In Vivo Reflectance Confocal Microscopy-Diagnostic Criteria for Actinic Cheilitis and Squamous Cell Carcinoma of the Lip
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. RCM Imaging and Analysis
2.3. Histopathology
2.4. Statistical Analysis
3. Results
3.1. RCM Features for Differentiating between AC and Lip SCC
3.2. Vascularization in AC and SCC Lesions
4. Discussions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- De Lucena, E.E.S.; Costa, D.C.B.; da Silveira, E.J.D.; Lima, K.C. Prevalence and factors associated to actinic cheilitis in beach workers. Oral Dis. 2012, 18, 575–579. [Google Scholar] [CrossRef]
- Kaugars, G.E.; Pillion, T.; Svirsky, J.A.; Page, D.G.; Burns, J.C.; Abbey, L.M. Actinic cheilitis: A review of 152 cases. Oral Surg. Oral Med. Oral Pathol. Oral Radiol Endodontology 1999, 88, 181–186. [Google Scholar] [CrossRef]
- Corso, F.; Wild, C.; Gouveia, L.; Ribas, M. Actinic cheilitis: Prevalence in dental clinics from pucpr, curitiba, brazil. Rev. Clin. Pesq Odontol 2006, 2, 277–281. [Google Scholar]
- Markopoulos, A.; Albanidou-Farmaki, E.; Kayavis, I. Actinic cheilitis: Clinical and pathologic characteristics in 65 cases. Oral Dis. 2004, 10, 212–216. [Google Scholar] [CrossRef]
- Cavalcante, A.S.R.; Anbinder, A.L.; Carvalho, Y.R. Actinic cheilitis: Clinical and histological features. J. Oral Maxillofac. Surg. 2008, 66, 498–503. [Google Scholar] [CrossRef]
- Savage, N.W.; McKay, C.; Faulkner, C. Actinic cheilitis in dental practice. Aust. Dent. J. 2010, 55 (Suppl. 1), 78–84. [Google Scholar] [CrossRef]
- Vieira, R.A.M.A.R.; Minicucci, E.M.; Marques, M.E.A.; Marques, S.A. Actinic cheilitis and squamous cell carcinoma of the lip: Clinical, histopathological and immunogenetic aspects. An. Bras. Dermatol. 2012, 87, 105–114. [Google Scholar] [CrossRef]
- Nico, M.M.S.; Rivitti, E.A.; Lourenço, S.V. Actinic cheilitis: Histologic study of the entire vermilion and comparison with previous biopsy. J. Cutan. Pathol. 2007, 34, 309–314. [Google Scholar] [CrossRef]
- Miranda, A.M.; Soares, L.G.; Ferrari, T.M.; Silva, D.G.; Falabella, M.E.; Tinoco, E. Prevalence of actinic cheilitis in a population of agricultural sugarcane workers. Acta Odontol. Latinoam. 2012, 25, 201–207. [Google Scholar]
- Miranda, A.M.; Ferrari, T.; Leite, T.; Domingos, T.; Cunha, K.; Dias, E. Value of videoroscopy in the detection of alterations of actinic cheilitis and the selection of biopsy areas. Med. Oral Patol. Oral Cir. Bucal. 2015, 20, e292–e297. [Google Scholar] [CrossRef]
- De Sarmento, D.J.S.; da Miguel, M.C.C.; Queiroz, L.M.; Godoy, G.P.; da Silveira, E.J. Actinic cheilitis: Clinicopathologic profile and association with degree of dysplasia. Int. J. Dermatol. 2014, 53, 466–472. [Google Scholar] [CrossRef]
- Lopes, M.L.; Junior, F.L.S.; Lima, K.C.; Oliveira, P.T.; Silveira, E.J. Clinicopathological profile and management of 161 cases of actinic cheilitis. An. Bras. Dermatol. 2015, 90, 505–512. [Google Scholar] [CrossRef]
- Picascia, D.D.; Robinson, J.K. Actinie cheilitis: A review of the etiology, differential diagnosis, and treatment. J. Am. Acad. Dermatol. 1987, 17, 255–264. [Google Scholar] [CrossRef]
- Ulrich, M.; Gonzalez, S.; Lange-Asschenfeldt, B.; Roewert-Huber, J.; Sterry, W.; Stockfleth, E.; Astner, S. Non-invasive diagnosis and monitoring of actinic cheilitis with reflectance confocal microscopy. J. Eur. Acad. Dermatol. Venereol. 2011, 25, 276–284. [Google Scholar] [CrossRef]
- Cooper, J.S.; Porter, K.; Mallin, K.; Hoffman, H.T.; Weber, R.S.; Ang, K.K.; Gay, E.G.; Langer, C.J. National cancer database report on cancer of the head and neck: 10-year update. Head Neck 2009, 31, 748–758. [Google Scholar] [CrossRef]
- Miranda, A.M.O.; Ferrari, T.M.; Calandro, T.L.L. Queilite actínica: Aspectos clínicos e prevalência encontrados em uma população rural do interior do brasil. Saúde E Pesquisa 2011, 4, 67–72. [Google Scholar]
- Cockerell, C.J. Pathology and pathobiology of the actinic (solar) keratosis. Br. J. Dermatol. 2003, 149, 34–36. [Google Scholar] [CrossRef]
- Holmes, C.; Foley, P.; Freeman, M.; Chong, A.H. Solar keratosis: Epidemiology, pathogenesis, presentation and treatment. Australas. J. Dermatol. 2007, 48, 67–76. [Google Scholar] [CrossRef]
- Wood, N.H.; Khammissa, R.; Meyerov, R.; Lemmer, J.; Feller, L. Actinic cheilitis: A case report and a review of the literature. Eur. J. Dent. 2011, 5, 101–106. [Google Scholar] [CrossRef]
- Kwon, N.H.; Kim, S.Y.; Kim, G.M. A case of metastatic squamous cell carcinoma arising from actinic cheilitis. Ann. Dermatol 2011, 23, 101–103. [Google Scholar] [CrossRef]
- de Abreu, M.A.M.M.; da Silva, O.M.P.; Pimentel, D.R.N.; Hirata, C.H.W.; Weckx, L.L.M.; de Alchorne, M.M.A.; Michalany, N.S. Actinic cheilitis adjacent to squamous carcinoma of the lips as an indicator of prognosis. Braz. J. Otorhinolaryngol. 2006, 72, 767–771. [Google Scholar] [CrossRef][Green Version]
- Glogau, R.G. The risk of progression to invasive disease. J. Am. Acad. Dermatol. 2000, 42, S23–S24. [Google Scholar] [CrossRef]
- Moy, R.L. Clinical presentation of actinic keratoses and squamous cell carcinoma. J. Am. Acad. Dermatol. 2000, 42, S8–S10. [Google Scholar] [CrossRef]
- Ion, A.; Popa, I.M.; Papagheorghe, L.M.L.; Lisievici, C.; Lupu, M.; Voiculescu, V.; Caruntu, C.; Boda, D. Proteomic approaches to biomarker discovery in cutaneous t-cell lymphoma. Dis. Markers 2016, 2016, 1–8. [Google Scholar] [CrossRef]
- Lupu, M.; Caruntu, C.; Ghita, M.A.; Voiculescu, V.; Voiculescu, S.; Rosca, A.E.; Caruntu, A.; Moraru, L.; Popa, I.M.; Calenic, B.; et al. Gene expression and proteome analysis as sources of biomarkers in basal cell carcinoma. Dis. Markers 2016, 2016, 1–9. [Google Scholar] [CrossRef]
- Voiculescu, V.; Calenic, B.; Ghita, M.; Lupu, M.; Caruntu, A.; Moraru, L.; Voiculescu, S.; Ion, A.; Greabu, M.; Ishkitiev, N.; et al. From normal skin to squamous cell carcinoma: A quest for novel biomarkers. Dis. Markers 2016, 2016, 1–14. [Google Scholar] [CrossRef]
- Solomon, I.; Voiculescu, V.M.; Caruntu, C.; Lupu, M.; Popa, A.; Ilie, M.A.; Albulescu, R.; Caruntu, A.; Tanase, C.; Constantin, C. Neuroendocrine factors and head and neck squamous cell carcinoma: An affair to remember. Dis. Markers 2018, 2018. [Google Scholar] [CrossRef]
- Lupu, M.; Caruntu, A.; Caruntu, C.; Boda, D.; Moraru, L.; Voiculescu, V.; Bastian, A. Non-invasive imaging of actinic cheilitis and squamous cell carcinoma of the lip. Mol. Clin. Oncol. 2018, 8, 640–646. [Google Scholar] [CrossRef]
- Lupu, M.; Căruntu, A.; Moraru, L.; Voiculescu, V.M.; Boda, D.; Tănase, C.; Căruntu, C. Non-invasive imaging techniques for early diagnosis of radiation-induced squamous cell carcinoma of the lip. Rom. J. Morphol. Embryol. 2018, 59, 949–953. [Google Scholar]
- Ridgway, J.M.; Armstrong, W.B.; Guo, S.; Mahmood, U.; Su, J.; Jackson, R.P.; Shibuya, T.; Crumley, R.L.; Gu, M.; Chen, Z.; et al. In vivo optical coherence tomography of the human oral cavity and oropharynx. Arch. Otolaryngol. Head Neck Surg. 2006, 132, 1074–1081. [Google Scholar] [CrossRef]
- Lupu, M.; Popa, I.M.; Voiculescu, V.M.; Boda, D.; Caruntu, C.; Zurac, S.; Giurcaneanu, C. A retrospective study of the diagnostic accuracy of in vivo reflectance confocal microscopy for basal cell carcinoma diagnosis and subtyping. J. Clin. Med. 2019, 8, 449. [Google Scholar] [CrossRef] [PubMed]
- Peppelman, M.; Nguyen, K.P.; Hoogedoorn, L.; van Erp, P.E.J.; Gerritsen, M.J.P. Reflectance confocal microscopy: Non-invasive distinction between actinic keratosis and squamous cell carcinoma. J. Eur. Acad. Dermatol. Venereol. 2014, 29, 1302–1309. [Google Scholar] [CrossRef] [PubMed]
- Guitera, P.; Menzies, S.W.; Longo, C.; Cesinaro, A.M.; Scolyer, R.A.; Pellacani, G. In vivo confocal microscopy for diagnosis of melanoma and basal cell carcinoma using a two-step method: Analysis of 710 consecutive clinically equivocal cases. J. Invest. Dermatol. 2012, 132, 2386–2394. [Google Scholar] [CrossRef] [PubMed]
- Langley, R.G.B.; Walsh, N.; Sutherland, A.E.; Propperova, I.; Delaney, L.; Morris, S.F.; Gallant, C. The diagnostic accuracy of in vivo confocal scanning laser microscopy compared to dermoscopy of benign and malignant melanocytic lesions: A prospective study. Dermatology 2007, 215, 365–372. [Google Scholar] [CrossRef] [PubMed]
- Wolberink, E.A.W.; van Erp, P.E.J.; Teussink, M.M.; van de Kerkhof, P.C.M.; Gerritsen, M.J.P. Cellular features of psoriatic skin: Imaging and quantification using in vivo reflectance confocal microscopy. Cytom. Part B Clin. Cytom. 2010, 80, 141–149. [Google Scholar] [CrossRef] [PubMed]
- Horn, M.; Gerger, A.; Ahlgrimm-Siess, V.; Weger, W.; Koller, S.; Kerl, H.; Samonigg, H.; Smolle, J.; Hofmann-Wellenhof, R. Discrimination of actinic keratoses from normal skin with reflectance mode confocal microscopy. Dermatol. Surg. 2008, 34, 620–625. [Google Scholar]
- Ulrich, M.; Forschner, T.; Röwert-Huber, J.; González, S.; Stockfleth, E.; Sterry, W.; Astner, S. Differentiation between actinic keratoses and disseminated superficial actinic porokeratoses with reflectance confocal microscopy. Br. J. Dermatol. 2007, 156, 47–52. [Google Scholar] [CrossRef]
- Aghassi, D.; Anderson, R.R.; Gonzlez, S. Confocal laser microscopic imaging of actinic keratoses in vivo: A preliminary report. J. Am. Acad. Dermatol. 2000, 43, 42–48. [Google Scholar] [CrossRef]
- Peppelman, M.; Wolberink, E.A.W.; Koopman, R.J.J.; van Erp, P.E.J.; Gerritsen, M.-J.P. In vivo reflectance confocal microscopy: A useful tool to select the location of a punch biopsy in a large, clinically indistinctive lesion. Case Rep. Dermatol. 2013, 5, 129–132. [Google Scholar] [CrossRef]
- Richtig, E.; Ahlgrimm-Siess, V.; Koller, S.; Gerger, A.; Horn, M.; Smolle, J.; Hofmann-Wellenhof, R. Follow-up of actinic keratoses after shave biopsy byin-vivoreflectance confocal microscopy-a pilot study. J. Eur. Acad. Dermatol. Venereol. 2010, 24, 293–298. [Google Scholar] [CrossRef]
- Ulrich, M.; Lange-Asschenfeldt, S.; González, S. In vivo reflectance confocal microscopy for early diagnosis of nonmelanoma skin cancer. Actas Dermosifiliogr. 2012, 103, 784–789. [Google Scholar] [CrossRef] [PubMed]
- Rajadhyaksha, M.; González, S.; Zavislan, J.M.; Rox Anderson, R.; Webb, R.H. In vivo confocal scanning laser microscopy of human skin ii: Advances in instrumentation and comparison with histology. J. Invest. Dermatol. 1999, 113, 293–303. [Google Scholar] [CrossRef]
- Langley, R.G.B.; Burton, E.; Walsh, N.; Propperova, I.; Murray, S.J. In vivo confocal scanning laser microscopy of benign lentigines: Comparison to conventional histology and in vivo characteristics of lentigo maligna. J. Am. Acad. Dermatol. 2006, 55, 88–97. [Google Scholar] [CrossRef] [PubMed]
- González, S.; González, E.; White, W.M.; Rajadhyaksha, M.; Anderson, R.R. Allergic contact dermatitis: Correlation of in vivo confocal imaging to routine histology. J. Am. Acad. Dermatol. 1999, 40, 708–713. [Google Scholar] [CrossRef]
- Ilie, M.A.; Caruntu, C.; Lixandru, D.; Tampa, M.; Georgescu, S.R.; Constantin, M.M.; Constantin, C.; Neagu, M.; Zurac, S.A.; Boda, D. In vivo confocal laser scanning microscopy imaging of skin inflammation: Clinical applications and research directions. Exp. Ther. Med. 2019, 17, 1004–1011. [Google Scholar] [CrossRef]
- Ilie, M.A.; Caruntu, C.; Lupu, M.; Lixandru, D.; Georgescu, S.-R.; Bastian, A.; Constantin, C.; Neagu, M.; Zurac, S.A.; Boda, D. Current and future applications of confocal laser scanning microscopy imaging in skin oncology. Oncol. Lett. 2019. [Google Scholar] [CrossRef]
- Ianoși, S.L.; Forsea, A.M.; Lupu, M.; Ilie, M.A.; Zurac, S.; Boda, D.; Ianosi, G.; Neagoe, D.; Tutunaru, C.; Popa, C.M. Role of modern imaging techniques for the in vivo diagnosis of lichen planus. Exp. Ther. Med. 2019, 17, 1052–1060. [Google Scholar] [CrossRef]
- Lupu, M.; Caruntu, C.; Popa, M.I.; Voiculescu, V.M.; Zurac, S.; Boda, D. Vascular patterns in basal cell carcinoma: Dermoscopic, confocal and histopathological perspectives (review). Oncol. Lett. 2019. [Google Scholar] [CrossRef]
- Lupu, M.; Popa, I.M.; Voiculescu, V.M.; Caruntu, A.; Caruntu, C. A systematic review and meta-analysis of the accuracy of in vivo reflectance confocal microscopy for the diagnosis of primary basal cell carcinoma. J. Clin. Med. 2019, 8, 1462. [Google Scholar] [CrossRef]
- Caruntu, C.; Boda, D.; Gutu, D.E.; Caruntu, A. In vivo reflectance confocal microscopy of basal cell carcinoma with cystic degeneration. Rom. J. Morphol. Embryol. 2014, 55, 1437–1441. [Google Scholar]
- Ghita, M.A.; Caruntu, C.; Rosca, A.E.; Kaleshi, H.; Caruntu, A.; Moraru, L.; Docea, A.O.; Zurac, S.; Boda, D.; Neagu, M.; et al. Reflectance confocal microscopy and dermoscopy for in vivo, non-invasive skin imaging of superficial basal cell carcinoma. Oncol. Lett. 2016, 11, 3019–3024. [Google Scholar] [CrossRef] [PubMed]
- Rishpon, A.; Kim, N.; Scope, A.; Porges, L.; Oliviero, M.C.; Braun, R.P.; Marghoob, A.A.; Fox, C.A.; Rabinovitz, H.S. Reflectance confocal microscopy criteria for squamous cell carcinomas and actinic keratoses. Arch. Dermatol. 2009, 145. [Google Scholar] [CrossRef] [PubMed]
- Braga, J.C.T.; Scope, A.; Klaz, I.; Mecca, P.; González, S.; Rabinovitz, H.; Marghoob, A.A. The significance of reflectance confocal microscopy in the assessment of solitary pink skin lesions. J. Am. Acad. Dermatol. 2009, 61, 230–241. [Google Scholar] [CrossRef] [PubMed]
- Bağcı, I.S.; Gürel, M.S.; Aksu, A.E.K.; Erdemir, A.T.; Yüksel, E.İ.; Başaran, Y.K. Reflectance confocal microscopic evaluation of nonmelanocytic lip lesions. Lasers Med. Sci. 2017, 32, 1497–1506. [Google Scholar] [CrossRef]
- Georgescu, S.R.; Mitran, C.I.; Mitran, M.I.; Caruntu, C.; Caruntu, A.; Lupu, M.; Matei, C.; Constantin, C.; Neagu, M. Tumour microenvironment in skin carcinogenesis. In Tumor Microenvironments in Organs; Springer: Berlin/Heidelberg, Germany, 2020; pp. 123–142. [Google Scholar]
- Hartmann, D.; Krammer, S.; Bachmann, M.R.; Mathemeier, L.; Ruzicka, T.; Bagci, I.S.; von Braunmühl, T. Ex vivo confocal microscopy features of cutaneous squamous cell carcinoma. J. Biophotonics 2018, 11, e201700318. [Google Scholar] [CrossRef]
- Ahlgrimm-Siess, V.; Cao, T.; Oliviero, M.; Hofmann-Wellenhof, R.; Rabinovitz, H.S.; Scope, A. The vasculature of nonmelanocytic skin tumors on reflectance confocal microscopy. Arch. Dermatol. 2011, 147, 264. [Google Scholar] [CrossRef]
- Skobe, M.; Rockwell, P.; Goldstein, N.; Vosseler, S.; Fusenig, N.E. Halting angiogenesis suppresses carcinoma cell invasion. Nat. Med. 1997, 3, 1222–1227. [Google Scholar] [CrossRef]
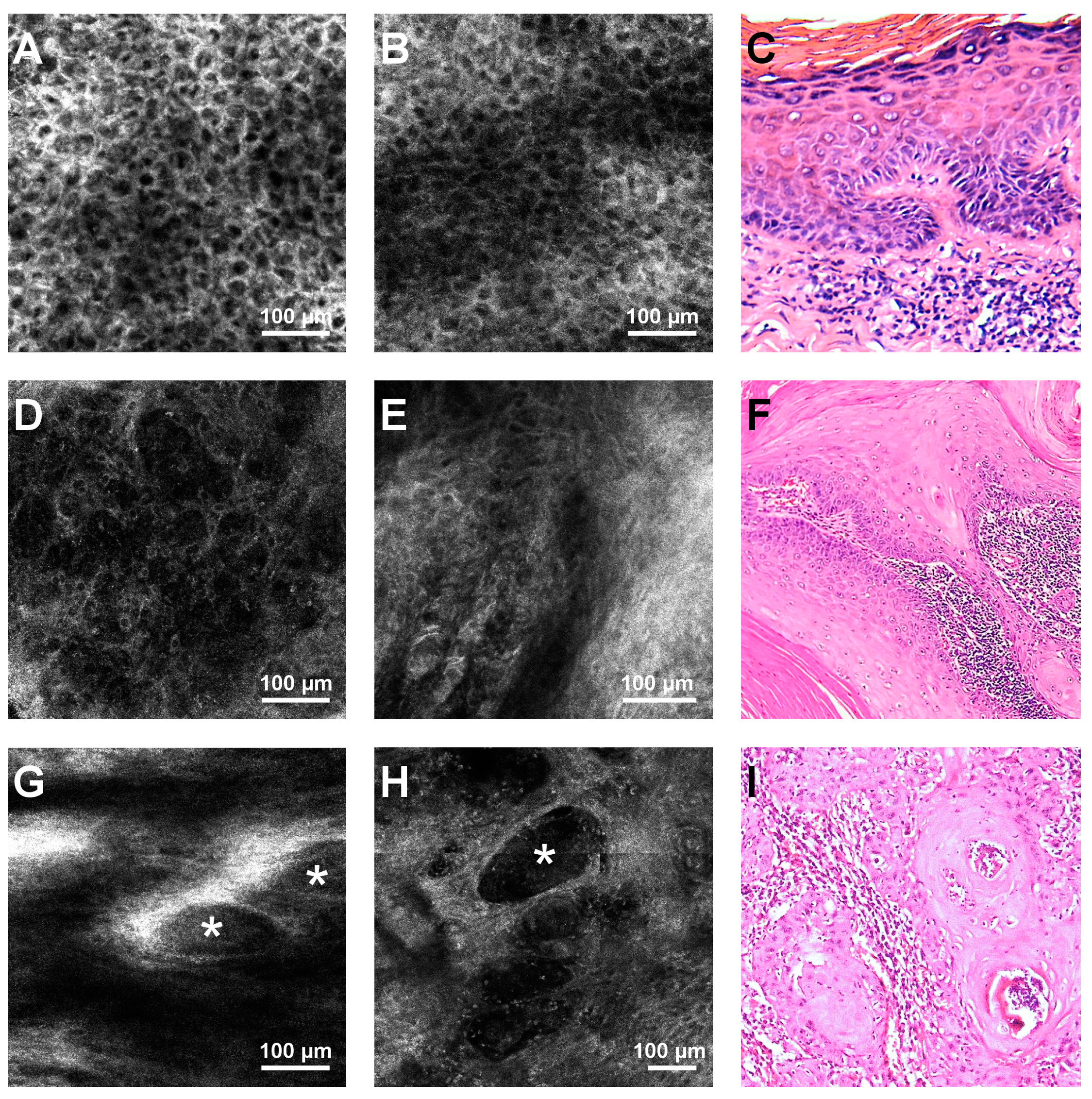
| Epidermis | |
| Ulceration | Dark areas, with irregular and well-defined borders, filled with amorphous material and cellular debris. |
| Hyperkeratosis/scale | Increased thickness of the stratum corneum seen as areas of amorphous, variably refractive material, and reduced resolution of deeper structures. |
| Parakeratosis | Presence of individual polygonal, sharply delineated, nucleated cells in the stratum corneum. |
| Atypical honeycomb pattern SG/SS * | Cells with irregular shape and size showing bright cell borders, arranged in a distorted fashion, deviating from the normal honeycomb pattern. |
| Architectural disarrangement SG/SS* | Disarray of the normal architecture of the superficial skin layers with unevenly dispersed hyper-refractive granular particles and cells, in which the honeycomb or cobblestone patterns are no longer visible. |
| Targetoid cells SS/SG* | A large cell resembling a target, either with a bright center and dark peripheral halo or a dark center and a bright rim surrounded by a dark peripheral halo. The first one corresponds histologically to large dyskeratotic keratinocytes separated from adjacent cells by a retraction halo, and the second type to dyskeratotic keratinocytes containing a pyknotic nucleus. |
| Dendritic cells | Large cells with obvious dendrites connected to them. |
| Dermal-epidermal junction | |
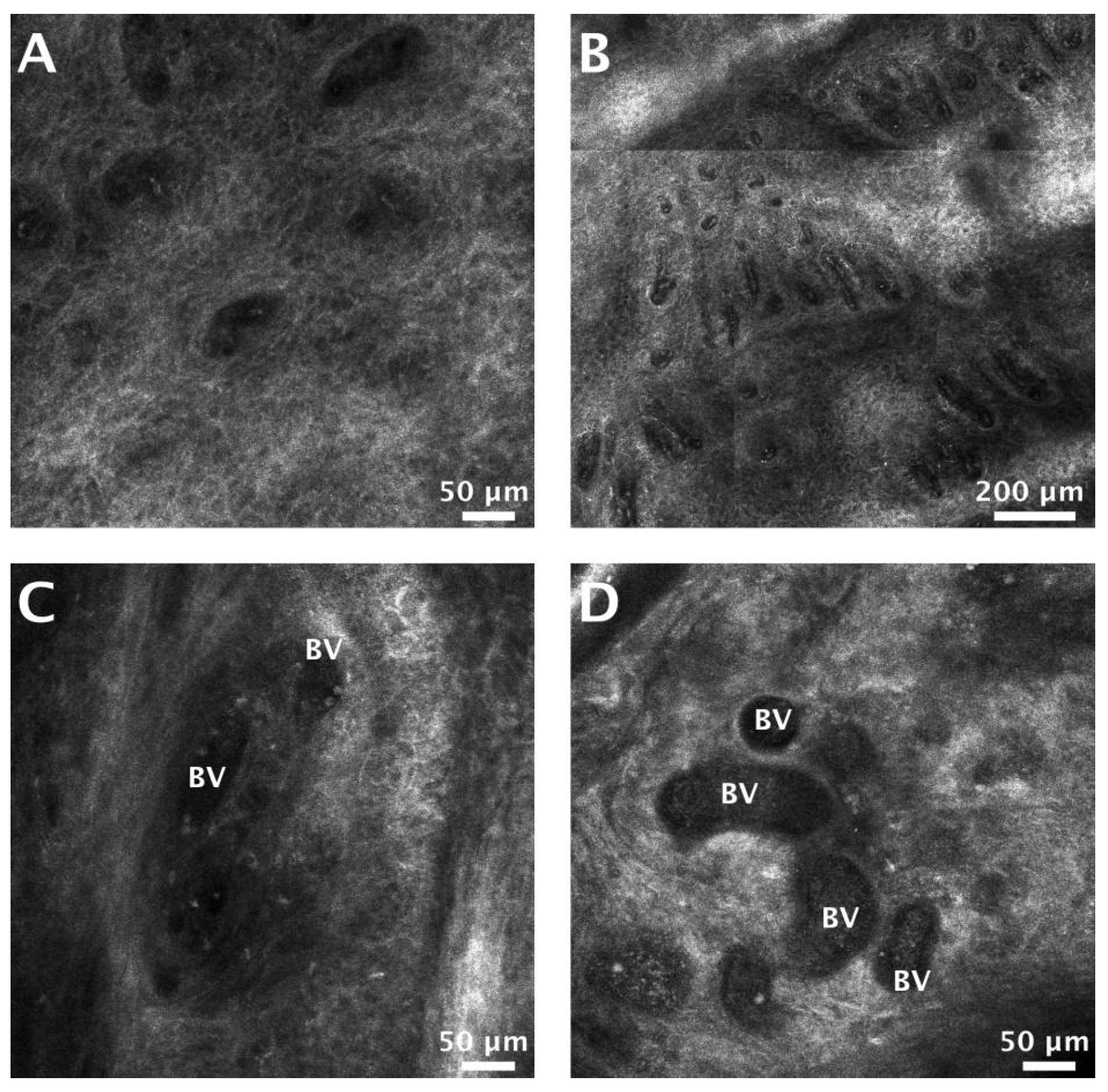
| Increased vessel diameter | Blood vessel diameter larger than 5 µm. |
| Increased vessel density | More than 5 blood vessels per 0.5 × 0.5 mm RCM image. |
| Dermis | |
| Solar elastosis | Lace-like material adjacent to hyper-refractive, thickened collagen bundles. |
| Inflammatory cells | Hyper-refractive, small structures, of 8–10 µm in diameter. |
| Dendritic cells | Large cells with obvious dendrites connected to them. |
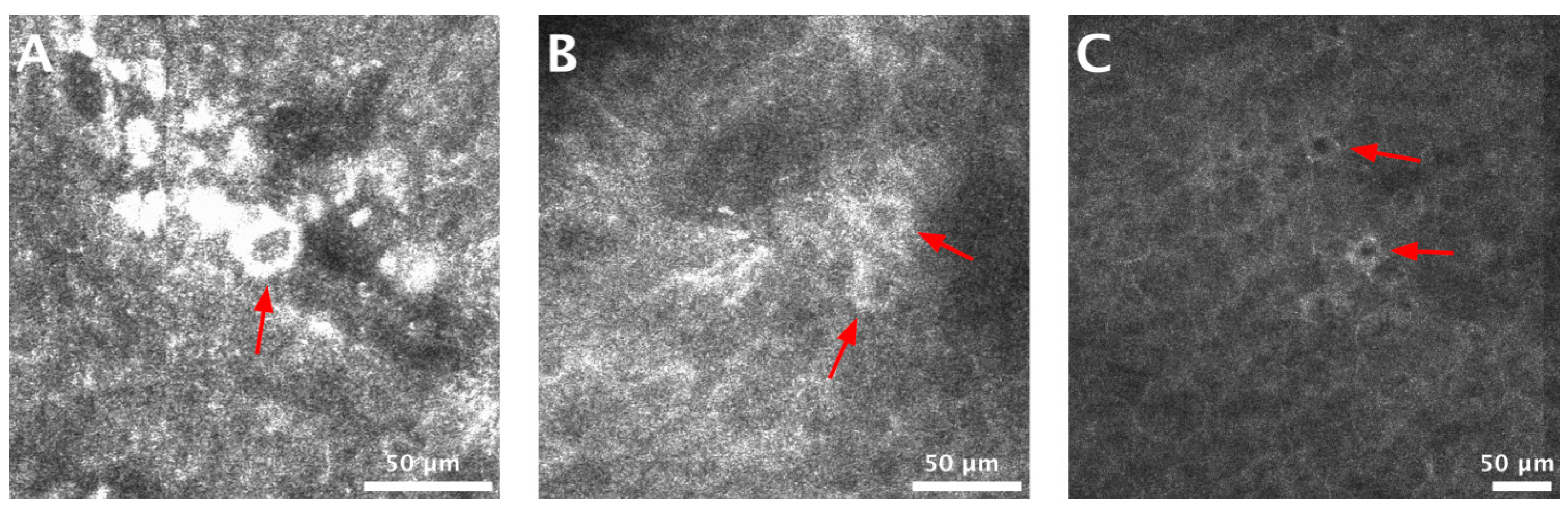
| Atypical keratinocytes (speckled/nucleated) | Round to polygonal cells with a dark nucleus and speckled appearance. |
| Nest-like structures | Defined, irregular, discohesive, aggregates of cells larger than inflammatory cells. |
| Keratin pearls | Whorl-shaped, hyper-refractive, speckled structures. |
| Sex | Age | Smoking | Immune Status | SCC Degree of Differentiation |
|---|---|---|---|---|
| male | 43 | yes | immunocompetent | moderately differentiated |
| female | 59 | no | immunocompetent | well differentiated |
| male | 80 | no | immunocompetent | well differentiated |
| male | 69 | no | immunocompetent | moderately differentiated |
| male | 71 | yes | immunocompetent | well differentiated |
| male | 66 | no | immunocompetent | moderately differentiated |
| male | 68 | yes | immunocompetent | moderately differentiated |
| male | 65 | no | immunocompetent | moderately differentiated |
| RCM Criteria, N (%) | Histopathological Diagnosis | |
|---|---|---|
| AC (N = 4) | Lip SCC (N = 8) | |
| Ulceration | 3 (75%) | 7 (87.5%) |
| Hyperkeratosis/scale | 4 (100%) | 7 (87.5%) |
| Parakeratosis | 3 (75%) | 3 (37.5%) |
| Atypical honeycomb pattern | 4 (100%) | 0 (0%) |
| Epidermal disarray | 0 (0%) | 8 (100%) |
| Target cells in the epidermis | 4 (100%) | 1 (12.5%) |
| Dendritic cells in the epidermis | 1 (25%) | 0 (0%) |
| Solar elastosis | 2 (50%) | 5 (62.5%) |
| Dermal inflammatory cells | 2 (50%) | 6 (75%) |
| Dendritic cells in the dermis | 0 (0%) | 3 (37.5%) |
| Atypical cells in the dermis | 0 (0%) | 4 (50%) |
| Tumor nests in the dermis | 0 (0%) | 4 (50%) |
| RCM Criteria, N (%) | Squamous Cell Carcinoma | p | |
|---|---|---|---|
| Well Differentiated (N = 3) | Moderately Differentiated (N = 5) | ||
| Ulceration | 3 (100%) | 4 (80%) | 0.408 |
| Hyperkeratosis/scale | 3 (100%) | 4 (80%) | 0.408 |
| Parakeratosis | 0 (0%) | 3 (60%) | 0.09 |
| Atypical honeycomb pattern | 0 (0%) | 0 (0%) | - |
| Epidermal disarray | 3 (100%) | 5 (100%) | - |
| Target cells in the epidermis | 1 (33.3%) | 0 (0%) | 0.168 |
| Dendritic cells in the epidermis | 0 (0%) | 0 (0%) | - |
| Solar elastosis | 2 (66.7%) | 3 (60%) | 0.85 |
| Dermal inflammatory cells | 3 (100%) | 3 (60%) | 0.206 |
| Dendritic cells in the dermis | 0 (0%) | 3 (60%) | 0.09 |
| Atypical cells in the dermis | 2 (66.7%) | 2 (40%) | 0.465 |
| Tumor nests in the dermis | 2 (66.7%) | 2 (40%) | 0.465 |
| RCM Criteria | p |
|---|---|
| Actinic cheilitis | |
| Atypical honeycomb pattern | 0.002 |
| Target cells in the epidermis | 0.01 |
| Lip squamous cell carcinoma | |
| Complete epidermal disarray | 0.002 |
| Histopathological Diagnosis | p | ||
|---|---|---|---|
| AC | Lip SCC | ||
| Mean ± SD | Mean ± SD | ||
| Mean blood vessel diameter (µm) | 19.26 ± 5.67 | 37.81 ± 12.77 | 0.006 |
| Mean number of blood vessels | 8.25 ± 1.89 | 8.88 ± 2.53 | 0.64 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lupu, M.; Caruntu, A.; Boda, D.; Caruntu, C. In Vivo Reflectance Confocal Microscopy-Diagnostic Criteria for Actinic Cheilitis and Squamous Cell Carcinoma of the Lip. J. Clin. Med. 2020, 9, 1987. https://doi.org/10.3390/jcm9061987
Lupu M, Caruntu A, Boda D, Caruntu C. In Vivo Reflectance Confocal Microscopy-Diagnostic Criteria for Actinic Cheilitis and Squamous Cell Carcinoma of the Lip. Journal of Clinical Medicine. 2020; 9(6):1987. https://doi.org/10.3390/jcm9061987
Chicago/Turabian StyleLupu, Mihai, Ana Caruntu, Daniel Boda, and Constantin Caruntu. 2020. "In Vivo Reflectance Confocal Microscopy-Diagnostic Criteria for Actinic Cheilitis and Squamous Cell Carcinoma of the Lip" Journal of Clinical Medicine 9, no. 6: 1987. https://doi.org/10.3390/jcm9061987
APA StyleLupu, M., Caruntu, A., Boda, D., & Caruntu, C. (2020). In Vivo Reflectance Confocal Microscopy-Diagnostic Criteria for Actinic Cheilitis and Squamous Cell Carcinoma of the Lip. Journal of Clinical Medicine, 9(6), 1987. https://doi.org/10.3390/jcm9061987








