Sjögren’s Syndrome Minor Salivary Gland CD4+ Memory T Cells Associate with Glandular Disease Features and Have a Germinal Center T Follicular Helper Transcriptional Profile
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Isolation of SG Memory CD4+ T Cells and cDNA Preparation
2.3. Flow Cytometry Analysis
2.4. Microarray Data Analysis
2.5. Salivary Gland Imaging
2.6. Other Statistical Analyses
3. Results
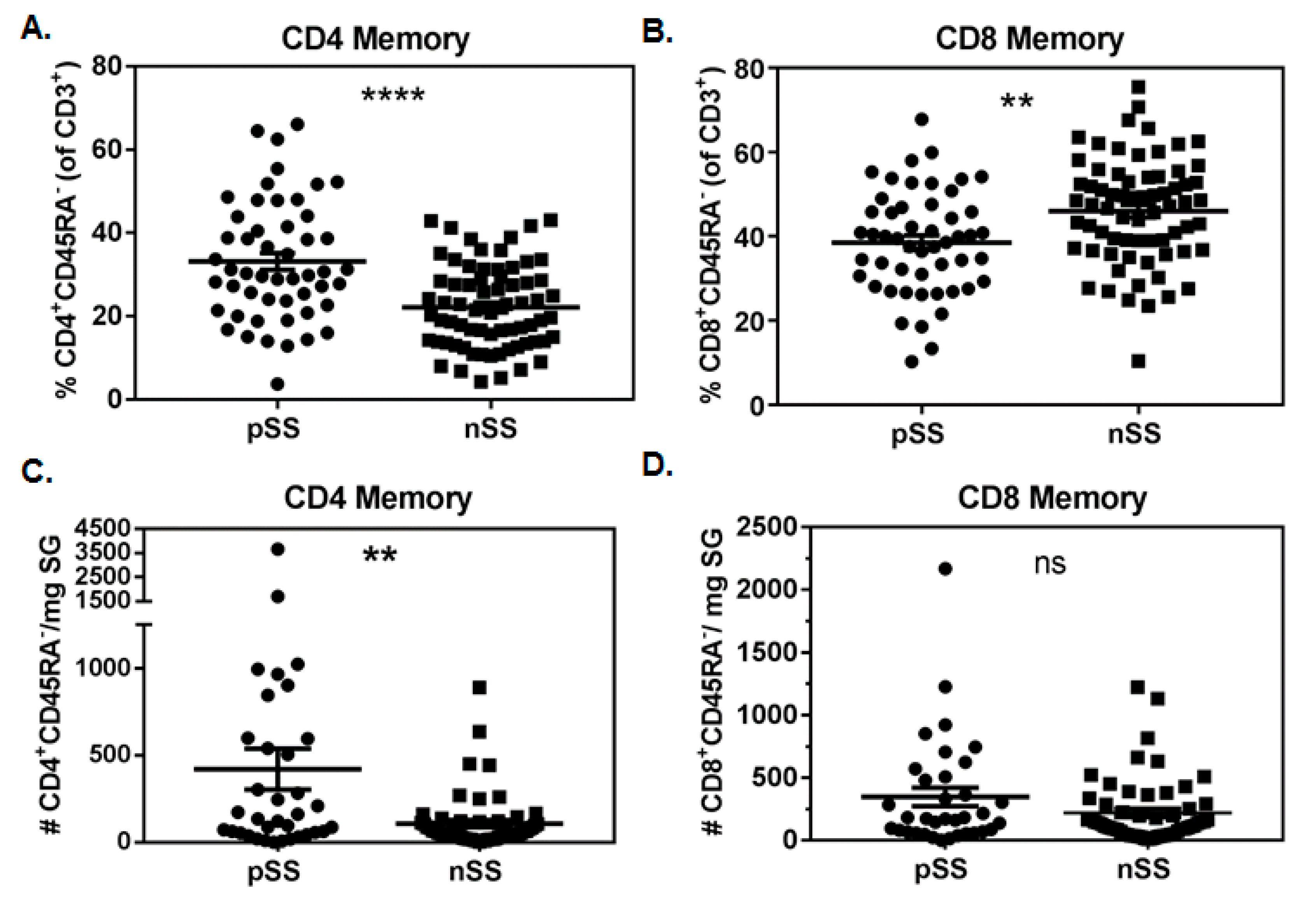
3.1. Memory CD4+ T Cells Are Increased in pSS SG
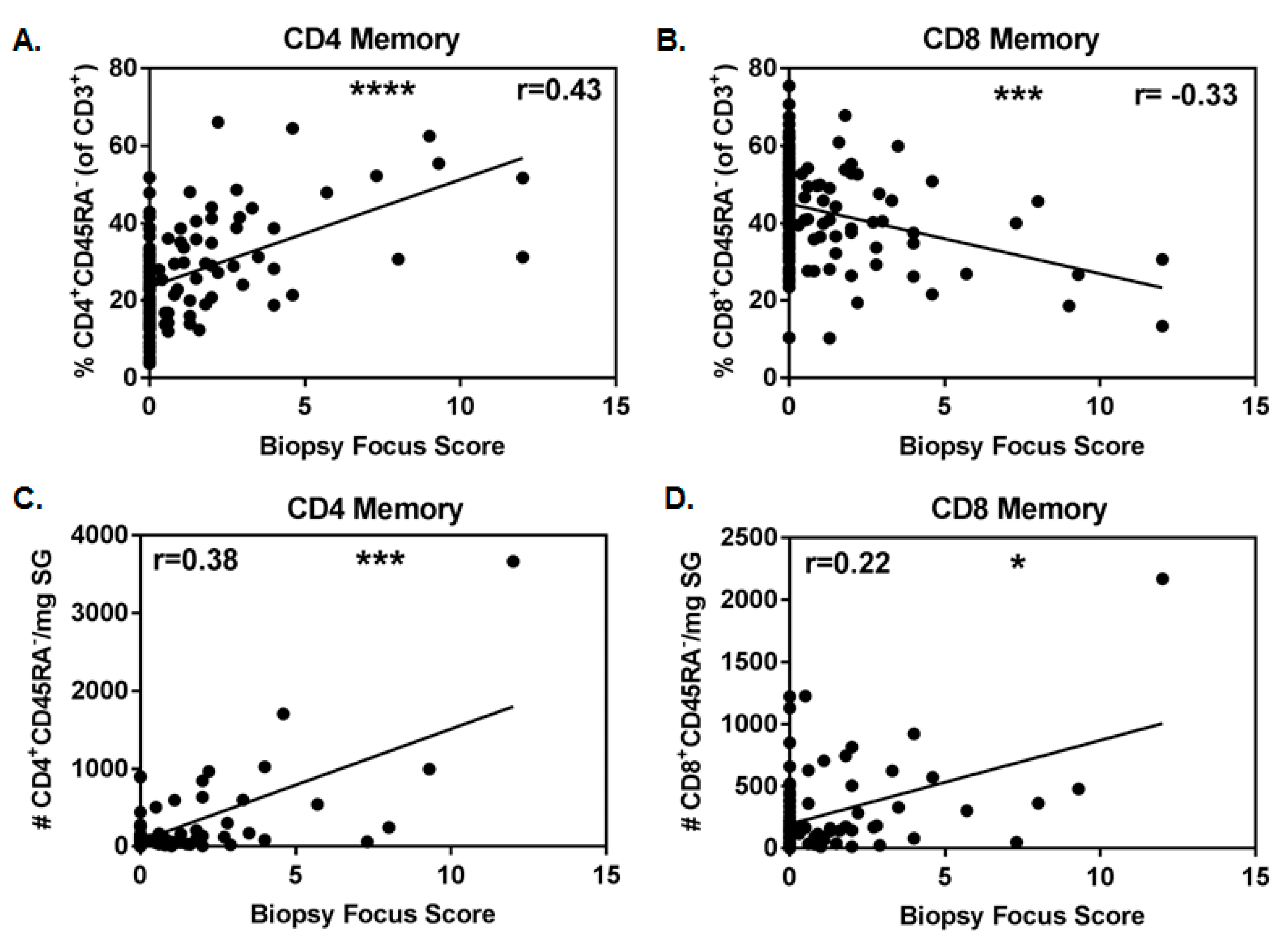
3.2. Numbers of SG CD4+ Antigen-Experienced T Cells Associate with Biopsy Focus Scores
3.3. Absolute Numbers of SG Antigen-Experienced T Cells Associate with the Extent of Salivary Gland Fibrosis
3.4. Proportions of SG CD4+ Memory T Cells Correlate with Clinical Features of SS
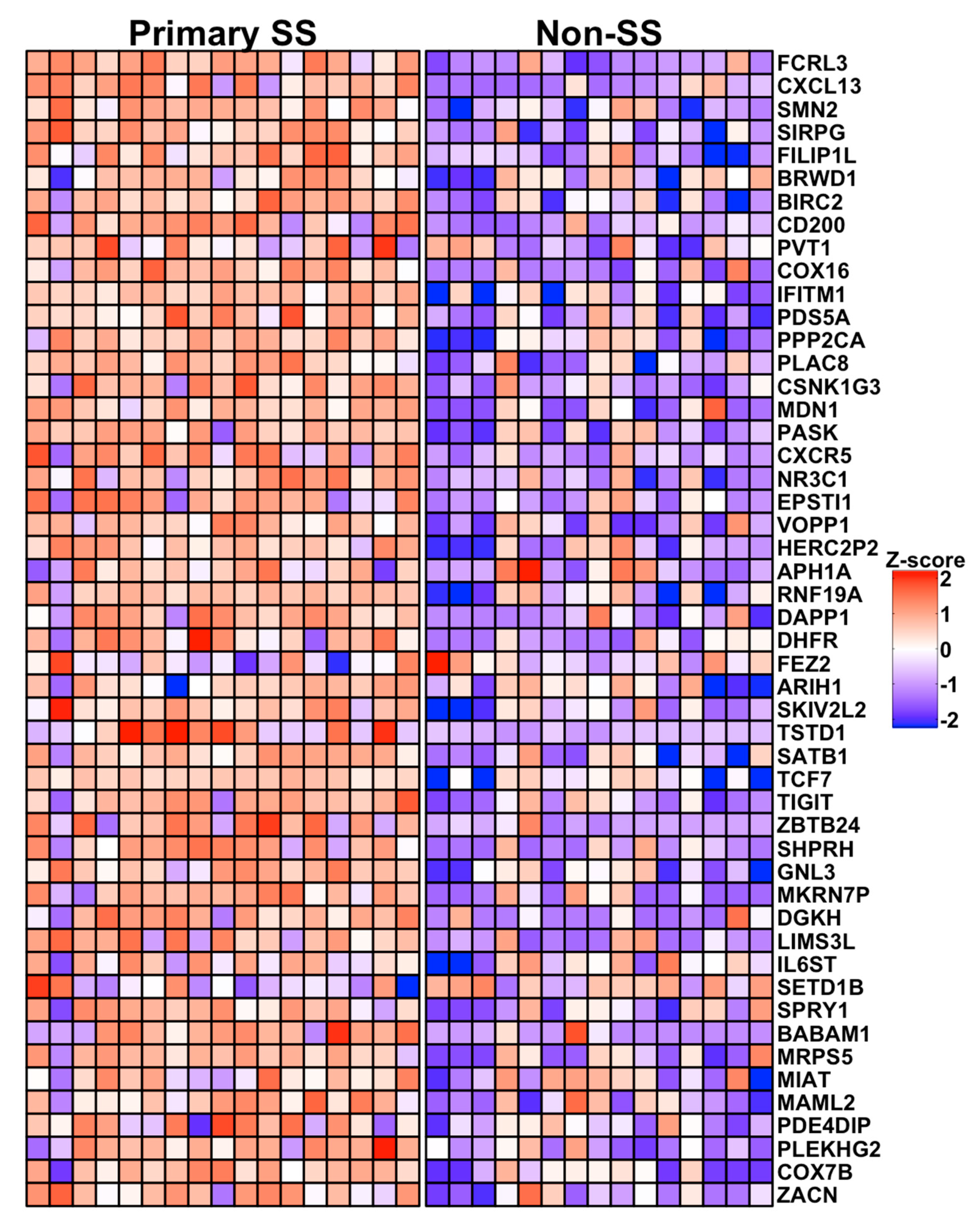
3.5. Characterization of the pSS CD4+CD45RA− SG T Cell Transcriptome
3.6. The Transcriptomes of pSS SG Memory CD4+ T Cells Include DEG for Homing, Interaction and Survival Functions
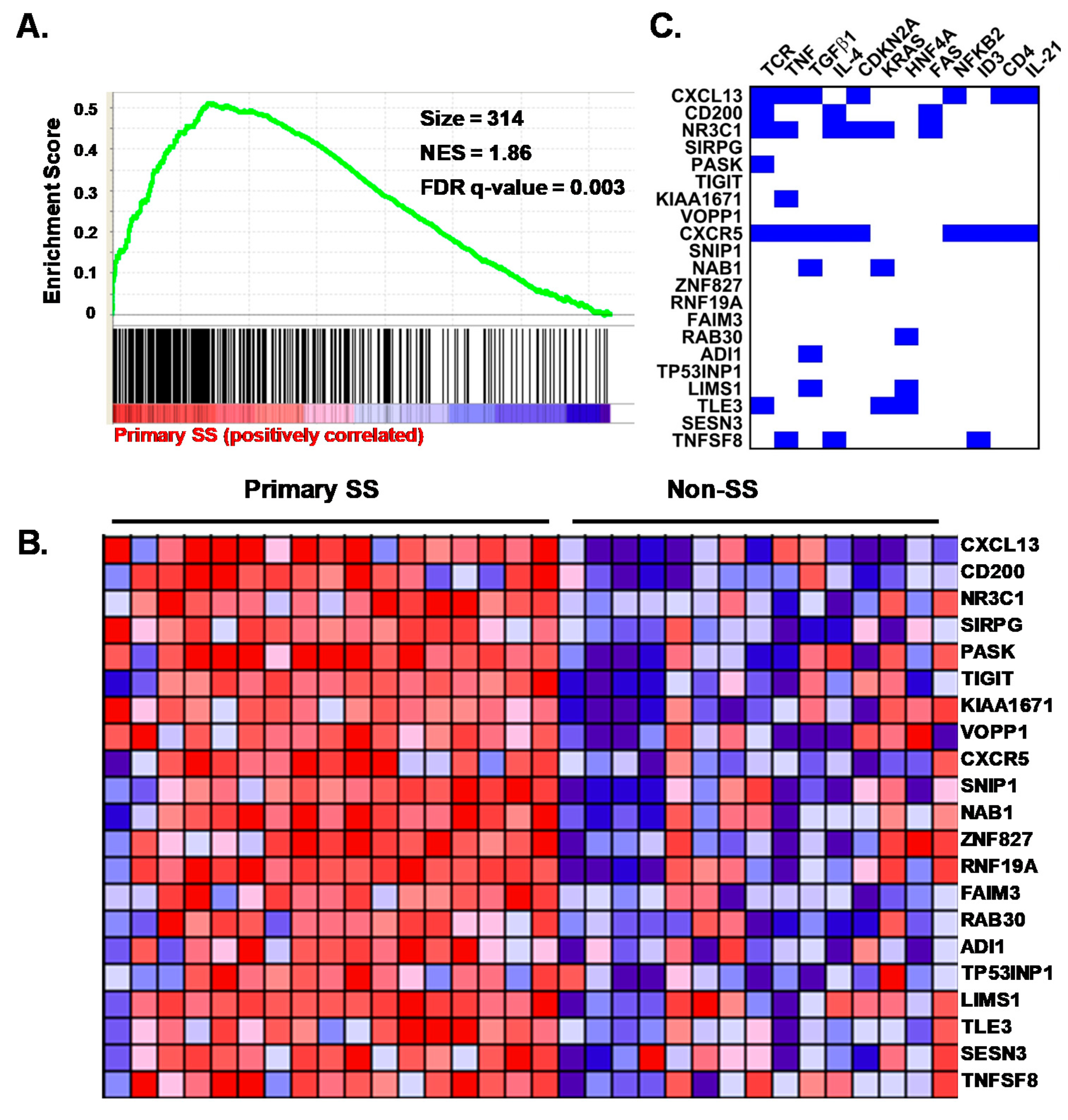
3.7. pSS SG Memory CD4+ T Cells Display a Germinal Center Tfh Cell Gene Signature
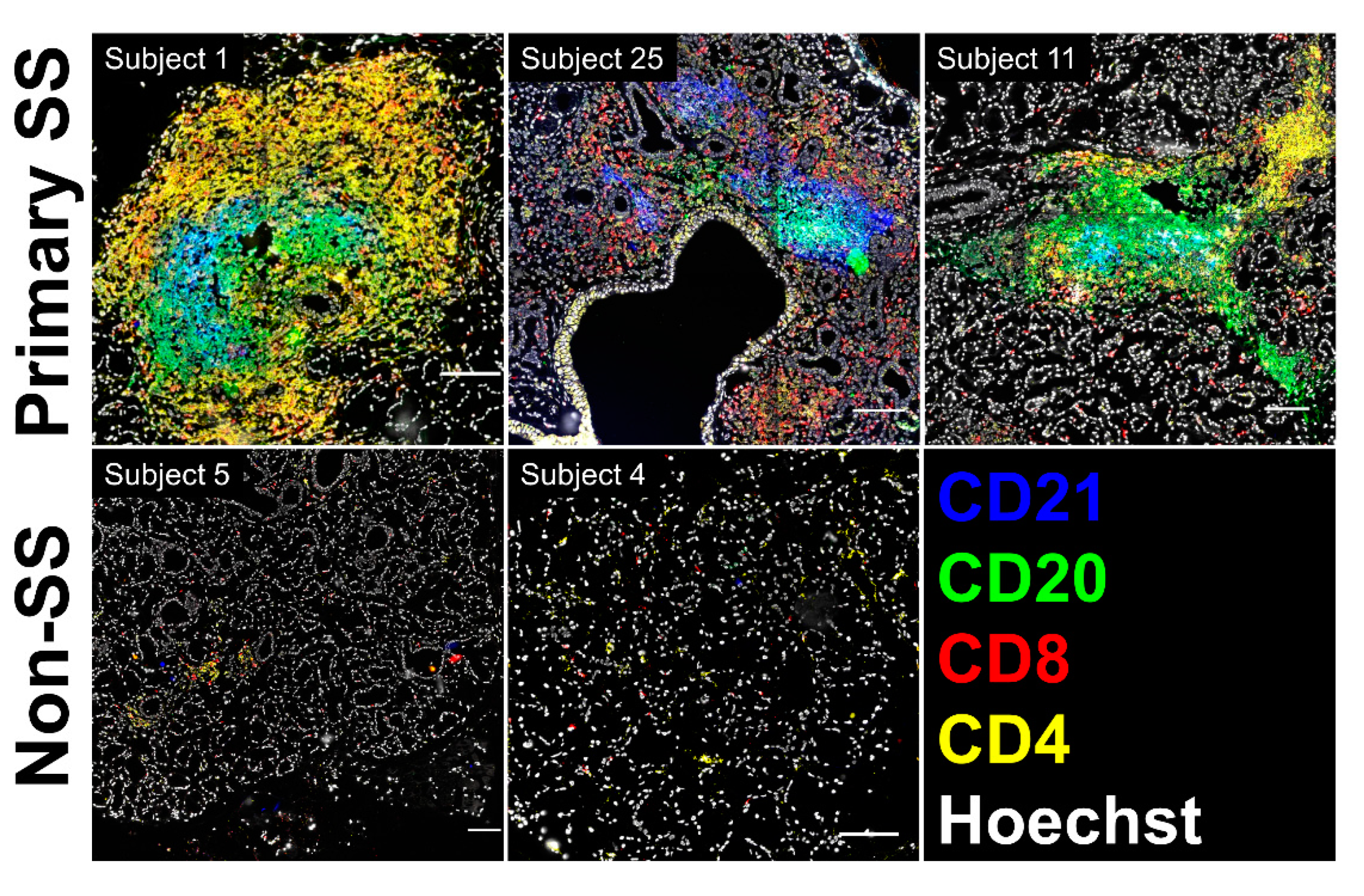
3.8. SG biopsies from pSS Subjects Showing SG Memory T Cell Tfh Gene Signatures Display Atypical Ectopic Lymphoid Structures
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Tapinos, N.I.; Polihronis, M.; Tzioufas, A.G.; Skopouli, F.N. Immunopathology of Sjogren’s syndrome. Ann. Med Interne (Paris) 1998, 149, 17–24. [Google Scholar] [PubMed]
- Singh, N.; Cohen, P.L. The T cell in Sjogren’s syndrome: Force majeure, not spectateur. J. Autoimmun. 2012, 39, 229–233. [Google Scholar] [CrossRef] [PubMed]
- Joachims, M.L.; Leehan, K.M.; Lawrence, C.; Pelikan, R.C.; Moore, J.S.; Pan, Z.; Rasmussen, A.; Radfar, L.; Lewis, D.M.; Grundahl, K.M.; et al. Single-cell analysis of glandular T cell receptors in Sjögren’s syndrome. JCI Insight 2016, 1, 85609. [Google Scholar] [CrossRef]
- Lessard, C.J.; Li, H.; Adrianto, I.; Ice, J.A.; Rasmussen, A.; Grundahl, K.M.; Kelly, J.; Dozmorov, M.; Miceli-Richard, C.; Bowman, S.; et al. Variants at multiple loci implicated in both innate and adaptive immune responses are associated with Sjögren’s syndrome. Nat. Genet. 2013, 45, 1284–1292. [Google Scholar] [CrossRef] [PubMed]
- Christodoulou, M.I.; Kapsogeorgou, E.K.; Moutsopoulos, H.M. Characteristics of the minor salivary gland infiltrates in Sjögren’s syndrome. J. Autoimmun. 2010, 34, 400–407. [Google Scholar] [CrossRef] [PubMed]
- Kapsogeorgou, E.K.; Christodoulou, M.I.; Panagiotakos, D.B.; Paikos, S.; Tassidou, A.; Tzioufas, A.G.; Moutsopoulos, H.M. Minor Salivary Gland Inflammatory Lesions in Sjögren Syndrome: Do They Evolve? J. Rheumatol. 2013, 40, 1566–1571. [Google Scholar] [CrossRef]
- Jonsson, M.V.; Skarstein, K.; Jonsson, R.; Brun, J.G. Serological implications of germinal center-like structures in primary Sjögren’s syndrome. J. Rheumatol. 2007, 34, 2044–2049. [Google Scholar]
- Theander, E.; Vasaitis, L.; Baecklund, E.; Nordmark, G.; Warfvinge, G.; Liedholm, R.; Brokstad, K.A.; Jonsson, R.; Jonsson, M.V. Lymphoid organisation in labial salivary gland biopsies is a possible predictor for the development of malignant lymphoma in primary Sjögren’s syndrome. Ann. Rheum. Dis. 2011, 70, 1363–1368. [Google Scholar] [CrossRef]
- Crotty, S. Follicular Helper CD4 T Cells (TFH). Annu. Rev. Immunol. 2011, 29, 621–663. [Google Scholar] [CrossRef]
- Tengner, P.; Halse, A.K.; Haga, H.J.; Jonsson, R.; Wahren-Herlenius, M. Detection of anti-Ro/SSA and anti-La/SSB autoantibody-producing cells in salivary glands from patients with Sjogren’s syndrome. Arthritis Rheum. 1998, 41, 2238–2248. [Google Scholar] [CrossRef]
- Mingueneau, M.; Boudaoud, S.; Haskett, S.; Reynolds, T.L.; Nocturne, G.; Norton, E.; Zhang, X.; Constant, M.; Park, D.; Wang, W.; et al. Cytometry by time-of-flight immunophenotyping identifies a blood Sjögren’s signature correlating with disease activity and glandular inflammation. J. Allergy Clin. Immunol. 2016, 137, 1809–1821. [Google Scholar] [CrossRef] [PubMed]
- Brookes, S.M.; Cohen, S.B.A.; Price, E.J.; Webb, L.M.C.; Feldmann, M.; Maini, R.N.; Venables, P.J.W. T cell clones from a Sjögren’s syndrome salivary gland biopsy produce high levels of IL-10. Clin. Exp. Immunol. 1996, 103, 268–272. [Google Scholar] [CrossRef] [PubMed]
- Szabó, K.; Papp, G.; Dezso, B.; Zeher, M. The Histopathology of Labial Salivary Glands in Primary Sjögren’s Syndrome: Focusing on Follicular Helper T Cells in the Inflammatory Infiltrates. Mediat. Inflamm. 2014, 2014, 1–11. [Google Scholar] [CrossRef]
- Sakai, A.; Sugawara, Y.; Kuroishi, T.; Sasano, T.; Sugawara, S. Identification of IL-18 and Th17 cells in salivary glands of patients with Sjögren’s syndrome, and amplification of IL-17-mediated secretion of inflammatory cytokines from salivary gland cells by IL-18. J. Immunol. 2008, 181, 2898–2906. [Google Scholar] [CrossRef]
- Christodoulou, M.I.; Kapsogeorgou, E.K.; Moutsopoulos, N.M.; Moutsopoulos, H.M. Foxp3+ T-Regulatory Cells in Sjögren’s Syndrome. Am. J. Pathol. 2008, 173, 1389–1396. [Google Scholar] [CrossRef]
- Sarigul, M.; Yazisiz, V.; Başsorgun, C.; Ulker, M.; Avci, A.; Erbasan, F.; Gelen, T.; Gorczynski, R.; Terzioğlu, E.; Ba?sorgun, C.; et al. The numbers of Foxp3 + Treg cells are positively correlated with higher grade of infiltration at the salivary glands in primary Sjögren’s syndrome. Lupus 2009, 19, 138–145. [Google Scholar] [CrossRef]
- Maehara, T.; Moriyama, M.; Hayashida, J.; Tanaka, A.; Shinozaki, S.; Kubo, Y.; Matsumura, K.; Nakamura, S. Selective localization of T helper subsets in labial salivary glands from primary Sjögren’s syndrome patients. Clin. Exp. Immunol. 2012, 169, 89–99. [Google Scholar] [CrossRef]
- Bolstad, A.I.; Eiken, H.G.; Rosenlund, B.; Alarcón-Riquelme, M.E.; Jonsson, R. Increased salivary gland tissue expression of Fas, Fas ligand, cytotoxic T lymphocyte–associated antigen 4, and programmed cell death 1 in primary Sjögren’s syndrome. Arthritis Rheum. 2003, 48, 174–185. [Google Scholar] [CrossRef]
- Hjelmervik, T.O.R.; Petersen, K.; Jonassen, I.; Jonsson, R.; Bolstad, A.I. Gene expression profiling of minor salivary glands clearly distinguishes primary Sjögren’s syndrome patients from healthy control subjects. Arthritis Rheum. 2005, 52, 1534–1544. [Google Scholar] [CrossRef]
- Gottenberg, J.-E.; Cagnard, N.; Lucchesi, C.; Letourneur, F.; Mistou, S.; Lazure, T.; Jacques, S.; Ba, N.; Ittah, M.; Lepajolec, C.; et al. Activation of IFN pathways and plasmacytoid dendritic cell recruitment in target organs of primary Sjogren’s syndrome. Proc. Natl. Acad. Sci. USA 2006, 103, 2770–2775. [Google Scholar] [CrossRef]
- Wakamatsu, E.; Nakamura, Y.; Matsumoto, I.; Goto, D.; Ito, S.; Tsutsumi, A.; Sumida, T. DNA microarray analysis of labial salivary glands of patients with Sjögren’s syndrome. Ann. Rheum. Dis. 2007, 66, 844–845. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Xu, P.; Wang, X.; Zhang, Z.; Zhao, W.; Li, Z.; Yang, G.; Liu, P. Identification of differentially expressed genes in primary Sjögren’s syndrome. J. Cell. Biochem. 2019, 120, 17368–17377. [Google Scholar] [CrossRef] [PubMed]
- Tandon, M.; Perez, P.; Burbelo, P.D.; Calkins, C.; Alevizos, I. Laser microdissection coupled with RNA-seq reveal cell-type and disease-specific markers in the salivary gland of Sjögren’s syndrome patients. Clin. Exp. Rheumatol. 2017, 35, 777–785. [Google Scholar]
- Rasmussen, A.; Ice, J.A.; Li, H.; Grundahl, K.; Kelly, J.; Radfar, L.; Stone, N.U.; Hefner, K.S.; Anaya, J.-M.; Rohrer, M.; et al. Comparison of the American-European Consensus Group Sjogren’s syndrome classification criteria to newly proposed American College of Rheumatology criteria in a large, carefully characterised sicca cohort. Ann. Rheum. Dis. 2013, 73, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Vitali, C.; Bombardieri, S.; Moutsopoulos, H.M.; Balestrieri, G.; Bencivelli, W.; Bernstein, R.M.; Bjerrum, K.B.; Braga, S.; Coll, J.; De Vita, S.; et al. Preliminary criteria for the classification of Sjögren’s syndrome. Results of a prospective concerted action supported by the European community. Arthritis Rheum. 1993, 36, 340–347. [Google Scholar] [CrossRef]
- Vitali, C.; Bombardieri, S.; Jonsson, R.; Moutsopoulos, H.M.; Alexander, E.L.; Carsons, S.E.; Daniels, T.E.; Fox, P.C.; Fox, R.I.; Kassan, S.S.; et al. Classification criteria for Sjogren’s syndrome: A revised version of the European criteria proposed by the American-European Consensus Group. Ann. Rheum. Dis. 2002, 61, 554–558. [Google Scholar] [CrossRef]
- Leehan, K.M.; Pezant, N.P.; Rasmussen, A.; Grundahl, K.; Moore, J.S.; Radfar, L.; Lewis, D.M.; Stone, N.U.; Lessard, C.J.; Rhodus, N.L.; et al. Minor salivary gland fibrosis in Sjögren’s syndrome is elevated, associated with focus score and not solely a consequence of aging. Clin. Exp. Rheumatol. 2017, 36, 80–88. [Google Scholar]
- Maier-Moore, J.S.; Koelsch, K.A.; Smith, K.; Lessard, C.J.; Radfar, L.; Lewis, D.; Kurien, B.T.; Wolska, N.; Deshmukh, U.; Rasmussen, A.; et al. Antibody-secreting cell specificity in labial salivary glands reflects the clinical presentation and serology in patients with Sjögren’s syndrome. Arthritis Rheumatol. 2014, 66, 3445–3456. [Google Scholar] [CrossRef]
- Kauffmann, A.; Gentleman, R.; Huber, W. Arrayqualitymetrics—A bioconductor package for quality assessment of microarray data. Bioinformatics 2009, 25, 415–416. [Google Scholar] [CrossRef]
- Smyth, G.K. Linear Models and Empirical Bayes Methods for Assessing Differential Expression in Microarray Experiments. Stat. Appl. Genet. Mol. Boil. 2004, 3, 1–25. [Google Scholar] [CrossRef]
- Subramanian, A.; Tamayo, P.; Mootha, V.K.; Mukherjee, S.; Ebert, B.L.; Gillette, M.A.; Paulovich, A.; Pomeroy, S.L.; Golub, T.R.; Lander, E.S.; et al. Gene set enrichment analysis: A knowledge-based approach for interpreting genome-wide expression profiles. Proc. Natl. Acad. Sci. USA 2005, 102, 15545–15550. [Google Scholar] [CrossRef] [PubMed]
- R. Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2013. [Google Scholar]
- Greenspan, J.; Daniels, T.; Talal, N.; Sylvester, R. The histopathology of Sjögren’s syndrome in labial salivary gland biopsies. Oral Surgery. Oral Med. Oral Pathol. 1974, 37, 217–229. [Google Scholar] [CrossRef]
- Bikker, A.; Van Woerkom, J.M.; Kruize, A.A.; Van Der Wurff-Jacobs, K.M.G.; Bijlsma, J.W.J.; Lafeber, F.P.J.G.; Van Roon, J.A.G. Clinical efficacy of leflunomide in primary Sjogren’s syndrome is associated with regulation of T-cell activity and upregulation of IL-7 receptor expression. Ann. Rheum. Dis. 2012, 71, 1934–1941. [Google Scholar] [CrossRef] [PubMed]
- Chtanova, T.; Tangye, S.G.; Newton, R.; Frank, N.; Hodge, M.R.; Rolph, M.S.; Mackay, C.R. T follicular helper cells express a distinctive transcriptional profile, reflecting their role as non-Th1/Th2 effector cells that provide help for B cells. J. Immunol. 2004, 173, 68–78. [Google Scholar] [CrossRef]
- Vella, L.A.; Buggert, M.; Manne, S.; Herati, R.S.; Sayin, I.; Kuri-Cervantes, L.; Brody, I.B.; O’Boyle, K.C.; Kaprielian, H.; Giles, J.R.; et al. T follicular helper cells in human efferent lymph retain lymphoid characteristics. J. Clin. Investig. 2019, 129, 3185–3200. [Google Scholar] [CrossRef]
- Godefroy, E.; Zhong, H.; Pham, P.; Friedman, D.; Yazdanbakhsh, K. TIGIT-positive circulating follicular helper T cells display robust B-cell help functions: Potential role in sickle cell alloimmunization. Haematologica 2015, 100, 1415–1425. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, Y.; Yang, J.; Zhang, L.; Sun, L.; Pan, H.-F.; Hirankarn, N.; Ying, D.; Zeng, S.; Lee, T.L.; et al. Three SNPs in chromosome 11q23.3 are independently associated with systemic lupus erythematosus in Asians. Hum. Mol. Genet. 2013, 23, 524–533. [Google Scholar] [CrossRef]
- Tan, W.; Sunahori, K.; Zhao, J.; Deng, Y.; Kaufman, K.M.; Kelly, J.A.; Langefeld, C.D.; Williams, A.H.; Comeau, M.E.; Ziegler, J.T.; et al. Association of PPP2CA polymorphisms with systemic lupus erythematosus susceptibility in multiple ethnic groups. Arthritis Rheum. 2011, 63, 2755–2763. [Google Scholar] [CrossRef]
- Bentham, J.; Morris, D.L.; Graham, D.S.C.; Pinder, C.L.; Tombleson, P.; Behrens, T.W.; Martin, J.; Fairfax, B.P.; Knight, J.C.; Chen, L.; et al. Genetic association analyses implicate aberrant regulation of innate and adaptive immunity genes in the pathogenesis of systemic lupus erythematosus. Nat. Genet. 2015, 47, 1457–1464. [Google Scholar] [CrossRef]
- Bajpai, U.D.; Swainson, L.A.; Mold, J.E.; Graf, J.D.; Imboden, J.B.; McCune, J.M. A functional variant in FCRL3 is associated with higher Fc receptor-like 3 expression on T cell subsets and rheumatoid arthritis disease activity. Arthritis Rheum. 2012, 64, 2451–2459. [Google Scholar] [CrossRef]
- Kim, J.; Lim, H.W.; Kim, J.; Rott, L.; Hillsamer, P.; Butcher, E.C. Unique gene expression program of human germinal center T helper cells. Blood 2004, 104, 1952–1960. [Google Scholar] [CrossRef] [PubMed]
- Fisher, B.A.; Jonsson, R.; Daniels, T.; Bombardieri, M.; Brown, R.M.; Morgan, P.; Bombardieri, S.; Ng, W.-F.; Tzioufas, A.G.; Vitali, C.; et al. Standardisation of labial salivary gland histopathology in clinical trials in primary Sjögren’s syndrome. Ann. Rheum. Dis. 2016, 76, 1161–1168. [Google Scholar] [CrossRef] [PubMed]
- Rao, D.A.; Gurish, M.F.; Marshall, J.; Slowikowski, K.; Fonseka, C.Y.; Liu, Y.; Donlin, L.T.; Henderson, L.A.; Wei, K.; Mizoguchi, F.; et al. Pathologically expanded peripheral T helper cell subset drives B cells in rheumatoid arthritis. Natural 2017, 542, 110–114. [Google Scholar] [CrossRef]
- Barone, F.; Bombardieri, M.; Manzo, A.; Blades, M.C.; Morgan, P.R.; Challacombe, S.; Valesini, G.; Pitzalis, C. Association of CXCL13 and CCL21 expression with the progressive organization of lymphoid-like structures in Sjögren’s syndrome. Arthritis Rheum. 2005, 52, 1773–1784. [Google Scholar] [CrossRef]
- Voigt, A.; Bohn, K.; Sukumaran, S.; Stewart, C.M.; Bhattacharya, I.; Nguyen, C.Q. Unique glandular ex-vivo Th1 and Th17 receptor motifs in Sjögren’s syndrome patients using single-cell analysis. Clin. Immunol. 2018, 192, 58–67. [Google Scholar] [CrossRef]
- Kennedy, M.K.; Willis, C.R.; Armitage, R.J. Deciphering CD30 ligand biology and its role in humoral immunity. Immunology 2006, 118, 143–152. [Google Scholar] [CrossRef] [PubMed]
- Sreeramkumar, V.; Hons, M.; Punzón, C.; Stein, J.V.; Sancho, D.; Fresno, M.; Cuesta, N. Efficient T-cell priming and activation requires signaling through prostaglandin E2 (EP) receptors. Immunol. Cell Boil. 2015, 94, 39–51. [Google Scholar] [CrossRef] [PubMed]
- DiToro, D.; Winstead, C.J.; Pham, D.; Witte, S.; Andargachew, R.; Singer, J.R.; Wilson, C.G.; Zindl, C.L.; Luther, R.J.; Silberger, D.J.; et al. Differential IL-2 expression defines developmental fates of follicular versus nonfollicular helper T cells. Science 2018, 361, e2933. [Google Scholar] [CrossRef] [PubMed]
- Schmitt, N.; Liu, Y.; Bentebibel, S.-E.; Munagala, I.; Bourdery, L.; Venuprasad, K.; Banchereau, J.; Ueno, H. The cytokine TGF-beta co-opts signaling via STAT3-STAT4 to promote the differentiation of human Tfh cells. Nat. Immunol. 2014, 15, 856–865. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; Wang, B.; Sun, X.; Li, H.; Ouyang, X.; Wei, J.; Dai, B.; Zhang, Y.; Li, X. Rheumatoid arthritis fibroblast-like synoviocytes co-cultured with PBMC increased peripheral CD4+CXCR5+ICOS+ T cell numbers. Clin. Exp. Immunol. 2017, 190, 384–393. [Google Scholar] [CrossRef] [PubMed]
- Ma, C.S.; Deenick, E.K.; Batten, M.; Tangye, S.G. The origins, function, and regulation of T follicular helper cells. J. Exp. Med. 2012, 209, 1241–1253. [Google Scholar] [CrossRef] [PubMed]
- Weinstein, J.S.; Herman, E.I.; Lainez, B.; Licona-Limón, P.; Esplugues, E.; Flavell, R.; Craft, J. TFH cells progressively differentiate to regulate the germinal center response. Nat. Immunol. 2016, 17, 1197–1205. [Google Scholar] [CrossRef] [PubMed]
| FACS Study | Microarray Study | |||||
|---|---|---|---|---|---|---|
| Demographics | pSS (n = 51) | nSS (n = 69) | p-Value | pSS (n = 17) | nSS (n = 15) | p-Value |
| Age (years) | 51 ± 1.9 | 49 ± 1.4 | 0.24 a | 51.8 ± 3.2 | 42.1 ± 2.2 | 0.022 a |
| Gender (% Fem) | 50/51 (98) | 64/69 (92.8) | 0.24 b | 16/17 (94) | 13/15 (87) | 0.58 b |
| Race (%) | ||||||
| White | 30/51 (59) | 35/69 (51) | 0.58 d | 11/17 (65) | 4/15 (24) | 0.09 d |
| NatAm/> one c | 19/51 (37) | 29/69 (42) | 5/17 (29) | 10/15 (66) | ||
| Black/Asian | 2/51 (4) | 5/69 (7) | 1/17 (6) | 1/15 (7) | ||
| Ethnicity | ||||||
| Hispanic | 2/51 (4) | 4/69 (6) | 1.0 b | 6/17 (35) | 7/15 (47) | 1.0 b |
| Clinical Features | pSS | nSS | p-value b | pSS | nSS | p-value b |
| FS+ (%) e | 32/51 (64) | 4/69 (6) | <0.0001 | 17/17 (100) | 0/15 (0) | <0.0001 |
| Anti-Ro+ (%) f | 28/51 (55) | 4/69 (6) | <0.0001 | 13/17 (76) | 0/15 (0) | <0.0001 |
| Anti-La+ (% +) f | 20/51 (39) | 1/69 (1) | <0.0001 | 4/17 (24) | 0/15 (0) | 0.104 |
| WUSF+ (% +) g | 30/51 (59) | 32/69 (46) | 0.19 | 11/17 (65) | 3/15 (20) | 0.016 |
| Schirmer’s+ (%) h | 19/51 (37) | 18/69 (26) | 0.23 | 6/17 (35) | 4/15 (27) | 0.712 |
| vBS+ (%) i | 35/51 (69) | 26/69 (38) | 0.0009 | 12/17 (71) | 3/15 (20) | 0.006 |
| Clinical Feature | Spearman’s Correlation a | Gamma Regression Model | ||
|---|---|---|---|---|
| r | p-Value | Model | Variable p-Value | |
| vBS b | 0.43 | 7.2 × 10−7 | x + age | 6.7 × 10−5 |
| x | 1.5 × 10−5 | |||
| Schirmer’s test c | −0.087 | 0.34 | x + age | 0.73 |
| x | 0.39 | |||
| WUSF d | −0.09 | 0.31 | x + age | 0.77 |
| x | 0.67 | |||
| Biopsy Focus Score | 0.43 | 7.3 × 10−7 | x + age | 9.0 × 10−7 |
| x | 2.3 × 10−8 | |||
| Anti-Ro Ab e | 0.40 | 2.6 × 10−5 | x + age | 1.1 × 10−5 |
| x | 3.0 × 10−6 | |||
| Serum IgG | 0.38 | 1.7 × 10−5 | x + age f | 9.0 × 10−7 |
| x | 1.4 × 10−5 | |||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Joachims, M.L.; Leehan, K.M.; Dozmorov, M.G.; Georgescu, C.; Pan, Z.; Lawrence, C.; Marlin, M.C.; Macwana, S.; Rasmussen, A.; Radfar, L.; et al. Sjögren’s Syndrome Minor Salivary Gland CD4+ Memory T Cells Associate with Glandular Disease Features and Have a Germinal Center T Follicular Helper Transcriptional Profile. J. Clin. Med. 2020, 9, 2164. https://doi.org/10.3390/jcm9072164
Joachims ML, Leehan KM, Dozmorov MG, Georgescu C, Pan Z, Lawrence C, Marlin MC, Macwana S, Rasmussen A, Radfar L, et al. Sjögren’s Syndrome Minor Salivary Gland CD4+ Memory T Cells Associate with Glandular Disease Features and Have a Germinal Center T Follicular Helper Transcriptional Profile. Journal of Clinical Medicine. 2020; 9(7):2164. https://doi.org/10.3390/jcm9072164
Chicago/Turabian StyleJoachims, Michelle L., Kerry M. Leehan, Mikhail G. Dozmorov, Constantin Georgescu, Zijian Pan, Christina Lawrence, M. Caleb Marlin, Susan Macwana, Astrid Rasmussen, Lida Radfar, and et al. 2020. "Sjögren’s Syndrome Minor Salivary Gland CD4+ Memory T Cells Associate with Glandular Disease Features and Have a Germinal Center T Follicular Helper Transcriptional Profile" Journal of Clinical Medicine 9, no. 7: 2164. https://doi.org/10.3390/jcm9072164
APA StyleJoachims, M. L., Leehan, K. M., Dozmorov, M. G., Georgescu, C., Pan, Z., Lawrence, C., Marlin, M. C., Macwana, S., Rasmussen, A., Radfar, L., Lewis, D. M., Stone, D. U., Grundahl, K., Scofield, R. H., Lessard, C. J., Wren, J. D., Thompson, L. F., Guthridge, J. M., Sivils, K. L., ... Farris, A. D. (2020). Sjögren’s Syndrome Minor Salivary Gland CD4+ Memory T Cells Associate with Glandular Disease Features and Have a Germinal Center T Follicular Helper Transcriptional Profile. Journal of Clinical Medicine, 9(7), 2164. https://doi.org/10.3390/jcm9072164





