Body Weight as a Determining Factor in the Predominance of Adverse Drug Reactions Induced by Fixed-Dose Adalimumab Injections in Female Patients in a Korean Hospital Setting
Abstract
1. Introduction
2. Experimental Section
2.1. EMR Data Collection
2.2. Analysis of Prescription and Laboratory Data
2.3. Statistics
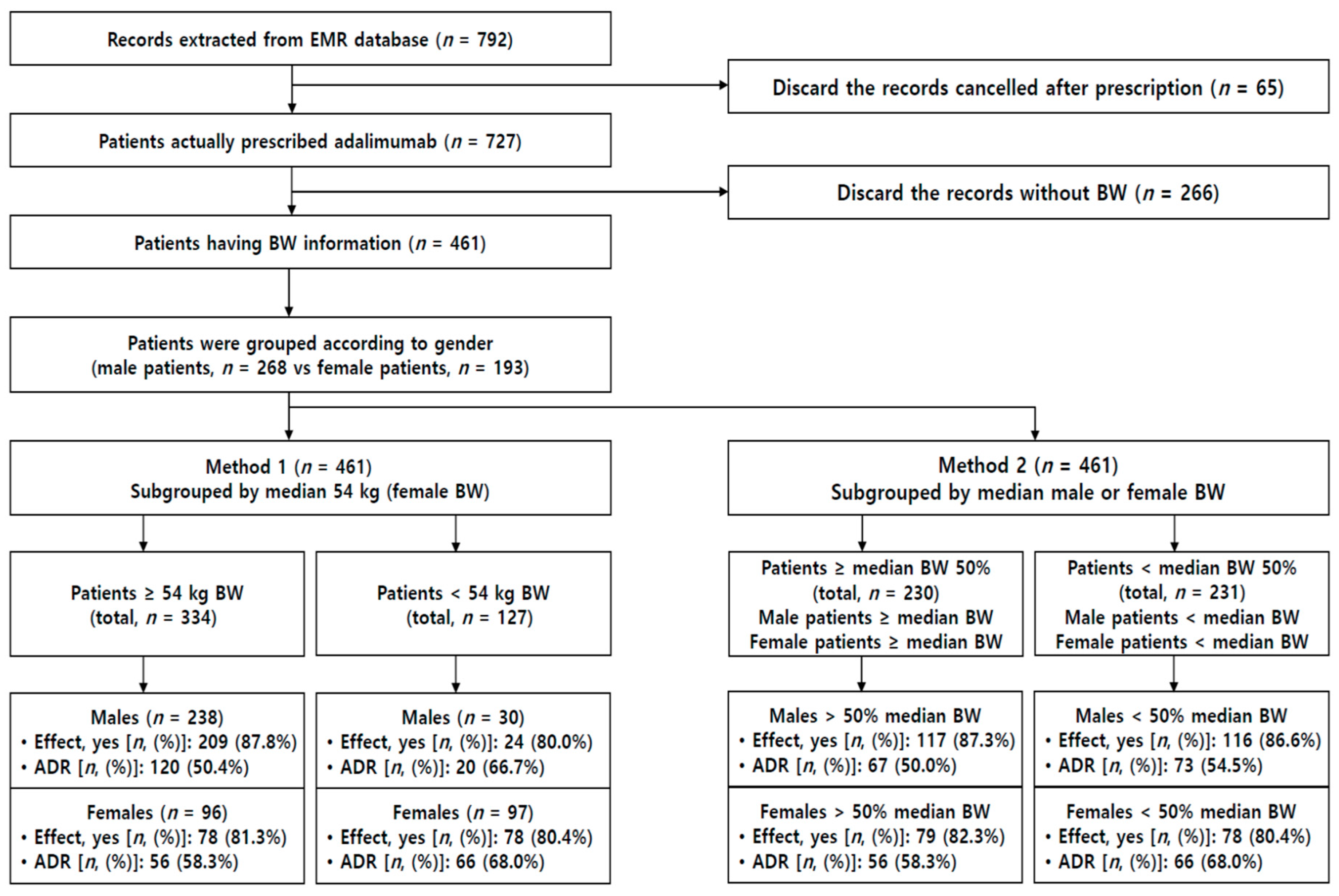
3. Results
3.1. Patient Information and Evaluation of Adalimumab Efficacy
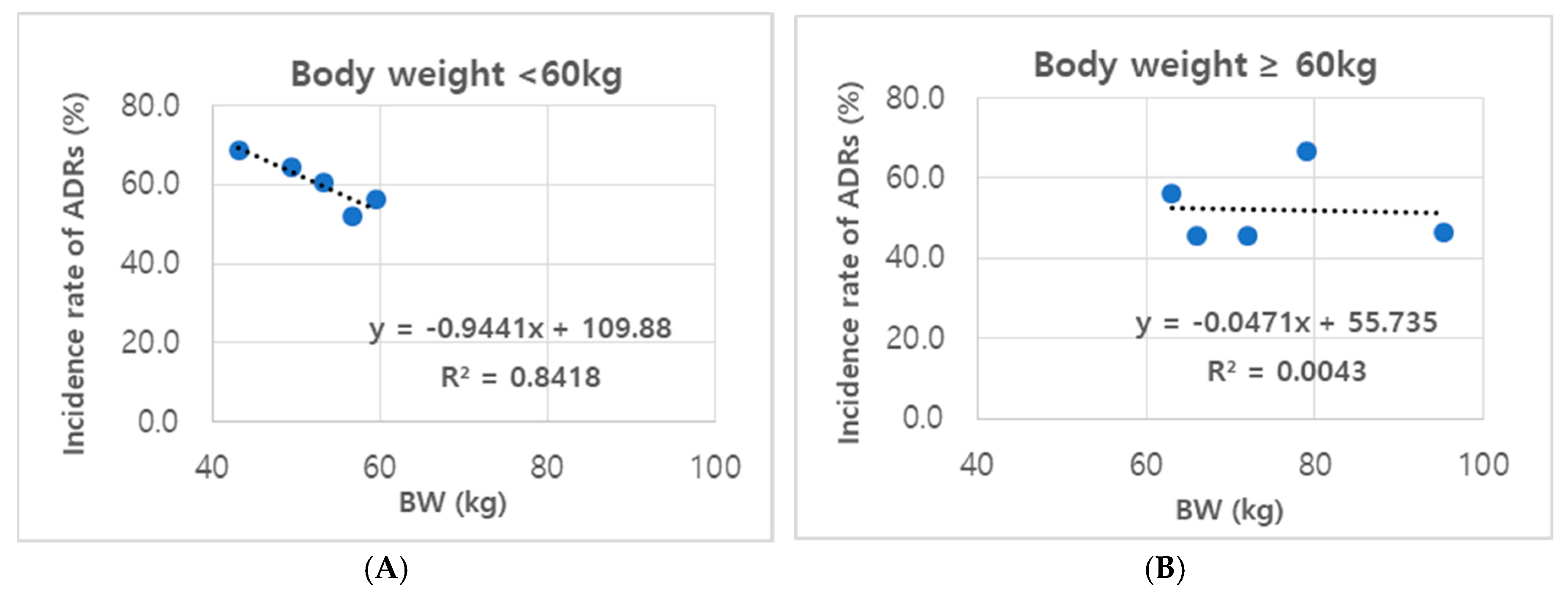
3.2. ADR Incidence Rates
3.3. ADR Types
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Lis, K.; Kuzawińska, O.; Bałkowiec-Iskra, E. Tumor necrosis factor inhibitors-state of knowledge. Arch. Med. Sci. 2014, 10, 1175–1185. [Google Scholar] [CrossRef]
- Cassinotti, A.; Sandro, A.; Porro, G.B. Adalimumab for the treatment of Crohn’s disease. Biologics 2008, 2, 763–777. [Google Scholar]
- Burmester, G.R.; Gordon, K.B.; Rosenbaum, J.T.; Arikan, D.; Lau, W.; Li, P.; Faccin, F.; Panaccione, R. Long-term safety of adalimumab in 29,967 adult patients from global clinical trials across multiple indications: An updated analysis. Adv. Ther. 2020, 37, 364–380. [Google Scholar] [CrossRef] [PubMed]
- FDA. Approval Package for BLA125057/S280/. Available online: https://www.accessdata.fda.gov/drugsatfdadocs/bla/2010/125057Orig1s0213.pdf (accessed on 25 July 2019).
- Datta, K.; Ghosh, R.K.; Ghosh, S.M. Serious neutropenia following etanercept administration in a 62 years female patient of rheumatoid arthritis. J. Assoc. Physicians India 2010, 58, 643–644. [Google Scholar] [PubMed]
- Tayar, J.H.; Lopez-Olivo, M.A.; Suarez-Almazor, M.E. Adalimumab: 8 years of experience in rheumatoid arthritis. Int. J. Clin. Rheumatol. 2013, 8, 165–184. [Google Scholar] [CrossRef]
- Chu, L.H.; Portugal, C.; Kawatkar, A.A.; Stohl, W.; Nichol, M.B. Racial/ethnic differences in the use of biologic disease-modifying antirheumatic drugs among California Medicaid rheumatoid arthritis patients. Arthritis Care Res. 2013, 65, 299–303. [Google Scholar] [CrossRef] [PubMed]
- Lie, M.R.; Kreijne, J.E.; van der Woude, C.J. Sex is associated with adalimumab side effects and drug survival in patients with Crohn’s disease. Inflamm. Bowel Dis. 2017, 23, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Vavricka, S.R.; Bentele, N.; Scharl, M.; Rogler, G.; Zeitz, J.; Frei, P.; Straumann, A.; Binek, J.; Schoepfer, A.M.; Fried, M.; et al. Systematic assessment of factors influencing preferences of Crohn’s disease patients in selecting an anti-tumor necrosis factor agent (CHOOSE TNF Trial). Inflamm. Bowel Dis. 2012, 18, 1523–1530. [Google Scholar] [CrossRef] [PubMed]
- Van der Valk, M.E.; Mangen, M.J.; Leenders, M.; Dijkstra, G.; van Bodegraven, A.A.; Fidder, H.H.; de Jong, D.J.; Pierik, M.; van der Woude, C.J.; Romberg-Camps, M.J.; et al. Healthcare costs of inflammatory bowel disease have shifted from hospitalization and surgery towards anti-TNFα therapy results from the COIN study. Gut 2014, 63, 72–79. [Google Scholar] [CrossRef]
- Nakase, H.; Motoya, S.; Matsumoto, T.; Watanabe, K.; Hisamatsu, T.; Yoshimura, N.; Ishida, T.; Kato, S.; Nakagawa, T.; Esaki, M.; et al. Significance of measurement of serum trough level and anti-drug antibody of adalimumab as personalized pharmacokinetics in patients with Crohn’s disease: A sub-analysis of the DIAMOND trial. Aliment. Pharmacol. Ther. 2017, 46, 873–882. [Google Scholar] [CrossRef]
- Luzier, A.B.; Killian, A.; Wilton, J.H.; Wilson, M.F.; Forrest, A.; Kazierad, D.J. Gender related effects on metoprolol pharmacokinetics and pharmacodynamics in healthy volunteers. Clin. Pharmacol. Ther. 1999, 66, 594–601. [Google Scholar] [CrossRef] [PubMed]
- Regitz-Zagrosek, V.; Seeland, U. Sex and gender differences in clinical medicine. In Sex and Gender Differences in Pharmacology; Springer: Berlin/Heidelberg, Germany, 2012; Volume 214, pp. 3–22. [Google Scholar]
- Schmetzer, O.; Florcken, A. Sex differences in the drug therapy for oncologic diseases. In Sex and Gender Differences in Pharmacology; Springer: Berlin/Heidelberg, Germany, 2012; Volume 214, pp. 411–442. [Google Scholar]
- Islam, M.; Iqbal, U.; Walther, B.A.; Nguyen, P.-A.; Li, Y.-C.; Dubey, N.K.; Poly, T.N.; Masud, J.H.B.; Atique, S.; Syed-Abdul, S. Gender based personalized pharmacotherapy: A systemic review. Arch. Gynecol. Obstet. 2017, 295, 1305–1317. [Google Scholar] [CrossRef] [PubMed]
- Soldin, O.P.; Mattison, D.R. Sex differences in pharmacokinetics and pharmacodynamics. Clin. Pharmacokinet. 2009, 49, 143–157. [Google Scholar] [CrossRef] [PubMed]
- Amderson, G.D. Gender differences in pharmacological response. Int. Rev. Neurobiol. 2008, 83, 1–10. [Google Scholar]
- Panaccione, R.; Loftus, E.V.; Binion, D.; McHugh, K.; Alam, S.; Chen, N.; Guerette, B.; Mulani, P.; Chao, J. Efficacy and safety of adalimumab in Canadian patients with moderate to severe Crohn’s disease: Results of the adalimumab in Canadian subjects with moderate to severe Crohn’s disease (ACCESS) trial. Can. J. Gastroenterol. 2011, 25, 419–425. [Google Scholar] [CrossRef]
- Bartley, E.J.; Fillingim, R.B. Sex differences in pain: A brief review of clinical and experimental finding. Br. J. Anaesth. 2013, 111, 52–58. [Google Scholar] [CrossRef]
- Peters, C.P.; Eshuis, E.J.; Toxopeüs, F.M.; Hellemons, M.E.; Jansen, J.M.; D’Haens, G.R.; Fockens, P.; Stokkers, P.C.; Tuynman, H.A.; van Bodegraven, A.A.; et al. Adalimumab for Crohn’s disease: Long-term sustained benefit in a population-based cohort of 438 patients. J. Crohn’s Colitis 2014, 8, 766–775. [Google Scholar] [CrossRef]
- National Institute for Health and Care Excellence. Infliximab and Adalimumab for Crohn’s Disease. Technology Appraisal Guidance. (Published Date: 19 May 2010). Available online: https://www.nice.org.uk/guidance/TA187 (accessed on 25 July 2019).
- Sato, E.; Tanaka, E.; Ochiai, M.; Shimizu, Y.; Kobayashi, A.; Shidara, K.; Hoshi, D.; Sugimoto, N.; Inoue, E.; Seto, Y.; et al. Chronological changes in baseline disease activity of patients with rheumatoid arthritis who received biologic DMARDs between 2003 and 2012. Mod. Rheumatol. 2015, 25, 350–357. [Google Scholar] [CrossRef]
- Fiorino, G.; Gilardi, D.; Correale, C.; Furfaro, F.; Roda, G.; Loy, L.; Argollo, M.; Allocca, M.; Peyrin-Biroulet, L.; Danese, S. Biosimilars of adalimumab: The upcoming challenge in IBD. Expert Opin. Biol. Ther. 2019, 19, 1023–1030. [Google Scholar] [CrossRef]
- Severs, M.; Oldenburg, B.; van Bodegraven, A.A.; Siersema, P.D.; Mangen, M.J.; Initiative of Crohn’s and Colitis. The economic impact of the introduction of biosimilars in inflammatory bowel disease. J. Crohn’s Colitis 2017, 11, 289–296. [Google Scholar] [CrossRef]
- Lesuis, N.; Befrits, R.; Nyberg, F.; van Vollenhoven, R.F. Gender and the treatment of immune mediated chronic inflammatory diseases: Rheumatoid arthritis, inflammatory bowel disease and psoriasis: An observational study. BMC Med. 2012, 10, 82. [Google Scholar] [CrossRef]
- Lin, P.T.; Wang, S.H.; Chi, C.C. Drug survival of biologics in treating psoriasis: A meta-analysis of real-world evidence. Sci. Rep. 2018, 8, 16068. [Google Scholar] [CrossRef] [PubMed]
- Rusman, T.; van Vollenhoven, R.F.; van der Horst-Bruinsma, I.E. Gender differences in axial spondyloarthritis: Women are not so lucky. Curr. Rheumatol. Rep. 2018, 20, 35. [Google Scholar] [CrossRef] [PubMed]
- Zelinkova, Z.; Bultman, E.; Vogelaar, L.; Bouziane, C.; Kuipers, E.J.; van der Woude, C.J. Sex-dimorphic adverse drug reactions to immune suppressive agents in inflammatory bowel disease. World J. Gastroenterol. 2012, 18, 6967–6973. [Google Scholar] [CrossRef] [PubMed]
- Panaccione, R.; Sandborn, W.J.; D’Haens, G.; Wolf, D.C.; Berg, S.; Maa, J.F.; Petersson, J.; Robinson, A.M. Clinical benefit of long-term adalimumab treatment in patients with Crohn’s disease following loss of response or intolerance to infliximab: 96-week efficacy data from GAIN/ADHERE Trials. J. Crohn’s Colitis 2018, 12, 930–938. [Google Scholar] [CrossRef]
- Hansel, K.; Bianchi, L.; Lanza, F.; Bini, V.; Stingeni, L. Adalimumab dose tapering in psoriasis: Predictive factors for maintenance of complete clearance. Acta Derm. Venereol. 2017, 97, 346–350. [Google Scholar] [CrossRef]
- Scheinfeld, N. Adalimumab: A review of side effect. Expert Opin. Drug Saf. 2005, 4, 637–641. [Google Scholar] [CrossRef]
- Harigai, M.; Tsuchiya, T.; Kawana, K.; Kurimoto, S. Long-term safety and effectiveness of adalimumab for the treatment of Japanese patients with rheumatoid arthritis: 3-year results from a postmarketing surveillance of 552 patients. Mod. Rheumatol. 2008, 28, 30–38. [Google Scholar] [CrossRef]
- Chao, Y.S.; Visintini, S. Biologics Dose Escalation for the Treatment of Inflammatory Bowel Disease: A Review of Clinical Effectiveness, Cost-effectiveness and Guidelines. 2018. Available online: http://creativecommons.org/licenses/by-nc-nd/4.0 (accessed on 25 July 2019).
- Humira (Adalimumab) Package Insert; AbbVie Inc.: North Chicago, IL, USA, 2013; Available online: https://www.humira.com (accessed on 25 July 2019).
- Hojgaard, P.; Blintgorg, B.; Kristensen, L.E.; Budbjornsson, B.; Love, T.J.; Dreyer, L. The influence of obesity on response to tumour necrosis factor-α inhibitors in psoriatic arthritis: Results from the DANBIO and ICEBIO registries. Rheumatology 2016, 55, 2191–2199. [Google Scholar] [CrossRef]
| Categories | |||
|---|---|---|---|
| Total (n = 461) | Male (n = 268) | Female (n = 193) | p-Values |
| Age (Median, (min-max)) * | 35.5 (19–78) | 48 (19–84) | p < 0.001 |
| BW (Median, (min-max)) * | 66.0 (36.8–120) | 54.0 (31.7–80.0) | p = 0.038 |
| Effect (n, (%)) | |||
| Yes | 233 (86.9%) | 156 (83.6%) | p = 0.075 |
| No * | 25 (9.3%) | 32 (13.5%) | p = 0.054 |
| Non-Judgment | 7 (2.6%) | 5 (2.9%) | p = 0.989 |
| Number of Patients with ADRs (n, (%)) * | 140 (52.2%) | 122 (63.2%) | p = 0.019 |
| Number of ADRs (n) | 223 | 199 | p = 0.155 |
| ADR per Patient (No. of Incidences/No. of Patients with ADR) | 1.6 | 1.6 | |
| Diagnosis | |||
| (1) Ankylosing Spondylitis (A) | |||
| Total (n = 149) | Male (n = 122) | Female (n = 27) | |
| Age (Median, (min-max)) | 36 (19–78) | 41 (19–67) | p = 0.401 |
| BW (Median, (min-max)) | 72.0 (50.0–120.0) | 58.4 (38.6–80.0) | p = 0.152 |
| Effect (n, (%)) | 109 (89.3%) | 22 (81.5%) | p = 0.257 |
| Number of Patients with ADRs (n, (%)) * | 74 (60.7%) | 22 (81.5%) | p = 0.041 |
| Number of ADRs (n) | 122 | 43 | p = 0.066 |
| ADR Cases per Patient | 1.6 | 2.0 | |
| (2) Rheumatoid Arthritis (R) | |||
| Total (n = 125) | Male (n = 38) | Female (n = 87) | |
| Age (Median, (min-max)) | 31.5 (19–78) | 54 (19–84) | p = 0.194 |
| BW (Median, (min-max)) | 65.0 (36.8–100.0) | 55.0 (31.7–74.0) | p = 0.626 |
| Effect (n, (%)) | 33 (86.8%) | 67 (77.0%) | p = 0.206 |
| Number of Patients with ADRs (n, (%)) | 19 (50.0%) | 56 (64.4%) | p = 0.131 |
| Number of ADRs (n) * | 36 | 79 | p = 0.037 |
| ADR Cases per Patient | 1.9 | 1.4 | |
| (3) Bechet’s Disease Plus Crohn’s Disease (B + C) | |||
| Total (n = 144) | Male (n = 91) | Female (n = 53) | |
| Age (Median, (min-max)) | 35 (19–71) | 36 (19–75) | p = 0.093 |
| BW (Median, (min-max)) | 60.3 (38.5–98.6) | 49.6 (38.3–76.6) | p = 0.241 |
| Effect (n, (%)) | 76 (83.5%) | 45 (84.9%) | p = 0.826 |
| Number of Patients with ADRs (n, (%)) | 37 (40.7%) | 30 (56.6%) | p = 0.064 |
| Number of ADRs (n) | 51 | 53 | p = 0.115 |
| ADR Cases per Patient | 1.4 | 1.8 | |
| (4) Psoriasis (P) | |||
| Total (n = 15) | Male (n = 8) | Female (n = 7) | |
| Age (Median, (min-max)) | 38 (25–67) | 58 (46–64) | p = 0.307 |
| BW (Median, (min-max)) | 71.5 (56.7–87.0) | 54.0 (48.2–65.0) | p = 0.449 |
| Effect (n, (%)) | 6 (75.0%) | 6 (85.7%) | p = 0.605 |
| Number of Patient with ADRs (n, (%)) | 5 (62.5%) | 5 (71.4%) | p = 0.714 |
| Number of ADRs (n) | 6 | 10 | p = 0.267 |
| ADR Cases per Patient | 1.2 | 2.0 | |
| (5) Two or More Other Diseases (Comorbid Diseases) | |||
| Total (n = 17) | Male (n = 6) | Female (n = 11) | |
| Age (Median, (min-max)) | 47 (22–60) | 37 (19–71) | p = 0.319 |
| BW (Median, (min-max)) | 57.9 (54.0–86.0) | 57.0 (34.6–63.2) | p = 0.386 |
| Effect (n, (%)) | 6 (100.0%) | 9 (81.8%) | p = 0.266 |
| Number of Patients with ADRs (n, (%)) | 5 (83.3%) | 7 (63.6%) | p = 0.394 |
| Number of ADRs (n) | 8 | 11 | p = 0.315 |
| ADR Cases per Patient | 1.6 | 1.6 | |
| (6) Others (E) | |||
| Total (n = 11) | Male (n = 3) | Female (n = 8) | |
| Age (Median, (min-max)) | 36 (36–48) | 26.5 (19–71) | p = 0.388 |
| BW [median, (min-max)) | 95.7 (86.8–109.2) | 54.1 (49.0–74.5) | p = 0.358 |
| Effect (n, (%)) | 3 (100.0%) | 7 (87.5%) | p = 0.521 |
| Number of Patients with ADRs (n, (%)) | 0 (0.0%) | 2 (25.0%) | p = 0.338 |
| Number of ADRs (n) | 0 | 3 | p = 0.632 |
| ADR Cases Per Patient | 0.0 | 1.5 |
| Males | Females | p-Values | |
|---|---|---|---|
| BW ≥ 54.0 kg (n = 334) | |||
| Number of Patients (n, (%)) | 238 (71.3%) | 96 (28.7%) | |
| Age (Median, (min–max)) * | 36 (19–78) | 50.5 (19–84) | p = 0.028 |
| BW (Median, (min-max)) | 67.5 (54.0–120.0) | 59.8 (54.0–80.0) | p = 0.215 |
| Effect, Yes (n, (%)) | 209 (87.8%) | 78 (81.3%) | p = 0.118 |
| Number of Patients with ADRs (n, (%)) * | 120 (50.4%) | 56 (58.3%) | p < 0.001 |
| ADR Cases per Patient (Incidences/Number of Patients with ADR) | 1.56 | 1.66 | p = 0.470 |
| BW < 54.0 kg (n = 127) | |||
| Number of Patients (n, (%)) | 30 (23.6%) | 97 (76.4%) | |
| Age (Median, (min-max)) | 30.5 (20–78) | 47 (19–79) | p = 0.271 |
| BW (Median, (min-max)) | 49.7 (36.8–53.5) | 49.0 (31.7–53.9) | p = 0.358 |
| Effect, Yes (n, (%)) | 24 (80.0%) | 78 (80.4%) | p = 0.960 |
| Number of Patients with ADRs (n, (%)) | 20 (66.7%) | 66 (68.0%) | p = 0.888 |
| ADR Cases per Patient (Incidences/Number of Patients with ADR) | 1.75 | 1.61 | p = 0.586 |
| Types of ADRs | Male | Female | p-Value | |
|---|---|---|---|---|
| Total Number of Patients (n = 461) (n, (%)) | 268 (58.1%) | 193 (41.9%) | ||
| Infection (n, (%)) | 59 (22.0%) | 44 (22.8%) | p = 0.768 | |
| Pain (n, (%)) | 18 (6.7%) | 16 (8.3%) | p = 0.993 | |
| Dermatologicals (n, (%)) | Skin * | 59 (22.0%) | 60 (31.1%) | p = 0.030 |
| Alopecia | 5 (1.9%) | 7 (3.6%) | p = 0.374 | |
| Gastrointestinals (n, (%)) | 26 (9.7%) | 17 (8.8%) | p = 0.473 | |
| Psychiatrics (n, (%)) * | 4 (1.5%) | 11 (5.0%) | p = 0.012 | |
| Abnormal Lab Test (n, (%)) | 32 (12.0%) | 30 (15.6%) | p = 0.625 | |
| Others (n, (%)) | 19 (7.1%) | 14 (7.3%) | p = 0.946 | |
| (1) Ankylosing Spondylitis (A) | ||||
| Number of Patients (n = 149) (n, (%)) | 122 (81.9%) | 27 (18.1%) | ||
| Infection (n, (%)) | 33 (27.0%) | 8 (29.6%) | p = 0.786 | |
| Pain (n, (%)) | 9 (7.4%) | 5 (18.5%) | p = 0.073 | |
| Dermatologicals (n, (%)) | Skin * | 32 (26.2%) | 13 (48.1%) | p = 0.025 |
| Alopecia | 2 (1.6%) | 0 (0.0%) | p = 0.503 | |
| Gastrointestinals (n, (%)) | 20 (16.4%) | 3 (11.1%) | p = 0.492 | |
| Psychiatrics (n, (%)) | 4 (3.3%) | 3 (1.6%) | p = 0.082 | |
| Abnormal Lab Test (n, (%)) | 17 (14.0%) | 7 (25.9%) | p = 0.125 | |
| Others (n, (%)) * | 5 (4.1%) | 4 (14.8%) | p = 0.034 | |
| (2) Rheumatoid Arthritis (R) | ||||
| Number of Patients (n = 125) (n, (%)) | 38 (30.4%) | 87 (69.6%) | ||
| Infection (n, (%)) | 9 (23.7%) | 19 (21.8%) | p = 0.820 | |
| Pain (n, (%)) | 4 (10.5%) | 7 (8.0%) | p = 0.653 | |
| Dermatologicals (n, (%)) | Skin | 8 (21.1%) | 22 (25.3%) | p = 0.481 |
| Alopecia | 1 (2.6%) | 4 (4.6%) | p = 0.873 | |
| Gastrointestinals (n, (%)) | 4 (10.5%) | 8 (9.2%) | p = 0.816 | |
| Psychiatrics (n, (%)) | 0 (0.0%) | 5 (1.1%) | p = 0.131 | |
| Abnormal Lab Test (n, (%)) | 3 (7.9%) | 6 (6.8%) | p = 0.843 | |
| Others (n, (%)) | 6 (15.8%) | 8 (9.2%) | p = 0.389 | |
| (3) Bechet’s Disease Plus Crohn’s Disease (B + C) | ||||
| Subtotal (n = 144) (n, (%)) | 91 (63.2%) | 53 (43.8%) | ||
| Infection (n, (%)) | 14 (15.4%) | 11 (20.8%) | p = 0.412 | |
| Pain (n, (%)) | 4 (4.4%) | 3 (5.7%) | p = 0.426 | |
| Dermatologicals (n, (%)) | Skin | 15 (16.5%) | 15 (28.3%) | p = 0.092 |
| Alopecia | 1 (1.1%) | 3 (5.7%) | p = 0.108 | |
| Gastrointestinals (n, (%)) | 2 (2.2%) | 3 (5.7%) | p = 0.274 | |
| Psychiatrics (n, (%)) | 0 (0.0%) | 2 (3.8%) | p = 0.062 | |
| Abnormal Lab Test (n, (%)) * | 10 (11.0%) | 14 (26.4%) | p = 0.017 | |
| Others (n, (%)) | 5 (5.5%) | 1 (1.9%) | p = 0.296 | |
| (4) Psoriasis (P) | ||||
| Subtotal (n = 15) (n, (%)) | 8 (53.3%) | 7 (46.7%) | ||
| Infection (n, (%)) | 2 (25.0%) | 2 (28.6%) | p = 0.876 | |
| Pain (n, (%)) | 0 (0.0%) | 0 (0.0%) | - | |
| Dermatologicals (n, (%)) | Skin | 1 (12.5%) | 3 (42.9%) | p = 0.185 |
| Alopecia | 1 (12.5%) | 0 (0.0%) | p = 0.333 | |
| Gastrointestinals (n, (%)) | 0 (0.0%) | 2 (28.6%) | p = 0.104 | |
| Psychiatrics (n, (%)) | 0 (0.0%) | 1 (14.3%) | p = 0.268 | |
| Abnormal Lab Test (n, (%)) | 1 (12.5%) | 2 (28.6%) | p = 0.438 | |
| Others (n, (%)) | 1 (12.5%) | 0 (0.0%) | p = 0.333 | |
| (5) Two or More Other Diseases (Comorbid Diseases) | ||||
| Subtotal (n = 17) (n, (%)) | 6 (35.3%) | 11 (64.7%) | ||
| Infection (n, (%)) | 1 (16.7%) | 3 (27.3%) | p = 0.159 | |
| Pain (n, (%)) | 1 (16.7%) | 1 (9.1%) | p = 0.643 | |
| Dermatologicals (n, (%)) | Skin | 3 (50.0%) | 7 (63.6%) | p = 0.585 |
| Alopecia | 0 (0.0%) | 0 (0.0%) | - | |
| Gastrointestinals (n, (%)) | 0 (0.0%) | 0 (0.0%) | - | |
| Psychiatrics (n, (%)) | 0 (0.0%) | 0 (0.0%) | - | |
| Abnormal Lab Test (n, %) | 1 (16.7%) | 1 (9.1%) | p = 0.643 | |
| Others (n, (%)) * | 2 (33.3%) | 0 (0.0%) | p = 0.041 | |
| (6) Others (E) | ||||
| Subtotal (n = 11) (n, (%)) | 3 (27.3%) | 8 (72.7%) | ||
| Infection (n, (%)) | 0 (0.0%) | 1 (12.5%) | p = 0.521 | |
| Pain (n, (%)) | 0 (0.0%) | 0 (0.0%) | - | |
| Dermatologicals (n, (%)) | Skin | 0 (0.0%) | 0 (0.0%) | - |
| Alopecia | 0 (0.0%) | 0 (0.0%) | - | |
| Gastrointestinals (n, (%)) | 0 (0.0%) | 1 (12.5%) | p = 0.521 | |
| Psychiatrics (n, (%)) | 0 (0.0%) | 0 (0.0%) | - | |
| Abnormal Lab Test (n, (%)) | 0 (0.0%) | 0 (0.0%) | - | |
| Others (n, (%)) | 0 (0.0%) | 1 (12.5%) | p = 0.521 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, K.S.; Choi, Y.H.; Moon, A.; Kim, S.G. Body Weight as a Determining Factor in the Predominance of Adverse Drug Reactions Induced by Fixed-Dose Adalimumab Injections in Female Patients in a Korean Hospital Setting. J. Clin. Med. 2020, 9, 461. https://doi.org/10.3390/jcm9020461
Kim KS, Choi YH, Moon A, Kim SG. Body Weight as a Determining Factor in the Predominance of Adverse Drug Reactions Induced by Fixed-Dose Adalimumab Injections in Female Patients in a Korean Hospital Setting. Journal of Clinical Medicine. 2020; 9(2):461. https://doi.org/10.3390/jcm9020461
Chicago/Turabian StyleKim, Kwi Suk, Young Hee Choi, Aree Moon, and Sang Geon Kim. 2020. "Body Weight as a Determining Factor in the Predominance of Adverse Drug Reactions Induced by Fixed-Dose Adalimumab Injections in Female Patients in a Korean Hospital Setting" Journal of Clinical Medicine 9, no. 2: 461. https://doi.org/10.3390/jcm9020461
APA StyleKim, K. S., Choi, Y. H., Moon, A., & Kim, S. G. (2020). Body Weight as a Determining Factor in the Predominance of Adverse Drug Reactions Induced by Fixed-Dose Adalimumab Injections in Female Patients in a Korean Hospital Setting. Journal of Clinical Medicine, 9(2), 461. https://doi.org/10.3390/jcm9020461








