Assessing the Impact of Reduced Travel on Exportation Dynamics of Novel Coronavirus Infection (COVID-19)
Abstract
1. Introduction
2. Methods
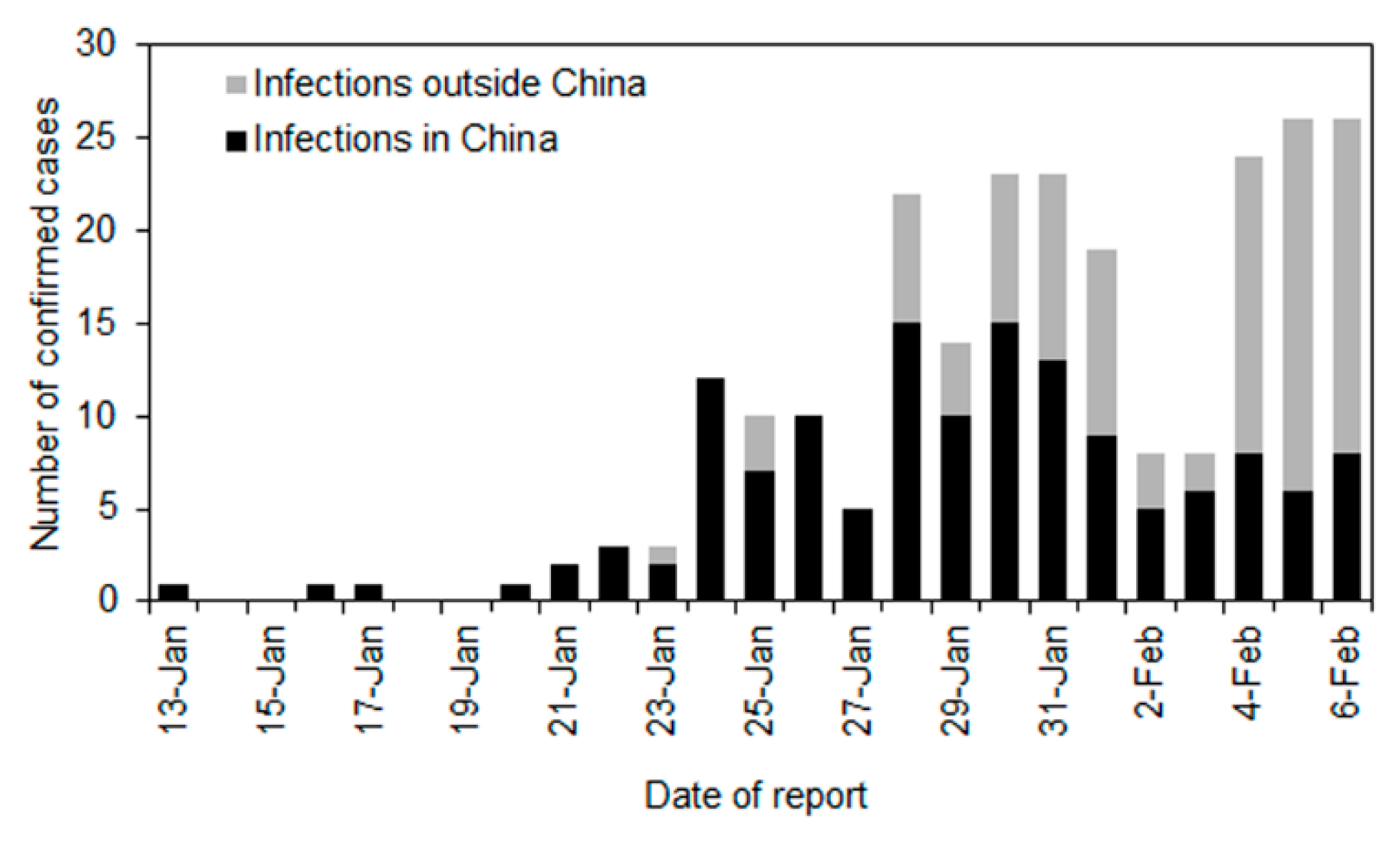
2.1. Epidemiological Data
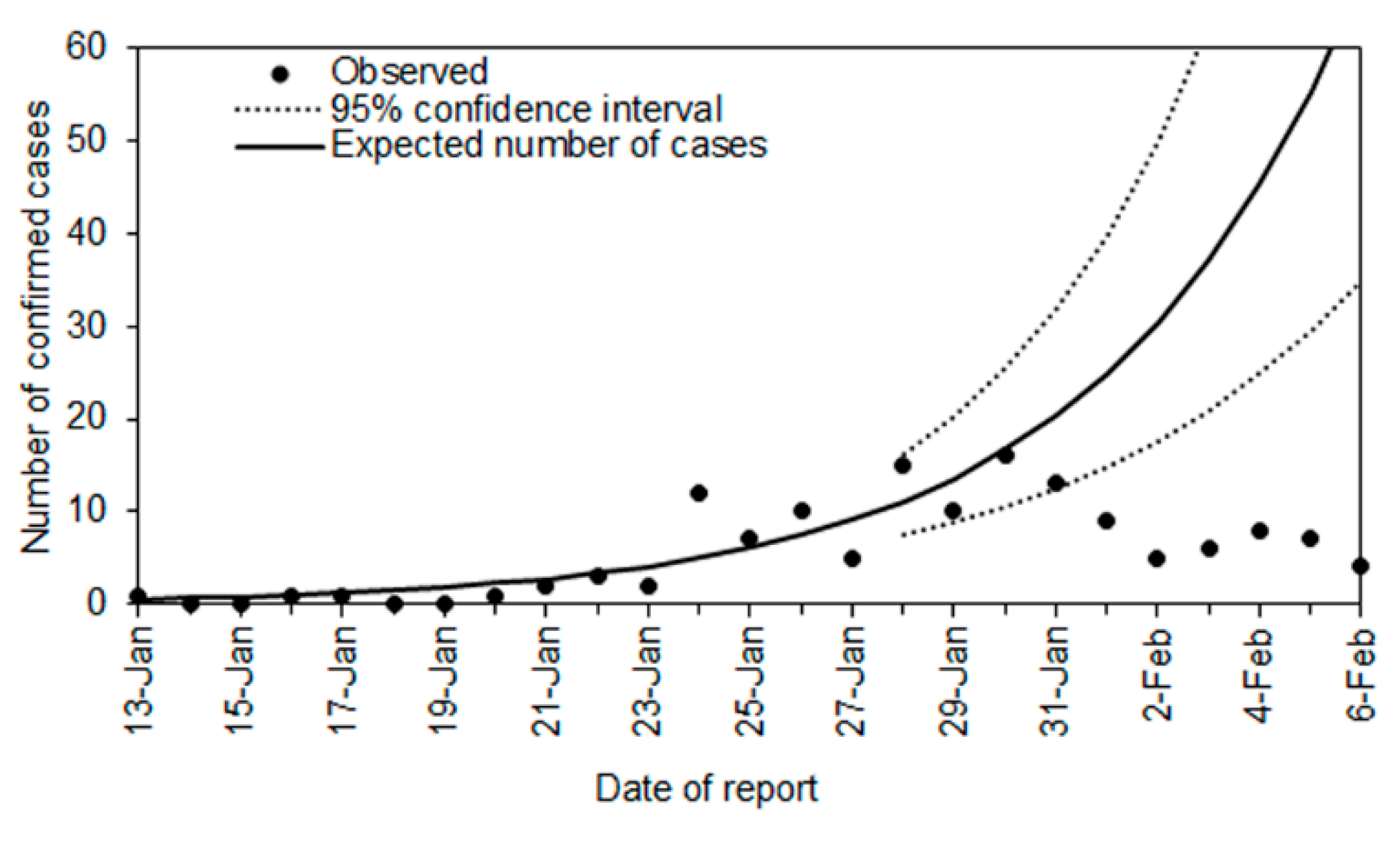
2.2. Statistical Model
2.2.1. Reduced Number of Exported Cases
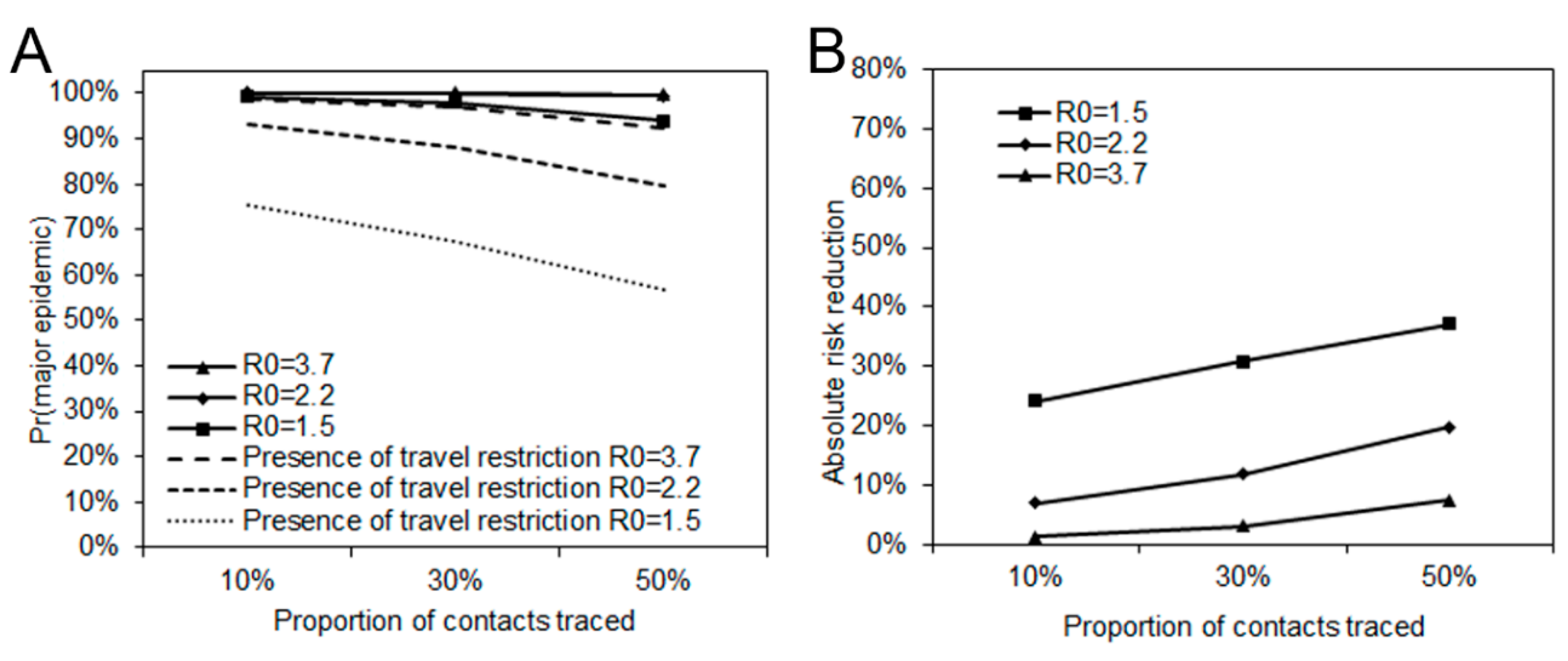
2.2.2. Reduced Probability of a Major Epidemic Overseas
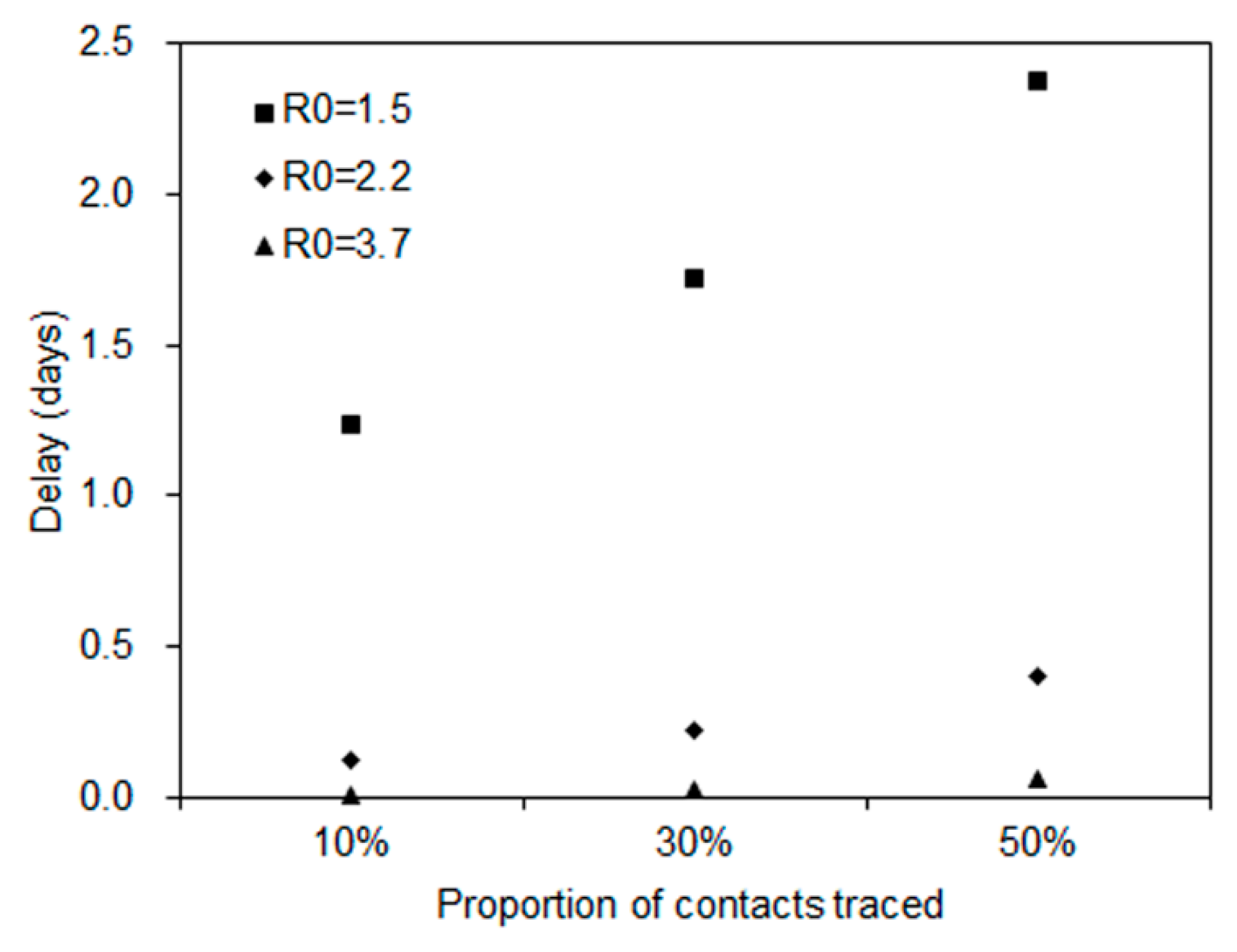
2.2.3. Time Delay to a Major Epidemic Gained from the Reduction in Travel Volume
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- World Health Organization. Novel Coronavirus—China, 2020. Available online: https://www.who.int/csr/don/12-january-2020-novel-coronavirus-china/en/ (accessed on 11 February 2020).
- World Health Organization. Novel Coronavirus (2019-nCoV) Situation Report-21, 2020. Available online: https://www.who.int/docs/default-source/coronaviruse/situation-reports/20200210-sitrep-21-ncov.pdf (accessed on 11 February 2020).
- Center for Disease Central and Prevention. 2019 Novel Coronavirus, Wuhan, China, 2020. Available online: https://www.cdc.gov/coronavirus/2019-nCoV/summary.html (accessed on 11 February 2020).
- Rothe, C.; Schunk, M.; Sothmann, P.; Bretzel, G.; Froeschl, G.; Wallrauch, C.; Zimmer, T.; Thiel, V.; Janke, C.; Guggemos, W.; et al. Transmission of 2019-nCoV Infection from an Asymptomatic Contact in Germany. N. Engl. J. Med. 2020. (In Press) [CrossRef] [PubMed]
- Chan, F.J.; Yuan, S.; Kok, K.H.; To, K.K.; Chu, H.; Yang, J.; Xing, F.; Liu, J.; Yip, C.C.; Poon, R.W.; et al. A familial cluster of pneumonia associated with the 2019 novel coronavirus indicating person-to-person transmission: A study of a family cluster. Lancet 2020. (In Press) [CrossRef]
- BBC NEWS. Coronavirus: Wuhan Shuts Public Transport over Outbreak, 2020. Available online: https://www.bbc.com/news/world-asia-china-51215348 (accessed on 11 February 2020).
- Barbera, J.; Macintyre, A.; Gostin, L.; Inglesby, T.; O’Toole, T.; DeAtley, C.; Tonat, K.; Layton, M. Large-scale quarantine following biological terrorism in the United States: Scientific examination, logistic and legal limits, and possible consequences. JAMA 2001, 286, 2711–2717. [Google Scholar] [CrossRef] [PubMed]
- Nishiura, H.; Wilson, N.; Baker, M.G. Quarantine for pandemic influenza control at the borders of small island nations. BMC Infect. Dis. 2009, 9, 27. [Google Scholar] [CrossRef] [PubMed]
- Barbisch, D.; Koenig, K.L.; Shih, F.-Y. Is There a Case for Quarantine? Perspectives from SARS to Ebola. Disaster Med. Public Health Prep. 2015, 9, 547–553. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, Y.-H.; King, C.-C.; Chen, C.W.S.; Ho, M.-S.; Hsu, S.-B.; Wu, Y.-C. Impact of quarantine on the 2003 SARS outbreak: A retrospective modeling study. J. Theor. Biol. 2007, 244, 729–736. [Google Scholar] [CrossRef] [PubMed]
- Pitman, R.J.; Cooper, B.S.; Trotter, C.L.; Gay, N.J.; Edmunds, W.J. Entry screening for severe acute respiratory syndrome (SARS) or influenza: Policy evaluation. BMJ 2005, 331, 1242–1243. [Google Scholar] [CrossRef] [PubMed]
- Samaan, G.; Patel, M.; Spencer, J.; Roberts, L. Border screening for SARS in Australia: What has been learnt? Med. J. Aust. 2004, 180, 220–223. [Google Scholar] [CrossRef] [PubMed]
- St John, R.K.; King, A.; de Jong, D.; Bodie-Collins, M.; Squires, S.G.; Tam, T.W.S. Border screening for SARS. Emerg. Infect. Dis. 2005, 11, 6–10. [Google Scholar] [CrossRef] [PubMed]
- Jung, S.; Akhmetzhanov, A.R.; Hayashi, K.; Linton, N.M.; Yang, Y.; Yuan, B.; Kobayashi, T.; Kinoshita, R.; Nishiura, H. Real time estimation of the risk of death from novel coronavirus (2019-nCoV) infection: Inference using exported cases. J. Clin. Med. 2020, 9, 523. [Google Scholar] [CrossRef]
- Britton, T. Stochastic epidemic models: A survey. Math. Biosci. 2010, 225, 24–35. [Google Scholar] [CrossRef] [PubMed]
- Quilty, B.J.; Clifford, S.; CMMID nCoV working group; Flasche, S.; Eggo, R.M. Effectiveness of airport screening at detecting travellers infected with novel coronavirus (2019-nCoV). Eurosurveillance 2020, 25, 2000080. [Google Scholar] [CrossRef] [PubMed]
- Riou, J.; Althaus, C.L. Pattern of early human-to-human transmission of Wuhan 2019 novel coronavirus (2019-nCoV), December 2019 to January 2020. Eurosurveillance 2020, 25, 2000058. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Guan, X.; Wu, P.; Wang, X.; Zhou, L.; Tong, Y.; Ren, R.; Leung, K.S.M.; Lau, E.H.Y.; Wong, J.Y.; et al. Early transmission dynamics in Wuhan, China, of novel coronavirus-infected pneumonia. N. Engl. J. Med. 2020. [Google Scholar] [CrossRef] [PubMed]
- Nishiura, H.; Kobayashi, T.; Miyama, T.; Suzuki, A.; Jung, S.; Hayashi, K.; Kinoshita, R.; Yang, Y.; Yuan, B.; Akhmetzhanov, A.R.; et al. Estimation of the Asymptomatic Ratio of Novel Coronavirus (2019-nCoV) Infections among Passengers on Evacuation Flights. 2020. Available online: https://www.medrxiv.org/content/10.1101/2020.02.03.20020248v1 (accessed on 13 February 2020).
- Chinazzi, M.; Davis, J.T.; Ajelli, M.; Gioannini, C.; Litvinova, M.; Merler, S.; Pastore, Y.; Piontti, A.; Rossi, L.; Sun, K.; et al. The Effect of Travel Restrictions on the Spread of the 2019 Novel Coronavirus (2019-nCoV) Outbreak. 2020. Available online: https://www.mobs-lab.org/uploads/6/7/8/7/6787877/modeling_travel_limitations_paper.pdf (accessed on 11 February 2020).
- Tian, H.; Li, Y.; Liu, Y.; Kraemer, M.U.G.; Chen, B.; Cai, J.; Li, B.; Xu, B.; Yang, Q.; Yang, P.; et al. Early Evaluation of the Wuhan City Travel Restrictions in Response to the 2019 Novel Coronavirus Outbreak. 2020. Available online: https://www.medrxiv.org/content/10.1101/2020.01.30.20019844v1 (accessed on 11 February 2020).
- World Health Organization. Statement on the Second Meeting of the International Health Regulations (2005) Emergency Committee Regarding the Outbreak of Novel Coronavirus (2019-nCoV), 2020. Available online: https://www.who.int/news-room/detail/30-01-2020-statement-on-the-second-meeting-of-the-international-health-regulations-(2005)-emergency-committee-regarding-the-outbreak-of-novel-coronavirus-(2019-ncov) (accessed on 11 February 2020).
- BBC NEWS. Coronavirus: US Bars Foreigners Who Recently Visited CHINA, 2020. Available online: https://www.bbc.com/news/world-us-canada-51335278 (accessed on 11 February 2020).
- Wu, J.T.; Leung, K.; Leung, G.M. Nowcasting and forecasting the potential domestic and international spread of the 2019-nCoV outbreak originating in Wuhan, China: A modelling study. Lancet 2020. (In Press) [CrossRef]
- Tang, B.; Wang, X.; Li, Q.; Bragazzi, N.L.; Tang, S.; Xiao, Y.; Wu, J. Estimation of the Transmission Risk of the 2019-nCoV and Its Implication for Public Health Interventions. J. Clin. Med. 2020, 9, 462. [Google Scholar] [CrossRef] [PubMed]
- Thompson, R.N. Novel Coronavirus Outbreak in Wuhan, China, 2020: Intense Surveillance Is Vital for Preventing Sustained Transmission in New Locations. J. Clin. Med. 2020, 9, 498. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Anzai, A.; Kobayashi, T.; Linton, N.M.; Kinoshita, R.; Hayashi, K.; Suzuki, A.; Yang, Y.; Jung, S.-m.; Miyama, T.; Akhmetzhanov, A.R.; et al. Assessing the Impact of Reduced Travel on Exportation Dynamics of Novel Coronavirus Infection (COVID-19). J. Clin. Med. 2020, 9, 601. https://doi.org/10.3390/jcm9020601
Anzai A, Kobayashi T, Linton NM, Kinoshita R, Hayashi K, Suzuki A, Yang Y, Jung S-m, Miyama T, Akhmetzhanov AR, et al. Assessing the Impact of Reduced Travel on Exportation Dynamics of Novel Coronavirus Infection (COVID-19). Journal of Clinical Medicine. 2020; 9(2):601. https://doi.org/10.3390/jcm9020601
Chicago/Turabian StyleAnzai, Asami, Tetsuro Kobayashi, Natalie M. Linton, Ryo Kinoshita, Katsuma Hayashi, Ayako Suzuki, Yichi Yang, Sung-mok Jung, Takeshi Miyama, Andrei R. Akhmetzhanov, and et al. 2020. "Assessing the Impact of Reduced Travel on Exportation Dynamics of Novel Coronavirus Infection (COVID-19)" Journal of Clinical Medicine 9, no. 2: 601. https://doi.org/10.3390/jcm9020601
APA StyleAnzai, A., Kobayashi, T., Linton, N. M., Kinoshita, R., Hayashi, K., Suzuki, A., Yang, Y., Jung, S.-m., Miyama, T., Akhmetzhanov, A. R., & Nishiura, H. (2020). Assessing the Impact of Reduced Travel on Exportation Dynamics of Novel Coronavirus Infection (COVID-19). Journal of Clinical Medicine, 9(2), 601. https://doi.org/10.3390/jcm9020601






