Markov State Modelling of Disease Courses and Mortality Risks of Patients with Community-Acquired Pneumonia
Abstract
1. Introduction
2. Materials and Method
2.1. Patient Data
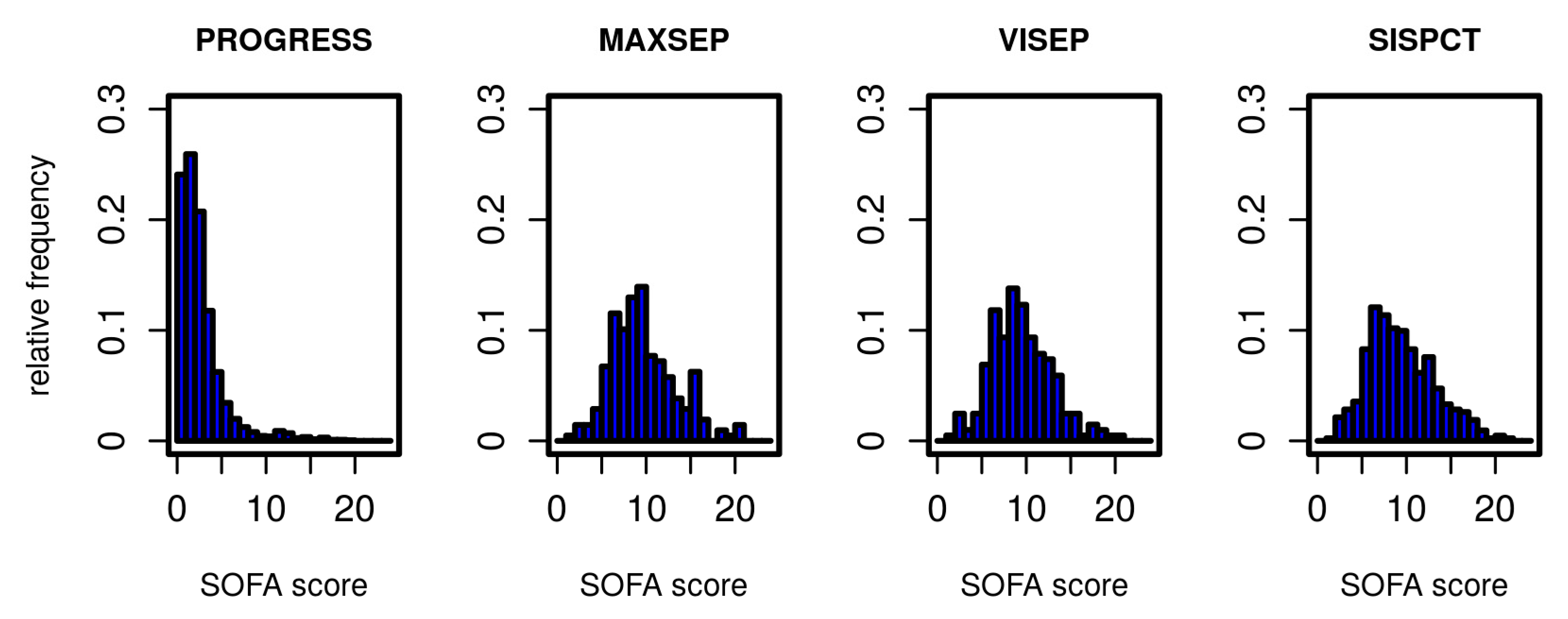
2.2. Defining States of CAP Severity
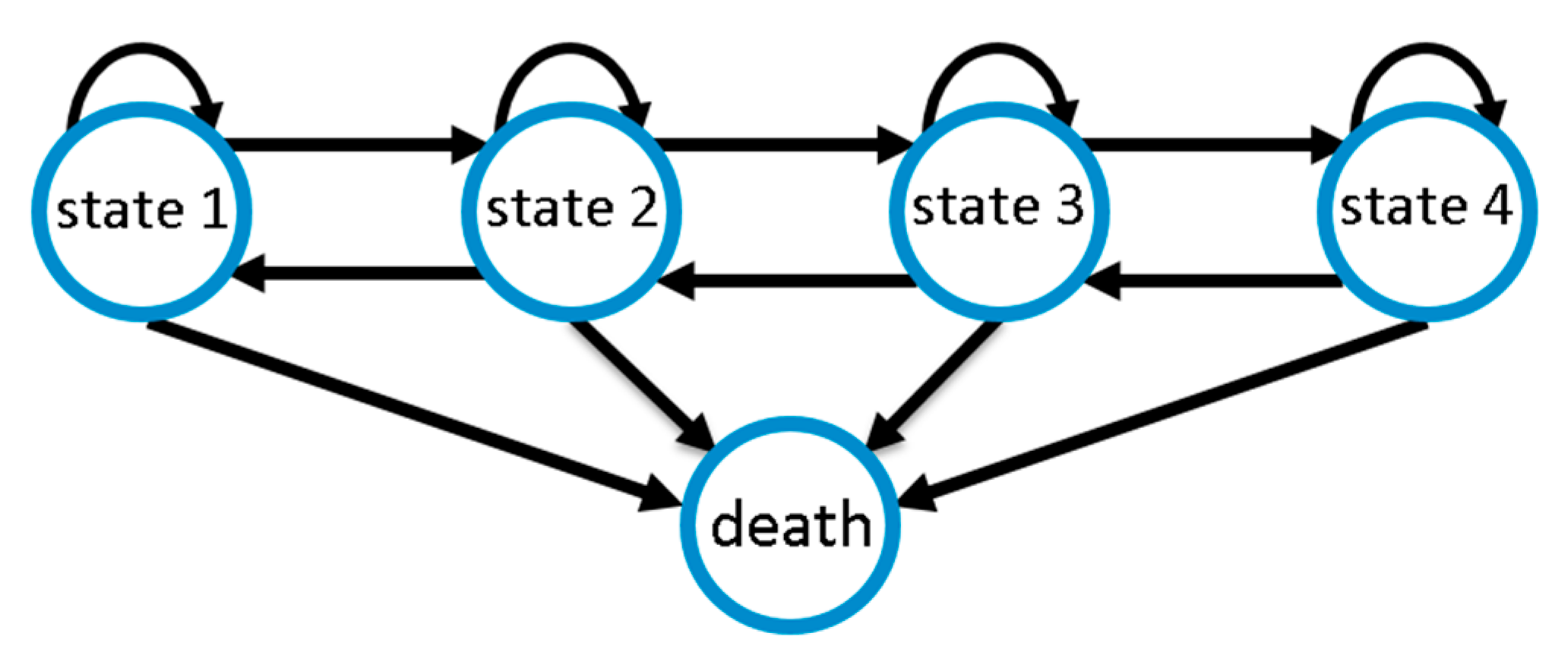
2.3. Establishing the Markov Model
2.4. Comparisons with Other Risk Scores
3. Results
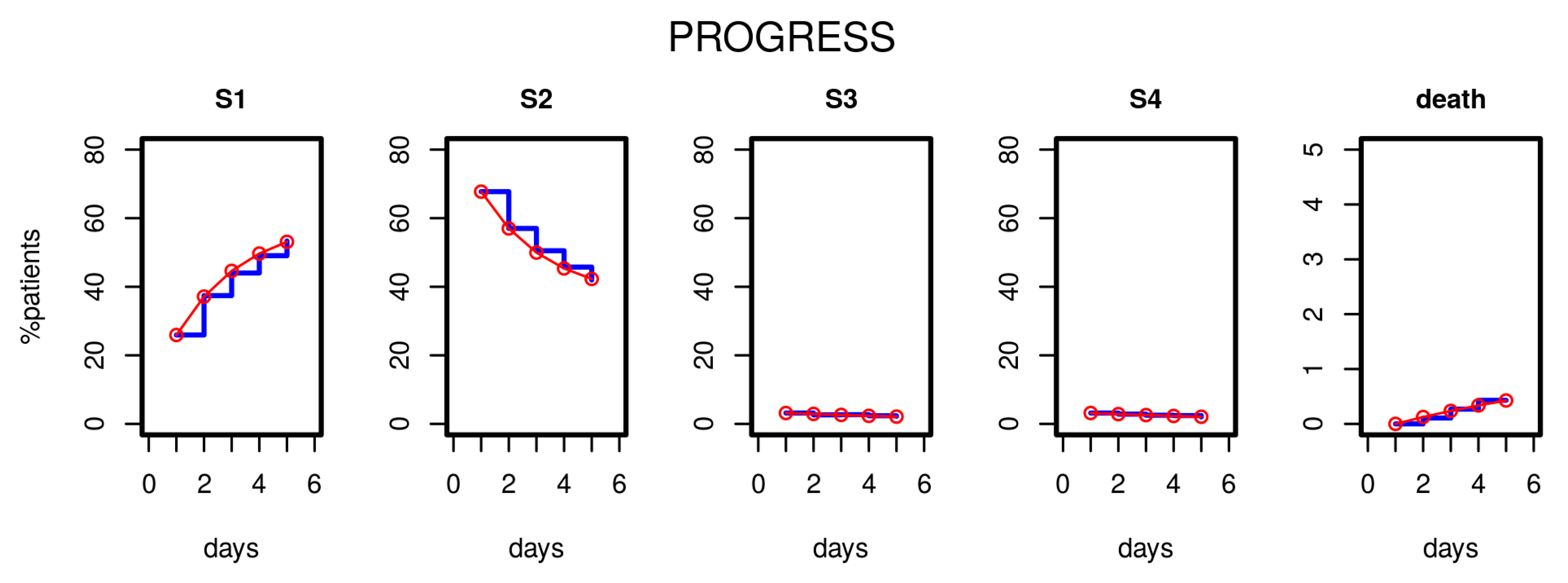
3.1. Comparison of Model and Data
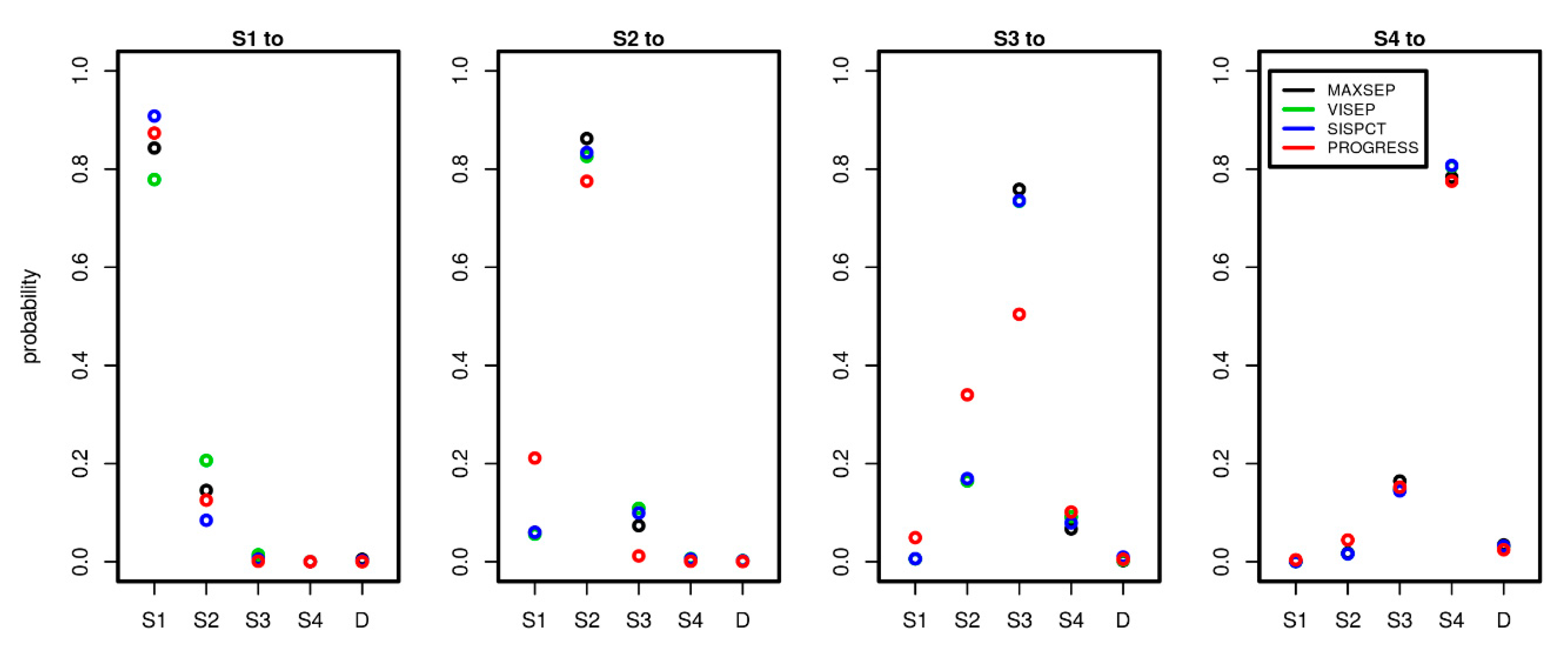
3.2. Transition Probability Matrices
3.3. Predicting 28 d Mortality
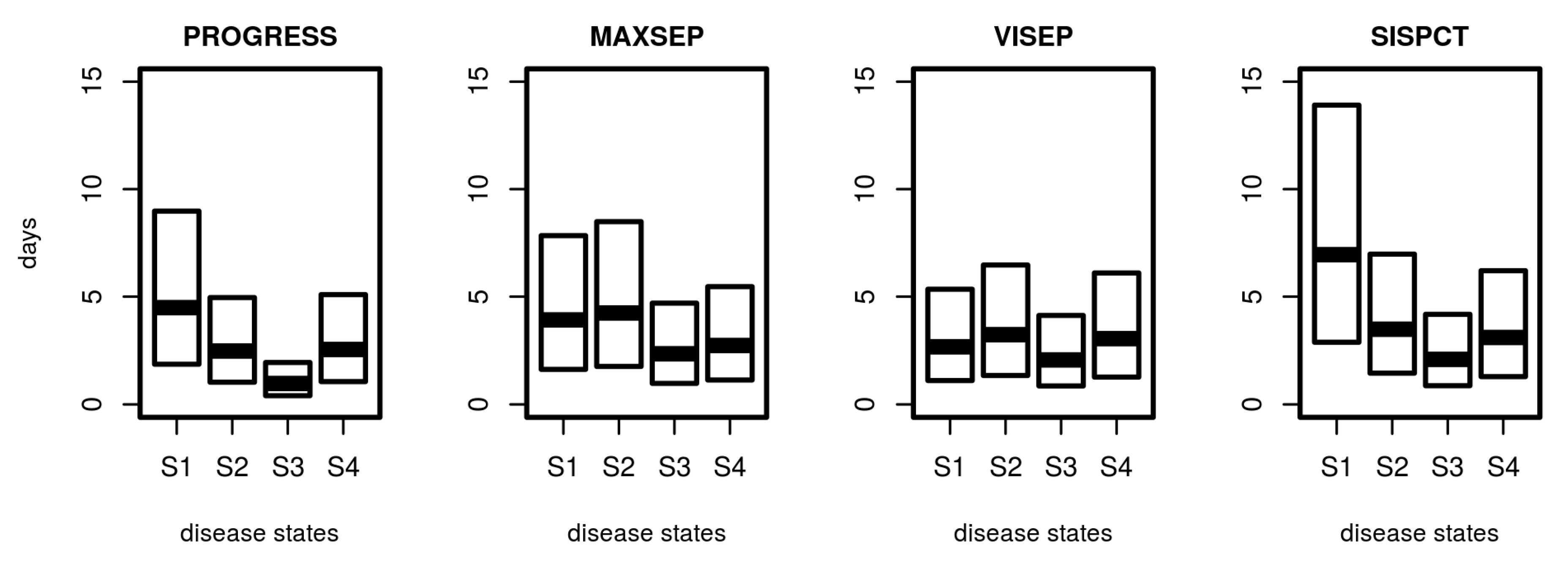
3.4. Distribution of Sojourn Times
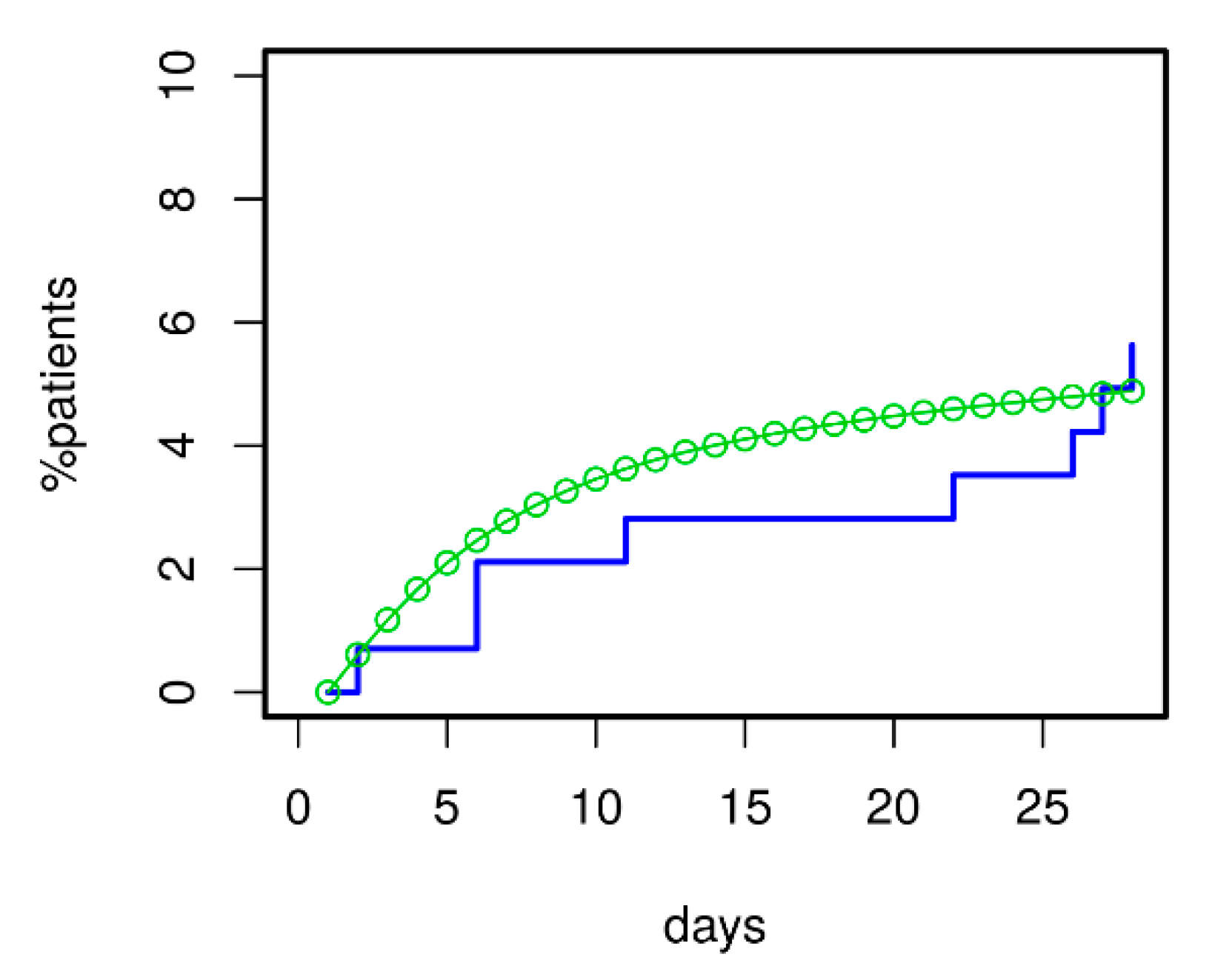
3.5. Prediction of Death for Patients with Initial Severe CAP
3.6. Clinical Utility of the Model
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
A.1. Details of Markov Modelling
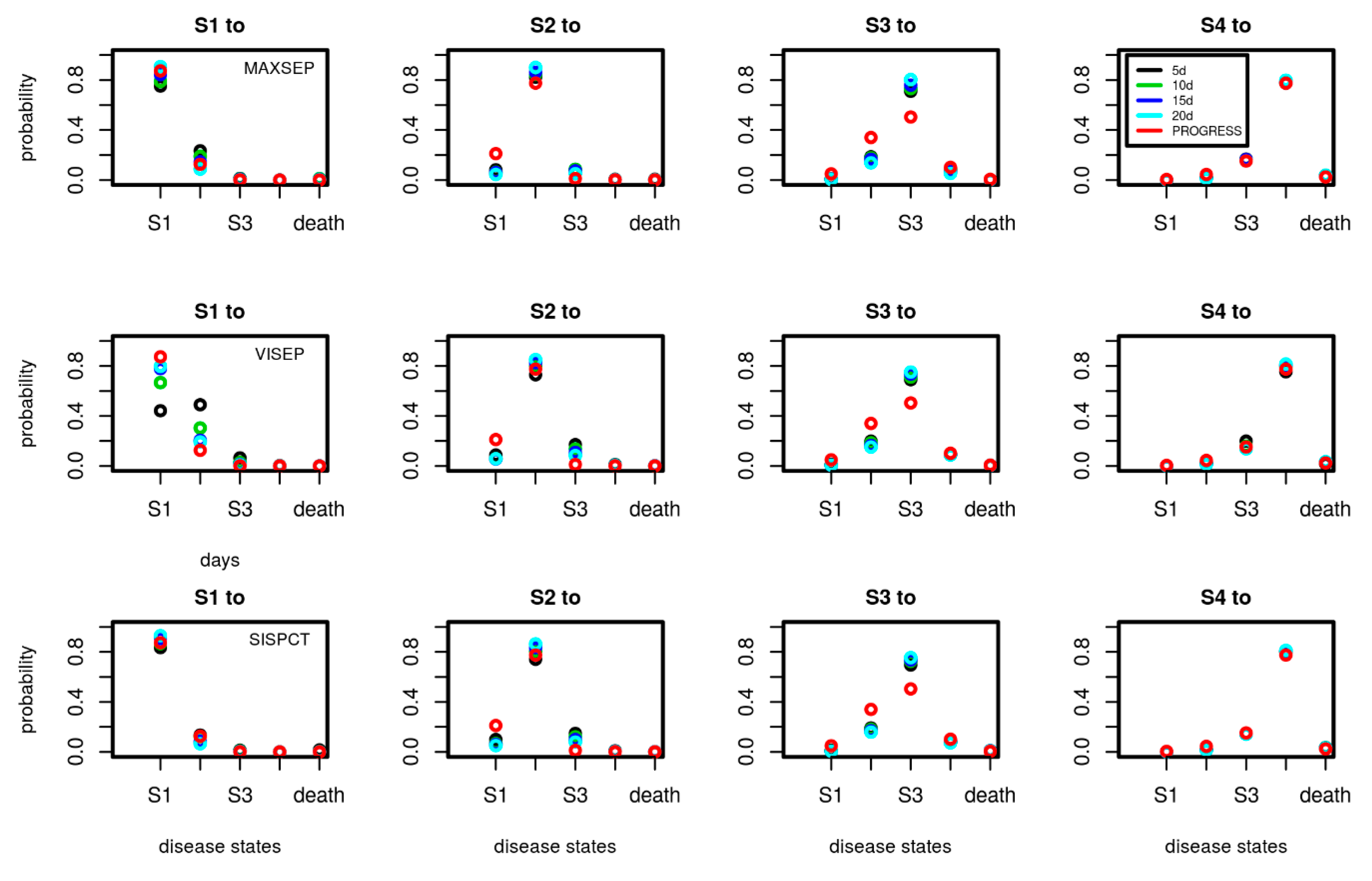
A.2. Transition Probabilities of SepNet Studies in Dependence on Number of Observation Days Used for Model-Calibration
References
- IQTIG—Institut für Qualitätssicherung und Transparenz im Gesundheitswesen. Qualitätsreport 2019. 2019. Available online: https://iqtig.org/downloads/berichte/2018/IQTIG_Qualitaetsreport-2019_2019-09-25.pdf (accessed on 7 December 2019).
- Dremsizov, T.; Clermont, G.; Kellum, J.A.; Kalassian, K.G.; Fine, M.J.; Angus, D.C. Severe sepsis in community-acquired pneumonia: When does it happen, and do systemic inflammatory response syndrome criteria help predict course? Chest 2006, 129, 968–978. [Google Scholar] [CrossRef]
- Ewig, S.; Birkner, N.; Strauss, R.; Schaefer, E.; Pauletzki, J.; Bischoff, H.; Schraeder, P.; Welte, T.; Hoeffken, G. New perspectives on community-acquired pneumonia in 388 406 patients. Results from a nationwide mandatory performance measurement programme in healthcare quality. Thorax 2009, 64, 1062–1069. [Google Scholar] [CrossRef]
- Kolditz, M.; Ewig, S.; Klapdor, B.; Schütte, H.; Winning, J.; Rupp, J.; Suttorp, N.; Welte, T.; Rohde, G. Community-acquired pneumonia as medical emergency: Predictors of early deterioration. Thorax 2015, 70, 551–558. [Google Scholar] [CrossRef]
- Ewig, S.; Welte, T. CRB-65 for the assessment of pneumonia severity: Who could ask for more? Thorax 2008, 63, 665–666. [Google Scholar] [CrossRef]
- Lim, W.S. Defining community acquired pneumonia severity on presentation to hospital: An international derivation and validation study. Thorax 2003, 58, 377–382. [Google Scholar] [CrossRef]
- Fine, M.J.; Auble, T.E.; Yealy, D.M.; Hanusa, B.H.; Weissfeld, L.A.; Singer, D.E.; Coley, C.M.; Marrie, T.J.; Kapoor, W.N. A prediction rule to identify low-risk patients with community-acquired pneumonia. N. Engl. J. Med. 1997, 336, 243–250. [Google Scholar] [CrossRef]
- Espana, P.P.; Capelastegui, A.; Gorordo, I.; Esteban, C.; Oribe, M.; Ortega, M.; Bilbao, A.; Quintana, J.M. Development and validation of a clinical prediction rule for severe community-acquired pneumonia. Am. J. Respir. Crit. Care Med. 2006, 174, 1249–1256. [Google Scholar] [CrossRef]
- Charles, P.G.P.; Wolfe, R.; Whitby, M.; Fine, M.J.; Fuller, A.J.; Stirling, R.; Wright, A.A.; Ramirez, J.A.; Christiansen, K.J.; Waterer, G.W.; et al. SMART-COP: A tool for predicting the need for intensive respiratory or vasopressor support in community-acquired pneumonia. Clin. Infect. Dis. 2008, 47, 375–384. [Google Scholar] [CrossRef]
- Mandell, L.A.; Wunderink, R.G.; Anzueto, A.; Bartlett, J.G.; Campbell, G.D.; Dean, N.C.; Dowell, S.F.; File, T.M., Jr.; Musher, D.M.; Niederman, M.S.; et al. Infectious Diseases Society of America/American Thoracic Society consensus guidelines on the management of community-acquired pneumonia in adults. Clin. Infect. Dis. 2007, 44, S27–S72. [Google Scholar] [CrossRef]
- Halm, E.A.; Fine, M.J.; Marrie, T.J.; Coley, C.M.; Kapoor, W.N.; Obrosky, D.S.; Singer, D.E. Time to clinical stability in patients hospitalized with community-acquired pneumonia: Implications for practice guidelines. JAMA 1998, 279, 1452–1457. [Google Scholar] [CrossRef]
- Akram, A.R.; Chalmers, J.D.; Taylor, J.K.; Rutherford, J.; Singanayagam, A.; Hill, A.T. An evaluation of clinical stability criteria to predict hospital course in community-acquired pneumonia. Clin. Microbiol. Infect. 2013, 19, 1174–1180. [Google Scholar] [CrossRef]
- Ewig, S.; Höffken, G.; Kern, W.V.; Rohde, G.; Flick, H.; Krause, R.; Ott, S.; Bauer, T.; Dalhoff, K.; Gatermann, S.; et al. Behandlung von erwachsenen Patienten mit ambulant erworbener Pneumonie und Prävention—Update 2016. Pneumologie 2016, 70, 151–200. [Google Scholar] [CrossRef]
- Aliberti, S.; Amir, A.; Peyrani, P.; Mirsaeidi, M.; Allen, M.; Moffett, B.K.; Myers, J.; Shaib, F.; Cirino, M.; Bordon, J.; et al. Incidence, etiology, timing, and risk factors for clinical failure in hospitalized patients with community-acquired pneumonia. Chest 2008, 134, 955–962. [Google Scholar] [CrossRef]
- Phua, J.; Ngerng, W.J.; Lim, T.K. The impact of a delay in intensive care unit admission for community-acquired pneumonia. Eur. Respir. J. 2010, 36, 826–833. [Google Scholar] [CrossRef]
- Peyrani, P.; Arnold, F.W.; Bordon, J.; Furmanek, S.; Luna, C.M.; Cavallazzi, R.; Ramirez, J. Incidence and Mortality of Adults Hospitalized With Community-Acquired Pneumonia According to Clinical Course. Chest 2019. [Google Scholar] [CrossRef]
- Chiang, C.L. Making annual indexes of health. Health Serv. Res. 1976, 11, 442–451. [Google Scholar]
- Kay, R. A Markov model for analysing cancer markers and disease states in survival studies. Biometrics 1986, 42, 855–865. [Google Scholar] [CrossRef]
- Klotz, J.H.; Sharpless, L.D. Estimation for a Markov Heart Transplant Model. J. R. Stat. Soc. Ser. D (Stat.) 1994, 43, 431–438. [Google Scholar] [CrossRef]
- Kumar, S.; Ghildayal, N.; Ghildayal, N. Markov chain decision model for urinary incontinence procedures. Int. J. Health Care Qual. Assur. 2017, 30, 187–202. [Google Scholar] [CrossRef]
- Ieva, F.; Jackson, C.H.; Sharples, L.D. Multi-state modelling of repeated hospitalisation and death in patients with heart failure: The use of large administrative databases in clinical epidemiology. Stat. Methods Med. Res. 2017, 26, 1350–1372. [Google Scholar] [CrossRef]
- Mathieu, E.; Loup, P.; Dellamonica, P.; Daures, J.P. Markov modelling of immunological and virological states in HIV-1 infected patients. Biom. J. 2005, 47, 834–846. [Google Scholar] [CrossRef]
- Rangel-Frausto, M.S.; Pittet, D.; Hwang, T.; Woolson, R.F.; Wenzel, R.P. The Dynamics of Disease Progression in Sepsis: Markov Modeling Describing the Natural History and the Likely Impact of Effective Antisepsis Agents. Clin. Infect. Dis. 1998, 185–190. [Google Scholar] [CrossRef]
- Ahnert, P.; Creutz, P.; Horn, K.; Schwarzenberger, F.; Kiehntopf, M.; Hossain, H.; Bauer, M.; Brunkhorst, F.M.; Reinhart, K.; Völker, U.; et al. Sequential organ failure assessment score is an excellent operationalization of disease severity of adult patients with hospitalized community acquired pneumonia—Results from the prospective observational PROGRESS study. Crit. Care 2019, 23, 110. [Google Scholar] [CrossRef]
- Ahnert, P.; Creutz, P.; Scholz, M.; Schütte, H.; Engel, C.; Hossain, H.; Chakraborty, T.; Bauer, M.; Kiehntopf, M.; Völker, U.; et al. PROGRESS—Prospective observational study on hospitalized community acquired pneumonia. BMC Pulm. Med. 2016, 16, 108. [Google Scholar] [CrossRef]
- Brunkhorst, F.M.; Oppert, M.; Marx, G.; Bloos, F.; Ludewig, K.; Putensen, C.; Nierhaus, A.; Jaschinski, U.; Meier-Hellmann, A.; Weyland, A.; et al. Effect of empirical treatment with moxifloxacin and meropenem vs meropenem on sepsis-related organ dysfunction in patients with severe sepsis: A randomized trial. JAMA 2012, 307, 2390–2399. [Google Scholar] [CrossRef]
- Brunkhorst, F.M.; Engel, C.; Bloos, F.; Meier-Hellmann, A.; Ragaller, M.; Weiler, N.; Moerer, O.; Gruendling, M.; Oppert, M.; Grond, S.; et al. Intensive insulin therapy and pentastarch resuscitation in severe sepsis. N. Engl. J. Med. 2008, 358, 125–139. [Google Scholar] [CrossRef]
- Bloos, F.; Trips, E.; Nierhaus, A.; Briegel, J.; Heyland, D.K.; Jaschinski, U.; Moerer, O.; Weyland, A.; Marx, G.; Gründling, M.; et al. Effect of Sodium Selenite Administration and Procalcitonin-Guided Therapy on Mortality in Patients With Severe Sepsis or Septic Shock: A Randomized Clinical Trial. JAMA Intern. Med. 2016, 176, 1266–1276. [Google Scholar] [CrossRef]
- Vincent, J.L.; Moreno, R.; Takala, J.; Willatts, S.; de Mendonça, A.; Bruining, H.; Reinhart, C.K.; Suter, P.M.; Thijs, L.G. The SOFA (Sepsis-related Organ Failure Assessment) score to describe organ dysfunction/failure. On behalf of the Working Group on Sepsis-Related Problems of the European Society of Intensive Care Medicine. Intensive Care Med. 1996, 22, 707–710. [Google Scholar] [CrossRef]
- Jackson, C.H. Multi-State Models for Panel Data: The msm Package for R. J. Stat. Softw. 2011, 38, 1–28. [Google Scholar] [CrossRef]
- Robin, X.; Turck, N.; Hainard, A.; Tiberti, N.; Lisacek, F.; Sanchez, J.-C.; Müller, M. pROC: An open-source package for R and S+ to analyze and compare ROC curves. BMC Bioinform. 2011, 12, 77. [Google Scholar] [CrossRef]
- Ferreira, F.L.; Bota, D.P.; Bross, A.; Mélot, C.; Vincent, J.L. Serial evaluation of the SOFA score to predict outcome in critically ill patients. JAMA 2001, 286, 1754–1758. [Google Scholar] [CrossRef] [PubMed]
- Chow, C.C.; Clermont, G.; Kumar, R.; Lagoa, C.; Tawadrous, Z.; Gallo, D.; Betten, B.; Bartels, J.; Constantine, G.; Fink, M.P.; et al. The acute inflammatory response in diverse shock states. Shock 2005, 24, 74–84. [Google Scholar] [CrossRef] [PubMed]
- Zuev, S.M.; Kingsmore, S.F.; Gessler, D.D.G. Sepsis progression and outcome: A dynamical model. Theor. Biol. Med. Model. 2006, 3, 8. [Google Scholar] [CrossRef] [PubMed]
- Schirm, S.; Ahnert, P.; Wienhold, S.; Mueller-Redetzky, H.; Nouailles-Kursar, G.; Loeffler, M.; Witzenrath, M.; Scholz, M. A Biomathematical Model of Pneumococcal Lung Infection and Antibiotic Treatment in Mice. PLoS ONE 2016, 11, e0156047. [Google Scholar] [CrossRef]



| Study | Age | Sex (m/f) | Initial SOFA | Type of Study | Maximum Observation Time (d) | 28d Mortality N (%) | Clinical trial.gov Identifier |
|---|---|---|---|---|---|---|---|
| PROGRESS | 62 (45,74) | 1081/782 | 2 (1,3) | Observational study | 5 | 37 (2.0%) | NCT02782013 |
| ssCAP * | 69 (55,76) | 104/38 | 7 (6,8) | Observational study | 1 | 8 (5.6%) | NCT02782013 |
| MAXSEP | 67 (57,74) | 152/56 | 10 (8,12) | Randomized trial | 22 | 39 (18.8%) | NCT00534287 |
| VISEP | 66 (57,74) | 138/65 | 10 (7.5,12) | Randomized trial | 22 | 54 (26.6%) | NCT00135473 |
| SISPCT | 67.5 (56,74) | 300/122 | 9 (7,12) | Randomized Trial | 22 | 97 (23.0%) | NCT00832039 |
| Disease State | SOFA Score (SC) Range |
|---|---|
| S1 | 0 ≤ SC ≤ 2 |
| S2 | 2 < SC ≤ 5 |
| S3 | 5 < SC ≤ 9 |
| S4 | 9 < SC ≤ 24 |
| death | --- |
| Disease State to From | S1 | S2 | S3 | S4 | Death |
|---|---|---|---|---|---|
| S1 | 2545 | 369 | 0 | 0 | 0 |
| S2 | 879 | 3180 | 49 | 7 | 2 |
| S3 | 1 | 83 | 99 | 18 | 1 |
| S4 | 0 | 5 | 36 | 158 | 5 |
| Disease State to From | S1 | S2 | S3 | S4 | Death |
|---|---|---|---|---|---|
| S1 | 172/109/425 | 28/28/35 | 3/3/6 | 0/0/1 | 1/0/1 |
| S2 | 52/47/100 | 739/664/1308 | 62/88/155 | 2/4/5 | 1/2/3 |
| S3 | 5/3/6 | 152/159/323 | 669/665/1286 | 60/85/142 | 2/3/17 |
| S4 | 0/1/0 | 8/6/7 | 120/126/242 | 548/650/1245 | 24/24/49 |
| Study | S1 | S2 | S3 | S4 |
|---|---|---|---|---|
| PROGRESS | 0.01 | 0.01 | 0.05 | 0.14 |
| MAXSEP | 0.10 | 0.10 | 0.13 | 0.25 |
| VISEP | 0.11 | 0.13 | 0.17 | 0.27 |
| SISPCT | 0.11 | 0.15 | 0.20 | 0.30 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Przybilla, J.; Ahnert, P.; Bogatsch, H.; Bloos, F.; Brunkhorst, F.M.; SepNet Critical Care Trials Group; PROGRESS study group; Bauer, M.; Loeffler, M.; Witzenrath, M.; et al. Markov State Modelling of Disease Courses and Mortality Risks of Patients with Community-Acquired Pneumonia. J. Clin. Med. 2020, 9, 393. https://doi.org/10.3390/jcm9020393
Przybilla J, Ahnert P, Bogatsch H, Bloos F, Brunkhorst FM, SepNet Critical Care Trials Group, PROGRESS study group, Bauer M, Loeffler M, Witzenrath M, et al. Markov State Modelling of Disease Courses and Mortality Risks of Patients with Community-Acquired Pneumonia. Journal of Clinical Medicine. 2020; 9(2):393. https://doi.org/10.3390/jcm9020393
Chicago/Turabian StylePrzybilla, Jens, Peter Ahnert, Holger Bogatsch, Frank Bloos, Frank M. Brunkhorst, SepNet Critical Care Trials Group, PROGRESS study group, Michael Bauer, Markus Loeffler, Martin Witzenrath, and et al. 2020. "Markov State Modelling of Disease Courses and Mortality Risks of Patients with Community-Acquired Pneumonia" Journal of Clinical Medicine 9, no. 2: 393. https://doi.org/10.3390/jcm9020393
APA StylePrzybilla, J., Ahnert, P., Bogatsch, H., Bloos, F., Brunkhorst, F. M., SepNet Critical Care Trials Group, PROGRESS study group, Bauer, M., Loeffler, M., Witzenrath, M., Suttorp, N., & Scholz, M. (2020). Markov State Modelling of Disease Courses and Mortality Risks of Patients with Community-Acquired Pneumonia. Journal of Clinical Medicine, 9(2), 393. https://doi.org/10.3390/jcm9020393






