Sedentary Time Accumulated in Bouts is Positively Associated with Disease Severity in Fibromyalgia: The Al-Ándalus Project
Abstract
1. Introduction
2. Experimental Section
2.1. Participants
2.2. Procedures
2.3. Measurements
2.3.1. Sociodemographic and Clinical Data
2.3.2. Cognitive Impairment
2.3.3. Anthropometry and Body Composition
2.3.4. 1990 ACR Fibromyalgia Diagnostic Criteria
2.3.5. Modified 2011 ACR Fibromyalgia Preliminary Diagnostic Criteria
2.3.6. The Severity of Fibromyalgia
2.3.7. Sedentary Time and Moderate-to-Vigorous Physical Activity
2.3.8. Physical Fitness
2.4. Statistical Analysis
3. Results
4. Discussion
Limitations and Strengths
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Tremblay, M.S.; Aubert, S.; Barnes, J.D.; Saunders, T.J.; Carson, V.; Latimer-Cheung, A.E.; Chastin, S.F.M.; Altenburg, T.M.; Chinapaw, M.J.M. Sedentary Behavior Research Network (SBRN)—Terminology Consensus Project process and outcome. Int. J. Behav. Nutr. Phys. Act. 2017, 14, 75. [Google Scholar] [CrossRef] [PubMed]
- Patterson, R.; McNamara, E.; Tainio, M.; de Sá, T.H.; Smith, A.D.; Sharp, S.J.; Edwards, P.; Woodcock, J.; Brage, S.; Wijndaele, K. Sedentary behaviour and risk of all-cause, cardiovascular and cancer mortality, and incident type 2 diabetes: A systematic review and dose response meta-analysis. Eur. J. Epidemiol. 2018, 33, 811–829. [Google Scholar] [CrossRef] [PubMed]
- Diaz, K.M.; Goldsmith, J.; Greenlee, H.; Strizich, G.; Qi, Q.; Mossavar-Rahmani, Y.; Vidot, D.C.; Buelna, C.; Brintz, C.E.; Elfassy, T.; et al. Prolonged, Uninterrupted Sedentary Behavior and Glycemic Biomarkers Among US Hispanic/Latino Adults. Circulation 2017, 136, 1362–1373. [Google Scholar] [CrossRef] [PubMed]
- Biswas, A.; Oh, P.I.; Faulkner, G.E.; Bajaj, R.R.; Silver, M.A.; Mitchell, M.S.; Alter, D. Sedentary time and its association with risk for disease incidence, mortality, and hospitalization in adults: A systematic review and meta-analysis. Ann. Intern. Med. 2015, 162, 123–132. [Google Scholar] [CrossRef] [PubMed]
- Committee 2018 Physical Activity Guidelines Advisory; 2018 Physical Activity Guidelines Advisory Committee Scientific Report; Department of Health and Human Services: Washington, DC, USA, 2018.
- Matthews, C.E.; George, S.M.; Moore, S.C.; Bowles, H.R.; Blair, A.; Park, Y.; Troiano, R.P.; Hollenbeck, A.; Schatzkin, A. Amount of time spent in sedentary behaviors and cause-specific mortality in US adults. Am. J. Clin. Nutr. 2012, 95, 437–445. [Google Scholar] [CrossRef]
- Dunstan, D.W.; Kingwell, B.A.; Larsen, R.; Healy, G.N.; Cerin, E.; Hamilton, M.T.; Shaw, J.E.; Bertovic, D.A.; Zimmet, P.Z.; Salmon, J.; et al. Breaking up prolonged sitting reduces postprandial glucose and insulin responses. Diabetes Care 2012, 35, 976–983. [Google Scholar] [CrossRef]
- Peddie, M.C.; Bone, J.L.; Rehrer, N.J.; Skeaff, C.M.; Gray, A.R.; Perry, T.L. Breaking prolonged sitting reduces postprandial glycemia in healthy, normal-weight adults: A randomized crossover trial. Am. J. Clin. Nutr. 2013, 98, 358–366. [Google Scholar] [CrossRef]
- Loprinzi, P.D. Sedentary behavior and medical multimorbidity. Physiol. Behav. 2015, 151, 395–397. [Google Scholar] [CrossRef]
- Mork, P.J.; Vasseljen, O.; Nilsen, T.I.L. Association between physical exercise, body mass index, and risk of fibromyalgia: Longitudinal data from the Norwegian Nord-Trøndela health study. Arthritis Care Res. 2010, 62, 611–617. [Google Scholar] [CrossRef]
- Wolfe, F.; Clauw, D.J.; Fitzcharles, M.A.; Goldenberg, D.L.; Häuser, W.; Katz, R.S.; Mease, P.; Russell, A.S.; Rusesell, I.J.; Winfield, J.B.; et al. Fibromyalgia criteria and severity scales for clinical and epidemiological studies: A modification of the ACR preliminary diagnostic criteria for fibromyalgia. J. Rheumatol. 2011, 38, 1113–1122. [Google Scholar] [CrossRef]
- Cimen, O.B.; Cimen, M.Y.B.; Yapici, Y.; Camdeviren, H. Arginase, NOS activities, and clinical features in fibromyalgia patients. Pain Med. 2009, 10, 813–818. [Google Scholar] [CrossRef] [PubMed]
- Segura-Jiménez, V.; Aparicio, V.A.; Álvarez-Gallardo, I.C.; Soriano-Maldonado, A.; Estévez-López, F.; Delgado-Fernández, M.; Carbonell-Baeza, A. Validation of the modified 2010 American College of Rheumatology diagnostic criteria for fibromyalgia in a Spanish population. Rheumatology 2014, 53, 1803–1811. [Google Scholar] [CrossRef] [PubMed]
- Estévez-López, F.; Segura-Jiménez, V.; Álvarez-Gallardo, I.C.; Borges-Cosic, M.; Pulido-Martos, M.; Carbonell-Baeza, A.; Aparicio, V.A.; Geenen, R.; Delgado-Fernández, M. Adaptation profiles comprising objective and subjective measures in fibromyalgia: The al-Ándalus project. Rheumatology 2017, 56, 2015–2024. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Acosta-Manzano, P.; Segura-Jiménez, V.; Coll-Risco, I.; Borges-Cosic, M.; Castro-Piñero, J.; Delgado-Fernández, M.; Aparicio, V.A. Association of sedentary time and physical fitness with ideal cardiovascular health in perimenopausal women: The FLAMENCO project. Maturitas 2019, 120, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Gavilán-Carrera, B.; Segura-Jiménez, V.; Estévez-López, F.; Álvarez-Gallardo, I.C.; Soriano-Maldonado, A.; Borges-Cosic, M.; Herrador-Colmenero, M. Acosta-Manzano, P. Delgado-Fernández, M. Association of objectively measured physical activity and sedentary time with health-related quality of life in women with fibromyalgia: The al-Ándalus project. J. Sport Heal. Sci. 2019, 8, 258–266. [Google Scholar] [CrossRef] [PubMed]
- Segura-Jiménez, V.; Borges-Cosic, M.; Soriano-Maldonado, A.; Estévez-López, F.; Álvarez-Gallardo, I.C.; Herrador-Colmenero, M.; Delgado-Fernández, M.; Ruiz, J.R. Association of sedentary time and physical activity with pain, fatigue, and impact of fibromyalgia: The al-Ándalus study. Scand J. Med. Sci. Sports 2017, 27, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Borges-Cosic, M.; Aparicio, V.A.; Estévez-López, F.; Soriano-Maldonado, A.; Acosta-Manzano, P.; Gavilán-Carrera, B.; Delgado-Fernández, M.; Geenen, R.; Segura-Jiménez, V. Sedentary time, physical activity, and sleep quality in fibromyalgia: The al-Ándalus project. Scand J. Med. Sci. Sports 2019, 29, 266–274. [Google Scholar] [CrossRef]
- Gavilán-Carrera, B.; Acosta-Manzano, P.; Soriano-Maldonado, A.; Borges-Cosic, M.; Aparicio, V.A.; Delgado-Fernández, M.; Segura-Jiménez, V. Sedentary Time, Physical Activity, and Sleep Duration: Associations with Body Composition in Fibromyalgia. The Al-Andalus Project. J. Clin. Med. 2019, 8, 1260. [Google Scholar] [CrossRef]
- Gavilán-Carrera, B.; Segura-Jiménez, V.; Mekary, R.A.; Borges-Cosic, M.; Acosta-Manzano, P.; Estévez-López, F.; Álvarez-Gallardo, I.C.; Geenen, R.; Delgado-Fernández, M. Substituting Sedentary Time with Physical Activity in Fibromyalgia and the Association With Quality of Life and Impact of the Disease: The al-Ándalus Project. Arthritis Care Res. 2019, 71, 281–289. [Google Scholar] [CrossRef]
- Ellingson, L.D.; Shields, M.R.; Stegner, A.J.; Cook, D.B. Physical activity, sustained sedentary behavior, and pain modulation in women with fibromyalgia. J. Pain 2012, 13, 195–206. [Google Scholar] [CrossRef]
- Macfarlane, G.J.; Kronisch, C.; Dean, L.E.; Atzeni, F.; Häuser, W.; Fluß, E.; Choy, E.; Kosek, E.; Amris, K.; Branco, J.; et al. EULAR revised recommendations for the management of fibromyalgia. Ann. Rheum. Dis. 2017, 76, 318–328. [Google Scholar] [CrossRef] [PubMed]
- Fitzcharles, M.A.; Ste-Marie, P.A.; Goldenberg, D.L.; Pereira, J.X.; Abbey, S.; Choinière, M.; Gordon, K.O.; Moulin, D.E.; Panopalis, P.; Proulx, J.; et al. Canadian Pain Society and Canadian Rheumatology Association Recommendations for Rational Care of Persons with Fibromyalgia. A Summary Report. J. Rheumatol. 2013, 40, 1388–1393. [Google Scholar] [CrossRef] [PubMed]
- Segura-Jiménez, V.; Álvarez-Gallardo, I.C.; Estévez-López, F.; Soriano-Maldonado, A.; Delgado-Fernández, M.; Ortega, F.B.; Aparicio, V.A.; Carbonell-Baeza, A.; Mota, J.; Silva, P.; et al. Differences in Sedentary Time and Physical Activity between Female Patients with Fibromyalgia and Healthy Controls: The al-Ándalus Project. Arthritis Rheumatol. 2015, 67, 3047–3057. [Google Scholar] [CrossRef] [PubMed]
- Segura-Jiménez, V.; Álvarez-Gallardo, I.C.; Carbonell-Baeza, A.; Aparicio, V.A.; Ortega, F.B.; Casimiro, A.J.; Delgado-Fernández, M. Fibromyalgia has a larger impact on physical health than on psychological health, yet both are markedly affected: The al-Ándalus project. Semin Arthritis Rheum. 2015, 44, 563–570. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, F.; Smythe, H.A.; Yunus, M.B.; Bennett, R.M.; Bombardier, C.; Goldenberg, D.L.; Tugwell, P.; Campbell, S.M.; Abeles, M.; Clark, P.; et al. The American College of Rheumatology 1990 criteria for the classification of fibromyalgia. Arthritis Rheum. 1990, 33, 160–172. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Andreu, J.; Ibáñez-Bosch, R.; Portero-Vázquez, A.; Masramon, X.; Rejas, J.; Gálvez, R. Cognitive impairment in patients with fibromyalgia syndrome as assessed by the mini-mental state examination. BMC Musculoskelet. Disord. 2009, 10, 162. [Google Scholar] [CrossRef] [PubMed]
- Malavolti, M.; Mussi, C.; Poli, M.; Fantuzzi, A.L.; Salvioli, G.; Battistini, N.; Bedogni, G. Cross-calibration of eight-polar bioelectrical impedance analysis versus dual-energy X-ray absorptiometry for the assessment of total and appendicular body composition in healthy subjects aged 21-82 years. Ann. Hum. Biol. 2003, 30, 380–391. [Google Scholar] [CrossRef]
- Segura-Jiménez, V.; Aparicio, V.A.; Álvarez-Gallardo, I.C.; Carbonell-Baeza, A.; Tornero-Quinones, I.; Delgado-Fernández, M. Does body composition differ between fibromyalgia patients and controls? The al-Andalus project. Clin. Exp. Rheumatol. 2015, 33, 25–32. [Google Scholar]
- Bennett, R.M.; Friend, R.; Jones, K.D.; Ward, R.; Han, B.K.; Ross, R.L. The Revised Fibromyalgia Impact Questionnaire (FIQR): Validation and psychometric properties. Arthritis Res. Ther. 2009, 11, R120. [Google Scholar] [CrossRef]
- Luciano, J.V.; Aguado, J.; Serrano-Blanco, A.; Calandre, E.P.; Rodriguez-Lopez, C.M. Dimensionality, reliability, and validity of the revised fibromyalgia impact questionnaire in two spanish samples. Arthritis Care Res. 2013, 65, 1682–1689. [Google Scholar] [CrossRef]
- Aguilar-Farías, N.; Brown, W.J.; Peeters, G.M.E.E. ActiGraph GT3X+ cut-points for identifying sedentary behaviour in older adults in free-living environments. J. Sci. Med. Sport 2014, 17, 293–299. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, J.E.; John, D.; Freedson, P.S. Validation and comparison of ActiGraph activity monitors. J. Sci. Med. Sport 2011, 14, 411–416. [Google Scholar] [CrossRef] [PubMed]
- Choi, L.; Ward, S.C.; Schnelle, J.F.; Buchowski, M.S. Assessment of wear/nonwear time classification algorithms for triaxial accelerometer. Med. Sci. Sports Exerc. 2012, 44, 2009–2016. [Google Scholar] [CrossRef] [PubMed]
- Diaz, K.M.; Howard, V.J.; Hutto, B.; Colabianchi, N.; Vena, J.E.; Safford, M.M.; Blair, S.N.; Hooker, S.P. Patterns of Sedentary Behavior and Mortality in U.S. Middle-Aged and Older Adults: A National Cohort Study. Ann. Intern. Med. 2017, 167, 465–475. [Google Scholar] [CrossRef]
- Segura-Jiménez, V.; Estévez-López, F.; Castro-Piñero, J.; Álvarez-Gallardo, I.C.; Soriano-Maldonado, A.; Borges-Cosic, M.; Delgado-Fernández, M. Association of Patterns of Moderate-to-Vigorous Physical Activity Bouts With Pain, Physical Fatigue, and Disease Severity in Women With Fibromyalgia: The al-Ándalus Project. Arch. Phys. Med. Rehabil. 2019, 100, 1234–1242.e1. [Google Scholar] [CrossRef]
- Rikli, R.; Jones, C. Development and Validation of a Functional Fitness Test for Community-Residing Older Adults. J. Aging Phys. Act. 1999, 7, 129–161. [Google Scholar] [CrossRef]
- Carbonell-Baeza, A.; Álvarez-Gallardo, I.C.; Segura-Jiménez, V.; Castro-Piñero, J.; Ruiz, J.R.; Delgado-Fernández, M.; Aparicio, V.A. Reliability and Feasibility of Physical Fitness Tests in Female Fibromyalgia Patients. Int. J. Sports Med. 2015, 36, 157–162. [Google Scholar] [CrossRef]
- Soriano-Maldonado, A.; Henriksen, M.; Segura-Jiménez, V.; Aparicio, V.A.; Carbonell-Baeza, A.; Delgado-Fernández, M.; Amris, K.; Ruiz, J.R. Association of Physical Fitness with Fibromyalgia Severity in Women: The al-Ándalus Project. Arch. Phys. Med. Rehabil. 2015, 96, 1599–1605. [Google Scholar] [CrossRef]
- Estévez-López, F.; Gray, C.M.; Segura-Jiménez, V.; Soriano-Maldonado, A.; Álvarez-Gallardo, I.C.; Arrayás-Grajera, M.J.; Carbonell-Baeza, A.; Aparicio, V.A.; Delgado-Fernández, M.; Pulido-Martos, M.; et al. Independent and combined association of overall physical fitness and subjective well-being with fibromyalgia severity: The al-Ándalus project. Qual. Life Res. 2015, 24, 1865–1873. [Google Scholar] [CrossRef]
- Pulido-Martos, M.; Luque-Reca, O.; Segura-Jiménez, V.; Álvarez-Gallardo, I.C.; Soriano-Maldonado, A.; Acosta-Manzano, P.; Gavilán-Carrera, B.; McVeigh, J.G.; Geenen, R.; Delgado-Fernández, M.; et al. Physical and psychological paths toward less severe fibromyalgia: A structural equation model. Ann. Phys. Rehabil. Med. 2019, 63, 46–52. [Google Scholar] [CrossRef]
- Shiroma, E.J.; Freedson, P.S.; Trost, S.G.; Lee, I.M. Patterns of accelerometer-assessed sedentary behavior in older women. JAMA 2013, 310, 2562–2563. [Google Scholar] [CrossRef] [PubMed]
- Diaz, K.M.; Howard, V.J.; Hutto, B.; Colabianchi, N.; Vena, J.E.; Blair, S.N.; Hooker, S.P. Patterns of Sedentary Behavior in US Middle-Age and Older Adults: The REGARDS Study. Med. Sci. Sports Exerc. 2016, 48, 430–438. [Google Scholar] [PubMed]
- Dutta, N.; Walton, T.; Pereira, M.A. One-year follow-up of a sit-stand workstation intervention to decrease sedentary time in office workers. Prev. Med. Rep. 2019, 13, 277–280. [Google Scholar] [CrossRef] [PubMed]
- Kerr, C.J.; Ploetz, T.; Charman, S.J.; Savory, L.A.; Bailey, D.P. Associations between prolonged sedentary time and breaks in sedentary time with cardiometabolic risk in 10–14-year-old children: The HAPPY study. J. Sports Sci. 2016, 35, 2164–2171. [Google Scholar]
- Korshøj, M.; Holtermann, A.; Jørgensen, M.B.; Aadahl, M.; Heiden, M.; Gupta, N. What Is the Effect on Obesity Indicators from Replacing Prolonged Sedentary Time with Brief Sedentary Bouts, Standing and Different Types of Physical Activity during Working Days? A Cross-Sectional Accelerometer-Based Study among Blue-Collar Workers. PLoS ONE 2016, 11, e0154935. [Google Scholar]
- Schultz, A.; Tangri, N.; Hiebert, B.; Arora, R.C.; Stammers, A.N.; Duhamel, T.A.; Clara, I.; Kehler, D.S.; Hay, J.L. The association between bouts of moderate to vigorous physical activity and patterns of sedentary behavior with frailty. Exp. Gerontol. 2018, 104, 28–34. [Google Scholar]
- Weijenberg, M.P.; Beets, G.L.; Lynch, B.M.; Winkler, E.A.H.; Van Roekel, E.H.; Kant, I.J.; Bours, M.J.L.; Meijer, K.; Sanduleanu, S.; Healy, G.N. Associations of sedentary time and patterns of sedentary time accumulation with health-related quality of life in colorectal cancer survivors. Prev. Med. Rep. 2016, 4, 262–269. [Google Scholar]
- Judice, P.B.; Silva, A.M.; Sardinha, L.B. Sedentary bout durations are associated with abdominal obesity in older adults. J. Nutr. Heal. Aging 2015, 19, 798–804. [Google Scholar] [CrossRef]
- Bennett, R.M.; Bushmakin, A.G.; Cappelleri, J.C.; Zlateva, G.; Sadosky, A.B. Minimal clinically important difference in the fibromyalgia impact questionnaire. J. Rheumatol. 2009, 36, 1304–1311. [Google Scholar] [CrossRef]
- Wang, C.; Schmid, C.H.; Fielding, R.A.; Harvey, W.F.; Reid, K.F.; Price, L.L.; Driban, J.B.; Kalish, R.; Rones, R.; McAlindon, T. Effect of tai chi versus aerobic exercise for fibromyalgia: Comparative effectiveness randomized controlled trial. BMJ 2018, 360, k851. [Google Scholar] [CrossRef]
- Martin, A.; Fitzsimons, C.; Jepson, R.; Saunders, D.H.; Van der Ploeg, H.P.; Teixeira, P.J.; Gray, C.M.; Mutrie, N. Interventions with potential to reduce sedentary time in adults: Systematic review and meta-analysis. Br. J. Sports Med. 2015, 49, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Compernolle, S.; Desmet, A.; Poppe, L.; Crombez, G.; De Bourdeaudhuij, I.; Cardon, G.; Van der Ploeg, H.P.; Van Dyck, D. Effectiveness of interventions using self- monitoring to reduce sedentary behavior in adults: A systematic review and meta- analysis. Int. J. Behav. Nutr. Phys. Act. 2019, 16, 63. [Google Scholar] [CrossRef] [PubMed]
- Gardner, B.; Smith, L.; Lorencatto, F.; Hamer, M.; Jh, S. How to reduce sitting time? A review of behaviour change strategies used in sedentary behaviour reduction interventions among adults. Health Psychol. Rev. 2016, 10, 89–112. [Google Scholar] [CrossRef] [PubMed]
- Gardiner, P.A.; Eakin, E.G.; Healy, G.N.; Owen, N. Feasibility of reducing older adults’ sedentary time. Am. J. Prev. Med. 2011, 41, 174–177. [Google Scholar] [CrossRef]
- Carter, S.E.; Jones, M.; Gladwell, V.F. Energy expenditure and heart rate response to breaking up sedentary time with three different physical activity interventions. Nutr. Metab. Cardiovasc. Dis. 2015, 25, 503–509. [Google Scholar] [CrossRef]
- Thyfault, J.P.; Du, M.; Kraus, W.E.; Levine, J.A.; Booth, F.W. Physiology of sedentary behavior and its relationship to health outcomes. Med. Sci. Sports Exerc. 2015, 47, 1301–1305. [Google Scholar] [CrossRef]
- Ellingson, L.D.; Kuffel, A.E.; Vack, N.J.; Cook, D.B. Active and sedentary behaviors influence feelings of energy and fatigue in women. Med. Sci. Sports Exerc. 2014, 46, 192–200. [Google Scholar] [CrossRef]
- McLoughlin, M.J.; Colbert, L.H.; Stegner, A.J.; Cook, D.B. Are women with fibromyalgia less physically active than healthy women? Med. Sci. Sports Exerc. 2011, 43, 905–912. [Google Scholar] [CrossRef]
| Clinical Variable | Mean | SD |
|---|---|---|
| Age (year) | 51.3 | (7.6) |
| Body mass index (kg/m2) | 28.5 | (5.4) |
| Fat percentage (%) | 40.0 | (7.6) |
| Tender points (11–18) | 15.1 | (4.6) |
| Pressure pain threshold (18–144 kg/cm2) | 50.0 | (21.9) |
| Widespread Pain Index (0–19) | 13.7 | (3.8) |
| Symptom Severity Score (0–9) | 8.0 | (2.2) |
| Polysymptomatic Distress (0–28) | 21.7 | (5.0) |
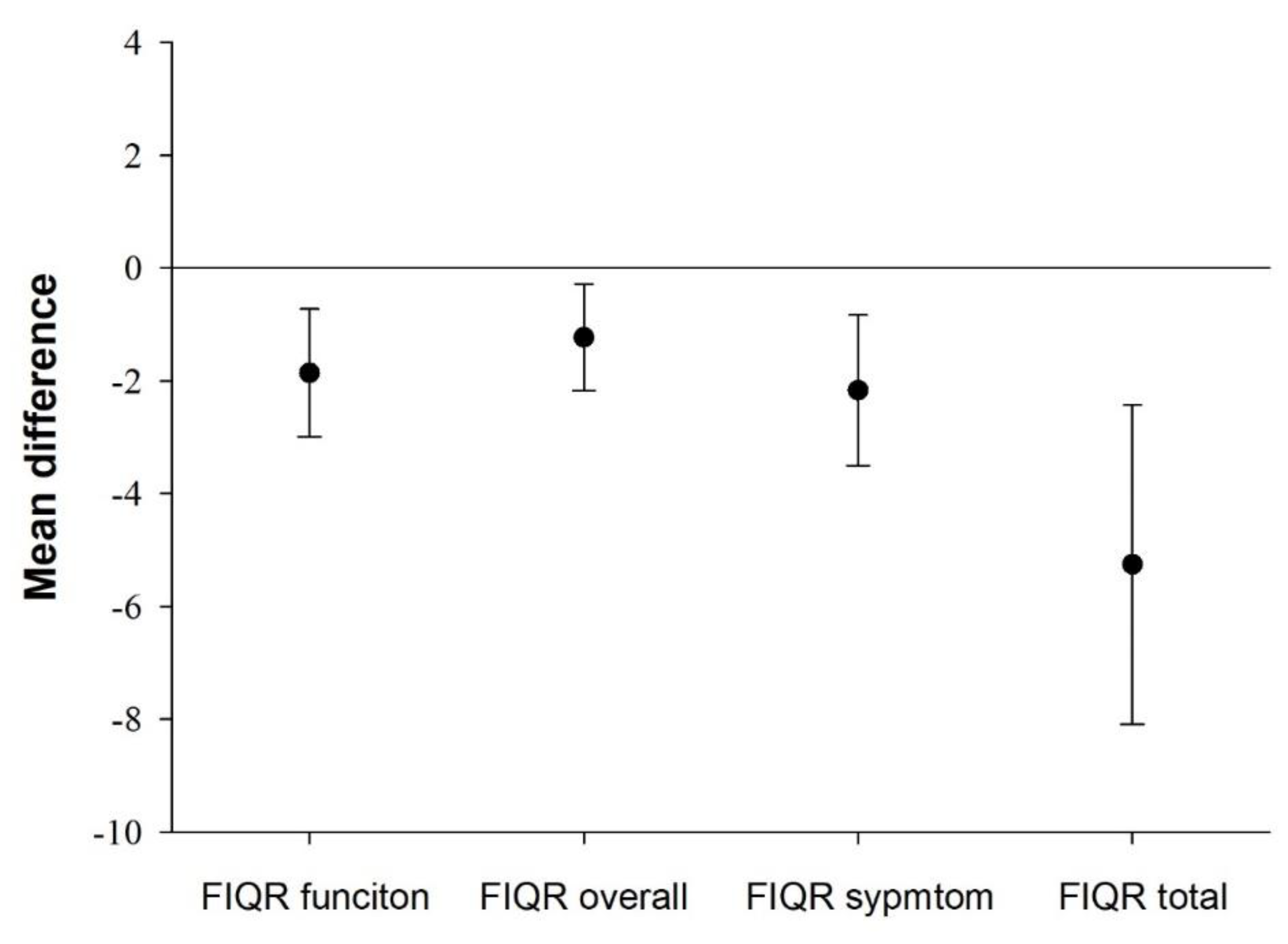
| FIQR Function (0–30) | 17.0 | (6.5) |
| FIQR Overall (0–20) | 12.2 | (5.3) |
| FIQR Symptoms (0–50) | 34.7 | (7.7) |
| FIQR Total Score (0–100) | 63.9 | (16.8) |
| Clinical and sociodemographic variable | n | % |
| Marital Status | ||
| Married | 340 | (75.4) |
| Not Married | 111 | (24.6) |
| Educational Level | ||
| Non-university | 390 | (86.5) |
| University | 61 | (13.5) |
| Current Occupational Status | ||
| Working | 126 | (27.9) |
| Housekeeper | 144 | (31.9) |
| Not Working | 181 | (40.1) |
| Variable | Mean | (SD) |
|---|---|---|
| Accelerometer wear time (min/day) | 923.3 | (75.1) |
| Sedentary time (min/day) | 458.3 | (104.2) |
| Percentage of sedentary time | 49.7 | (10.9) |
| Time in ≥30-min sedentary bout (min/day) | 129.4 | (81.2) |
| Percentage of time in ≥30-min sedentary bout | 14.0 | (8.7) |
| Time in ≥60-min sedentary bout (min/day) | 50.7 | (49.8) |
| Percentage of time in ≥60-min sedentary bout | 5.5 | (5.4) |
| Percentage of MVPA | 4.9 | (3.3) |
| Percentage of bouted MVPA | 0.6 | (0.7) |
| FIQR Function | FIQR Overall | FIQR Symptoms | FIQR Total | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Variables | β | B | (95% CI) | Adj. R2 | β | B | (95% CI) | Adj. R2 | β | B | (95% CI) | Adj. R2 | β | B | (95% CI) | Adj. R2 | |
| Percentage of ST in ≥30-min bout | Model 1 | 0.17 | 9.08 | (4.18; 13.98) | 0.04 | 0.21 | 9.12 | (5.12; 13.11) | 0.05 | 0.21 | 13.26 | (7.49; 19.03) | 0.06 | 0.23 | 31.46 | (19.02; 43.90) | 0.07 |
| Model 2 | 0.15 | 8.02 | (3.12; 12.93) | 0.06 | 0.19 | 8.38 | (4.37; 12.40) | 0.06 | 0.19 | 12.24 | (6.44; 18.04) | 0.07 | 0.21 | 28.64 | (16.20; 41.09) | 0.09 | |
| Model 3 | 0.10 | 5.08 | (0.29; 9.86) | 0.14 | 0.13 | 5.75 | (1.79; 9.71) | 0.11 | 0.14 | 8.59 | (2.96; 14.22) | 0.16 | 0.14 | 19.94 | (8.01; 31.88) | 0.19 | |
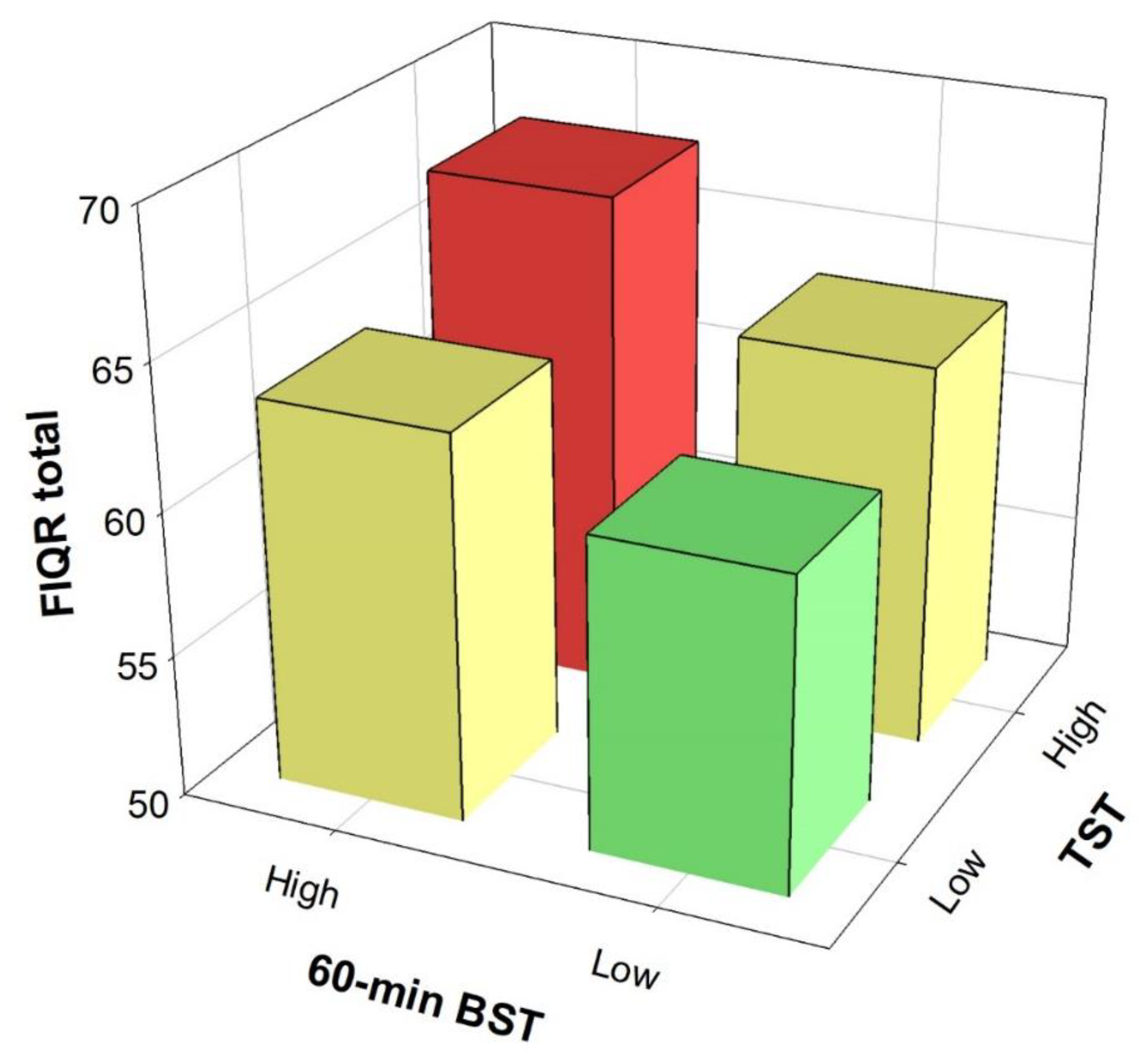
| Percentage of ST in ≥60-min bout | Model 1 | 0.15 | 11.13 | (4.36; 17.89) | 0.04 | 0.18 | 11.18 | (5.64; 16.71) | 0.04 | 0.21 | 18.29 | (10.34; 26.23) | 0.06 | 0.21 | 40.59 | (23.39; 57.78) | 0.07 |
| Model 2 | 0.13 | 9.88 | (3.14; 16.63) | 0.06 | 0.17 | 10.30 | (4.77; 15.83) | 0.06 | 0.19 | 17.10 | (9.14; 25.05) | 0.08 | 0.19 | 37.28 | (20.16; 54.40) | 0.09 | |
| Model 3 | 0.08 | 6.03 | (−0.53; 12.60) | 0.14 | 0.12 | 7.52 | (2.08; 12.96) | 0.12 | 0.14 | 12.37 | (4.66; 20.07) | 0.16 | 0.15 | 46.01 | (19.46; 72.57) | 0.20 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Segura-Jiménez, V.; Gavilán-Carrera, B.; Acosta-Manzano, P.; Cook, D.B.; Estévez-López, F.; Delgado-Fernández, M. Sedentary Time Accumulated in Bouts is Positively Associated with Disease Severity in Fibromyalgia: The Al-Ándalus Project. J. Clin. Med. 2020, 9, 733. https://doi.org/10.3390/jcm9030733
Segura-Jiménez V, Gavilán-Carrera B, Acosta-Manzano P, Cook DB, Estévez-López F, Delgado-Fernández M. Sedentary Time Accumulated in Bouts is Positively Associated with Disease Severity in Fibromyalgia: The Al-Ándalus Project. Journal of Clinical Medicine. 2020; 9(3):733. https://doi.org/10.3390/jcm9030733
Chicago/Turabian StyleSegura-Jiménez, Víctor, Blanca Gavilán-Carrera, Pedro Acosta-Manzano, Dane B Cook, Fernando Estévez-López, and Manuel Delgado-Fernández. 2020. "Sedentary Time Accumulated in Bouts is Positively Associated with Disease Severity in Fibromyalgia: The Al-Ándalus Project" Journal of Clinical Medicine 9, no. 3: 733. https://doi.org/10.3390/jcm9030733
APA StyleSegura-Jiménez, V., Gavilán-Carrera, B., Acosta-Manzano, P., Cook, D. B., Estévez-López, F., & Delgado-Fernández, M. (2020). Sedentary Time Accumulated in Bouts is Positively Associated with Disease Severity in Fibromyalgia: The Al-Ándalus Project. Journal of Clinical Medicine, 9(3), 733. https://doi.org/10.3390/jcm9030733







