guimarães, J.P.; Guimarães, F.C.; Neves, A.C.C. Palpation and pressure pain threshold: Reliability and validity in patients with temporomandibular disorders. Cranio Pract. 2008, 26, 202–210. [Google Scholar] [CrossRef]
- Su, N.; Liu, Y.; Yang, X.; Shen, J.; Wang, H. Correlation between oral health-related quality of life and clinical dysfunction index in patients with temporomandibular joint osteoarthritis. J. Oral Sci. 2016, 58, 483–490. [Google Scholar] [CrossRef]
- Hurst, H.; Bolton, J. Assessing the clinical significance of change scores recorded on subjective outcome measures. J. Manip. Physiol. Ther. 2004, 27, 26–35. [Google Scholar] [CrossRef] [PubMed]
- Anderson, M.J.; Braak, C.T. Permutation tests for multi-factorial analysis of variance. J. Stat. Comput. Sim. 2003, 73, 85–113. [Google Scholar] [CrossRef]
- Anderson, M.J. Permutational multivariate analysis of variance (PERMANOVA). Wiley Statsref Stat. Ref. Online 2014, 1–15. [Google Scholar] [CrossRef]
- Ialongo, C. Understanding the effect size and its measures. Biochem. Med. 2016, 150–163. [Google Scholar] [CrossRef]
- Cohen, J. Eta-Squared and partial eta-squared in fixed factor anova designs. Educ. Psychol. Meas. 1973, 33, 107–112. [Google Scholar] [CrossRef]
- Jensen, M.P.; Smith, D.G.; Ehde, D.M.; Robinsin, L.R. Pain site and the effects of amputation pain: Further clarification of the meaning of mild, moderate, and severe pain. Pain 2001, 91, 317–322. [Google Scholar] [CrossRef]
- Lee, J.S. Clinically important change in the visual analog scale after adequate pain control. Acad. Emerg. Med. 2003, 10, 1128–1130. [Google Scholar] [CrossRef]
- Kalichman, L. Massage therapy for fibromyalgia symptoms. Rheumatol. Int. 2010, 30, 1151–1157. [Google Scholar] [CrossRef]
- Dhanani, N.M.; Caruso, T.J.; Carinci, A.J. Complementary and alternative medicine for pain: An evidence-based review. Curr. Pain Headache Rep. 2011, 15, 39–46. [Google Scholar] [CrossRef]
- Marfurt, C.F.; Rajchert, D.M. Trigeminal primary afferent projections to non-trigeminal areas of the rat central nervous system. J. Comp. Neurol. 1991, 303, 489–511. [Google Scholar] [CrossRef]
- Kuć, J.; Szarejko, K.D.; Aleksandrowicz, K.; Gołębiewska, M. The role of soft tissue mobilization in reducing orofacial and general complaints in a patient with kimmerle anomaly and temporomandibular joint disorder: A case report. Cranio 2019, 1–14. [Google Scholar] [CrossRef]
- Bartsch, T. Increased responses in trigeminocervical nociceptive neurons to cervical input after stimulation of the dura mater. Brain 2003, 126, 1801–1813. [Google Scholar] [CrossRef]
- Wall, P.; Melzack, R. Textbook of Pain, 2nd ed.; Churchill Livingstone: Edinburgh, UK, 1989. [Google Scholar]
- Herpich, C.M.; Junior, E.C.L.; Gomes, C.A.F.D.P.; Glória, I.P.D.S.; Amaral, A.P.; Amaral, M.D.F.D.R.S.; Politti, F.; Biasotto-Gonzalez, D.A. Immediate and short-term effects of phototherapy on pain, muscle activity, and joint mobility in women with temporomandibular disorder: A randomized, double-blind, placebo-controlled, clinical trial. Disabil. Rehabil. 2017, 40, 2318–2324. [Google Scholar] [CrossRef] [PubMed]
- Barão, V.A.R.; Gallo, A.K.G.; Zuim, P.R.J.; Garcia, A.R.; Assunção, W.G. Effect of occlusal splint treatment on the temperature of different muscles in patients with TMD. J. Prosthodont. Res. 2011, 55, 19–23. [Google Scholar] [CrossRef] [PubMed]
- Ariji, E.; Katsumata, A.; Hiraiwa, Y.; Izumi, M.; Sakuma, S.; Shimizu, M.; Kurita, K.; Ariji, E. Masseter muscle sonographic features as indices for evaluating efficacy of massage treatment. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2010, 110, 517–526. [Google Scholar] [CrossRef] [PubMed]
- Sefton, J.M.; Yarar, C.; Berry, J.W.; Pascoe, D.D. Therapeutic massage of the neck and shoulders produces changes in peripheral blood flow when assessed with dynamic infrared thermography. J. Altern. Complement. Med. 2010, 16, 723–732. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.Y.; Wang, X.G.; Dong, J.; Zhang, J.F.; Lü, Y.L. Effect of occlusal splints for the management of patients with myofascial pain: A randomized, controlled, double-blind study. Chin. Med. J. 2013, 126, 2270–2275. [Google Scholar]
- Gomes, C.A.F.D.P.; Politti, F.; Andrade, D.V.; De Sousa, D.F.M.; Herpich, C.M.; Dibai-Filho, A.V.; Gonzalez, T.D.O.; Gonzalez, D.A.B. Effects of massage therapy and occlusal splint therapy on mandibular range of motion in individuals with temporomandibular disorder: A randomized clinical trial. J. Manip. Physiol. Ther. 2014, 37, 164–169. [Google Scholar] [CrossRef]
- Cuccia, A.M.; Caradonna, C.; Annunziata, V.; Caradonna, D. Osteopathic manual therapy versus conventional conservative therapy in the treatment of temporomandibular disorders: A randomized controlled trial. J. Bodyw. Mov. Ther. 2010, 14, 179–184. [Google Scholar] [CrossRef]
- Ferguson, L.; Scheman, J. Patient global impression of change scores within the context of a chronic pain rehabilitation program. J. Pain 2009, 10, S73. [Google Scholar] [CrossRef]
- Gagnon, C.M.; Scholten, P.; Atchison, J. Multidimensional Patient Impression of Change Following Interdisciplinary Pain Management. Pain Pract. 2018, 18, 997–1010. [Google Scholar] [CrossRef]
- Geisser, M.E.; Clauw, D.J.; Strand, V.; Gendreau, M.R.; Palmer, R.; Williams, D.A. Contributions of change in clinical status parameters to patient global impression of change (PGIC) scores among persons with fibromyalgia treated with milnacipran. Pain 2010, 149, 373–378. [Google Scholar] [CrossRef]
| Inclusion Criteria | Exclusion Criteria |
|---|---|
|
|
| Variables | CG (n = 8) | EG (n = 8) | p-Value |
|---|---|---|---|
| Age (years) | 29.8 ± 14.6 | 30.0 ± 11.6 | 0.967 |
| Gender (%male/%female) | 0/100 | 37.5/62.5 | N.A. |
| BMI | 20.5 ± 2.2 | 23.0 ± 2.0 | 0.031 * |
| VAS a | 4.8 ± 2.3 | 3.6 ± 1.9 | 0.238 |
| Helkimo Scale | 9.0 ± 1.7 | 9.8 ± 2.3 | 0.407 |
| Algometry b | |||
| Right Masseter | 4.7 ± 0.8 | 4.9 ± 0.8 | 0.741 |
| Left Masseter | 4.5 ± 0.9 | 4.8 ± 0.7 | 0.452 |
| Right Temporal | 4.5 ± 0.8 | 4.6 ± 0.6 | 0.859 |
| Left Temporal | 4.9 ± 1.0 | 4.9 ± 0.9 | 0.837 |
| Right Sternocleidomastoid | 3.8 ± 0.8 | 4.0 ± 0.7 | 0.640 |
| Left Sternocleidomastoid | 4.0 ± 0.8 | 4.2 ± 0.8 | 0.657 |
| Pain characteristics | |||
| When it started (years) | 6.8 ± 4.0 | 4.5 ± 2.4 | 0.202 |
| Intensity (VAS last 2 weeks) | 5.3 ± 1.8 | 6.0 ± 1.9 | 0.294 |
| Frequency (%monthly/%weekly/%daily) | 12.5/50/37.5 | 0/75/25 | 0.270 |
| Severity (%mild/%moderate/%severe) c | 12.5/87.5/0 | 25/50/25 | 0.176 |
| Pain Location (%never/%sometimes/%frequently) | |||
| Head | 50/25/25 | 0/75/25 | 0.020 * |
| Jaw | 0/37.5/62.5 | 0/50/50 | 0.317 |
| Cervical | 0/62.5/37.5 | 12.5/37.5/50 | 0.319 |
| Shoulders | 75/25/0 | 62.5/25/12.5 | 0.582 |
| Variables | Groups | T0 | T1 | T2 | Mean Difference (95%CI); Effect Size (d) | ||||
|---|---|---|---|---|---|---|---|---|---|
| Within-Group Differences | Between-Groups Differences | ||||||||
| T0–T1 | T0–T2 | T1–T2 | T1 | T2 | |||||
| VAS a | CG | 4.8 ± 2.3 | 5.0 ± 2.1 | 4.0 ± 2.3 | −0.2 (−0.6 to 2.1) | 0.8 (−1.1 to 0.6) | 1.0 (−0.3 to 2.3) | −4.5 (−6.3 to −2.7); d = 0.8 | −2.8 (−4.7 to −0.8); d = 0.8 |
| EG | 3.6 ± 1.9 | 0.5 ± 0.5 * | 1.3 ± 0.9 *,† | 3.1 (1.7 to 4.4); d = 0.3 | 2.4 (1.1 to 3.7); d = 0.4 | −0,8 (−1.3 to 0.2); d = 0.5 | |||
| Helkimo Index | CG | 9.0 ± 1.7 | 9.6 ± 1.8 | 9.1 ± 1.9 | −0.6 (−1.5 to 0.3) | −0.1 (−0.9 to 0.7) | 0.5 (−0.1 to 0.9) | −4.2 (−6.9 to −1.6); d = 1.2 | −3.2 (−5.6 to −0.9); d = 1.1 |
| EG | 9.8 ± 2.3 | 5.4 ± 2.9 * | 5.9 ± 2.4 * | 4.4 (2.7 to 6.0); d = 0.4 | 3.9 (1.5 to 6.3); d = 0.4 | −0.5 (−2.1 to 1.1) | |||
| Algometry b | |||||||||
| Right Masseter | CG | 4.7 ± 0.8 | 4.8 ± 0.8 | 4.8 ± 0.8 | −0.1 (−0.3 to 0.2) | −0.1 (−0.4 to 0.2) | −0.0 (−0.2 to 0.1) | 1.1 (0.3 to 1.9); d = 0.4 | 1.2 (0.4 to 2.1); d = 0.4 |
| EG | 4.9 ± 0.8 | 5.9 ± 0.7 * | 6.0 ± 0.9 * | −1.0 (−1.4 to −0.6); d = 0.4 | −1.1 (−1.7 to −0.7) d = 0.4 | −0.1 (−0.4 to −0.1) | |||
| Left Masseter | CG | 4.5 ± 0.9 | 4.4 ± 0.7 | 4.4 ± 0.6 | 0.1 (−0.2 to 0.5) | 0.1 (−0.3 to 0.6) | − 0.0 (−0.2 to 0.1) | 1.9 (1.1 to 2.6); d = 0.4 | 1.5 (0.8 to 2.2); d = 0.3 |
| EG | 4.8 ± 0.7 | 6.3 ± 0.8 * | 5.9 ± 0.7 * | −1.5 (−1.8 to −1.0); d = 0.4 | −1.1 (−1.6 to −0.6); d = 0.4 | 0.4 (−0.2 to 0.9) | |||
| Right Temporal | CG | 4.5 ± 0.8 | 4.8 ± 0.9 | 4.8 ± 0.9 | −0.3 (−0.7 to 0.3) | −0.3 (−0.7 to 0.1) | −0.0 (−0.4 to 0.2) | 1.7 (0.7 to 2.9); d = 0.5 | 1.4 (0.5 to 2.3); d = 0.4) |
| EG | 4.6 ± 0.6 | 6.5 ± 1.0 * | 6.2 ± 0.8 *,† | −1.9 (−2.7 to −1.2); d = 0.3 | −1.6 (−2.3 to −0.9): d = 0.3 | 0.3 (−0.0 to 0.7); d = 0.5 | |||
| Left Temporal | CG | 4.9 ± 1.0 | 4.4 ± 0.8 | 4.7 ± 0.9 | 0.4 (−0.1 to 1.0) | 0.2 (−0.2 to 0.5) | −0.3 (−0.7 to 0.2) | 2.5 (1.4 to 3.5); d = 0.5 | 1.6 (0.6 to 2.5); d = 0.4 |
| EG | 4.9 ± 0.9 | 6.9 ± 1.1 * | 6.3 ± 0.8 *,† | −2.0 (−2.8 to −1.2); d = 0.4 | −1.4 (−1.8 to 0.9); d = 0.4 | 0.6 (0.1 to 1.1); d = 0.5 | |||
| Right SCM | CG | 3.8 ± 0.8 | 3.9 ± 0.6 | 3.9 ± 0.5 | −0.1 (−0.5 to 0.2) | −0.1 (0.5 to 0.4) | 0.0 (−0.2 to 0.4) | 1.6 (0.7 to 2.3); d = 0.4 | 1.5 (0.6 to 2.3); d = 0.4 |
| EG | 4.0 ± 0.7 | 5.5 ± 0.9 * | 5.4 ± 1.0 * | −1.5 (−1.9 to −1.1); d = 0.4 | −1.4 (−1.8 to −0.8); d = 0.4 | 0.1 (−0.4 to 0.6) | |||
| Left SCM | CG | 4.0 ± 0.8 | 4.0 ± 0.6 | 3.9 ± 0.5 | −0.0 (−0.5 to 0.4) | 0.1 (−0.3 to 0.4) | 0.1 (−0.1 to 0.3) | 1.5 (0.6 to 2.2); d = 0.4 | 1.3 (0.4 to 2.2); d = 0.4 |
| EG | 4.2 ± 0.8 | 5.5 ± 0.9 * | 5.2 ± 1.0 * | −1.3 (−1.8 to −0.8); d = 0.4 | −1.0 (−1.8 to −0.3); d = 0.4 | 0.3 (−0.4 to 0.9) | |||
| CG (n = 8) | EG (n = 8) | |||||
|---|---|---|---|---|---|---|
| T0 | T1 | T2 | T0 | T1 | T2 | |
| Helkimo Index, frequency, (%) | ||||||
| Absence (0 points) | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) |
| Mild (1–4 points) | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) | 4 (50%) | 3 (37.5%) |
| Moderate (5–9 points) | 6 (75%) | 5 (62.5%) | 5 (62.5%) | 4 (50%) | 4 (50% | 5 (62.5%) |
| Severe (10–25 points) | 2 (25%) | 3 (37.5%) | 3 (37.5%) | 4 (50%) | 0 (0%) | 0 (0%) |
| PGICS, frequency, (%) | ||||||
| 1 = “very much improved” | 0 (0%) | 2 (25%) | ||||
| 2 = “much improved” | 0 (0%) | 4 (50%) | ||||
| 3 = “minimally improved” | 2 (25%) | 0 (0%) | ||||
| 4 = “no change” | 2 (25%) | 1 (12.5%) | ||||
| 5 = “minimally worse” | 4 (50%) | 1 (12.5%) | ||||
| 6 = “much worse” | 0 (0%) | 0 (0%) | ||||
| 7 = “very much worse” | 0 (0%) | 0 (0%) | ||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Espí-López, G.V.; Arnal-Gómez, A.; Cuerda del Pino, A.; Benavent-Corai, J.; Serra-Añó, P.; Inglés, M. Effect of Manual Therapy and Splint Therapy in People with Temporomandibular Disorders: A Preliminary Study. J. Clin. Med. 2020, 9, 2411. https://doi.org/10.3390/jcm9082411
Espí-López GV, Arnal-Gómez A, Cuerda del Pino A, Benavent-Corai J, Serra-Añó P, Inglés M. Effect of Manual Therapy and Splint Therapy in People with Temporomandibular Disorders: A Preliminary Study. Journal of Clinical Medicine. 2020; 9(8):2411. https://doi.org/10.3390/jcm9082411
Chicago/Turabian StyleEspí-López, Gemma Victoria, Anna Arnal-Gómez, Alba Cuerda del Pino, José Benavent-Corai, Pilar Serra-Añó, and Marta Inglés. 2020. "Effect of Manual Therapy and Splint Therapy in People with Temporomandibular Disorders: A Preliminary Study" Journal of Clinical Medicine 9, no. 8: 2411. https://doi.org/10.3390/jcm9082411
APA StyleEspí-López, G. V., Arnal-Gómez, A., Cuerda del Pino, A., Benavent-Corai, J., Serra-Añó, P., & Inglés, M. (2020). Effect of Manual Therapy and Splint Therapy in People with Temporomandibular Disorders: A Preliminary Study. Journal of Clinical Medicine, 9(8), 2411. https://doi.org/10.3390/jcm9082411








