A U-Shaped Relationship between the Prevalence of Frailty and Body Mass Index in Community-Dwelling Japanese Older Adults: The Kyoto–Kameoka Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. BMI and Other Variables
2.3. Definition of Frailty
2.4. Statistical Analysis
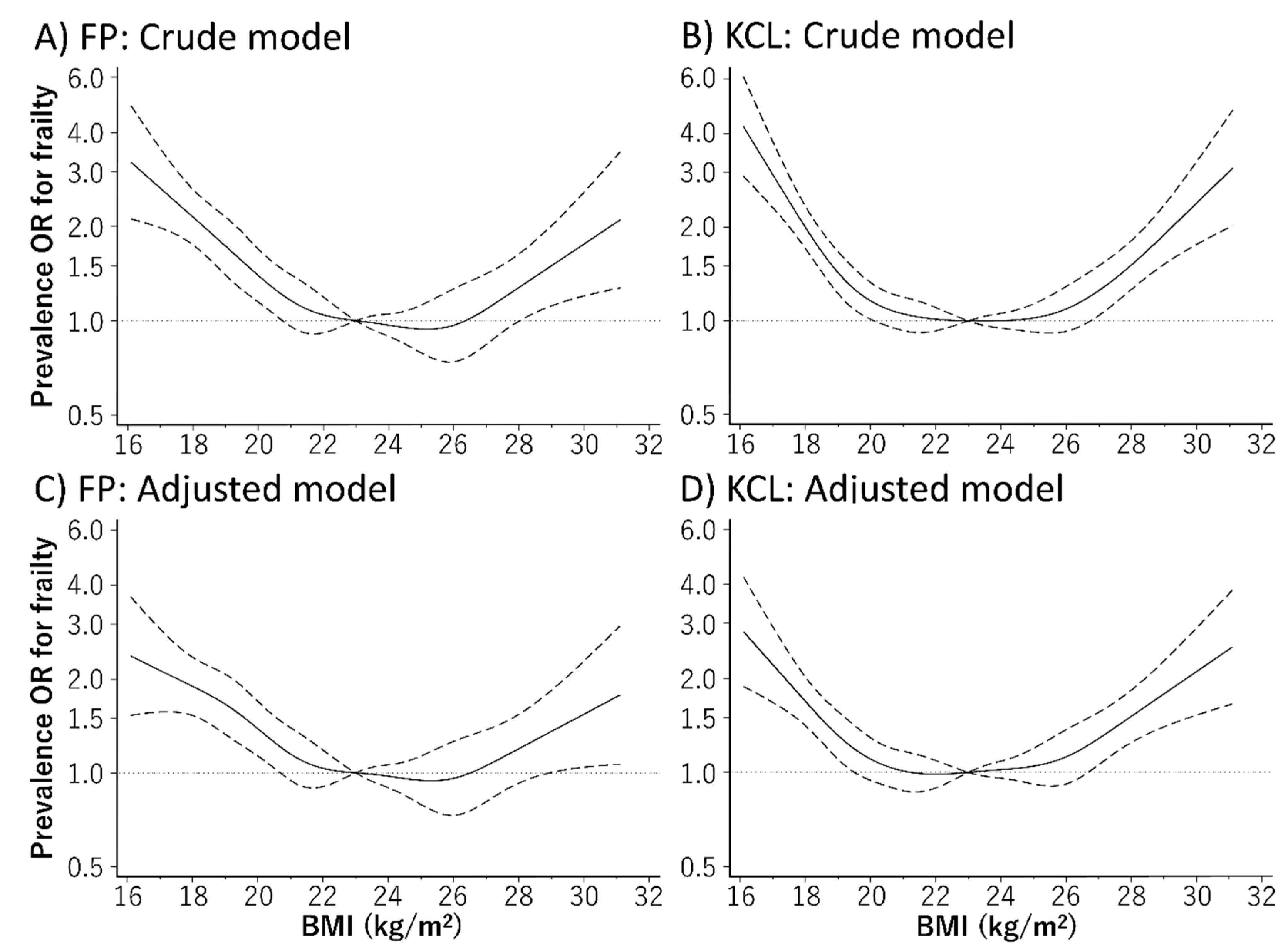
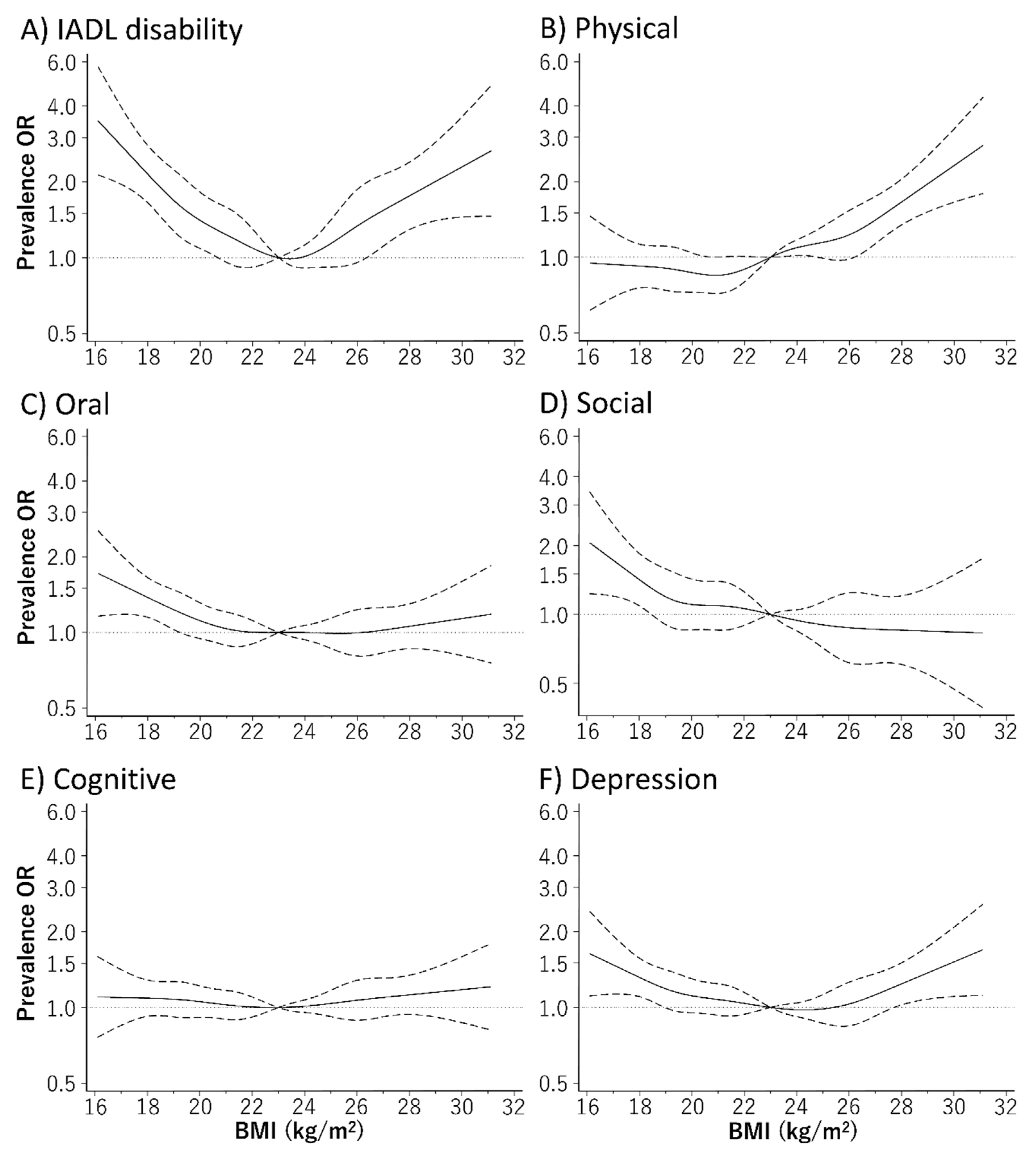
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Clegg, A.; Young, J.; Iliffe, S.; Rikkert, M.O.; Rockwood, K. Frailty in elderly people. Lancet 2013, 381, 752–762. [Google Scholar] [CrossRef]
- Hoogendijk, E.O.; Afilalo, J.; Ensrud, K.E.; Kowal, P.; Onder, G.; Fried, L.P. Frailty: Implications for clinical practice and public health. Lancet 2019, 394, 1365–1375. [Google Scholar] [CrossRef]
- Dent, E.; Martin, F.C.; Bergman, H.; Woo, J.; Romero-Ortuno, R.; Walston, J.D. Management of frailty: Opportunities, challenges, and future directions. Lancet 2019, 394, 1376–1386. [Google Scholar] [CrossRef]
- Nanri, H.; Yamada, Y.; Yoshida, T.; Okabe, Y.; Nozawa, Y.; Itoi, A.; Yoshimura, E.; Watanabe, Y.; Yamaguchi, M.; Yokoyama, K.; et al. Sex Difference in the Association between Protein Intake and Frailty: Assessed Using the Kihon Checklist Indexes Among Older Adults. J. Am. Med. Dir. Assoc. 2018, 19, 801–805. [Google Scholar] [CrossRef]
- Malmstrom, T.K.; Miller, D.K.; Morley, J.E. A comparison of four frailty models. J. Am. Geriatr. Soc. 2014, 62, 721–726. [Google Scholar] [CrossRef]
- Satake, S.; Shimokata, H.; Senda, K.; Kondo, I.; Toba, K. Validity of Total Kihon Checklist Score for Predicting the Incidence of 3-Year Dependency and Mortality in a Community-Dwelling Older Population. J. Am. Med. Dir. Assoc. 2017, 18, 552.e1–552.e6. [Google Scholar] [CrossRef]
- Rockwood, K.; Andrew, M.; Mitnitski, A. A comparison of two approaches to measuring frailty in elderly people. J. Gerontol. A Biol. Sci. Med. Sci. 2007, 62, 738–743. [Google Scholar] [CrossRef]
- Yamada, M.; Arai, H. Predictive Value of Frailty Scores for Healthy Life Expectancy in Community-Dwelling Older Japanese Adults. J. Am. Med. Dir. Assoc. 2015, 16, 1002. [Google Scholar] [CrossRef]
- Matsuo, T.; Sairenchi, T.; Iso, H.; Irie, F.; Tanaka, K.; Fukasawa, N.; Ota, H.; Muto, T. Age- and gender-specific BMI in terms of the lowest mortality in Japanese general population. Obesity (Silver Spring) 2008, 16, 2348–2355. [Google Scholar] [CrossRef]
- Global, B.M.I.M.C.; Di Angelantonio, E.; Bhupathiraju Sh, N.; Wormser, D.; Gao, P.; Kaptoge, S.; Berrington de Gonzalez, A.; Cairns, B.J.; Huxley, R.; Jackson Ch, L.; et al. Body-mass index and all-cause mortality: Individual-participant-data meta-analysis of 239 prospective studies in four continents. Lancet 2016, 388, 776–786. [Google Scholar] [CrossRef]
- Lee, Y.; Kim, J.; Han, E.S.; Ryu, M.; Cho, Y.; Chae, S. Frailty and body mass index as predictors of 3-year mortality in older adults living in the community. Gerontology 2014, 60, 475–482. [Google Scholar] [CrossRef] [PubMed]
- Brunner, E.J.; Shipley, M.J.; Ahmadi-Abhari, S.; Valencia Hernandez, C.; Abell, J.G.; Singh-Manoux, A.; Kawachi, I.; Kivimaki, M. Midlife contributors to socioeconomic differences in frailty during later life: A prospective cohort study. Lancet Public Health 2018, 3, e313–e322. [Google Scholar] [CrossRef]
- Hanlon, P.; Nicholl, B.I.; Jani, B.D.; Lee, D.; McQueenie, R.; Mair, F.S. Frailty and pre-frailty in middle-aged and older adults and its association with multimorbidity and mortality: A prospective analysis of 493 737 UK Biobank participants. Lancet Public Health 2018, 3, e323–e332. [Google Scholar] [CrossRef]
- Hubbard, R.E.; Lang, I.A.; Llewellyn, D.J.; Rockwood, K. Frailty, body mass index, and abdominal obesity in older people. J. Gerontol. A Biol. Sci. Med. Sci. 2010, 65, 377–381. [Google Scholar] [CrossRef]
- Collaboration, N.C.D.R.F. Rising rural body-mass index is the main driver of the global obesity epidemic in adults. Nature 2019, 569, 260–264. [Google Scholar] [CrossRef]
- Yamada, Y.; Nanri, H.; Watanabe, Y.; Yoshida, T.; Yokoyama, K.; Itoi, A.; Date, H.; Yamaguchi, M.; Miyake, M.; Yamagata, E.; et al. Prevalence of Frailty Assessed by Fried and Kihon Checklist Indexes in a Prospective Cohort Study: Design and Demographics of the Kyoto-Kameoka Longitudinal Study. J. Am. Med. Dir. Assoc. 2017, 18, 733. [Google Scholar] [CrossRef]
- Watanabe, D.; Nanri, H.; Sagayama, H.; Yoshida, T.; Itoi, A.; Yamaguchi, M.; Yokoyama, K.; Watanabe, Y.; Goto, C.; Ebine, N.; et al. Estimation of Energy Intake by a Food Frequency Questionnaire: Calibration and Validation with the Doubly Labeled Water Method in Japanese Older People. Nutrients 2019, 11. [Google Scholar] [CrossRef]
- Watanabe, D.; Nanri, H.; Yoshida, T.; Yamaguchi, M.; Sugita, M.; Nozawa, Y.; Okabe, Y.; Itoi, A.; Goto, C.; Yamada, Y.; et al. Validation of Energy and Nutrition Intake in Japanese Elderly Individuals Estimated Based on a Short Food Frequency Questionnaire Compared against a 7-day Dietary Record: The Kyoto-Kameoka Study. Nutrients 2019, 11. [Google Scholar] [CrossRef]
- Ambagtsheer, R.C.; Visvanathan, R.; Dent, E.; Yu, S.; Schultz, T.J.; Beilby, J.M.D. Commonly used screening instruments to identify frailty among community-dwelling older people in a general practice (primary care) setting: A study of diagnostic test accuracy. J. Gerontol. A Biol. Sci. Med. Sci. 2019. [Google Scholar] [CrossRef]
- Sasazuki, S.; Inoue, M.; Tsuji, I.; Sugawara, Y.; Tamakoshi, A.; Matsuo, K.; Wakai, K.; Nagata, C.; Tanaka, K.; Mizoue, T.; et al. Body mass index and mortality from all causes and major causes in Japanese: Results of a pooled analysis of 7 large-scale cohort studies. J. Epidemiol. 2011, 21, 417–430. [Google Scholar] [CrossRef]
- Sewo Sampaio, P.Y.; Sampaio, R.A.; Yamada, M.; Ogita, M.; Arai, H. Validation and translation of the Kihon Checklist (frailty index) into Brazilian Portuguese. Geriatr. Gerontol. Int. 2014, 14, 561–569. [Google Scholar] [CrossRef] [PubMed]
- van Buuren, S.; Groothuis-Oudshoorn, K. Multivariate Imputation by Chained Equations in R. J. Stat. Softw. 2011, 45, 1–67. [Google Scholar] [CrossRef]
- Hozawa, A.; Hirata, T.; Yatsuya, H.; Murakami, Y.; Kuriyama, S.; Tsuji, I.; Sugiyama, D.; Satoh, A.; Tanaka-Mizuno, S.; Miura, K.; et al. Association Between Body Mass Index and All-Cause Death in Japanese Population: Pooled Individual Participant Data Analysis of 13 Cohort Studies. J. Epidemiol. 2019, 29, 457–463. [Google Scholar] [CrossRef]
- Lee, I.M.; Shiroma, E.J.; Kamada, M.; Bassett, D.R.; Matthews, C.E.; Buring, J.E. Association of Step Volume and Intensity with All-Cause Mortality in Older Women. JAMA Intern. Med. 2019. [Google Scholar] [CrossRef] [PubMed]
- Cederholm, T.; Jensen, G.L.; Correia, M.; Gonzalez, M.C.; Fukushima, R.; Higashiguchi, T.; Baptista, G.; Barazzoni, R.; Blaauw, R.; Coats, A.; et al. GLIM criteria for the diagnosis of malnutrition—A consensus report from the global clinical nutrition community. Clin. Nutr. 2019, 38, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Rojer, A.G.; Kruizenga, H.M.; Trappenburg, M.C.; Reijnierse, E.M.; Sipila, S.; Narici, M.V.; Hogrel, J.Y.; Butler-Browne, G.; McPhee, J.S.; Paasuke, M.; et al. The prevalence of malnutrition according to the new ESPEN definition in four diverse populations. Clin. Nutr. 2016, 35, 758–762. [Google Scholar] [CrossRef]
- Volkert, D.; Beck, A.M.; Cederholm, T.; Cruz-Jentoft, A.; Goisser, S.; Hooper, L.; Kiesswetter, E.; Maggio, M.; Raynaud-Simon, A.; Sieber, C.C.; et al. ESPEN guideline on clinical nutrition and hydration in geriatrics. Clin. Nutr. 2019, 38, 10–47. [Google Scholar] [CrossRef]
- Wright, L.; Hickson, M.; Frost, G. Eating together is important: Using a dining room in an acute elderly medical ward increases energy intake. J. Hum. Nutr. Diet. 2006, 19, 23–26. [Google Scholar] [CrossRef]
- Suzuki, H.; Kanazawa, M.; Komagamine, Y.; Iwaki, M.; Jo, A.; Amagai, N.; Minakuchi, S. The effect of new complete denture fabrication and simplified dietary advice on nutrient intake and masticatory function of edentulous elderly: A randomized-controlled trial. Clin. Nutr. 2018, 37, 1441–1447. [Google Scholar] [CrossRef]
- Ministry of Health, Labour and Welfare of Japan Dietary Reference Intakes for Japanese. 2020. Available online: https://www.mhlw.go.jp/stf/seisakunitsuite/bunya/kenkou_iryou/kenkou/eiyou/syokuji_kijyun.html (accessed on 4 March 2020).
- Niederstrasser, N.G.; Rogers, N.T.; Bandelow, S. Determinants of frailty development and progression using a multidimensional frailty index: Evidence from the English Longitudinal Study of Ageing. PLoS ONE 2019, 14, e0223799. [Google Scholar] [CrossRef]
- Hall, K.D.; Sacks, G.; Chandramohan, D.; Chow, C.C.; Wang, Y.C.; Gortmaker, S.L.; Swinburn, B.A. Quantification of the effect of energy imbalance on bodyweight. Lancet 2011, 378, 826–837. [Google Scholar] [CrossRef]
- Yamada, Y.; Colman, R.J.; Kemnitz, J.W.; Baum, S.T.; Anderson, R.M.; Weindruch, R.; Schoeller, D.A. Long-term calorie restriction decreases metabolic cost of movement and prevents decrease of physical activity during aging in rhesus monkeys. Exp. Gerontol 2013, 48, 1226–1235. [Google Scholar] [CrossRef] [PubMed]
- Yamada, Y.; Kemnitz, J.W.; Weindruch, R.; Anderson, R.M.; Schoeller, D.A.; Colman, R.J. Caloric Restriction and Healthy Life Span: Frail Phenotype of Nonhuman Primates in the Wisconsin National Primate Research Center Caloric Restriction Study. J. Gerontol. A Biol. Sci. Med. Sci. 2018, 73, 273–278. [Google Scholar] [CrossRef] [PubMed]
- Nakao, Y.M.; Miyamoto, Y.; Ueshima, K.; Nakao, K.; Nakai, M.; Nishimura, K.; Yasuno, S.; Hosoda, K.; Ogawa, Y.; Itoh, H.; et al. Effectiveness of nationwide screening and lifestyle intervention for abdominal obesity and cardiometabolic risks in Japan: The metabolic syndrome and comprehensive lifestyle intervention study on nationwide database in Japan (MetS ACTION-J study). PLoS ONE 2018, 13, e0190862. [Google Scholar] [CrossRef]
| Whole Cohort (n = 7191) b | Subcohort (n = 1169) c | |||
|---|---|---|---|---|
| Age (years) d | 73.4 | (6.2) | 72.7 | (5.4) |
| Women (n (%)) e | 3788 | (52.7) | 585 | (50.0) |
| BMI (kg/m2) d | 22.7 | (3.5) | 22.6 | (2.8) |
| Alcohol drinker (n (%)) e | 4691 | (65.2) | 812 | (69.5) |
| Current smoker (n (%)) e | 769 | (10.7) | 94 | (8.0) |
| Living alone (n (%)) e | 813 | (11.3) | 123 | (10.5) |
| HSE (n (%)) e | 2442 | (34.0) | 440 | (37.6) |
| Education ≥13 y (n (%)) e | 1607 | (22.3) | 313 | (26.8) |
| MVPA (n (%)) e | 3160 | (43.9) | 621 | (53.1) |
| Denture use (n (%)) e | 4332 | (60.2) | 692 | (59.2) |
| No medication (n (%)) e | 1591 | (22.1) | 240 | (20.5) |
| Hypertension (n (%)) e | 2789 | (38.8) | 474 | (40.5) |
| Stroke (n (%)) e | 269 | (3.7) | 33 | (2.8) |
| Heart disease (n (%)) e | 914 | (12.7) | 131 | (11.2) |
| Diabetes (n (%)) e | 763 | (10.6) | 112 | (9.6) |
| Hyperlipidemia (n (%)) e | 723 | (10.1) | 139 | (11.9) |
| Self-Reported a | Measurement (M) a | Mean Difference (95% CI) b | Correlation c | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Baseline Survey (BS) | Additional Survey (AS) | BS and AS | BS and M | BS and AS | BS and M | |||||||
| Total (n = 1169) | ||||||||||||
| Height (cm) | 157.8 | (8.6) | 157.7 | (8.5) | 156.9 | (8.7) | −0.1 | (−0.2 to 0.0) | −0.9 | (−1.0 to −0.8) | 0.970 | 0.970 |
| Body weight (kg) | 56.4 | (9.6) | 56.8 | (9.8) | 56.8 | (10.0) | 0.4 | (0.2 to 0.6) | 0.4 | (0.3 to 0.5) | 0.958 | 0.965 |
| BMI (kg/m2) | 22.6 | (2.8) | 22.8 | (2.9) | 23.0 | (3.0) | 0.2 | (0.1 to 0.3) | 0.4 | (0.3 to 0.5) | 0.898 | 0.915 |
| Men (n = 584) | ||||||||||||
| Height (cm) | 164.2 | (5.9) | 164.0 | (6.1) | 163.5 | (6.0) | −0.2 | (−0.4 to −0.1) | −0.7 | (−0.9 to −0.5) | 0.930 | 0.940 |
| Body weight (kg) | 61.8 | (8.4) | 62.3 | (8.4) | 62.4 | (8.8) | 0.5 | (0.3 to 0.7) | 0.6 | (0.3 to 0.8) | 0.958 | 0.952 |
| BMI (kg/m2) | 22.9 | (2.6) | 23.1 | (2.6) | 23.3 | (2.8) | 0.2 | (0.1 to 0.3) | 0.4 | (0.3 to 0.5) | 0.910 | 0.916 |
| Women (n = 585) | ||||||||||||
| Height (cm) | 151.4 | (5.4) | 151.4 | (5.3) | 150.4 | (5.5) | 0.0 | (−0.1 to 0.1) | −1.0 | (−1.1 to −0.8) | 0.940 | 0.920 |
| Body weight (kg) | 51.0 | (7.5) | 51.3 | (7.8) | 51.3 | (7.8) | 0.3 | (0.1 to 0.6) | 0.3 | (0.1 to 0.5) | 0.915 | 0.947 |
| BMI (kg/m2) | 22.2 | (3.0) | 22.4 | (3.1) | 22.7 | (3.1) | 0.2 | (0.1 to 0.3) | 0.5 | (0.3 to 0.6) | 0.888 | 0.912 |
| <75 years (n = 770) | ||||||||||||
| Height (cm) | 158.6 | (8.3) | 158.4 | (8.2) | 157.8 | (8.4) | −0.2 | (−0.3 to −0.1) | −0.8 | (−0.9 to −0.7) | 0.980 | 0.983 |
| Body weight (kg) | 57.2 | (9.6) | 57.5 | (9.8) | 57.5 | (10.0) | 0.3 | (0.1 to 0.5) | 0.3 | (0.2 to 0.5) | 0.957 | 0.964 |
| BMI (kg/m2) | 22.6 | (2.8) | 22.8 | (2.9) | 23.0 | (3.0) | 0.2 | (0.1 to 0.3) | 0.4 | (0.3 to 0.5) | 0.908 | 0.928 |
| ≥75 years (n = 399) | ||||||||||||
| Height (cm) | 156.2 | (8.9) | 156.2 | (8.9) | 155.2 | (9.1) | 0.0 | (−0.3 to 0.2) | −1.0 | (−1.3 to −0.7) | 0.954 | 0.946 |
| Body weight (kg) | 54.9 | (9.4) | 55.4 | (9.5) | 55.4 | (9.8) | 0.5 | (0.2 to 0.7) | 0.5 | (0.3 to 0.7) | 0.958 | 0.967 |
| BMI (kg/m2) | 22.4 | (2.8) | 22.6 | (2.9) | 22.9 | (3.0) | 0.2 | (0.1 to 0.3) | 0.5 | (0.4 to 0.6) | 0.879 | 0.889 |
| BMI Categories (kg/m2) | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| <18.5 (n = 562) | 18.5–19.9 (n = 787) | 20.0–22.4 (n = 2195) | 22.5–24.9 (n = 2218) | 25.0–27.4 (n = 1006) | ≥27.5 (n = 423) | |||||||
| Age (years) b | 75.6 | (6.8) | 74.0 | (6.7) | 73.3 | (6.2) | 72.9 | (5.8) | 72.8 | (5.9) | 72.8 | (5.8) |
| Women (n (%)) c | 388 | (69.0) | 465 | (59.1) | 1205 | (54.9) | 1012 | (45.6) | 469 | (46.6) | 249 | (58.9) |
| BMI (kg/m2) b | 17.3 | (1.0) | 19.3 | (1.5) | 21.3 | (1.6) | 23.7 | (1.9) | 26.1 | (2.3) | 29.6 | (2.2) |
| Alcohol drinker (n (%)) c | 304 | (54.1) | 484 | (61.5) | 1443 | (65.7) | 1504 | (67.8) | 685 | (68.1) | 271 | (64.1) |
| Current smoker (n (%)) c | 71 | (12.6) | 85 | (10.8) | 252 | (11.5) | 239 | (10.8) | 90 | (9.0) | 32 | (7.6) |
| Living alone (n (%)) c | 63 | (11.2) | 98 | (12.5) | 250 | (11.4) | 257 | (11.6) | 104 | (10.3) | 41 | (9.7) |
| HSE (n (%)) c | 184 | (32.7) | 271 | (34.4) | 779 | (35.5) | 738 | (33.3) | 347 | (34.5) | 123 | (29.1) |
| Education ≥13 y (n (%)) c | 104 | (18.5) | 181 | (23.0) | 467 | (21.3) | 535 | (24.1) | 225 | (22.4) | 95 | (22.5) |
| MVPA (n (%)) c | 186 | (33.1) | 341 | (43.3) | 1002 | (45.7) | 1045 | (47.1) | 441 | (43.8) | 145 | (34.3) |
| Denture use (n (%)) c | 370 | (65.8) | 484 | (61.5) | 1323 | (60.3) | 1311 | (59.1) | 600 | (59.6) | 244 | (57.7) |
| No medication (n (%)) c | 138 | (24.6) | 210 | (26.7) | 529 | (24.1) | 477 | (21.5) | 182 | (18.1) | 55 | (13.0) |
| Hypertension (n (%)) c | 126 | (22.4) | 227 | (28.8) | 774 | (35.3) | 934 | (42.1) | 507 | (50.4) | 221 | (52.3) |
| Stroke (n (%)) c | 21 | (3.7) | 27 | (3.4) | 76 | (3.5) | 90 | (4.1) | 37 | (3.7) | 18 | (4.3) |
| Heart disease (n (%)) c | 52 | (9.3) | 90 | (11.4) | 258 | (11.8) | 289 | (13.0) | 148 | (14.7) | 77 | (18.2) |
| Diabetes (n (%)) c | 41 | (7.3) | 63 | (8.0) | 213 | (9.7) | 236 | (10.6) | 127 | (12.6) | 83 | (19.6) |
| Hyperlipidemia (n (%)) c | 34 | (6.1) | 72 | (9.2) | 209 | (9.5) | 232 | (10.5) | 110 | (10.9) | 66 | (15.6) |
| BMI Categories (kg/m2) | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| <18.5 (n = 562) | 18.5–19.9 (n = 787) | 20.0–22.4 (n = 2195) | 22.5–24.9 (n = 2218) | 25.0–27.4 (n = 1006) | ≥27.5 (n = 423) | |||||||
| BMI (kg/m2) | 17.3 | (1.0) | 19.3 | (0.4) | 21.3 | (0.7) | 23.6 | (0.7) | 26.0 | (0.7) | 29.5 | (2.1) |
| FP model | ||||||||||||
| Case (n (%)) | 142 | (25.3) | 154 | (19.6) | 313 | (14.3) | 276 | (12.4) | 127 | (12.6) | 82 | (19.4) |
| Crude model | 2.34 | (1.84 to 2.97) | 1.69 | (1.34 to 2.12) | 1.15 | (0.96 to 1.38) | 1.00 | (Ref) | 0.98 | (0.78 to 1.25) | 1.66 | (1.25 to 2.20) |
| Model 1 b | 2.07 | (1.62 to 2.65) | 1.63 | (1.29 to 2.05) | 1.13 | (0.94 to 1.36) | 1.00 | (Ref) | 1.01 | (0.80 to 1.29) | 1.73 | (1.30 to 2.31) |
| Model 2 c | 2.04 | (1.58 to 2.63) | 1.69 | (1.33 to 2.14) | 1.16 | (0.96 to 1.41) | 1.00 | (Ref) | 1.00 | (0.78 to 1.27) | 1.54 | (1.15 to 2.07) |
| KCL | ||||||||||||
| Case (n (%)) | 312 | (55.5) | 297 | (37.7) | 750 | (34.2) | 723 | (32.6) | 345 | (34.3) | 208 | (49.2) |
| Crude model | 2.58 | (2.14 to 3.12) | 1.25 | (1.06 to 1.48) | 1.07 | (0.95 to 1.22) | 1.00 | (Ref) | 1.08 | (0.92 to 1.26) | 2.00 | (1.62 to 2.47) |
| Model 1 b | 2.08 | (1.70 to 2.54) | 1.13 | (0.94 to 1.35) | 1.01 | (0.88 to 1.15) | 1.00 | (Ref) | 1.10 | (0.93 to 1.30) | 2.13 | (1.71 to 2.66) |
| Model 2 c | 2.04 | (1.65 to 2.54) | 1.16 | (0.96 to 1.41) | 1.03 | (0.90 to 1.19) | 1.00 | (Ref) | 1.07 | (0.90 to 1.28) | 1.86 | (1.47 to 2.35) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Watanabe, D.; Yoshida, T.; Watanabe, Y.; Yamada, Y.; Kimura, M. A U-Shaped Relationship between the Prevalence of Frailty and Body Mass Index in Community-Dwelling Japanese Older Adults: The Kyoto–Kameoka Study. J. Clin. Med. 2020, 9, 1367. https://doi.org/10.3390/jcm9051367
Watanabe D, Yoshida T, Watanabe Y, Yamada Y, Kimura M. A U-Shaped Relationship between the Prevalence of Frailty and Body Mass Index in Community-Dwelling Japanese Older Adults: The Kyoto–Kameoka Study. Journal of Clinical Medicine. 2020; 9(5):1367. https://doi.org/10.3390/jcm9051367
Chicago/Turabian StyleWatanabe, Daiki, Tsukasa Yoshida, Yuya Watanabe, Yosuke Yamada, and Misaka Kimura. 2020. "A U-Shaped Relationship between the Prevalence of Frailty and Body Mass Index in Community-Dwelling Japanese Older Adults: The Kyoto–Kameoka Study" Journal of Clinical Medicine 9, no. 5: 1367. https://doi.org/10.3390/jcm9051367
APA StyleWatanabe, D., Yoshida, T., Watanabe, Y., Yamada, Y., & Kimura, M. (2020). A U-Shaped Relationship between the Prevalence of Frailty and Body Mass Index in Community-Dwelling Japanese Older Adults: The Kyoto–Kameoka Study. Journal of Clinical Medicine, 9(5), 1367. https://doi.org/10.3390/jcm9051367






