GDNF, A Neuron-Derived Factor Upregulated in Glial Cells during Disease
Abstract
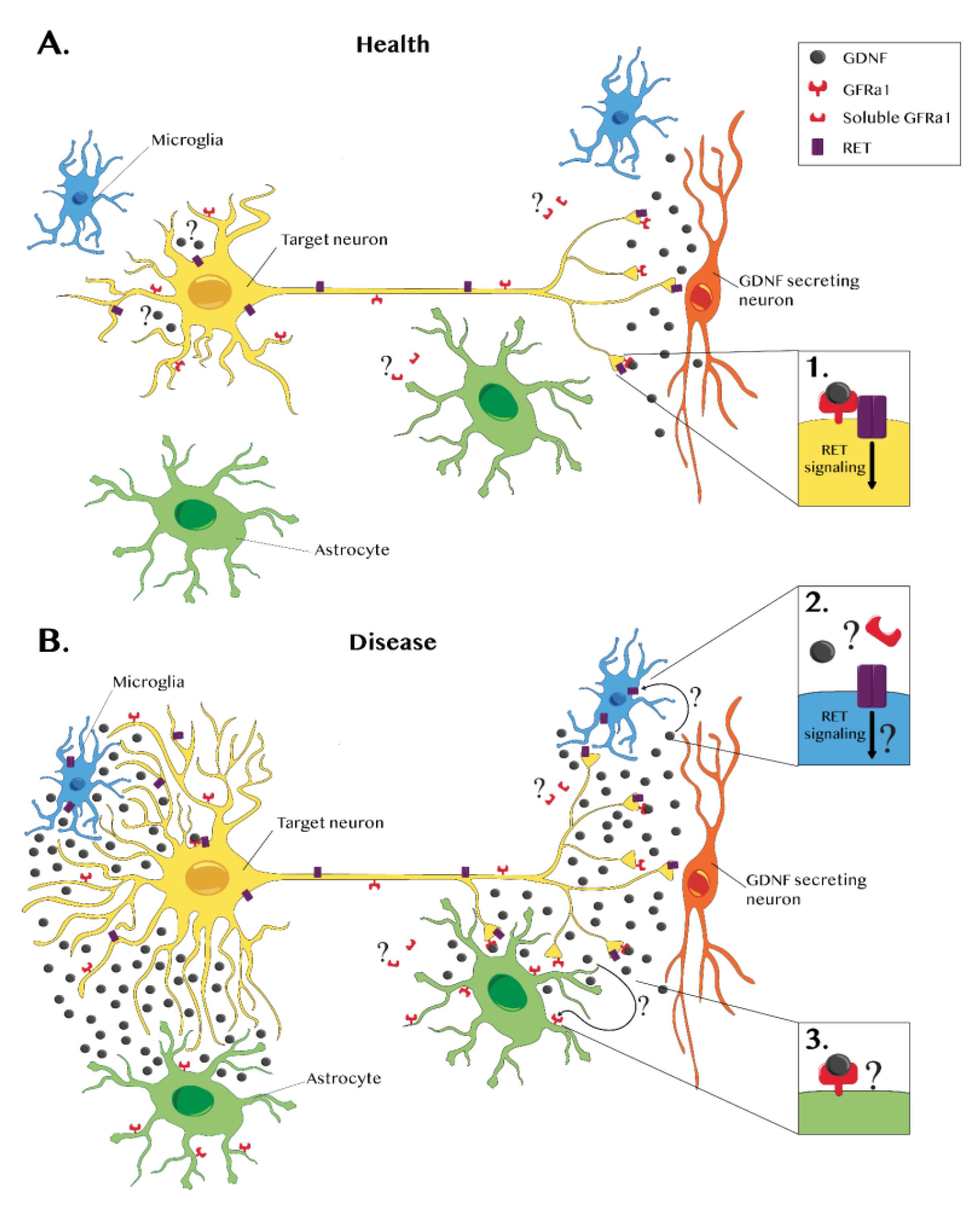
1. Introduction
2. Glial Cells Express GDNF during Disease
3. Glial Cells Express GDNF Receptors during Disease
4. Conclusions and Further Prospects
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lin, L.F.; Doherty, D.H.; Lile, J.D.; Bektesh, S.; Collins, F. GDNF: A glial cell line-derived neurotrophic factor for midbrain dopaminergic neurons. Science 1993, 260, 1130–1132. [Google Scholar] [CrossRef] [PubMed]
- Kirik, D.; Rosenblad, C.; Bjorklund, A.; Mandel, R.J. Long-term rAAV-mediated gene transfer of GDNF in the rat Parkinson’s model: Intrastriatal but not intranigral transduction promotes functional regeneration in the lesioned nigrostriatal system. J. Neurosci. 2000, 20, 4686–4700. [Google Scholar] [CrossRef] [PubMed]
- Ramaswamy, S.; McBride, J.L.; Han, I.; Berry-Kravis, E.M.; Zhou, L.; Herzog, C.D.; Gasmi, M.; Bartus, R.T.; Kordower, J.H. Intrastriatal CERE-120 (AAV-Neurturin) protects striatal and cortical neurons and delays motor deficits in a transgenic mouse model of Huntington’s disease. Neurobiol. Dis. 2009, 34, 40–50. [Google Scholar] [CrossRef] [PubMed]
- Blits, B.; Carlstedt, T.P.; Ruitenberg, M.J.; de Winter, F.; Hermens, W.T.; Dijkhuizen, P.A.; Claasens, J.W.; Eggers, R.; van der Sluis, R.; Tenenbaum, L.; et al. Rescue and sprouting of motoneurons following ventral root avulsion and reimplantation combined with intraspinal adeno-associated viral vector-mediated expression of glial cell line-derived neurotrophic factor or brain-derived neurotrophic factor. Exp. Neurol. 2004, 189, 303–316. [Google Scholar] [CrossRef] [PubMed]
- Georgievska, B.; Kirik, D.; Bjorklund, A. Aberrant sprouting and downregulation of tyrosine hydroxylase in lesioned nigrostriatal dopamine neurons induced by long-lasting overexpression of glial cell line derived neurotrophic factor in the striatum by lentiviral gene transfer. Exp. Neurol. 2002, 177, 461–474. [Google Scholar] [CrossRef] [PubMed]
- Eggers, R.; Hendriks, W.T.; Tannemaat, M.R.; van Heerikhuize, J.J.; Pool, C.W.; Carlstedt, T.P.; Zaldumbide, A.; Hoeben, R.C.; Boer, G.J.; Verhaagen, J. Neuroregenerative effects of lentiviral vector-mediated GDNF expression in reimplanted ventral roots. Mol. Cell Neurosci. 2008, 39, 105–117. [Google Scholar] [CrossRef] [PubMed]
- Tenenbaum, L.; Humbert-Claude, M. Glial Cell Line-Derived Neurotrophic Factor Gene Delivery in Parkinson’s Disease: A Delicate Balance between Neuroprotection, Trophic Effects, and Unwanted Compensatory Mechanisms. Front. Neuroanat. 2017, 11, 29. [Google Scholar] [CrossRef]
- Eggers, R.; de Winter, F.; Hoyng, S.A.; Hoeben, R.C.; Malessy, M.J.A.; Tannemaat, M.R.; Verhaagen, J. Timed GDNF gene therapy using an immune-evasive gene switch promotes long distance axon regeneration. Brain 2019, 142, 295–311. [Google Scholar] [CrossRef]
- Marks, W.J., Jr.; Bartus, R.T.; Siffert, J.; Davis, C.S.; Lozano, A.; Boulis, N.; Vitek, J.; Stacy, M.; Turner, D.; Verhagen, L.; et al. Gene delivery of AAV2-neurturin for Parkinson’s disease: A double-blind, randomised, controlled trial. Lancet Neurol. 2010, 9, 1164–1172. [Google Scholar] [CrossRef]
- Lang, A.E.; Gill, S.; Patel, N.K.; Lozano, A.; Nutt, J.G.; Penn, R.; Brooks, D.J.; Hotton, G.; Moro, E.; Heywood, P.; et al. Randomized controlled trial of intraputamenal glial cell line-derived neurotrophic factor infusion in Parkinson disease. Ann. Neurol. 2006, 59, 459–466. [Google Scholar] [CrossRef]
- Heiss, J.D.; Lungu, C.; Hammoud, D.A.; Herscovitch, P.; Ehrlich, D.J.; Argersinger, D.P.; Sinharay, S.; Scott, G.; Wu, T.; Federoff, H.J.; et al. Trial of magnetic resonance-guided putaminal gene therapy for advanced Parkinson’s disease. Mov. Disord. 2019, 34, 1073–1078. [Google Scholar] [CrossRef] [PubMed]
- Whone, A.; Luz, M.; Boca, M.; Woolley, M.; Mooney, L.; Dharia, S.; Broadfoot, J.; Cronin, D.; Schroers, C.; Barua, N.U.; et al. Randomized trial of intermittent intraputamenal glial cell line-derived neurotrophic factor in Parkinson’s disease. Brain 2019, 142, 512–525. [Google Scholar] [CrossRef] [PubMed]
- Bartus, R.T.; Kordower, J.H.; Johnson, E.M., Jr.; Brown, L.; Kruegel, B.R.; Chu, Y.; Baumann, T.L.; Lang, A.E.; Olanow, C.W.; Herzog, C.D. Post-mortem assessment of the short and long-term effects of the trophic factor neurturin in patients with α-synucleinopathies. Neurobiol. Dis. 2015, 78, 162–171. [Google Scholar] [CrossRef] [PubMed]
- Ernsberger, U. The role of GDNF family ligand signalling in the differentiation of sympathetic and dorsal root ganglion neurons. Cell Tissue Res. 2008, 333, 353–371. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Pozas, E.; Ibanez, C.F. GDNF and GFRalpha1 promote differentiation and tangential migration of cortical GABAergic neurons. Neuron 2005, 45, 701–713. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Guo, H.; Hellard, D.T.; Katz, D.M. Glial cell line-derived neurotrophic factor (GDNF) is required for differentiation of pontine noradrenergic neurons and patterning of central respiratory output. Neuroscience 2005, 130, 95–105. [Google Scholar] [CrossRef]
- Hellmich, H.L.; Kos, L.; Cho, E.S.; Mahon, K.A.; Zimmer, A. Embryonic expression of glial cell-line derived neurotrophic factor (GDNF) suggests multiple developmental roles in neural differentiation and epithelial-mesenchymal interactions. Mech. Dev. 1996, 54, 95–105. [Google Scholar] [CrossRef]
- Hidalgo-Figueroa, M.; Bonilla, S.; Gutierrez, F.; Pascual, A.; Lopez-Barneo, J. GDNF is predominantly expressed in the PV+ neostriatal interneuronal ensemble in normal mouse and after injury of the nigrostriatal pathway. J. Neurosci. 2012, 32, 864–872. [Google Scholar] [CrossRef]
- Kumar, A.; Kopra, J.; Varendi, K.; Porokuokka, L.L.; Panhelainen, A.; Kuure, S.; Marshall, P.; Karalija, N.; Harma, M.A.; Vilenius, C.; et al. GDNF Overexpression from the native locus reveals its role in the nigrostriatal dopaminergic system function. PLoS Genet. 2015, 11, e1005710. [Google Scholar] [CrossRef]
- Pochon, N.A.; Menoud, A.; Tseng, J.L.; Zurn, A.D.; Aebischer, P. Neuronal GDNF expression in the adult rat nervous system identified by in situ hybridization. Eur. J. Neurosci. 1997, 9, 463–471. [Google Scholar] [CrossRef]
- Widenfalk, J.; Nosrat, C.; Tomac, A.; Westphal, H.; Hoffer, B.; Olson, L. Neurturin and glial cell line-derived neurotrophic factor receptor-β (GDNFR-β), novel proteins related to GDNF and GDNFR-α with specific cellular patterns of expression suggesting roles in the developing and adult nervous system and in peripheral organs. J. Neurosci. 1997, 17, 8506–8519. [Google Scholar] [CrossRef] [PubMed]
- Trupp, M.; Belluardo, N.; Funakoshi, H.; Ibanez, C.F. Complementary and overlapping expression of glial cell line-derived neurotrophic factor (GDNF), c-ret proto-oncogene, and GDNF receptor-α indicates multiple mechanisms of trophic actions in the adult rat CNS. J. Neurosci. 1997, 17, 3554–3567. [Google Scholar] [CrossRef] [PubMed]
- Lonka-Nevalaita, L.; Lume, M.; Leppanen, S.; Jokitalo, E.; Peranen, J.; Saarma, M. Characterization of the intracellular localization, processing, and secretion of two glial cell line-derived neurotrophic factor splice isoforms. J. Neurosci. 2010, 30, 11403–11413. [Google Scholar] [CrossRef] [PubMed]
- Bizon, J.L.; Lauterborn, J.C.; Gall, C.M. Subpopulations of striatal interneurons can be distinguished on the basis of neurotrophic factor expression. J. Comp. Neurol. 1999, 408, 283–298. [Google Scholar] [CrossRef]
- Enterria-Morales, D.; Lopez-Lopez, I.; Lopez-Barneo, J.; d’Anglemont de Tassigny, X. Role of glial cell line-derived neurotrophic factor in the maintenance of adult mesencephalic catecholaminergic neurons. Mov. Disord. 2020. [Google Scholar] [CrossRef] [PubMed]
- Poyhonen, S.; Er, S.; Domanskyi, A.; Airavaara, M. Effects of neurotrophic factors in glial cells in the central nervous system: Expression and properties in neurodegeneration and injury. Front. Physiol. 2019, 10, 486. [Google Scholar] [CrossRef]
- Saavedra, A.; Baltazar, G.; Duarte, E.P. Driving GDNF expression: The green and the red traffic lights. Prog. Neurobiol. 2008, 86, 186–215. [Google Scholar] [CrossRef][Green Version]
- Sotoyama, H.; Iwakura, Y.; Oda, K.; Sasaoka, T.; Takei, N.; Kakita, A.; Enomoto, H.; Nawa, H. Striatal hypodopamine phenotypes found in transgenic mice that overexpress glial cell line-derived neurotrophic factor. Neurosci. Lett. 2017, 654, 99–106. [Google Scholar] [CrossRef]
- Nosrat, C.A.; Tomac, A.; Lindqvist, E.; Lindskog, S.; Humpel, C.; Stromberg, I.; Ebendal, T.; Hoffer, B.J.; Olson, L. Cellular expression of GDNF mRNA suggests multiple functions inside and outside the nervous system. Cell Tissue Res. 1996, 286, 191–207. [Google Scholar] [CrossRef]
- Golden, J.P.; DeMaro, J.A.; Osborne, P.A.; Milbrandt, J.; Johnson, E.M., Jr. Expression of neurturin, GDNF, and GDNF family-receptor mRNA in the developing and mature mouse. Exp. Neurol. 1999, 158, 504–528. [Google Scholar] [CrossRef]
- Ortega-de San Luis, C.; Pascual, A. Simultaneous detection of both GDNF and GFRα1 expression patterns in the mouse central nervous system. Front. Neuroanat. 2016, 10, 73. [Google Scholar] [CrossRef]
- Ubhi, K.; Rockenstein, E.; Mante, M.; Inglis, C.; Adame, A.; Patrick, C.; Whitney, K.; Masliah, E. Neurodegeneration in a transgenic mouse model of multiple system atrophy is associated with altered expression of oligodendroglial-derived neurotrophic factors. J. Neurosci. 2010, 30, 6236–6246. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Merienne, N.; Meunier, C.; Schneider, A.; Seguin, J.; Nair, S.S.; Rocher, A.B.; Le Gras, S.; Keime, C.; Faull, R.; Pellerin, L.; et al. Cell-type-specific gene expression profiling in adult mouse brain reveals normal and disease-state signatures. Cell Rep. 2019, 26, 2477–2493 e2479. [Google Scholar] [CrossRef] [PubMed]
- Marco, S.; Canudas, A.M.; Canals, J.M.; Gavalda, N.; Perez-Navarro, E.; Alberch, J. Excitatory amino acids differentially regulate the expression of GDNF, neurturin, and their receptors in the adult rat striatum. Exp. Neurol. 2002, 174, 243–252. [Google Scholar] [CrossRef] [PubMed]
- Bresjanac, M.; Antauer, G. Reactive astrocytes of the quinolinic acid-lesioned rat striatum express GFRα1 as well as GDNF in vivo. Exp. Neurol. 2000, 164, 53–59. [Google Scholar] [CrossRef]
- Iravani, M.M.; Sadeghian, M.; Leung, C.C.; Jenner, P.; Rose, S. Lipopolysaccharide-induced nigral inflammation leads to increased IL-1β tissue content and expression of astrocytic glial cell line-derived neurotrophic factor. Neurosci. Lett. 2012, 510, 138–142. [Google Scholar] [CrossRef]
- Nakagawa, T.; Schwartz, J.P. Gene expression profiles of reactive astrocytes in dopamine-depleted striatum. Brain Pathol. 2004, 14, 275–280. [Google Scholar] [CrossRef]
- Katoh-Semba, R.; Tsuzuki, M.; Miyazaki, N.; Yoshida, A.; Nakajima, H.; Nakagawa, C.; Kitajima, S.; Matsuda, M. Distribution and immunohistochemical localization of GDNF protein in selected neural and non-neural tissues of rats during development and changes in unilateral 6-hydroxydopamine lesions. Neurosci. Res. 2007, 59, 277–287. [Google Scholar] [CrossRef]
- Patel, D.; Jana, A.; Roy, A.; Pahan, K. Cinnamon and its metabolite protect the nigrostriatum in a mouse model of Parkinson’s disease via astrocytic GDNF. J. Neuroimmune Pharmacol. 2019, 14, 503–518. [Google Scholar] [CrossRef]
- Tokumine, J.; Kakinohana, O.; Cizkova, D.; Smith, D.W.; Marsala, M. Changes in spinal GDNF, BDNF, and NT-3 expression after transient spinal cord ischemia in the rat. J. Neurosci. Res. 2003, 74, 552–561. [Google Scholar] [CrossRef]
- Batchelor, P.E.; Porritt, M.J.; Martinello, P.; Parish, C.L.; Liberatore, G.T.; Donnan, G.A.; Howells, D.W. Macrophages and microglia produce local trophic gradients that stimulate axonal sprouting toward but not beyond the wound edge. Mol. Cell Neurosci. 2002, 21, 436–453. [Google Scholar] [CrossRef] [PubMed]
- Batchelor, P.E.; Liberatore, G.T.; Wong, J.Y.; Porritt, M.J.; Frerichs, F.; Donnan, G.A.; Howells, D.W. Activated macrophages and microglia induce dopaminergic sprouting in the injured striatum and express brain-derived neurotrophic factor and glial cell line-derived neurotrophic factor. J. Neurosci. 1999, 19, 1708–1716. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, M.; Nitta, A.; Fukumitsu, H.; Nomoto, H.; Shen, L.; Furukawa, S. Inflammation-induced GDNF improves locomotor function after spinal cord injury. Neuroreport 2005, 16, 99–102. [Google Scholar] [CrossRef] [PubMed]
- Liberatore, G.T.; Wong, J.Y.; Porritt, M.J.; Donnan, G.A.; Howells, D.W. Expression of glial cell line-derived neurotrophic factor (GDNF) mRNA following mechanical injury to mouse striatum. Neuroreport 1997, 8, 3097–3101. [Google Scholar] [CrossRef]
- Ahn, M.; Jin, J.K.; Moon, C.; Matsumoto, Y.; Koh, C.S.; Shin, T. Glial cell line-derived neurotrophic factor is expressed by inflammatory cells in the sciatic nerves of Lewis rats with experimental autoimmune neuritis. J. Peripher. Nerv. Syst. 2010, 15, 104–112. [Google Scholar] [CrossRef]
- Chen, L.W.; Zhang, J.P.; Kwok-Yan Shum, D.; Chan, Y.S. Localization of nerve growth factor, neurotrophin-3, and glial cell line-derived neurotrophic factor in nestin-expressing reactive astrocytes in the caudate-putamen of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-treated C57/Bl mice. J. Comp. Neurol. 2006, 497, 898–909. [Google Scholar] [CrossRef]
- Kitamura, Y.; Inden, M.; Minamino, H.; Abe, M.; Takata, K.; Taniguchi, T. The 6-hydroxydopamine-induced nigrostriatal neurodegeneration produces microglia-like NG2 glial cells in the rat substantia nigra. Glia 2010, 58, 1686–1700. [Google Scholar] [CrossRef]
- Smirkin, A.; Matsumoto, H.; Takahashi, H.; Inoue, A.; Tagawa, M.; Ohue, S.; Watanabe, H.; Yano, H.; Kumon, Y.; Ohnishi, T.; et al. Iba1+/NG2+ macrophage-like cells expressing a variety of neuroprotective factors ameliorate ischemic damage of the brain. J. Cereb. Blood Flow Metab. 2010, 30, 603–615. [Google Scholar] [CrossRef]
- Sawada, H.; Hishida, R.; Hirata, Y.; Ono, K.; Suzuki, H.; Muramatsu, S.; Nakano, I.; Nagatsu, T.; Sawada, M. Activated microglia affect the nigro-striatal dopamine neurons differently in neonatal and aged mice treated with 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine. J. Neurosci. Res. 2007, 85, 1752–1761. [Google Scholar] [CrossRef]
- Kanthasamy, A.; Jin, H.; Charli, A.; Vellareddy, A.; Kanthasamy, A. Environmental neurotoxicant-induced dopaminergic neurodegeneration: A potential link to impaired neuroinflammatory mechanisms. Pharmacol. Ther. 2019, 197, 61–82. [Google Scholar] [CrossRef]
- Batchelor, P.E.; Liberatore, G.T.; Porritt, M.J.; Donnan, G.A.; Howells, D.W. Inhibition of brain-derived neurotrophic factor and glial cell line-derived neurotrophic factor expression reduces dopaminergic sprouting in the injured striatum. Eur. J. Neurosci. 2000, 12, 3462–3468. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Guderyon, M.J.; Li, Y.; Ge, G.; Bhattacharjee, A.; Ballard, C.; He, Z.; Masliah, E.; Clark, R.A.; O’Connor, J.C.; et al. Non-toxic HSC transplantation-based macrophage/microglia-mediated GDNF delivery for Parkinson’s disease. Mol. Ther. Methods Clin. Dev. 2020, 17, 83–98. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Li, X.; Ge, G.; Liu, J.; Biju, K.C.; Laing, S.D.; Qian, Y.; Ballard, C.; He, Z.; Masliah, E.; et al. GDNF-expressing macrophages mitigate loss of dopamine neurons and improve Parkinsonian symptoms in MitoPark mice. Sci. Rep. 2018, 8, 5460. [Google Scholar] [CrossRef] [PubMed]
- Georgievska, B.; Kirik, D.; Bjorklund, A. Overexpression of glial cell line-derived neurotrophic factor using a lentiviral vector induces time- and dose-dependent downregulation of tyrosine hydroxylase in the intact nigrostriatal dopamine system. J. Neurosci. 2004, 24, 6437–6445. [Google Scholar] [CrossRef]
- Paratcha, G.; Ledda, F.; Baars, L.; Coulpier, M.; Besset, V.; Anders, J.; Scott, R.; Ibanez, C.F. Released GFRα1 potentiates downstream signaling, neuronal survival, and differentiation via a novel mechanism of recruitment of c-Ret to lipid rafts. Neuron 2001, 29, 171–184. [Google Scholar] [CrossRef]
- Mikaels-Edman, A.; Baudet, C.; Ernfors, P. Soluble and bound forms of GFRα1 elicit different GDNF-independent neurite growth responses in primary sensory neurons. Dev. Dyn. 2003, 227, 27–34. [Google Scholar] [CrossRef]
- Airaksinen, M.S.; Titievsky, A.; Saarma, M. GDNF family neurotrophic factor signaling: Four masters, one servant? Mol. Cell Neurosci. 1999, 13, 313–325. [Google Scholar] [CrossRef]
- Hoffer, B.J.; Hoffman, A.; Bowenkamp, K.; Huettl, P.; Hudson, J.; Martin, D.; Lin, L.F.; Gerhardt, G.A. Glial cell line-derived neurotrophic factor reverses toxin-induced injury to midbrain dopaminergic neurons in vivo. Neurosci. Lett. 1994, 182, 107–111. [Google Scholar] [CrossRef]
- Tomac, A.; Widenfalk, J.; Lin, L.F.; Kohno, T.; Ebendal, T.; Hoffer, B.J.; Olson, L. Retrograde axonal transport of glial cell line-derived neurotrophic factor in the adult nigrostriatal system suggests a trophic role in the adult. Proc. Natl. Acad. Sci. USA 1995, 92, 8274–8278. [Google Scholar] [CrossRef]
- Henderson, C.E.; Phillips, H.S.; Pollock, R.A.; Davies, A.M.; Lemeulle, C.; Armanini, M.; Simmons, L.; Moffet, B.; Vandlen, R.A.; Simpson, L.L.C. GDNF: A potent survival factor for motoneurons present in peripheral nerve and muscle. Science 1994, 266, 1062–1064. [Google Scholar] [CrossRef]
- Arenas, E.; Trupp, M.; Akerud, P.; Ibanez, C.F. GDNF prevents degeneration and promotes the phenotype of brain noradrenergic neurons in vivo. Neuron 1995, 15, 1465–1473. [Google Scholar] [CrossRef]
- Rodrigues, D.M.; Li, A.Y.; Nair, D.G.; Blennerhassett, M.G. Glial cell line-derived neurotrophic factor is a key neurotrophin in the postnatal enteric nervous system. Neurogastroenterol. Motil. 2011, 23, e44–e56. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Okada, T.; Shimazaki, K.; Sheykholeslami, K.; Nomoto, T.; Muramatsu, S.I.; Mizukami, H.; Kume, A.; Xiao, S.; Ichimura, K.; et al. Protection against aminoglycoside-induced ototoxicity by regulated AAV vector-mediated GDNF gene transfer into the cochlea. Mol. Ther. 2008, 16, 474–480. [Google Scholar] [CrossRef]
- Cao, J.P.; Wang, H.J.; Yu, J.K.; Yang, H.; Xiao, C.H.; Gao, D.S. Involvement of NCAM in the effects of GDNF on the neurite outgrowth in the dopamine neurons. Neurosci. Res. 2008, 61, 390–397. [Google Scholar] [CrossRef]
- Paratcha, G.; Ledda, F.; Ibanez, C.F. The neural cell adhesion molecule NCAM is an alternative signaling receptor for GDNF family ligands. Cell 2003, 113, 867–879. [Google Scholar] [CrossRef]
- Ledda, F.; Paratcha, G.; Sandoval-Guzman, T.; Ibanez, C.F. GDNF and GFRalpha1 promote formation of neuronal synapses by ligand-induced cell adhesion. Nat. Neurosci. 2007, 10, 293–300. [Google Scholar] [CrossRef] [PubMed]
- Sergaki, M.C.; Ibanez, C.F. GFRα1 regulates purkinje cell migration by counteracting NCAM function. Cell Rep. 2017, 18, 367–379. [Google Scholar] [CrossRef]
- Chen, J.; Kawamura, T.; Sethi, M.K.; Zaia, J.; Repunte-Canonigo, V.; Sanna, P.P. Heparan sulfate: Resilience factor and therapeutic target for cocaine abuse. Sci. Rep. 2017, 7, 13931. [Google Scholar] [CrossRef]
- Bespalov, M.M.; Sidorova, Y.A.; Tumova, S.; Ahonen-Bishopp, A.; Magalhaes, A.C.; Kulesskiy, E.; Paveliev, M.; Rivera, C.; Rauvala, H.; Saarma, M. Heparan sulfate proteoglycan syndecan-3 is a novel receptor for GDNF, neurturin, and artemin. J. Cell Biol. 2011, 192, 153–169. [Google Scholar] [CrossRef]
- Sariola, H.; Saarma, M. Novel functions and signalling pathways for GDNF. J. Cell Sci. 2003, 116, 3855–3862. [Google Scholar] [CrossRef]
- Piltonen, M.; Bespalov, M.M.; Ervasti, D.; Matilainen, T.; Sidorova, Y.A.; Rauvala, H.; Saarma, M.; Mannisto, P.T. Heparin-binding determinants of GDNF reduce its tissue distribution but are beneficial for the protection of nigral dopaminergic neurons. Exp. Neurol. 2009, 219, 499–506. [Google Scholar] [CrossRef] [PubMed]
- Walker, D.G.; Beach, T.G.; Xu, R.; Lile, J.; Beck, K.D.; McGeer, E.G.; McGeer, P.L. Expression of the proto-oncogene Ret, a component of the GDNF receptor complex, persists in human substantia nigra neurons in Parkinson’s disease. Brain Res. 1998, 792, 207–217. [Google Scholar] [CrossRef]
- Boscia, F.; Esposito, C.L.; Di Crisci, A.; de Franciscis, V.; Annunziato, L.; Cerchia, L. GDNF selectively induces microglial activation and neuronal survival in CA1/CA3 hippocampal regions exposed to NMDA insult through Ret/ERK signalling. PLoS ONE 2009, 4, e6486. [Google Scholar] [CrossRef] [PubMed]
- Ryu, H.; Jeon, G.S.; Cashman, N.R.; Kowall, N.W.; Lee, J. Differential expression of c-Ret in motor neurons versus non-neuronal cells is linked to the pathogenesis of ALS. Lab. Invest. 2011, 91, 342–352. [Google Scholar] [CrossRef][Green Version]
- Lee, J.; Hyeon, S.J.; Im, H.; Ryu, H.; Kim, Y.; Ryu, H. Astrocytes and microglia as non-cell autonomous players in the pathogenesis of ALS. Exp. Neurobiol. 2016, 25, 233–240. [Google Scholar] [CrossRef]
- Widenfalk, J.; Lundstromer, K.; Jubran, M.; Brene, S.; Olson, L. Neurotrophic factors and receptors in the immature and adult spinal cord after mechanical injury or kainic acid. J. Neurosci. 2001, 21, 3457–3475. [Google Scholar] [CrossRef]
- Defaux, A.; Zurich, M.G.; Honegger, P.; Monnet-Tschudi, F. Inflammatory responses in aggregating rat brain cell cultures subjected to different demyelinating conditions. Brain Res. 2010, 1353, 213–224. [Google Scholar] [CrossRef]
- Boscia, F.; Esposito, C.L.; Casamassa, A.; de Franciscis, V.; Annunziato, L.; Cerchia, L. The isolectin IB4 binds RET receptor tyrosine kinase in microglia. J. Neurochem. 2013, 126, 428–436. [Google Scholar] [CrossRef]
- Moore, M.W.; Klein, R.D.; Farinas, I.; Sauer, H.; Armanini, M.; Phillips, H.; Reichardt, L.F.; Ryan, A.M.; Carver-Moore, K.; Rosenthal, A. Renal and neuronal abnormalities in mice lacking GDNF. Nature 1996, 382, 76–79. [Google Scholar] [CrossRef]
- Sanchez, M.P.; Silos-Santiago, I.; Frisen, J.; He, B.; Lira, S.A.; Barbacid, M. Renal agenesis and the absence of enteric neurons in mice lacking GDNF. Nature 1996, 382, 70–73. [Google Scholar] [CrossRef]
- Pascual, A.; Hidalgo-Figueroa, M.; Piruat, J.I.; Pintado, C.O.; Gomez-Diaz, R.; Lopez-Barneo, J. Absolute requirement of GDNF for adult catecholaminergic neuron survival. Nat. Neurosci. 2008, 11, 755–761. [Google Scholar] [CrossRef] [PubMed]
- Kopra, J.; Vilenius, C.; Grealish, S.; Harma, M.A.; Varendi, K.; Lindholm, J.; Castren, E.; Voikar, V.; Bjorklund, A.; Piepponen, T.P.; et al. GDNF is not required for catecholaminergic neuron survival in vivo. Nat. Neurosci. 2015, 18, 319–322. [Google Scholar] [CrossRef] [PubMed]
- Drinkut, A.; Tereshchenko, Y.; Schulz, J.B.; Bahr, M.; Kugler, S. Efficient gene therapy for Parkinson’s disease using astrocytes as hosts for localized neurotrophic factor delivery. Mol. Ther. 2012, 20, 534–543. [Google Scholar] [CrossRef] [PubMed]
- Brown, G.C.; Neher, J.J. Microglial phagocytosis of live neurons. Nat. Rev. Neurosci. 2014, 15, 209–216. [Google Scholar] [CrossRef]
- Sawada, H.; Suzuki, H.; Nagatsu, T.; Sawada, M. Neuroprotective and neurotoxic phenotypes of activated microglia in neonatal mice with respective MPTP- and ethanol-induced brain injury. Neurodegener. Dis. 2010, 7, 64–67. [Google Scholar] [CrossRef]
- London, A.; Cohen, M.; Schwartz, M. Microglia and monocyte-derived macrophages: Functionally distinct populations that act in concert in CNS plasticity and repair. Front. Cell Neurosci. 2013, 7, 34. [Google Scholar] [CrossRef]
- Mansour, R.M.; Ahmed, M.A.E.; El-Sahar, A.E.; El Sayed, N.S. Montelukast attenuates rotenone-induced microglial activation/p38 MAPK expression in rats: Possible role of its antioxidant, anti-inflammatory and antiapoptotic effects. Toxicol. Appl. Pharmacol. 2018, 358, 76–85. [Google Scholar] [CrossRef]
- Bian, Y.; Zhao, X.; Li, M.; Zeng, S.; Zhao, B. Various roles of astrocytes during recovery from repeated exposure to different doses of lipopolysaccharide. Behav. Brain Res. 2013, 253, 253–261. [Google Scholar] [CrossRef]
- Woodbury, D.; Schaar, D.G.; Ramakrishnan, L.; Black, I.B. Novel structure of the human GDNF gene. Brain Res. 1998, 803, 95–104. [Google Scholar] [CrossRef]
- Tanaka, M.; Ito, S.; Matsushita, N.; Mori, N.; Kiuchi, K. Promoter analysis and characteristics of the 5′-untranslated region of the mouse glial cell line-derived neurotrophic factor gene. Brain Res. Mol. Brain Res. 2000, 85, 91–102. [Google Scholar] [CrossRef]
- Baecker, P.A.; Lee, W.H.; Verity, A.N.; Eglen, R.M.; Johnson, R.M. Characterization of a promoter for the human glial cell line-derived neurotrophic factor gene. Brain Res. Mol. Brain Res. 1999, 69, 209–222. [Google Scholar] [CrossRef]
- Whone, A.L.; Boca, M.; Luz, M.; Woolley, M.; Mooney, L.; Dharia, S.; Broadfoot, J.; Cronin, D.; Schroers, C.; Barua, N.U.; et al. Extended treatment with glial cell line-derived neurotrophic factor in Parkinson’s disease. J. Parkinsons Dis. 2019, 9, 301–313. [Google Scholar] [CrossRef] [PubMed]
- Bartus, R.T.; Herzog, C.D.; Chu, Y.; Wilson, A.; Brown, L.; Siffert, J.; Johnson, E.M., Jr.; Olanow, C.W.; Mufson, E.J.; Kordower, J.H. Bioactivity of AAV2-neurturin gene therapy (CERE-120): Differences between Parkinson’s disease and nonhuman primate brains. Mov. Disord. 2011, 26, 27–36. [Google Scholar] [CrossRef] [PubMed]
- Su, X.; Kells, A.P.; Huang, E.J.; Lee, H.S.; Hadaczek, P.; Beyer, J.; Bringas, J.; Pivirotto, P.; Penticuff, J.; Eberling, J.; et al. Safety evaluation of AAV2-GDNF gene transfer into the dopaminergic nigrostriatal pathway in aged and parkinsonian rhesus monkeys. Hum. Gene Ther. 2009, 20, 1627–1640. [Google Scholar] [CrossRef] [PubMed]
- Tereshchenko, J.; Maddalena, A.; Bahr, M.; Kugler, S. Pharmacologically controlled, discontinuous GDNF gene therapy restores motor function in a rat model of Parkinson’s disease. Neurobiol. Dis. 2014, 65, 35–42. [Google Scholar] [CrossRef]
- Cheng, S.; Tereshchenko, J.; Zimmer, V.; Vachey, G.; Pythoud, C.; Rey, M.; Liefhebber, J.; Raina, A.; Streit, F.; Mazur, A.; et al. Therapeutic efficacy of regulable GDNF expression for Huntington’s and Parkinson’s disease by a high-induction, background-free “GeneSwitch” vector. Exp. Neurol. 2018, 309, 79–90. [Google Scholar] [CrossRef] [PubMed]
- Quintino, L.; Namislo, A.; Davidsson, M.; Breger, L.S.; Kavanagh, P.; Avallone, M.; Elgstrand-Wettergren, E.; Isaksson, C.; Lundberg, C. Destabilizing domains enable long-term and inert regulation of GDNF expression in the brain. Mol. Ther. Methods Clin. Dev. 2018, 11, 29–39. [Google Scholar] [CrossRef]
- Chtarto, A.; Humbert-Claude, M.; Bockstael, O.; Das, A.T.; Boutry, S.; Breger, L.S.; Klaver, B.; Melas, C.; Barroso-Chinea, P.; Gonzalez-Hernandez, T.; et al. A regulatable AAV vector mediating GDNF biological effects at clinically-approved sub-antimicrobial doxycycline doses. Mol. Ther. Methods Clin. Dev. 2016, 5, 16027. [Google Scholar] [CrossRef]
- Quintino, L.; Manfre, G.; Wettergren, E.E.; Namislo, A.; Isaksson, C.; Lundberg, C. Functional neuroprotection and efficient regulation of GDNF using destabilizing domains in a rodent model of Parkinson’s disease. Mol. Ther. 2013, 21, 2169–2180. [Google Scholar] [CrossRef]
- Hoyng, S.A.; Gnavi, S.; de Winter, F.; Eggers, R.; Ozawa, T.; Zaldumbide, A.; Hoeben, R.C.; Malessy, M.J.; Verhaagen, J. Developing a potentially immunologically inert tetracycline-regulatable viral vector for gene therapy in the peripheral nerve. Gene Ther. 2014, 21, 549–557. [Google Scholar] [CrossRef]
- Eggers, R.; de Winter, F.; Arkenaar, C.; Tannemaat, M.R.; Verhaagen, J. Enhanced regeneration and reinnervation following timed GDNF gene therapy in a cervical ventral root avulsion. Exp. Neurol. 2019, 321, 113037. [Google Scholar] [CrossRef] [PubMed]
| Cell Type | Disease Model | Methods Used | References |
|---|---|---|---|
| Macrophages/Microglia | Striatal mechanical injury | ISH + immunohistochemistry | [41,42,44] |
| Experimental autoimmune neuritis | Double immunofluorescence | [45] | |
| LPS-induced inflammation | Double immunofluorescence | [36,43] | |
| Astrocytes | Quinolic acid lesion | Double immunofluorescence | [34,35] |
| LPS-induced inflammation | Double immunofluorescence | [36] | |
| 6-OHDA | ISH + immunofluorescence | [37] | |
| Double immunofluorescence | [38] | ||
| MPTP | Double immunofluorescence | [39] | |
| Spinal cord ischemia | Double immunofluorescence | [40] | |
| Nestin-Expressing Reactive Astrocytes | MPTP | Double immunofluorescence | [46] |
| Microglia-Like NG2-Expressing Cells | 6-OHDA | Double immunofluorescence | [47] |
| Receptor | Cell Type | Disease/Lesion | Methods | Reference |
|---|---|---|---|---|
| GFRα1 | Astrocytes | Rat striatum treated with quinolinic acid | Double immunofluorescence | [35] |
| Rat striatum treated with quinolinic acid or kainic acid | GFRα1 immunoreactivity, morphology | [34] | ||
| Spinal cord mechanical injury | GFRα1 immunoreactivity, localization in white matter, morphology. | [76] | ||
| RET | Microglia | Human PD and aging | Single immunohistochemistry, morphology | [72] |
| RET, pRET | Microglia | ALS transgenic mice | Double immunofluorescence. | [74] |
| pRET | Microglia? | Rat hippocampal slices treated with NMDA and exogenous GDNF | Immunofluorescence combined with isolectin IB4 * | [73] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Duarte Azevedo, M.; Sander, S.; Tenenbaum, L. GDNF, A Neuron-Derived Factor Upregulated in Glial Cells during Disease. J. Clin. Med. 2020, 9, 456. https://doi.org/10.3390/jcm9020456
Duarte Azevedo M, Sander S, Tenenbaum L. GDNF, A Neuron-Derived Factor Upregulated in Glial Cells during Disease. Journal of Clinical Medicine. 2020; 9(2):456. https://doi.org/10.3390/jcm9020456
Chicago/Turabian StyleDuarte Azevedo, Marcelo, Sibilla Sander, and Liliane Tenenbaum. 2020. "GDNF, A Neuron-Derived Factor Upregulated in Glial Cells during Disease" Journal of Clinical Medicine 9, no. 2: 456. https://doi.org/10.3390/jcm9020456
APA StyleDuarte Azevedo, M., Sander, S., & Tenenbaum, L. (2020). GDNF, A Neuron-Derived Factor Upregulated in Glial Cells during Disease. Journal of Clinical Medicine, 9(2), 456. https://doi.org/10.3390/jcm9020456




