Which Child with Asthma is a Candidate for Biological Therapies?
Abstract
1. Introduction
- Is the eosinophil always the “bad guy” or could there be a down side to the aggressive, anti-eosinophil strategies which have been effective in adults?
- Is paediatric severe, therapy resistant asthma (STRA) the same as adult disease?
- What is a truly severe disease in childhood, in other words, is it only children with STRA who should receive these medications?
- How should we evaluate children referred for biological therapies?
- What are the paediatric data on the biologicals, and how do we match the right biological to the right child?
2. The Eosinophil: A Janus Cell, Facing Both Ways?
3. Adult and Paediatric STRA: Similarities and Differences
4. Are Sputum Phenotypes Stable in Paediatric Asthma?
5. What Is True STRA in Children?
6. How Should We Evaluate Children for Biological Therapies?
- Is there ongoing airway inflammation, and if so, what is the phenotype/endotype?
- Is any inflammation present steroid sensitive? (For example, corticosteroids are very effective against eosinophilic inflammation, but not in neutrophilic disease)
- Is there a disconnect between the degree of inflammation and the level of symptoms?
- Is there evidence of persistent airflow limitation?
7. What Are the Paediatric Data, and Who Should Get What Biologic?
7.1. Omalizumab
7.2. Mepolizumab
8. Limitations of Current Clinical Trials
9. Summary and Conclusions: Where from Here?
Conflicts of Interest
References
- Pavord, I.D.; Beasley, R.; Agusti, A.; Anderson, G.P.; Bel, E.; Brusselle, G.; Frey, U. After asthma—redefining airways diseases. Lancet 2018, 391, 350–400. [Google Scholar] [CrossRef]
- Brown, H.M. Treatment of chronic asthma with prednisolone; significance of eosinophils in the sputum. Lancet 1958, 2, 1245–1247. [Google Scholar] [CrossRef]
- Leckie, M.J.; ten Brinke, A.; Khan, J.; Diamant, Z.; O’Connor, B.J.; Walls, C.M.; Hansel, T.T. Effects of an interleukin-5 Blocking Monoclonal Antibody on Eosinophils, Airway Hyper-Responsiveness, and the Late Asthmatic Response. Lancet 2000, 356, 2144–2148. [Google Scholar] [CrossRef]
- Nair, P.; Pizzichini, M.M.; Kjarsgaard, M.; Inman, M.D.; Efthimiadis, A.; Pizzichini, E.; O’Byrne, P.M. Mepolizumab for Prednisone-Dependent Asthma with Sputum Eosinophilia. N. Engl. J. Med. 2009, 360, 985–993. [Google Scholar] [CrossRef]
- Haldar, P.; Brightling, C.E.; Hargadon, B.; Gupta, S.; Monteiro, W.; Sousa, A.; Pavord, I.D. Mepolizumab and exacerbations of refractory eosinophilic asthma. N. Engl. J. Med. 2009, 360, 973–984. [Google Scholar] [CrossRef]
- De Boeck, K.; Amaral, M.D. Progress in Therapies for Cystic Fibrosis. Lancet Respir. Med. 2016, 4, 662–674. [Google Scholar] [CrossRef]
- Ramsey, B.W.; Davies, J.; McElvaney, N.G.; Tullis, E.; Bell, S.C.; Dřevínek, P.; Moss, R. VX08-770-102 Study Group. A CFTR Potentiator in Patients with Cystic Fibrosis and the G551D Mutation. N. Engl. J. Med. 2011, 365, 1663–1672. [Google Scholar] [CrossRef] [PubMed]
- Holguin, F.; Cardet, J.C.; Chung, K.F.; Diver, S.; Ferreira, D.S.; Fitzpatrick, A.; Gaga, M.; Kellermeyer, L.; Khurana, S.; Knight, S.; et al. Management of Severe Asthma: A European Respiratory Society/American Thoracic Society Guideline. Eur. Respir. J. 2019, 26, 1900588. [Google Scholar] [CrossRef]
- Pavord, I.D.; Korn, S.; Howarth, P.; Bleecker, E.R.; Buhl, R.; Keene, O.N.; Chanez, P. Mepolizumab for severe eosinophilic asthma (DREAM): A multicentre, double-blind, placebo-controlled trial. Lancet 2012, 380, 651–659. [Google Scholar] [CrossRef]
- Pavlidis, S.; Takahashi, K.; Kwong, F.N.K.; Xie, J.; Hoda, U.; Sun, K.; Chanez, P. “T2-high” in severe asthma related to blood eosinophil, exhaled nitric oxide and serum periostin. Eur. Respir. J. 2018, 53, 1800938. [Google Scholar] [CrossRef] [PubMed]
- Kuo, C.H.S.; Pavlidis, S.; Loza, M.; Baribaud, F.; Rowe, A.; Pandis, I.; Sterk, P.J. T-helper Cell Type 2 (Th2) and non-Th2 Molecular Phenotypes of Asthma Using Sputum Transcriptomics in U-BIOPRED. Eur. Respir. J. 2017, 49, 28179442. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.B.; Weller, P.F. Pivotal advance: Eosinophils mediate early alum adjuvant-elicited B cell priming and IgM production. J. Leukoc. Biol. 2008, 83, 817–821. [Google Scholar] [CrossRef] [PubMed]
- Fröhlich, A.; Steinhauser, G.; Scheel, T.; Roch, T.; Fillatreau, S.; Lee, J.J.; Berek, C. Eosinophils are required for the maintenance of plasma cells in the bone marrow. Nat. Immunol. 2011, 12, 151–159. [Google Scholar]
- Xenakis, J.J.; Howard, E.D.; Smith, K.M.; Olbrich, C.L.; Huang, Y.; Anketell, D.; Spencer, L.A. Resident intestinal eosinophils constitutively express antigen presentation markers and include two phenotypically distinct subsets of eosinophils. Immunology 2018, 154, 298–308. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; Molofsky, A.B.; Liang, H.E.; Ricardo-Gonzalez, R.R.; Jouihan, H.A.; Bando, J.K.; Locksley, R.M. Eosinophils sustain adipose alternatively activated macrophages associated with glucose homeostasis. Science 2011, 332, 243–247. [Google Scholar] [CrossRef]
- Qiu, Y.; Nguyen, K.D.; Odegaard, J.I.; Cui, X.; Tian, X.; Locksley, R.M.; Chawla, A. Eosinophils and type 2 cytokine signaling in macrophages orchestrate development of functional beige fat. Cell 2014, 157, 1292–1308. [Google Scholar] [CrossRef]
- Sabogal Piñeros, Y.S.; Bal, S.M.; Dijkhuis, A.; Majoor, C.J.; Dierdorp, B.S.; Dekker, T.; Koenderman, L. Eosinophils Capture Viruses, a Capacity That Is Defective in Asthma. Allergy 2019, 74, 1898–1909. [Google Scholar] [CrossRef]
- Gupta, A.; Ikeda, M.; Geng, B.; Azmi, J.; Price, R.G.; Bradford, E.S.; Yancey, S.W.; Steinfeld, J. Long-term Safety and Pharmacodynamics of Mepolizumab in Children with Severe Asthma With an Eosinophilic Phenotype. J. Allergy Clin. Immunol. 2019, 144, 1336–1342. [Google Scholar] [CrossRef]
- Saglani, S.; Lui, S.; Ullmann, N.; Campbell, G.A.; Sherburn, R.T.; Mathie, S.A.; Denney, L.; Bossley, C.J.; Oates, T.; Walker, S.A.; et al. IL-33 promotes airway remodeling in pediatric patients with severe, steroid-resistant asthma. J. Allergy Clin. Immunol. 2013, 132, 676–685. [Google Scholar] [CrossRef]
- Castanhinha, S.; Sherburn, R.; Walker, S.; Gupta, A.; Bossley, C.J.; Buckley, J.; Ullmann, N.; Grychtol, R.; Campbell, G.; Maglione, M.; et al. Pediatric severe asthma with fungal sensitization is mediated by steroid-resistant IL-33. J. Allergy Clin. Immunol. 2015, 136, 312–322. [Google Scholar] [CrossRef]
- Fitzpatrick, A.M. National Institutes of Health/National Heart, Lung and Blood Institute’s Severe Asthma Research Program. The molecular phenotype of severe asthma in children. J. Allergy Clin. Immunol. 2010, 125, 851–857. [Google Scholar] [CrossRef] [PubMed]
- Wisniewski, J.A.; Muehling, L.M.; Eccles, J.D.; Capaldo, B.J.; Agrawal, R.; Shirley, D.A.; Teague, W.G. TH1 signatures are present in the lower airways of children with severe asthma, regardless of allergic status. J. Allergy Clin. Immunol. 2018, 141, 2048–2060. [Google Scholar] [CrossRef] [PubMed]
- Fleming, L.; Tsartsali, L.; Wilson, N.; Regamey, N.; Bush, A. Sputum inflammatory phenotypes are not stable in children with asthma. Thorax 2012, 67, 675–681. [Google Scholar] [CrossRef] [PubMed]
- Wark, P.A.B.; Simpson, J.; Hensley, M.J.; Gibson, P.G. Airway Inflammation in Thunderstorm Asthma. Clin. Exp. Allergy 2002, 32, 1750–1756. [Google Scholar] [CrossRef] [PubMed]
- Petsky, H.L.; Cates, C.J.; Kew, K.M.; Chang, A.B. Tailoring Asthma Treatment on Eosinophilic Markers (Exhaled Nitric Oxide or Sputum Eosinophils): A Systematic Review and Meta-Analysis. Thorax 2018, 73, 1110–1119. [Google Scholar] [CrossRef]
- Van den Berge, M.; Pauw, R.G.; de Monchy, J.G.; van Minnen, C.A.; Postma, D.S.; Kerstjens, H.A. Beneficial effects of treatment with anti-IgE antibodies (Omalizumab) in a patient with severe asthma and negative skin-prick test results. Chest 2011, 139, 190–193. [Google Scholar] [CrossRef]
- Fitzpatrick, A.M.; Jackson, D.J.; Mauger, D.T.; Boehmer, S.J.; Phipatanakul, W.; Sheehan, W.J.; Covar, R. NIH/NHLBI AsthmaNet. Individualized Therapy for Persistent Asthma in Young Children. J. Allergy Clin. Immunol. 2016, 138, 1608–1618.e12. [Google Scholar] [CrossRef]
- Ullmann, N.; Bossley, C.J.; Fleming, L.; Silvestri, M.; Bush, A.; Saglani, S. Blood eosinophil counts rarely reflect airway eosinophilia in children with severe asthma. Allergy 2013, 68, 402–406. [Google Scholar] [CrossRef]
- Fleming, L.; Koo, M.; Bossley, C.J.; Nagakumar, P.; Bush, A.; Saglani, S. The utility of a multidomain assessment of steroid response for predicting clinical response to omalizumab. J. Allergy Clin. Immunol. 2016, 138, 292–294. [Google Scholar] [CrossRef]
- Fleming, L.; Tsartsali, L.; Wilson, N.; Regamey, N.; Bush, A. Longitudinal Relationship between Sputum Eosinophils and Exhaled Nitric Oxide in Children with Asthma. Am. J. Respir. Crit. Care Med. 2013, 188, 400–402. [Google Scholar] [CrossRef]
- Shrimanker, R.; Keene, O.; Hynes, G.; Wenzel, S.; Yancey, S.; Pavord, I.D. Prognostic and predictive value of blood eosinophil count, fractional exhaled nitric oxide, and their combination in severe asthma: A post hoc analysis. Am. J. Respir. Crit. Care Med. 2019, 200, 1308–1311. [Google Scholar] [CrossRef] [PubMed]
- Bracken, M.B.; Fleming, L.; Hall, P.; Van Stiphout, N.; Bossley, C.J.; Biggart, E.; Wilson, N.M.; Bush, A. The importance of nurse led home visits in the assessment of children with problematic asthma. Arch. Dis. Child 2009, 94, 780–784. [Google Scholar] [CrossRef] [PubMed]
- Strunk, R.C.; Bacharier, L.B.; Phillips, B.R.; Szefler, S.J.; Zeiger, R.S.; Chinchilli, V.M.; Morgan, W.J. CARE Network. Azithromycin or montelukast as inhaled corticosteroid-sparing agents in moderate-to-severe childhood asthma study. J. Allergy Clin. Immunol. 2008, 122, 1138–1144. [Google Scholar] [CrossRef] [PubMed]
- Szefler, S.J.; Mitchell, H.; Sorkness, C.A.; Gergen, P.J.; T O’Connor, G.; Morgan, W.J.; Eggleston, P.A. Management of asthma based on exhaled nitric oxide in addition to guideline-based treatment for inner-city adolescents and young adults: A randomised controlled trial. Lancet 2008, 372, 1065–1072. [Google Scholar] [CrossRef]
- Lemanske, R.F., Jr.; Mauger, D.T.; Sorkness, C.A.; Jackson, D.J.; Boehmer, S.J.; Martinez, F.D.; Covar, R.A. Childhood Asthma Research and Education (CARE) Network of the National Heart, Lung, and Blood Institute. Step-up therapy for children with uncontrolled asthma receiving inhaled corticosteroids. N. Engl. J. Med. 2010, 362, 975–985. [Google Scholar] [CrossRef]
- Chung, K.F.; Wenzel, S.E.; Brozek, J.L.; Bush, A.; Castro, M.; Sterk, P.J.; Adcock, I.M.; Bateman, E.D.; Bel, E.H.; Bleecker, E.R.; et al. International ERS/ATS guidelines on definition, evaluation and treatment of severe asthma. Eur. Respir. J. 2014, 43, 343–373. [Google Scholar] [CrossRef]
- Available online: https://www.rcplondon.ac.uk/file/868/download?token=JQzyNWUs (accessed on 23 April 2020).
- Suissa, S.; Ernst, P.; Boivin, J.F.; Horwitz, R.I.; Habbick, B.; Cockroft, D.; Blais, L.; McNutt, M.; Buist, A.S.; Spitzer, W.O. A cohort analysis of excess mortality in asthma and the use of inhaled beta-agonists. Am. J. Respir. Crit. Care Med. 1994, 149, 604–610. [Google Scholar] [CrossRef]
- Spitzer, W.O.; Suissa, S.; Ernst, P.; Horwitz, R.I.; Habbick, B.; Cockcroft, D.; Rebuck, A.S. The use of β-agonists and the risk of death and near death from asthma. N. Engl. J. Med. 1992, 326, 501–506. [Google Scholar] [CrossRef]
- Bush, A.; Saglani, S. Management of severe asthma in children. Lancet 2010, 376, 814–825. [Google Scholar] [CrossRef]
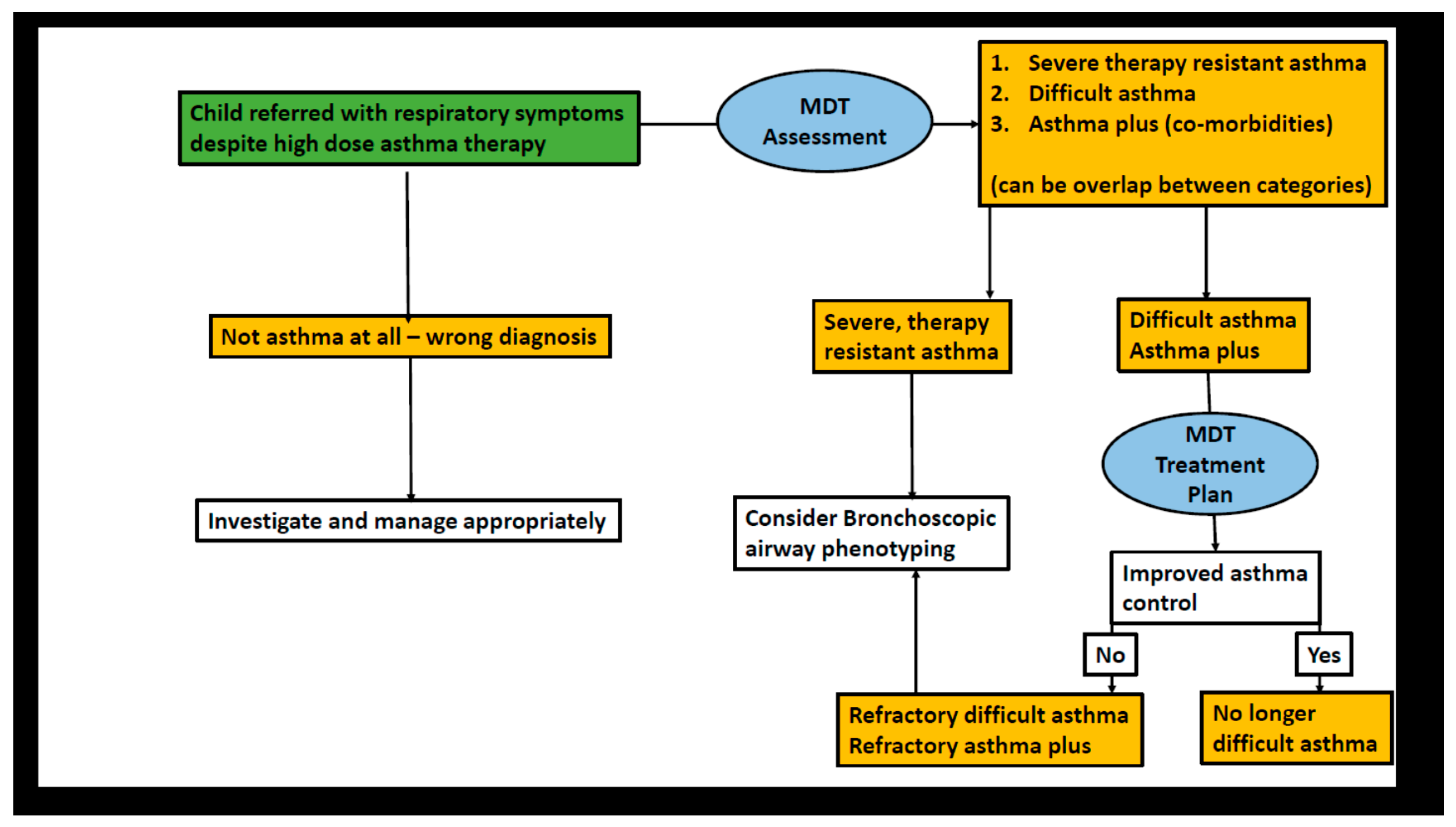
- Cook, J.; Beresford, F.; Fainardi, V.; Hall, P.; Housley, G.; Jamalzadeh, A.; Nightingale, M.; Winch, D.; Bush, A.; Fleming, L.; et al. Managing the paediatric patient with refractory asthma: A multidisciplinary approach. J. Asthma Allergy 2017, 10, 123–130. [Google Scholar] [CrossRef]
- Bush, A.; Fleming, L.; Saglani, S. Severe asthma in children. Respirology 2017, 22, 886–897. [Google Scholar] [CrossRef] [PubMed]
- Halvorsen, T.; Walsted, E.S.; Bucca, C.; Bush, A.; Cantarella, G.; Friedrich, G.; Herth, F.J.F.; Hull, J.H.; Jung, H.; Maat, R.; et al. Inducible laryngeal obstruction: An official joint European Respiratory Society and European Laryngological Society statement. Eur. Respir. J. 2017, 50, 1602221. [Google Scholar] [CrossRef] [PubMed]
- Sharples, J.; Gupta, A.; Fleming, L.; Bossley, C.J.; Bracken-King, M.; Hall, P.; Hayward, A.; Puckey, M.; Balfour-Lynn, I.M.; Rosenthal, M.; et al. Long-term effectiveness of a staged assessment for paediatric problematic severe asthma [Research Letter]. Eur. Respir. J. 2012, 40, 264–267. [Google Scholar] [CrossRef] [PubMed]
- Bush, A.; Saglani, S.; Fleming, L. Severe asthma: Looking beyond the amount of medication. Lancet Respir. Med. 2017, 5, 844–846. [Google Scholar] [CrossRef]
- Desai, D.; Newby, C.; Symon, F.A.; Haldar, P.; Shah, S.; Gupta, S.; Herath, A. Elevated sputum interleukin-5 and submucosal eosinophilia in obese individuals with severe asthma. Am. J. Respir. Crit. Care Med. 2013, 188, 657–663. [Google Scholar] [CrossRef] [PubMed]
- Van Huisstede, A.; Rudolphus, A.; van Schadewijk, A.; Cabezas, M.C.; Mannaerts, G.H.; Taube, C.; Braunstahl, G.J. Bronchial and systemic inflammation in morbidly obese subjects with asthma: A biopsy study. Am. J. Respir. Crit. Care Med. 2014, 190, 951–954. [Google Scholar] [CrossRef] [PubMed]
- Forno, E.; Weiner, D.J.; Mullen, J.; Sawicki, G.; Kurland, G.; Han, Y.Y.; Cloutier, M.M.; Canino, G.; Weiss, S.T.; Litonjua, A.A.; et al. Obesity and Airway Dysanapsis in Children with and without Asthma. Am. J. Respir. Crit. Care Med. 2017, 195, 314–323. [Google Scholar] [CrossRef] [PubMed]
- Peters, M.C.; McGrath, K.W.; Hawkins, G.A.; Hastie, A.T.; Levy, B.D.; Israel, E.; Johansson, M.W. National Heart, Lung, and Blood Institute Severe Asthma Research Program. Plasma interleukin-6 concentrations, metabolic dysfunction, and asthma severity: A cross-sectional analysis of two cohorts. Lancet Respir. Med. 2016, 4, 574–584. [Google Scholar] [CrossRef]
- Johansson, H.; Norlander, K.; Berglund, L.; Janson, C.; Malinovschi, A.; Nordvall, L.; Emtner, M. Prevalence of exercise-induced bronchoconstriction and exercise-induced laryngeal obstruction in a general adolescent population. Thorax 2015, 70, 57–63. [Google Scholar] [CrossRef]
- Vogelberg, C.; Szefler, S.J.; Vrijlandt, E.J.; Boner, A.L.; Engel, M.; El Azzi, G.; Hamelmann, E.H. Tiotropium add-on therapy is safe and reduces seasonal worsening in paediatric asthma patients. Eur. Respir. J. 2019, 53, 31097514. [Google Scholar] [CrossRef]
- Lazarus, S.C.; Krishnan, J.A.; King, T.S.; Lang, J.E.; Blake, K.V.; Covar, R.; Dyer, A.M. National Heart, Lung, and Blood Institute AsthmaNet. Mometasone or Tiotropium in Mild Asthma with a Low Sputum Eosinophil Level. N. Engl. J. Med. 2019, 380, 2009–2019. [Google Scholar] [CrossRef] [PubMed]
- Normansell, R.; Walker, S.; Milan, S.J.; Walters, E.H.; Nair, P. Omalizumab for asthma in adults and children. Cochrane Database Syst. Rev. 2014, 1, CD003559. [Google Scholar] [CrossRef] [PubMed]
- Buelo, A.; McLean, S.; Julious, S.; Flores-Kim, J.; Bush, A.; Henderson, J.; Paton, J.Y.; Sheikh, A.; Shields, M.; Pinnock, H. At-risk children with asthma (ARC): A systematic review. Thorax 2018, 73, 813–824. [Google Scholar] [CrossRef] [PubMed]
- Jochmann, A.; Artusio, L.; Jamalzadeh, A.; Nagakumar, P.; Delgado-Eckert, E.; Saglani, S.; Bush, A.; Frey, U.; Fleming, L. Electronic monitoring of adherence to inhaled corticosteroids: An essential tool in identifying severe asthma in children. Eur. Respir. J. 2017, 50, 1700910. [Google Scholar] [CrossRef]
- Busse, W.W.; Morgan, W.J.; Gergen, P.J.; Mitchell, H.E.; Gern, J.E.; Liu, A.H.; Gruchalla, R.S.; Kattan, M.; Teach, S.J.; Pongracic, J.A.; et al. Randomized trial of omalizumab (anti-IgE) for asthma in inner-city children. N. Engl. J. Med. 2011, 364, 1005–1015. [Google Scholar] [CrossRef]
- Teach, S.J.; Gill, M.A.; Togias, A.; Sorkness, C.A.; Arbes, S.J., Jr.; Calatroni, A.; Kercsmar, C.M. Preseasonal Treatment with Either Omalizumab or an Inhaled Corticosteroid Boost to Prevent Fall Asthma Exacerbations. J. Allergy Clin. Immunol. 2015, 136, 1476–1485. [Google Scholar] [CrossRef]
- Saglani, S.; Bush, A.; Carroll, W.; Cunningham, S.; Fleming, L.; Gaillard, E.; Roberts, G. Biologics for paediatric severe asthma: Trick or TREAT? Lancet Respir. Med. 2019, 7, 294–296. [Google Scholar] [CrossRef]
- Russell, R.J.; Chachi, L.; FitzGerald, J.M.; Backer, V.; Olivenstein, R.; Titlestad, I.L.; Leaker, B. MESOS study investigators. Effect of Tralokinumab, an interleukin-13 Neutralising Monoclonal Antibody, on Eosinophilic Airway Inflammation in Uncontrolled Moderate-To-Severe Asthma (MESOS): A Multicentre, Double-Blind, Randomised, Placebo-Controlled Phase 2 Trial. Lancet Respir. Med. 2018, 6, 499–510. [Google Scholar]
- Saglani, S.; Mathie, S.A.; Gregory, L.G.; Bell, M.J.; Bush, A.; Lloyd, C.M. Pathophysiological Features of Asthma Develop in Parallel in House Dust Mite Exposed Neonatal Mice. Am. J. Respir. Cell Mol. Biol. 2009, 41, 281–289. [Google Scholar] [CrossRef]
- Turner, S.; Custovic, A.; Ghazal, P.; Grigg, J.; Gore, M.; Henderson, J.; Lloyd, C.M.; Marsland, B.; Power, U.F.; Roberts, G.; et al. Pulmonary epithelial barrier and immunological functions at birth and in early life - key determinants of the development of asthma? A description of the protocol for the Breathing Together study. Wellcome Open Res. 2018, 3, 60. [Google Scholar] [CrossRef]
- Guilbert, T.W.; Morgan, W.J.; Zeiger, R.S.; Mauger, D.T.; Boehmer, S.J.; Szefler, S.J.; Bloomberg, G.R. Long-term inhaled corticosteroids in preschool children at high risk for asthma. N. Engl. J. Med. 2006, 354, 1985–1997. [Google Scholar] [CrossRef] [PubMed]
- Murray, C.S.; Woodcock, A.; Langley, S.J.; Morris, J.; Custovic, A. Secondary prevention of asthma by the use of Inhaled Fluticasone propionate in Wheezy INfants (IFWIN): Double-blind, randomised, controlled study. Lancet 2006, 368, 754–762. [Google Scholar] [CrossRef]
- Bisgaard, H.; Hermansen, M.N.; Loland, L.; Halkjaer, L.B.; Buchvald, F. Intermittent inhaled corticosteroids in infants with episodic wheezing. N. Engl. J. Med. 2006, 354, 1998–2005. [Google Scholar] [CrossRef] [PubMed]
- Guilbert, T.W.; Morgan, W.J.; Krawiec, M.; Lemanske, R.F., Jr.; Sorkness, C.; Szefler, S.J.; Strunk, R.C. The Prevention of Early Asthma in Kids study: Design, rationale and methods for the Childhood Asthma Research and Education network. Control. Clin. Trials 2004, 25, 286–310. [Google Scholar] [CrossRef]
- Devulapalli, C.S.; Carlsen, K.C.L.; Håland, G.; Munthe-Kaas, M.C.; Pettersen, M.; Mowinckel, P.; Carlsen, K.H. Severity of Obstructive Airways Disease by Age 2 Years Predicts Asthma at 10 Years of Age. Thorax 2008, 63, 8–13. [Google Scholar] [CrossRef]
| Level of Medication | Asthma Functional Deficit |
|---|---|
| Asthma which is only controlled or uncontrolled on therapy with ≥ 800 mcg/day BDP equivalent plus additional controllers (LABA, LTRA. Theophylline) or failed trials of these agents | Poor symptom control, e.g., Asthma Control Test (ACT) <20 |
| ≥2 bursts of systemic corticosteroids (≥3 days each) in the previous year | |
| Serious exacerbations (≥1 hospitalisation or PICU stay) in the previous year | |
| Airflow limitation: FEV1 < 80% predicted following SABA and LABA withhold |
| Issue to be Addressed | Tests Performed |
|---|---|
| Symptom pattern | ACT or c-ACT, asthma attacks and prednisolone bursts, unscheduled emergency visits; evidence of severity of symptoms at emergency presentation School attendance and impact of symptoms at school |
| Breathing pattern disorder | Physiotherapy assessment Consider asking parents to make a video of breathing pattern Consider laryngoscopy during exercise |
| Psychosocial factors | Questionnaires relating to treatment burden, anxiety and depression, quality of life |
| Physiology | Spirometry before and after bronchodilator Lung clearance index |
| Allergic sensitization | Total IgE skin prick tests and specific IgE to grass and tree pollen, house dust mite, cockroach, cat and dog, aspergillus, alternaria and cladosporium and any likely relevant other antigens Not food allergens unless a suggestive clinical history |
| Airway inflammation | FeNO Induced sputum cytospin for eosinophil count Peripheral blood eosinophil count |
| Nicotine exposure (tobacco or vaping, passive or active) | Urine cotinine |
| Medication adherence | Prescription uptake Serum prednisolone and theophylline levels if prescribed; serum inhaled corticosteroid levels if available (usually only in a research context) Electronic monitoring |
| Tests | First Visit | Second Visit | Third Visit |
|---|---|---|---|
| Non-invasive | Assessment of current symptoms Spirometry before and after SABA LCI FENO Induced sputum eosinophils | Assessment of current symptoms Spirometry before and after SABA LCI FENO Induced sputum eosinophils | Assessment of current symptoms Spirometry before and after SABA LCI FENO Induced sputum eosinophils |
| Invasive | Fibreoptic bronchocopy, BAL, endobronchial biopsy | ||
| Actions | Intramuscular triamcinolone (steroid trial) | Assess steroid responsiveness Develop bespoke treatment plan | Assess response to treatment |
© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bush, A. Which Child with Asthma is a Candidate for Biological Therapies? J. Clin. Med. 2020, 9, 1237. https://doi.org/10.3390/jcm9041237
Bush A. Which Child with Asthma is a Candidate for Biological Therapies? Journal of Clinical Medicine. 2020; 9(4):1237. https://doi.org/10.3390/jcm9041237
Chicago/Turabian StyleBush, Andrew. 2020. "Which Child with Asthma is a Candidate for Biological Therapies?" Journal of Clinical Medicine 9, no. 4: 1237. https://doi.org/10.3390/jcm9041237
APA StyleBush, A. (2020). Which Child with Asthma is a Candidate for Biological Therapies? Journal of Clinical Medicine, 9(4), 1237. https://doi.org/10.3390/jcm9041237





