Circulating Irisin and esRAGE as Early Biomarkers of Decline of Metabolic Health
Abstract
1. Introduction
2. Experimental Section
2.1. Source Population
2.2. Blood Collection and Biochemical Parameters
2.3. Anthropometric Measures
2.4. Enzyme-Linked Immunosorbent Assay (ELISA)
2.5. Glycated Albumin Quantification
2.6. Statistical Analysis
3. Results
3.1. Demographic, Anthropometric and Clinical Characteristics of the Individuals Included in the Study
3.2. Correlations of Irisin and esRAGE with Clinical Parameters
3.3. Multivariate Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Brenmoehl, J.; Albrecht, E.; Komolka, K.; Schering, L.; Langhammer, M.; Hoeflich, A.; Maak, S. Irisin Is Elevated in Skeletal Muscle and Serum of Mice Immediately after Acute Exercise. Int. J. Boil. Sci. 2014, 10, 338–349. [Google Scholar] [CrossRef] [PubMed]
- Hofmann, T.; Elbelt, U.; Stengel, A. Irisin as a muscle-derived hormone stimulating thermogenesis—A critical update. Peptides 2014, 54, 89–100. [Google Scholar] [CrossRef] [PubMed]
- Liu, J. Irisin as an exercise-stimulated hormone binding crosstalk between organs. Eur. Rev. Med. Pharmacol. Sci. 2015, 19, 316–321. [Google Scholar] [PubMed]
- Xin, C.; Liu, J.; Zhang, J.; Zhu, D.; Wang, H.; Xiong, L.; Lee, Y.; Ye, J.; Lian, K.; Xu, C.; et al. Irisin improves fatty acid oxidation and glucose utilization in type 2 diabetes by regulating the AMPK signaling pathway. Int. J. Obes. (Lond.) 2016, 40, 443–451. [Google Scholar] [CrossRef] [PubMed]
- Benedini, S.; Dozio, E.; Invernizzi, P.L.; Vianello, E.; Banfi, G.; Terruzzi, I.; Luzi, L.; Romanelli, M.M.C. Irisin: A Potential Link between Physical Exercise and Metabolism—An Observational Study in Differently Trained Subjects, from Elite Athletes to Sedentary People. J. Diabetes Res. 2017, 2017, 1–7. [Google Scholar] [CrossRef]
- Ye, L.; Xu, M.; Hu, M.; Zhang, H.; Tan, X.; Li, Q.; Shen, B.; Huang, J. TRPV4 is involved in irisin-induced endothelium-dependent vasodilation. Biochem. Biophys. Res. Commun. 2018, 495, 41–45. [Google Scholar] [CrossRef]
- Zhang, D.; Xie, T.; Leung, P.S. Irisin Ameliorates Glucolipotoxicity-Associated beta-Cell Dysfunction and Apoptosis via AMPK Signaling and Anti-Inflammatory Actions. Cell Physiol. Biochem. 2018, 51, 924–937. [Google Scholar] [CrossRef]
- Deng, X.; Huang, W.; Peng, J.; Zhu, T.T.; Sun, X.L.; Zhou, X.Y.; Yang, H.; Xiong, J.F.; He, H.Q.; Xu, Y.H.; et al. Irisin Alleviates Advanced Glycation End Products-Induced Inflammation and Endothelial Dysfunction via Inhibiting ROS-NLRP3 Inflammasome Signaling. Inflammation 2018, 41, 260–275. [Google Scholar] [CrossRef]
- Ramasamy, R.; Yan, S.F.; Herold, K.; Clynes, R.; Schmidt, A.M. Receptor for advanced glycation end products: Fundamental roles in the inflammatory response: Winding the way to the pathogenesis of endothelial dysfunction and atherosclerosis. Ann. N. Y. Acad. Sci. 2008, 1126, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Yan, S.F.; Ramasamy, R.; Schmidt, A.M. The receptor for advanced glycation endproducts (RAGE) and cardiovascular disease. Expert Rev. Mol. Med. 2009, 11, e9. [Google Scholar] [CrossRef]
- Choi, K.; Yoo, H.; Kim, H.; Lee, K.; Seo, J.; Kim, S.; Kim, N.; Choi, D.; Baik, S. Association between endogenous secretory RAGE, inflammatory markers and arterial stiffness. Int. J. Cardiol. 2009, 132, 96–101. [Google Scholar] [CrossRef] [PubMed]
- Di Pino, A.; Urbano, F.; Zagami, R.M.; Filippello, A.; Di Mauro, S.; Piro, S.; Purrello, F.; Rabuazzo, A.M. Low Endogenous Secretory Receptor for Advanced Glycation End-Products Levels Are Associated With Inflammation and Carotid Atherosclerosis in Prediabetes. J. Clin. Endocrinol. Metab. 2016, 101, 1701–1709. [Google Scholar] [CrossRef] [PubMed]
- Du, R.; Zhang, R.Y.; Lu, L.; Shen, Y.; Pu, L.J.; Zhu, Z.B.; Zhang, Q.; Hu, J.; Yang, Z.K.; Ding, F.H.; et al. Increased glycated albumin and decreased esRAGE levels in serum are related to negative coronary artery remodeling in patients with type 2 diabetes: An Intravascular ultrasound study. Cardiovasc. Diabetol. 2018, 17, 149. [Google Scholar] [CrossRef] [PubMed]
- Raucci, A.; Cugusi, S.; Antonelli, A.; Barabino, S.M.; Monti, L.; Bierhaus, A.; Reiss, K.; Saftig, P.; Bianchi, M.E. A soluble form of the receptor for advanced glycation endproducts (RAGE) is produced by proteolytic cleavage of the membrane-bound form by the sheddase a disintegrin and metalloprotease 10 (ADAM10). FASEB J. 2008, 22, 3716–3727. [Google Scholar] [CrossRef]
- Ueno, H.; Koyama, H.; Shoji, T.; Monden, M.; Fukumoto, S.; Tanaka, S.; Otsuka, Y.; Mima, Y.; Morioka, T.; Mori, K.; et al. Receptor for advanced glycation end-products (RAGE) regulation of adiposity and adiponectin is associated with atherogenesis in apoE-deficient mouse. Atherosclerosis 2011, 211, 431–436. [Google Scholar] [CrossRef]
- Zhao, D.; Wang, Y.; Xu, Y. Decreased serum endogenous secretory receptor for advanced glycation endproducts and increased cleaved receptor for advanced glycation endproducts levels in patients with atrial fibrillation. Int. J. Cardiol. 2012, 158, 471–472. [Google Scholar] [CrossRef]
- Monden, M.; Koyama, H.; Otsuka, Y.; Morioka, T.; Mori, K.; Shoji, T.; Mima, Y.; Motoyama, K.; Fukumoto, S.; Shioi, A.; et al. Receptor for advanced glycation end products regulates adipocyte hypertrophy and insulin sensitivity in mice: Involvement of Toll-like receptor 2. Diabetes 2013, 62, 478–489. [Google Scholar] [CrossRef]
- Miura, J.; Yamamoto, Y.; Osawa, M.; Watanabe, T.; Yonekura, H.; Uchigata, Y.; Yamamoto, H.; Iwamoto, Y. Endogenous Secretory Receptor for Advanced Glycation Endproducts Levels Are Correlated With Serum Pentosidine and CML in Patients With Type 1 Diabetes. Arter. Thromb. Vasc. Boil. 2007, 27, 253–254. [Google Scholar] [CrossRef][Green Version]
- Choi, Y.-K.; Kim, M.-K.; Bae, K.H.; Seo, H.-A.; Jeong, J.-Y.; Lee, W.-K.; Kim, J.-G.; Lee, I.-K.; Park, K.-G. Serum irisin levels in new-onset type 2 diabetes. Diabetes Res. Clin. Pr. 2013, 100, 96–101. [Google Scholar] [CrossRef]
- Liu, J.-J.; Wong, M.D.; Toy, W.C.; Tan, C.S.; Liu, S.; Ng, X.W.; Tavintharan, S.; Sum, C.F.; Lim, S.C. Lower circulating irisin is associated with type 2 diabetes mellitus. J. Diabetes Complicat. 2013, 27, 365–369. [Google Scholar] [CrossRef]
- Rana, K.S.; Pararasa, C.; Afzal, I.; Nagel, D.A.; Hill, E.J.; Bailey, C.J.; Griffiths, H.R.; Kyrou, I.; Randeva, H.S.; Bellary, S.; et al. Plasma irisin is elevated in type 2 diabetes and is associated with increased E-selectin levels. Cardiovasc. Diabetol. 2017, 16, 147. [Google Scholar] [CrossRef] [PubMed]
- Elizondo-Montemayor, L.; Mendoza-Lara, G.; Gutierrez-DelBosque, G.; Peschard-Franco, M.; Nieblas, B.; Garcia-Rivas, G. Relationship of Circulating Irisin with Body Composition, Physical Activity, and Cardiovascular and Metabolic Disorders in the Pediatric Population. Int. J. Mol. Sci. 2018, 19, 3727. [Google Scholar] [CrossRef] [PubMed]
- Saber, G.Y.; Kasabri, V.; Saleh, M.I.; Suyagh, M.; Halaseh, L.; Jaber, R.; Abu-Hassan, H.; Alalawi, S. Increased irisin versus reduced fibroblast growth factor1 (FGF1) in relation to adiposity, atherogenicity and hematological indices in metabolic syndrome patients with and without prediabetes. Horm. Mol. Boil. Clin. Investig. 2019, 38. [Google Scholar] [CrossRef] [PubMed]
- Malavazos, A.E.; Corsi, M.M.; Ermetici, F.; Coman, C.; Sardanelli, F.; Rossi, A.; Morricone, L.; Ambrosi, B. Proinflammatory cytokines and cardiac abnormalities in uncomplicated obesity: Relationship with abdominal fat deposition. Nutr. Metab. Cardiovasc. Dis. 2007, 17, 294–302. [Google Scholar] [CrossRef] [PubMed]
- Dozio, E.; Dogliotti, G.; Malavazos, A.; Bandera, F.; Cassetti, G.; Vianello, E.; Zelaschi, R.; Barassi, A.; Pellissero, G.; Solimene, U.; et al. IL-18 level in patients undergoing coronary artery bypass grafting surgery or valve replacement: Which link with epicardial fat depot? Int. J. Immunopathol. Pharmacol. 2012, 25, 1011–1020. [Google Scholar] [CrossRef]
- Matthews, D.R.; Hosker, J.P.; Rudenski, A.S.; A Naylor, B.; Treacher, D.F.; Turner, R.C. Homeostasis model assessment: Insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985, 28, 412–419. [Google Scholar] [CrossRef]
- Kahn, H.S. The “lipid accumulation product” performs better than the body mass index for recognizing cardiovascular risk: A population-based comparison. BMC Cardiovasc. Disord. 2005, 5, 26. [Google Scholar] [CrossRef]
- Browning, L.M.; Hsieh, S.D.; Ashwell, M. A systematic review of waist-to-height ratio as a screening tool for the prediction of cardiovascular disease and diabetes: 0·5 could be a suitable global boundary value. Nutr. Res. Rev. 2010, 23, 247–269. [Google Scholar] [CrossRef]
- Alberti, K.; Zimmet, P.; Shaw, J. Metabolic Syndrome--A New World-Wide Definition. A Consensus Statement from the International Diabetes Federation. Diabet. Met. 2006, 23, 469–480. [Google Scholar] [CrossRef]
- Yamamoto, Y.; Miura, J.; Sakurai, S.; Watanabe, T.; Yonekura, H.; Tamei, H.; Matsuki, H.; Obata, K.-I.; Uchigata, Y.; Iwamoto, Y.; et al. Assaying soluble forms of receptor for advanced glycation end products. Arter. Thromb. Vasc. Biol. 2007, 27, 33–34. [Google Scholar] [CrossRef]
- Tang, S.C.; Yeh, S.J.; Tsai, L.K.; Hu, C.J.; Lien, L.M.; Peng, G.S.; Yang, W.S.; Chiou, H.Y.; Jeng, J.S. Cleaved but not endogenous secretory RAGE is associated with outcome in acute ischemic stroke. Neurology 2016, 86, 270–276. [Google Scholar] [CrossRef]
- Miranda, E.R.; Fuller, K.N.; Perkins, R.K.; Kroeger, C.M.; Trepanowski, J.F.; Varady, K.A.; Haus, J.M. Endogenous secretory RAGE increases with improvements in body composition and is associated with markers of adipocyte health. Nutr. Metab. Cardiovasc. Dis. 2018, 28, 1155–1165. [Google Scholar] [CrossRef]
- Kouzuma, T.; Usami, T.; Yamakoshi, M.; Takahashi, M.; Imamura, S. An enzymatic method for the measurement of glycated albumin in biological samples. Clin. Chim. Acta 2002, 324, 61–71. [Google Scholar] [CrossRef]
- Kouzuma, T.; Uemastu, Y.; Usami, T.; Imamura, S. Study of glycated amino acid elimination reaction for an improved enzymatic glycated albumin measurement method. Clin. Chim. Acta 2004, 346, 135–143. [Google Scholar] [CrossRef]
- Kohzuma, T.; Yamamoto, T.; Uematsu, Y.; Shihabi, Z.K.; Freedman, B.I. Basic performance of an enzymatic method for glycated albumin and reference range determination. J. Diabetes Sci. Technol. 2011, 5, 1455–1462. [Google Scholar] [CrossRef]
- Jang, H.B.; Kim, H.-J.; Kang, J.H.; Park, S.I.; Park, K.H.; Lee, H.-J. Association of circulating irisin levels with metabolic and metabolite profiles of Korean adolescents. Metabolism 2017, 73, 100–108. [Google Scholar] [CrossRef]
- Nigro, E.; Scudiero, O.; Monaco, M.L.; Polito, R.; Schettino, P.; Grandone, A.; Perrone, L.; Del Giudice, E.M.; Daniele, A. Adiponectin profile and Irisin expression in Italian obese children: Association with insulin-resistance. Cytokine 2017, 94, 8–13. [Google Scholar] [CrossRef]
- Park, K.H.; Zaichenko, L.; Brinkoetter, M.; Thakkar, B.; Sahin-Efe, A.; Joung, K.E.; Tsoukas, M.A.; Geladari, E.V.; Huh, J.Y.; Dincer, F.; et al. Circulating irisin in relation to insulin resistance and the metabolic syndrome. J. Clin. Endocrinol. Metab. 2013, 98, 4899–4907. [Google Scholar] [CrossRef]
- Chaudhuri, J.; Bains, Y.; Guha, S.; Kahn, A.; Hall, D.; Bose, N.; Gugliucci, A.; Kapahi, P. The Role of Advanced Glycation End Products in Aging and Metabolic Diseases: Bridging Association and Causality. Cell Metab. 2018, 28, 337–352. [Google Scholar] [CrossRef]
- Arsov, S.; Graaff, R.; Van Oeveren, W.; Stegmayr, B.; Sikole, A.; Rakhorst, G.; Smit, A.J. Advanced glycation end-products and skin autofluorescence in end-stage renal disease: A review. Clin. Chem. Lab. Med. 2014, 52, 11–20. [Google Scholar] [CrossRef]
- Karumanchi, D.K.; Karunaratne, N.; Lurio, L.; Dillon, J.P.; Gaillard, E.R. Non-enzymatic glycation of alpha-crystallin as an in vitro model for aging, diabetes and degenerative diseases. Amino Acids 2015, 47, 2601–2608. [Google Scholar] [CrossRef]
- Limongi, D.; Baldelli, S. Redox Imbalance and Viral Infections in Neurodegenerative Diseases. Oxidative Med. Cell. Longev. 2016, 2016, 1–13. [Google Scholar] [CrossRef]
- Basta, G. Receptor for advanced glycation endproducts and atherosclerosis: From basic mechanisms to clinical implications. Atherosclerosis. 2008, 196, 9–21. [Google Scholar] [CrossRef]
- Dozio, E.; Vianello, E.; Briganti, S.; Lamont, J.; Tacchini, L.; Schmitz, G.; Corsi Romanelli, M.M. Expression of the Receptor for Advanced Glycation End Products in Epicardial Fat: Link with Tissue Thickness and Local Insulin Resistance in Coronary Artery Disease. J. Diabetes Res. 2016, 2016, 1–8. [Google Scholar] [CrossRef]
- Dozio, E.; Vianello, E.; Bandera, F.; Longhi, E.; Brizzola, S.; Nebuloni, M.; Romanelli, M.M.C. Soluble Receptor for Advanced Glycation End Products: A Protective Molecule against Intramyocardial Lipid Accumulation in Obese Zucker Rats? Mediat. Inflamm. 2019, 2019, 1–8. [Google Scholar] [CrossRef]
- Vazzana, N.; Santilli, F.; Cuccurullo, C.; Davì, G. Soluble forms of RAGE in internal medicine. Intern. Emerg. Med. 2009, 4, 389–401. [Google Scholar] [CrossRef]
- Li, Z.; Wang, G.; Zhu, Y.-J.; Li, C.-G.; Tang, Y.-Z.; Jiang, Z.-H.; Yang, M.; Ni, C.-L.; Chen, L.-M.; Niu, W.-Y. The relationship between circulating irisin levels and tissues AGE accumulation in type 2 diabetes patients. Biosci. Rep. 2017, 37. [Google Scholar] [CrossRef]
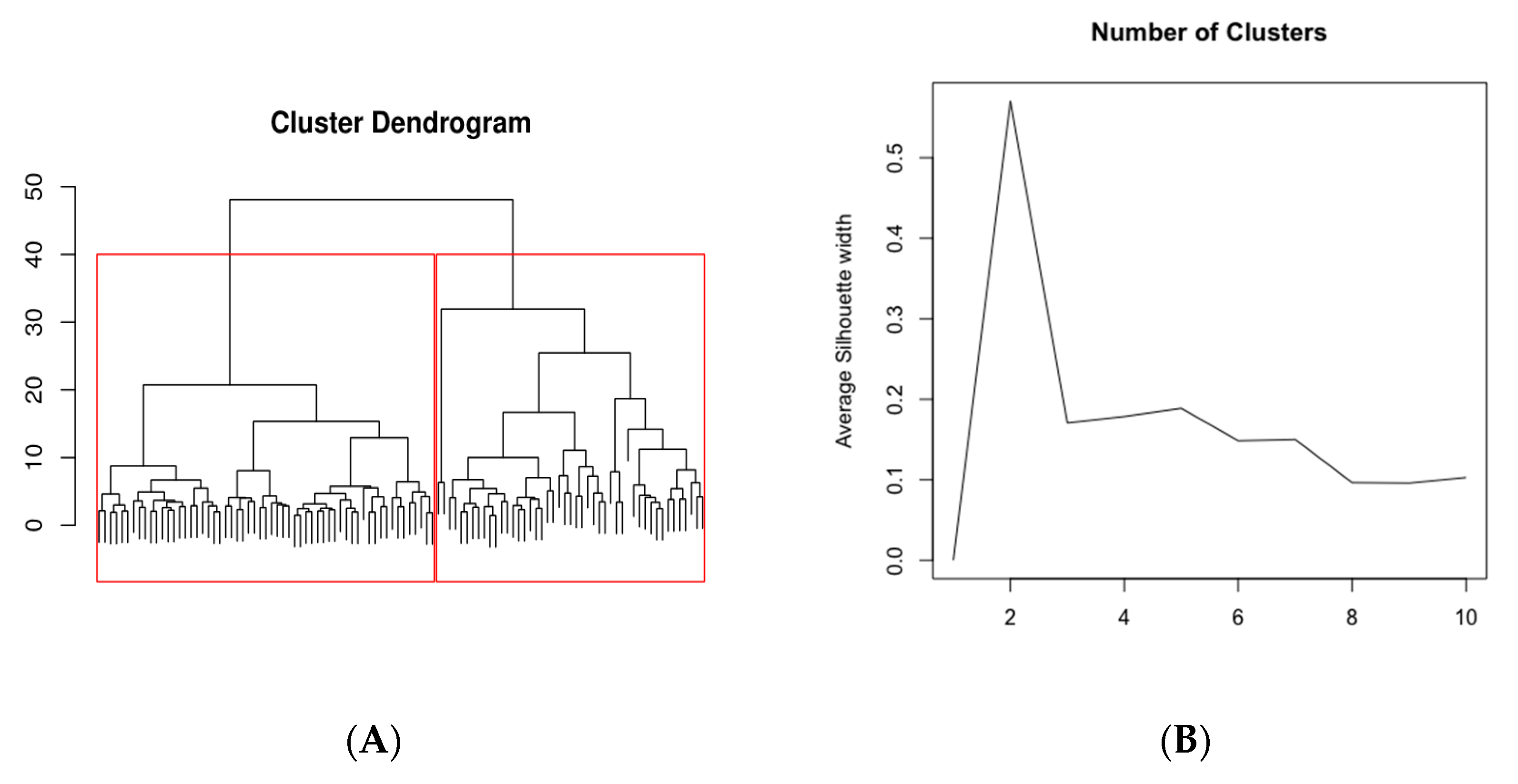
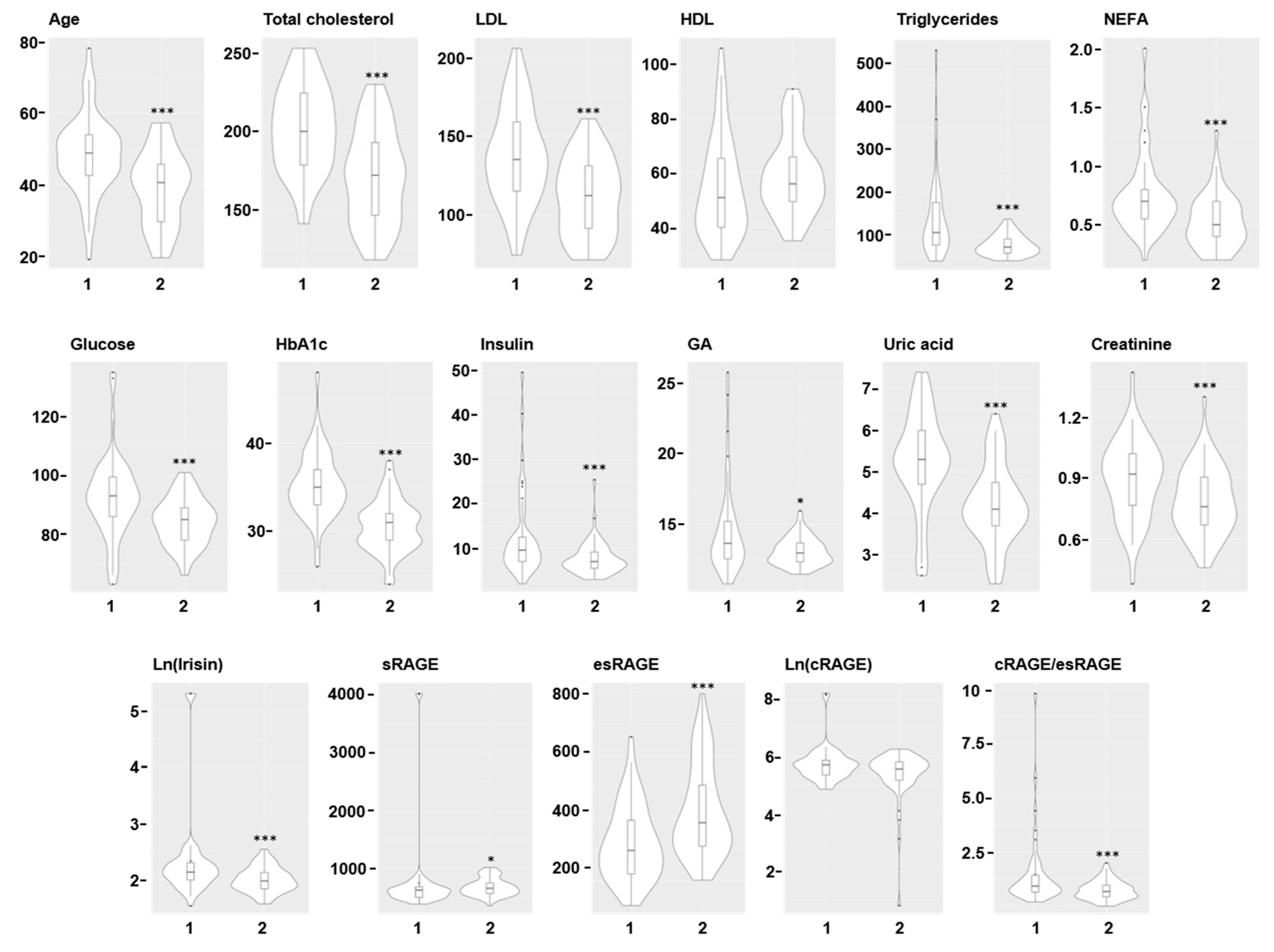
| Variable | n = 106 |
|---|---|
| Age (years) | 43.00 ± 11.72, 43.30 (35.73–51.06) |
| Male gender (n, %) | 58, 54.72% |
| BMI | 25.33 ± 4.72, 24.68 (21.66–27.54) |
| WC (cm) | 87.76 ± 15.20, 85.00 (75.00–99.50) |
| WHtR | 0.52 ± 0.08, 0.51 (0.46–0.57) |
| Fasting glucose (mg/dL) | 88.25 ± 12.46, 87.50 (80.75–94.00) |
| Fasting insulin (microU/mL) | 9.71 ± 7.16, 7.52 (5.80–10.96) |
| HbA1c (mmoL/moL) | 32.61 ± 4.04, 32.00 (29.00–35.00) |
| HOMA-IR | 2.24 ± 2.12, 1.62 (1.14–2.44) |
| LAP index | 35.02 ± 41.22, 19.62 (11.65–35.51) |
| Total Cholesterol (mg/dL) | 185.00 ± 33.04, 181.00 (162.00–212.00) |
| LDL-Cholesterol (mg/dL) | 122.80 ± 30.11, 121.00 (99.00–142.00) |
| HDL-Cholesterol (mg/dL) | 57.01 ± 16.23, 53.50 (43.00–67.00) |
| Triglycerides (mg/dL) | 102.30 ± 70.48, 85.50 (60.00–115.30) |
| Creatinine (mg/dL) | 0.83 ± 1.19, 0.84 (0.69–0.96) |
| GFR (ml/min/1.73 m2) | 84.09 ± 8.70, 89.00 (80.75–90.00) |
| Uric acid (mg/dL) | 4.68 ± 1.18, 4.70 (3.80–5.43) |
| NEFA (mg/dL) | 0.62 ± 0.30, 0.60 (0.40–0.70) |
| Obesity (n, %) | 13, 12.26% |
| Central obesity (n, %) | 53, 50.00% |
| Smoking (n, %) | 24, 22.64% |
| Irisin (ng/mL) | 13.44 ± 31.99, 7.89 (6.54–7.63) |
| sRAGE (pg/mL) | 720.00 ± 478.60, 642.20 (554.00–746.70) |
| esRAGE (pg/mL) | 377.00 ± 143.20, 341.10 (271.70–462.80) |
| cRAGE (pg/mL) | 343.10 ± 458.10, 286.70 (202.60–343.30) |
| cRAGE/esRAGE | 1.06 ± 1.20,0.82 (0.49–1.22) |
| GA (%) | 13.68 ± 2.34, 13.13 (12.38–14.09) |
| IRISIN | esRAGE | |||
|---|---|---|---|---|
| r | p-Value | r | p-Value | |
| Age | 0.387 | <0.0001 | −0.146 | 0.1358 |
| BMI | 0.141 | 0.149 | −0.421 | <0.0001 |
| Waist | 0.131 | 0.184 | −0.368 | <0.0001 |
| WHtR | 0.136 | 0.169 | −0.435 | <0.0001 |
| WHR | ||||
| Fasting glucose | 0.200 | 0.050 | −0.165 | 0.092 |
| HbA1c | 0.115 | 0.239 | −0.218 | 0.025 |
| Fasting insulin | 0.018 | 0.855 | −0.282 | 0.003 |
| Total cholesterol | 0.305 | 0.002 | −0.100 | 0.310 |
| HDL cholesterol | −0.169 | 0.083 | 0.207 | 0.033 |
| LDL cholesterol | 0.264 | 0.006 | −0.093 | 0.343 |
| Triglycerides | 0.266 | 0.006 | −0.210 | 0.030 |
| Uric Acid | 0.153 | 0.117 | −0.253 | 0.009 |
| Creatinine | 0.037 | 0.708 | −0.038 | 0.699 |
| GFR | −0.003 | 0.978 | −0.059 | 0.551 |
| NEFA | 0.075 | 0.442 | −0.177 | 0.069 |
| HOMA-IR | 0.060 | 0.540 | −0.261 | 0.007 |
| LAP index | 0.200 | 0.045 | −0.359 | <0.001 |
| sRAGE | −0.263 | 0.006 | 0.614 | <0.0001 |
| esRAGE | −0.200 | 0.050 | - | - |
| cRAGE | −0.119 | 0.224 | −0.329 | <0.001 |
| cRAGE/esRAGE | 0.005 | 0.961 | −0.743 | <0.001 |
| GA | 0.276 | 0.004 | 0.123 | 0.210 |
| GA/sRAGE | 0.440 | <0.0001 | −0.496 | <0.0001 |
| GA/esRAGE | 0.327 | <0.0001 | −0.909 | <0.0001 |
| GA/cRAGE | 0.269 | 0.006 | 0.344 | <0.001 |
| irisin | - | - | -0.200 | 0.050 |
| Variable | Cluster 1 | Cluster 2 | OR (95% CI) |
|---|---|---|---|
| Females | 15/47 (0.32) | 33/59 (0.56) | 2.71 (1.22–6.03) |
| Alcohol consumption | |||
| Moderate | 12/47 (0.26) | 19/59 (0.32) | 1.11 (0.44–2.87) |
| Yes | 15/47 (0.32) | 13/59 (0.22) | 0.61 (0.23–1.57) |
| Smoking | 10/47 (0.21) | 14/59 (0.24) | 1.15 (0.46–2.89) |
| WC ≥ 94 cm (male), ≥ 80 cm (female) | 33/45 (0.73) | 19/59 (0.32) | 0.17 (0.07–0.41) |
| WHtR ≥ 0.5 | 36/45 (0.80) | 22/59 (0.37) | 0.15 (0.06–0.37) |
| HOMA-IR ≥ 2.5 | 17/47 (0.36) | 7/59 (0.12) | 0.24 (0.09–0.64) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dozio, E.; Vianello, E.; Sitzia, C.; Ambrogi, F.; Benedini, S.; Gorini, S.; Rampoldi, B.; Rigolini, R.; Tacchini, L.; Corsi Romanelli, M.M. Circulating Irisin and esRAGE as Early Biomarkers of Decline of Metabolic Health. J. Clin. Med. 2020, 9, 454. https://doi.org/10.3390/jcm9020454
Dozio E, Vianello E, Sitzia C, Ambrogi F, Benedini S, Gorini S, Rampoldi B, Rigolini R, Tacchini L, Corsi Romanelli MM. Circulating Irisin and esRAGE as Early Biomarkers of Decline of Metabolic Health. Journal of Clinical Medicine. 2020; 9(2):454. https://doi.org/10.3390/jcm9020454
Chicago/Turabian StyleDozio, Elena, Elena Vianello, Clementina Sitzia, Federico Ambrogi, Stefano Benedini, Silvia Gorini, Benedetta Rampoldi, Roberta Rigolini, Lorenza Tacchini, and Massimiliano Marco Corsi Romanelli. 2020. "Circulating Irisin and esRAGE as Early Biomarkers of Decline of Metabolic Health" Journal of Clinical Medicine 9, no. 2: 454. https://doi.org/10.3390/jcm9020454
APA StyleDozio, E., Vianello, E., Sitzia, C., Ambrogi, F., Benedini, S., Gorini, S., Rampoldi, B., Rigolini, R., Tacchini, L., & Corsi Romanelli, M. M. (2020). Circulating Irisin and esRAGE as Early Biomarkers of Decline of Metabolic Health. Journal of Clinical Medicine, 9(2), 454. https://doi.org/10.3390/jcm9020454








