Associations between Disease Activity/Severity and Damage and Health-Related Quality of Life in Adult Patients with Localized Scleroderma—A Comparison of LoSCAT and Visual Analogue Scales
Abstract
1. Introduction
2. Experimental Section
2.1. Materials and Methods
2.1.1. Study Design and Patient Selection
2.1.2. LoSCAT and VASs
2.1.3. HRQoL
2.1.4. Statistical Analysis
3. Results
3.1. Study Participants Characteristics
3.2. Reliability and Concurrent Validity of LoSCAT and VASs
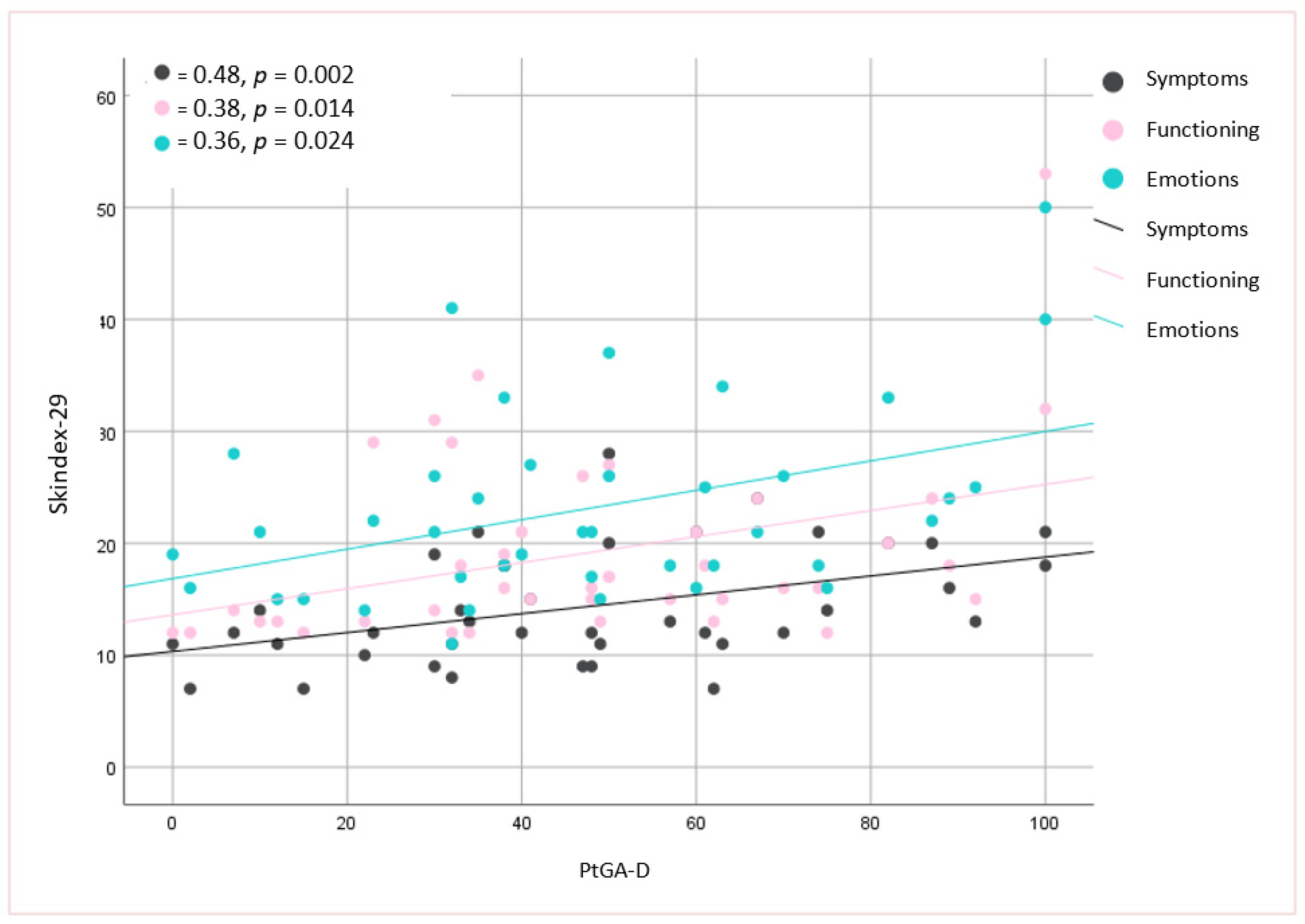
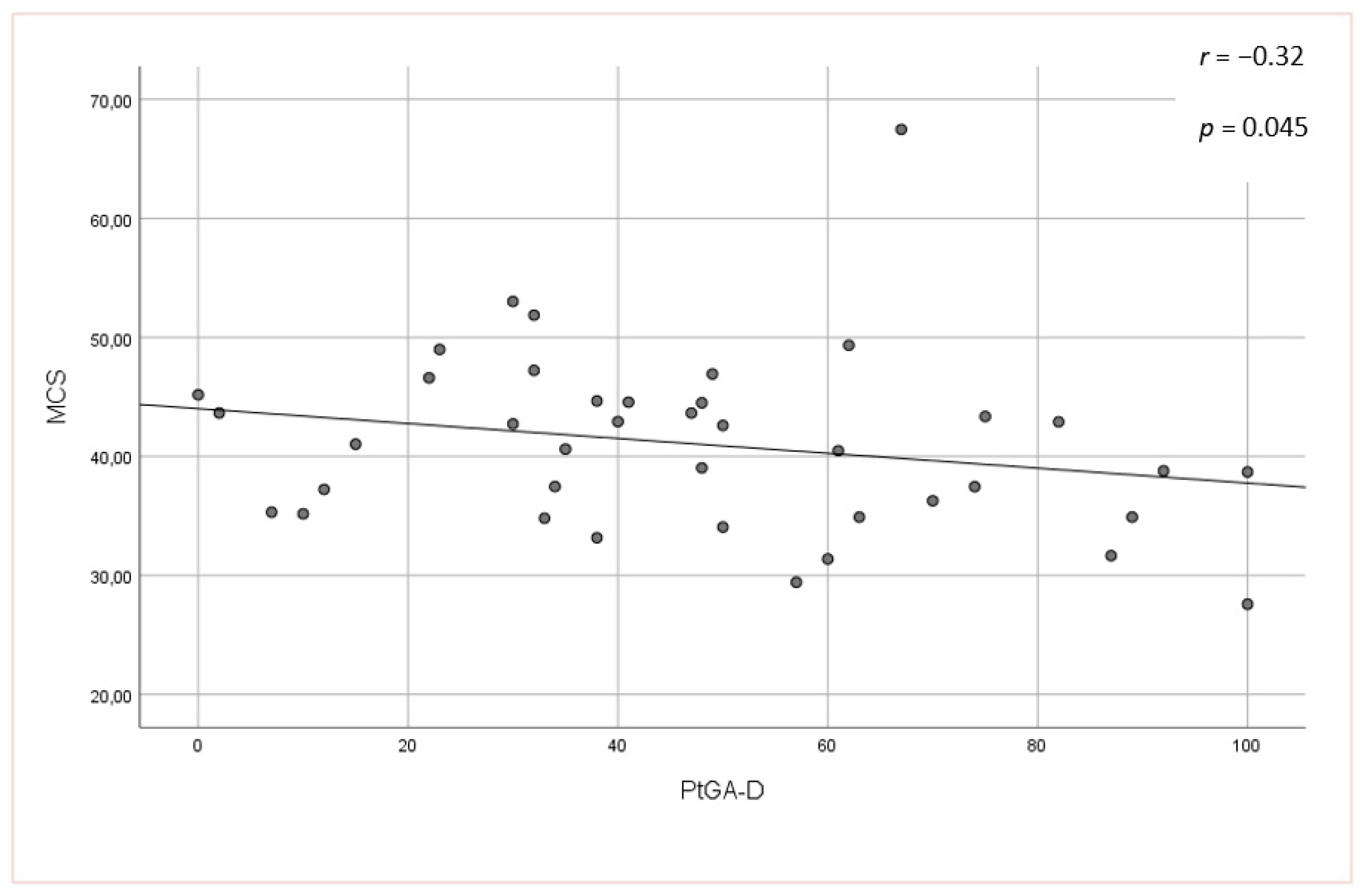
3.3. Relationship between Clinical Assessment and HRQoL
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Lis-Święty, A.; Skrzypek-Salamon, A.; Ranosz-Janicka, I.; Brzezińska-Wcisło, L. Localized scleroderma: Clinical and epidemiological features with emphasis on adulthood- versus childhood-onset disease differences. J. Eur. Acad. Dermatol. Venereol. 2017, 31, 1595–1603. [Google Scholar] [CrossRef] [PubMed]
- Kreuter, A.; Krieg, T.; Worm, M.; Wenzel, J.; Moinzadeh, P.; Kuhn, A. German guidelines for the diagnosis and therapy of localized scleroderma. J. Dtsch. Dermatol. Ges. 2016, 14, 199–216. [Google Scholar] [CrossRef]
- Arkachaisri, T.; Vilaiyuk, S.; Li, S.; O’Neil, K.M.; Pope, E.; Higgins, G.C. The localized scleroderma skin severity index and physician global assessment of disease activity: A work in progress toward development of localized scleroderma outcome measures. J. Rheumatol. 2013, 36, 2819–2829. [Google Scholar] [CrossRef] [PubMed]
- Zulian, F.; Vallongo, C.; Woo, P.; Russo, R.; Ruperto, N.; Harper, J. Localized scleroderma in childhood is not just a skin disease. Arthritis Rheum. 2005, 52, 2873–2881. [Google Scholar] [CrossRef] [PubMed]
- Marzano, A.V.; Menni, S.; Parodi, A.; Borghi, A.; Fuligni, A.; Fabbri, P.; Caputo, R. Localized scleroderma in adults and children. Clinical and laboratory investigations on 239 cases. Eur. J. Dermatol. 2003, 13, 171–176. [Google Scholar] [PubMed]
- Kreuter, A.; Wischnewski, J.; Terras, S.; Altmeyer, P.; Stücker, M.; Gambichler, T. Coexistence of lichen sclerosus and morphea: A retrospective analysis of 472 patients with localized scleroderma from a German tertiary referral center. J. Am. Acad. Dermatol. 2012, 67, 1157–1162. [Google Scholar] [CrossRef] [PubMed]
- Lis-Święty, A.; Janicka, I.; Skrzypek-Salamon, A.; Brzezińska-Wcisło, L. A systematic review of tools for determining activity of localized scleroderma in paediatric and adult patients. J. Eur. Acad. Dermatol. Venereol. 2017, 31, 30–37. [Google Scholar] [CrossRef] [PubMed]
- Arkachaisri, T.; Vilaiyuk, S.; Torok, K.S.; Medsger, T.A., Jr. Development and initial validation of the localized scleroderma skin damage index and physician global assessment of disease damage: A proof-of-concept study. Rheumatology (Oxford) 2010, 49, 373–381. [Google Scholar] [CrossRef] [PubMed]
- Kroft, E.B.; de Jong, E.M.; Evers, A.W. Physical Burden of Symptoms in Patients with Localized Scleroderma and Eosinophilic Fasciitis. Arch. Dermatol. 2008, 144, 1394–1395. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Klimas, N.K.; Shedd, A.D.; Bernstein, I.H.; Jacobe, H. Health-related quality of life in morphoea. Br. J. Dermatol. 2015, 172, 1329–1337. [Google Scholar] [CrossRef] [PubMed]
- Das, S.; Bernstein, I.; Jacobe, H. Correlates of self-reported quality of life in adults and children with morphea. J. Am. Acad Dermatol. 2014, 70, 904–910. [Google Scholar] [CrossRef] [PubMed]
- Condie, D.; Grabell, D.; Jacobe, H. Comparison of outcomes in adults with pediatric-onset morphea and those with adult-onset morphea: A cross-sectional study from the morphea in adults and children cohort. Arthritis Rheumatol. 2014, 66, 3496–3504. [Google Scholar] [CrossRef] [PubMed]
- Ardalan, K.; Zigler, C.K.; Torok, K.S. Predictors of Longitudinal Quality of Life in Juvenile Localized Scleroderma. Arthritis Care Res (Hoboken) 2017, 69, 1082–1087. [Google Scholar] [CrossRef] [PubMed]
- Kreuter, A.; Hyun, J.; Stucker, M.; Sommer, A.; Altmeyer, P.; Gambichler, T. A randomized controlled study of low-dose UVA1, medium-dose UVA1, and narrowband UVB phototherapy in the treatment of localized scleroderma. J. Am. Acad. Dermatol. 2006, 54, 440–447. [Google Scholar] [CrossRef] [PubMed]
- Skrzypek-Salamon, A.; Lis-Swiety, A.; Ranosz-Janicka, I.; Brzezińska-Wcisło, L. Localized Scleroderma Cutaneous Assessment Tool (LoSCAT) adapted for use in adult patients: Report from an initial validation study. Health Qual Life Outcomes 2018, 16, 185. [Google Scholar] [CrossRef] [PubMed]
- Prinsen, C. Interpretation of Skindex-29 scores: Cutoffs for mild, moderate, and severe impairment of health-related quality of life. Ther. Drug Monit. 2007, 29, 566–570. [Google Scholar] [CrossRef] [PubMed]
- Dancey, C.P.; Reidy, J. Pearson Education; Statistics without Maths for Psychology: Using SPSS for Windows; Prentice Hall: London, UK, 2007. [Google Scholar]
- Szramka-Pawlak, B.; Danczak-Pazdrowska, A.; Rzepa, T.; Szewczyk, A.; Sadowska-Przytocka, A.; Zaba, R. Health-related quality of life, optimism, and coping strategies in persons suffering from localized scleroderma. Psychol Health Med. 2013, 18, 654–663. [Google Scholar] [CrossRef] [PubMed]
- Bali, G.; Kárpáti, S.; Sárdy, M.; Brodszky, V.; Hidvégi, B.; Rencz, F. Association between quality of life and clinical characteristics in patients with morphea. Qual Life Res. 2018, 27, 2525–2532. [Google Scholar] [CrossRef] [PubMed]

| Variable | Level of Localized Scleroderma Activity | ||||
|---|---|---|---|---|---|
| Lack | Mild | Moderate | High | Very High | |
| New or enlarged skin lesions | - | - | + | + | + |
| Erythema | - | + | + | + | + |
| Skin thickness | - | +/− | +/− | +/− | +/− |
| Subjective symptoms (e.g., itch, pain, burning sensations) | - | - | - | +/− | +/− |
| General symptoms (e.g., weakness, arthralgia, uveitis) * | - | - | - | + | + |
| Laboratory signs (e.g., elevated erythrocyte sedimentation rate and/or C-reactive protein level) * | - | - | - | - | + |
| Variable | Level of Damage in Localized Scleroderma | ||||
|---|---|---|---|---|---|
| Lack | Mild | Moderate | High | Very High | |
| Dyspigmentation | - | + | + | + | + |
| Dermal atrophy | - | - | + | + | + |
| Subcutaneous atrophy | - | - | +/− | + | + |
| Muscle atrophy | - | - | - | +/− | +/− |
| Bone atrophy | - | - | - | +/− | +/− |
| Functional limitations due to extracutaneous manifestations (e.g., orthopedic, neurological, eye complications) | - | - | - | - | + |
| Decreased HRQoL * | |||||
| Item | Min-Max | Mean (±SD) |
|---|---|---|
| mLoSSI | 0–29 | 7.15 (±7.21) |
| New or enlarged skin lesions (N/E) | 0–6 | 0.83 (±1.66) |
| Erythema (ER) | 0–19 | 3.48 (±3.78) |
| Skin thickness (ST) | 0–15 | 2.85 (±3.534) |
| LoSDI | 0–52 | 10.42 (±9.82) |
| Dermal atrophy (DAT) | 0–25 | 4.28 (±4.64) |
| Subcutaneous atrophy (SAT) | 0–12 | 1.55 (±2.76) |
| Dyspigmentation (DP) | 0–15 | 4.6 (±3.59) |
| PhysGA-A | 0–76 | 31.35 (±21.58) |
| PhysGA-D | 0–88 | 39.20 (±15.42) |
| PtGA-S | 0–100 | 42.48 (±31.42) |
| PtGA-D | 0–100 | 47.63 (±26.83) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lis-Święty, A.; Skrzypek-Salamon, A.; Ranosz-Janicka, I.; Brzezińska-Wcisło, L. Associations between Disease Activity/Severity and Damage and Health-Related Quality of Life in Adult Patients with Localized Scleroderma—A Comparison of LoSCAT and Visual Analogue Scales. J. Clin. Med. 2020, 9, 756. https://doi.org/10.3390/jcm9030756
Lis-Święty A, Skrzypek-Salamon A, Ranosz-Janicka I, Brzezińska-Wcisło L. Associations between Disease Activity/Severity and Damage and Health-Related Quality of Life in Adult Patients with Localized Scleroderma—A Comparison of LoSCAT and Visual Analogue Scales. Journal of Clinical Medicine. 2020; 9(3):756. https://doi.org/10.3390/jcm9030756
Chicago/Turabian StyleLis-Święty, Anna, Alina Skrzypek-Salamon, Irmina Ranosz-Janicka, and Ligia Brzezińska-Wcisło. 2020. "Associations between Disease Activity/Severity and Damage and Health-Related Quality of Life in Adult Patients with Localized Scleroderma—A Comparison of LoSCAT and Visual Analogue Scales" Journal of Clinical Medicine 9, no. 3: 756. https://doi.org/10.3390/jcm9030756
APA StyleLis-Święty, A., Skrzypek-Salamon, A., Ranosz-Janicka, I., & Brzezińska-Wcisło, L. (2020). Associations between Disease Activity/Severity and Damage and Health-Related Quality of Life in Adult Patients with Localized Scleroderma—A Comparison of LoSCAT and Visual Analogue Scales. Journal of Clinical Medicine, 9(3), 756. https://doi.org/10.3390/jcm9030756





