Affinity-Enhanced CTC-Capturing Hydrogel Microparticles Fabricated by Degassed Mold Lithography
Abstract
1. Introduction
2. Experimental Section
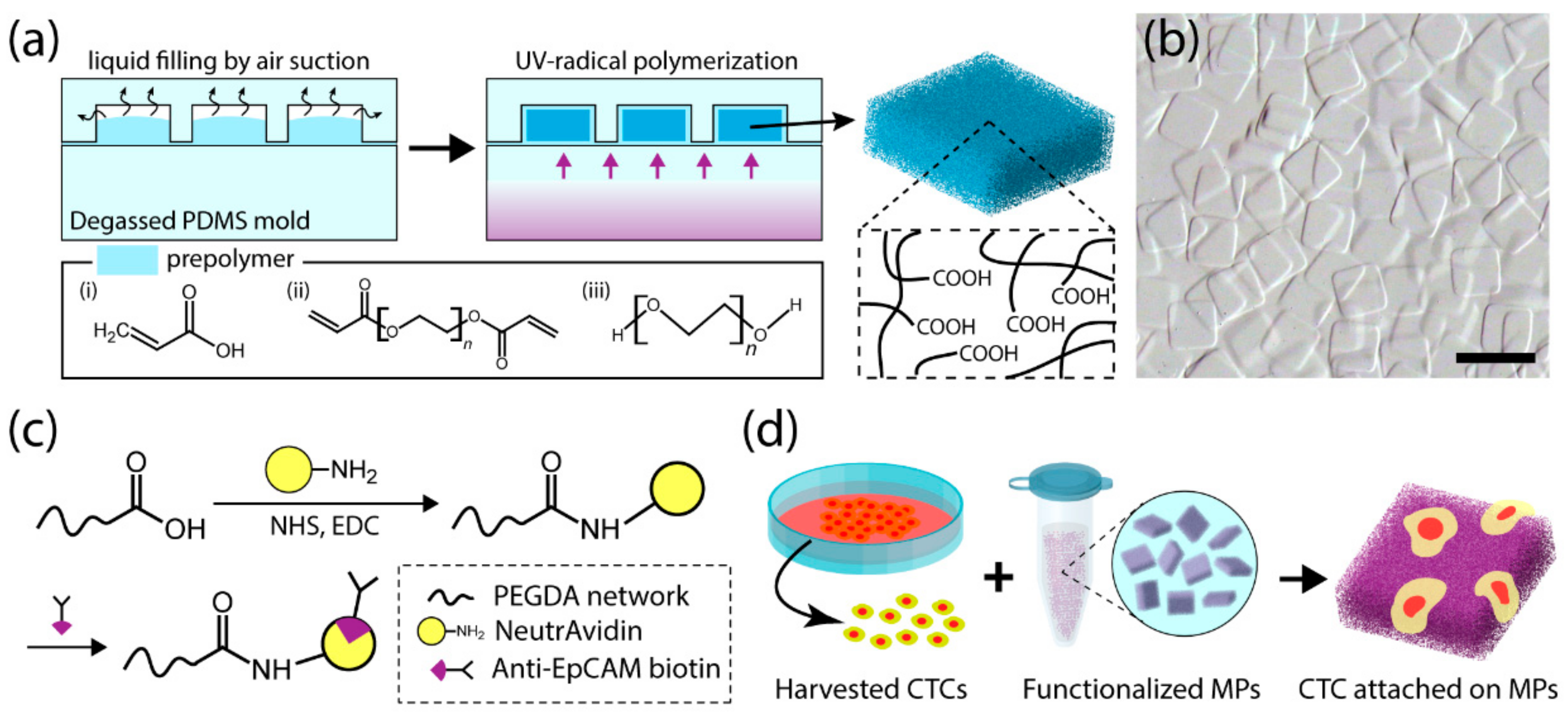
2.1. Fabrication of the Micromold and Microfluidic Device
2.2. DML Setup
2.3. SFL Setup
2.4. Functionalization of Hydrogel Microparticle
2.5. Fluorescence Analysis
2.6. Cell Culture
2.7. Cell Affinity Test
2.8. Image Analysis
3. Results
3.1. Functionalized Hydrogel Microparticle Synthesis via DML
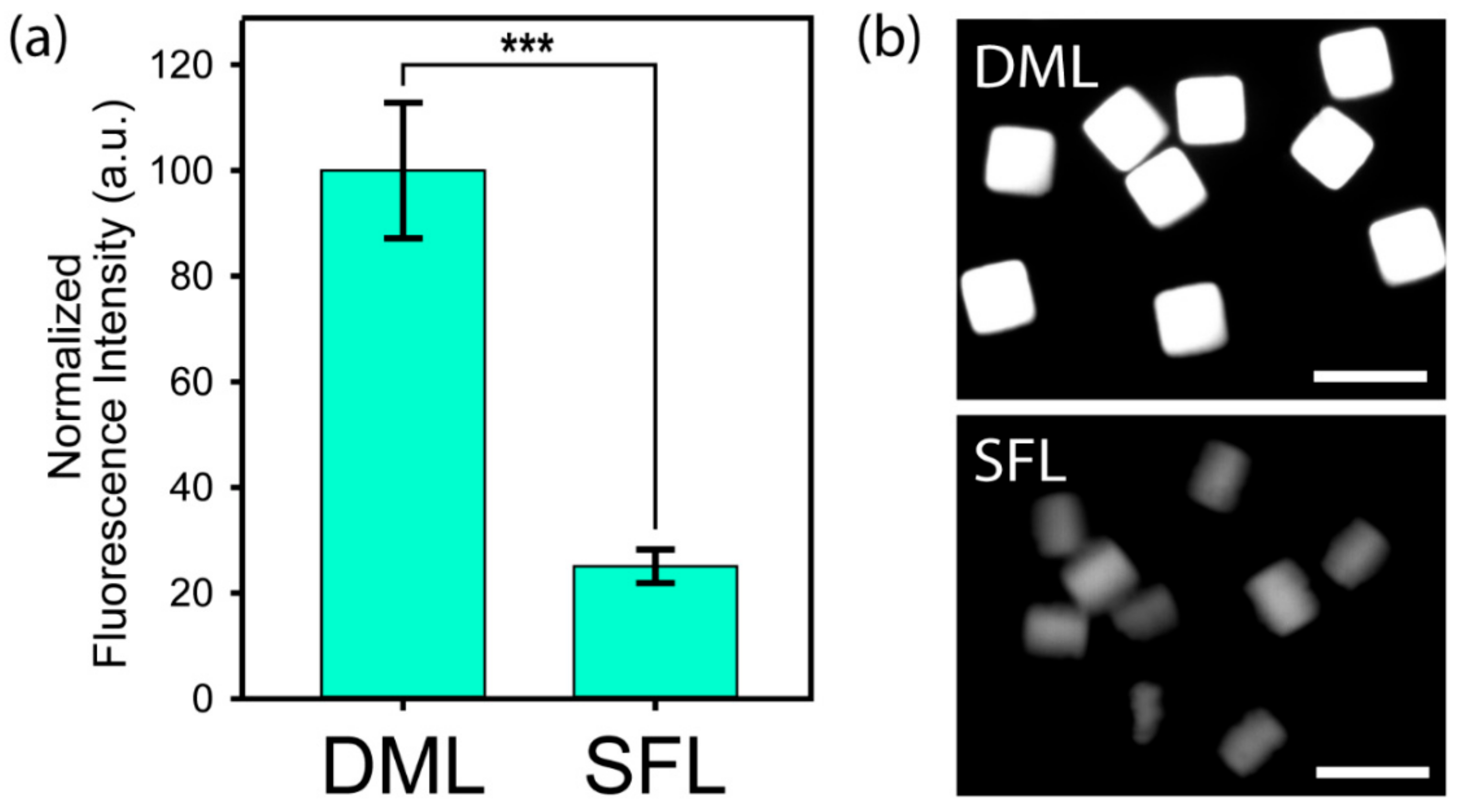
3.2. Comparison of the Particle Properties between DML and SFL
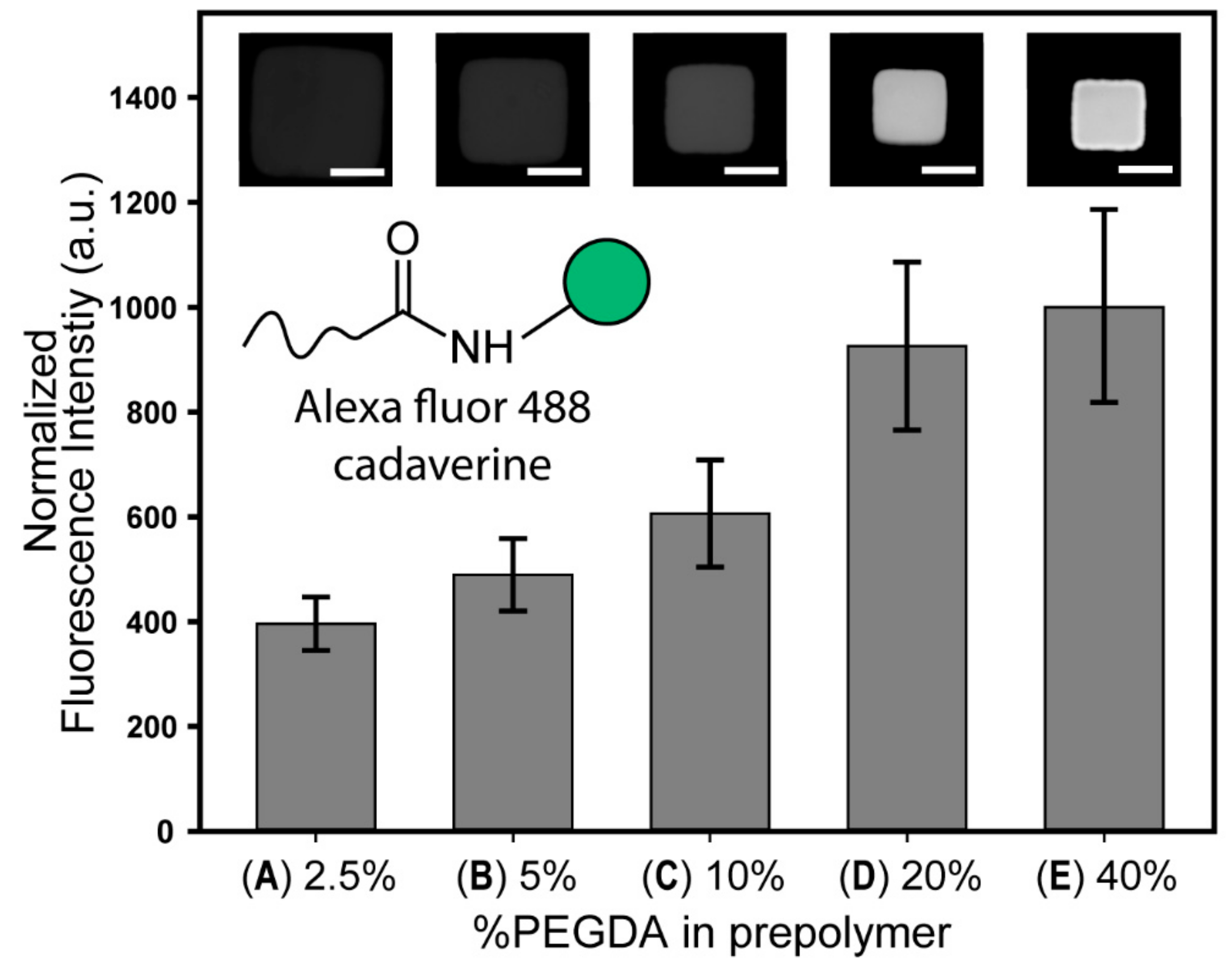
3.3. Qualitative Analysis of Surface Carboxyl Group
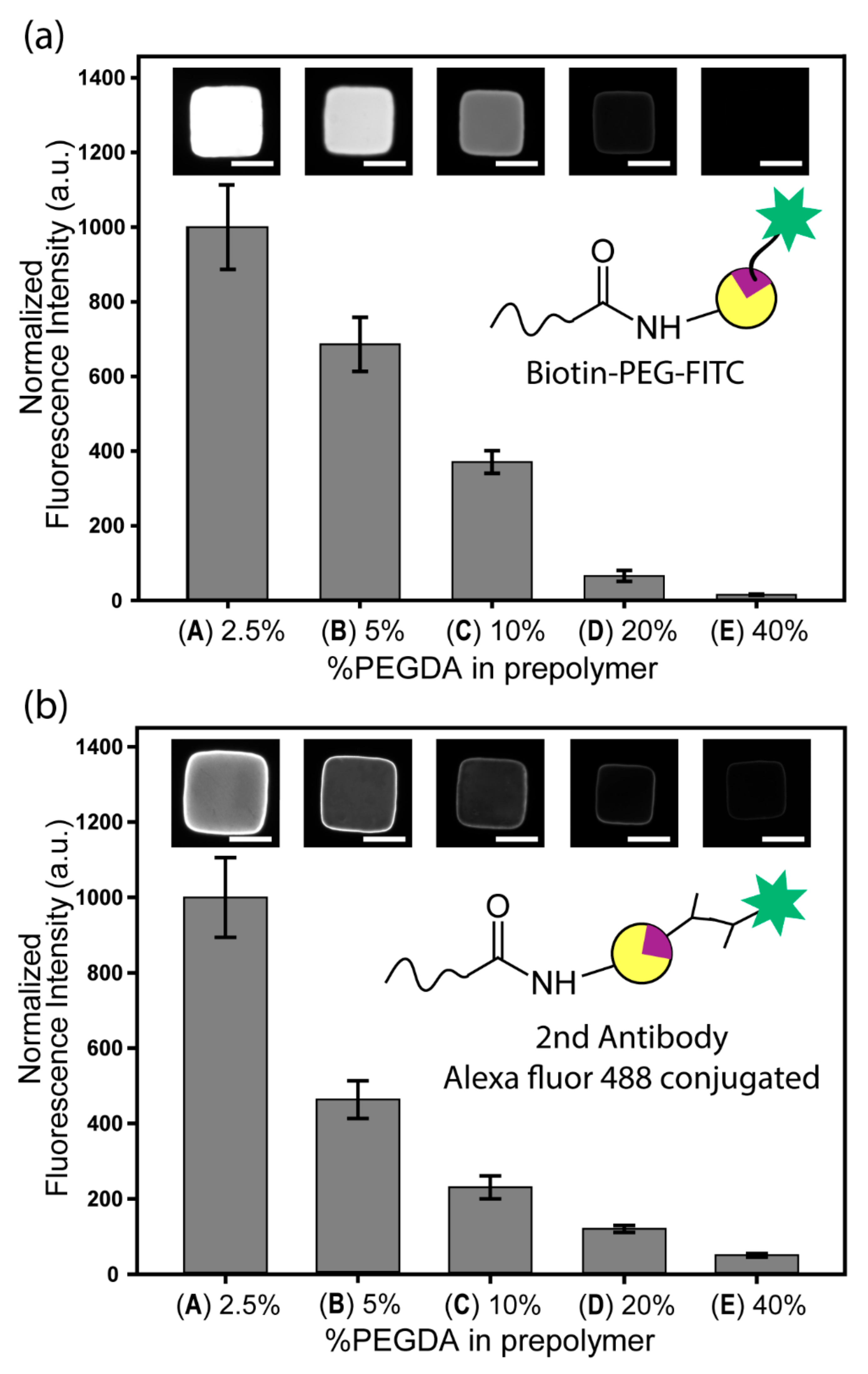
3.4. Qualitative Analysis of NeutrAvidin and Anti-EpCAM Functionalization
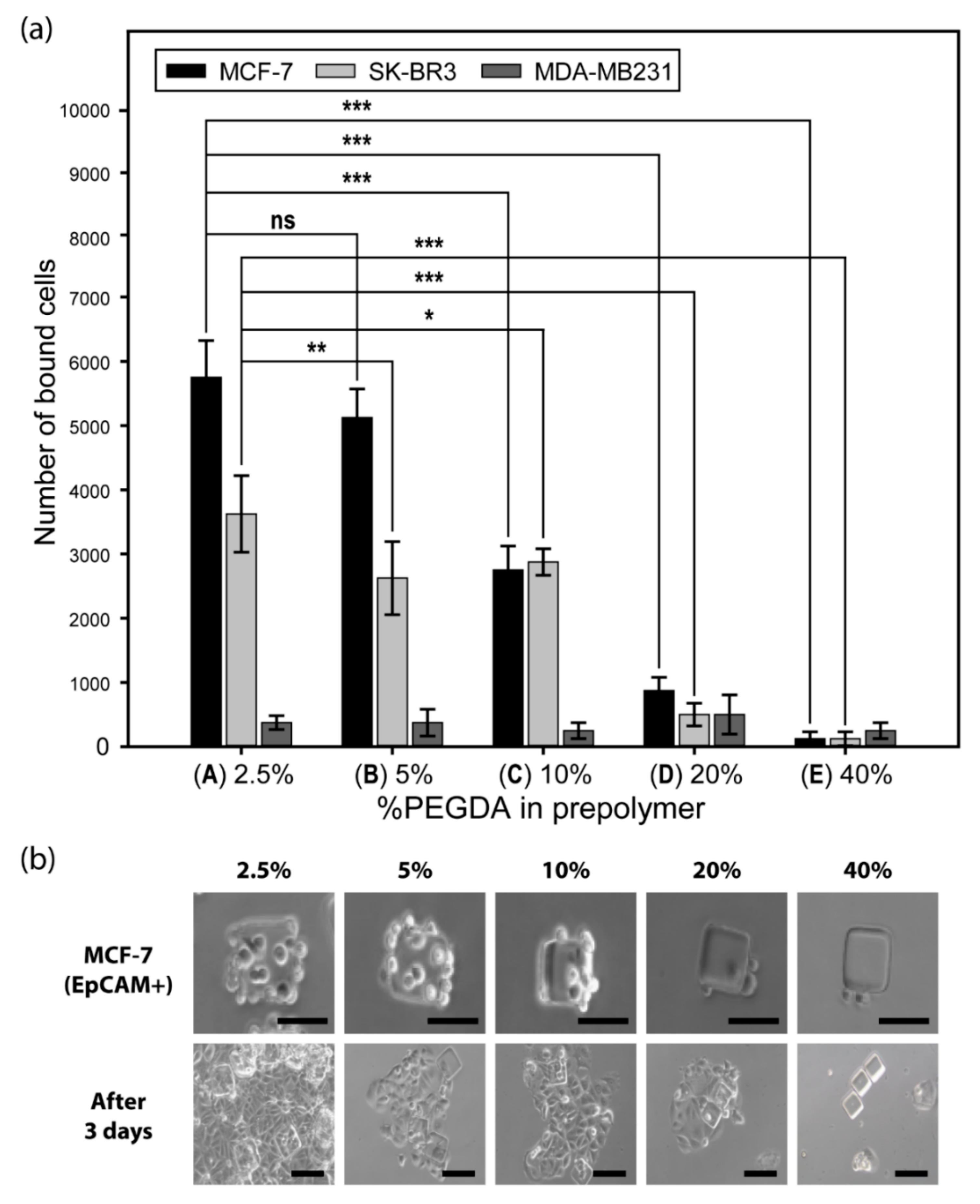
3.5. Cell Affinity Test
4. Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Alix-Panabières, C.; Pantel, K. Circulating tumor cells: Liquid biopsy of cancer. Clin. Chem. 2013, 59, 110–118. [Google Scholar] [CrossRef]
- Gradilone, A.; Naso, G.; Raimondi, C.; Cortesi, E.; Gandini, O.; Vincenzi, B.; Saltarelli, R.; Chiapparino, E.; Spremberg, F.; Cristofanilli, M. Circulating tumor cells (CTCs) in metastatic breast cancer (MBC): Prognosis, drug resistance and phenotypic characterization. Ann. Oncol. 2010, 22, 86–92. [Google Scholar] [CrossRef]
- Cristofanilli, M.; Budd, G.T.; Ellis, M.J.; Stopeck, A.; Matera, J.; Miller, M.C.; Reuben, J.M.; Doyle, G.V.; Allard, W.J.; Terstappen, L.W. Circulating tumor cells, disease progression, and survival in metastatic breast cancer. N. Engl. J. Med. 2004, 351, 781–791. [Google Scholar] [CrossRef] [PubMed]
- Thangavel, H.; Angelis, C.D.; Vasaikar, S.; Bhat, R.; Jolly, M.K.; Nagi, C.; Creighton, C.J.; Chen, F.; Dobrolecki, L.E.; George, J.T. A CTC-cluster-specific signature derived from OMICS analysis of patient-derived xenograft tumors predicts outcomes in basal-like breast cancer. J. Clin. Med. 2019, 8, 1772. [Google Scholar] [CrossRef] [PubMed]
- Jee, H.-G.; Kim, B.; Kim, M.; Yu, H.W.; Choi, J.Y.; Kim, S.-J.; Lee, K.E. Expression of SLC5A5 in circulating tumor cells may distinguish follicular thyroid carcinomas from adenomas: Implications for blood-based preoperative diagnosis. J. Clin. Med. 2019, 8, 257. [Google Scholar] [CrossRef] [PubMed]
- Allard, W.J.; Matera, J.; Miller, M.C.; Repollet, M.; Connelly, M.C.; Rao, C.; Tibbe, A.G.; Uhr, J.W.; Terstappen, L.W. Tumor cells circulate in the peripheral blood of all major carcinomas but not in healthy subjects or patients with nonmalignant diseases. Clin. Cancer Res. 2004, 10, 6897–6904. [Google Scholar] [CrossRef]
- Alix-Panabières, C.; Pantel, K. Challenges in circulating tumour cell research. Nat. Rev. Cancer 2014, 14, 623–631. [Google Scholar] [CrossRef] [PubMed]
- Bidard, F.-C.; Peeters, D.J.; Fehm, T.; Nolé, F.; Gisbert-Criado, R.; Mavroudis, D.; Grisanti, S.; Generali, D.; Garcia-Saenz, J.A.; Stebbing, J. Clinical validity of circulating tumour cells in patients with metastatic breast cancer: A pooled analysis of individual patient data. Lancet Oncol. 2014, 15, 406–414. [Google Scholar] [CrossRef]
- Bidard, F.-C.; Michiels, S.; Riethdorf, S.; Mueller, V.; Esserman, L.J.; Lucci, A.; Naume, B.; Horiguchi, J.; Gisbert-Criado, R.; Sleijfer, S. Circulating tumor cells in breast cancer patients treated by neoadjuvant chemotherapy: A meta-analysis. J. Natl. Cancer Inst. 2018, 110, 560–567. [Google Scholar] [CrossRef]
- Shen, Z.; Wu, A.; Chen, X. Current detection technologies for circulating tumor cells. Chem. Soc. Rev. 2017, 46, 2038–2056. [Google Scholar] [CrossRef]
- Adams, D.L.; Martin, S.S.; Alpaugh, R.K.; Charpentier, M.; Tsai, S.; Bergan, R.C.; Ogden, I.M.; Catalona, W.; Chumsri, S.; Tang, C.-M. Circulating giant macrophages as a potential biomarker of solid tumors. Proc. Natl. Acad. Sci. USA 2014, 111, 3514–3519. [Google Scholar] [CrossRef] [PubMed]
- Khoo, B.L.; Lee, S.C.; Kumar, P.; Tan, T.Z.; Warkiani, M.E.; Ow, S.G.; Nandi, S.; Lim, C.T.; Thiery, J.P. Short-term expansion of breast circulating cancer cells predicts response to anti-cancer therapy. OncoTarget 2015, 6, 15578–15593. [Google Scholar] [CrossRef] [PubMed]
- Freidin, M.B.; Tay, A.; Freydina, D.V.; Chudasama, D.; Nicholson, A.G.; Rice, A.; Anikin, V.; Lim, E. An assessment of diagnostic performance of a filter-based antibody-independent peripheral blood circulating tumour cell capture paired with cytomorphologic criteria for the diagnosis of cancer. Lung Cancer 2014, 85, 182–185. [Google Scholar] [CrossRef] [PubMed]
- Sarioglu, A.F.; Aceto, N.; Kojic, N.; Donaldson, M.C.; Zeinali, M.; Hamza, B.; Engstrom, A.; Zhu, H.; Sundaresan, T.K.; Miyamoto, D.T. A microfluidic device for label-free, physical capture of circulating tumor cell clusters. Nat. Methods 2015, 12, 685–691. [Google Scholar] [CrossRef]
- Kim, T.-H.; Lim, M.; Park, J.; Oh, J.M.; Kim, H.; Jeong, H.; Lee, S.J.; Park, H.C.; Jung, S.; Kim, B.C. FAST: Size-selective, clog-free isolation of rare cancer cells from whole blood at a liquid—Liquid interface. Anal. Chem. 2016, 89, 1155–1162. [Google Scholar] [CrossRef]
- Marrinucci, D.; Bethel, K.; Kolatkar, A.; Luttgen, M.S.; Malchiodi, M.; Baehring, F.; Voigt, K.; Lazar, D.; Nieva, J.; Bazhenova, L. Fluid biopsy in patients with metastatic prostate, pancreatic and breast cancers. Phys. Biol. 2012, 9, 016003. [Google Scholar] [CrossRef]
- Somlo, G.; Lau, S.K.; Frankel, P.; Hsieh, H.B.; Liu, X.; Yang, L.; Krivacic, R.; Bruce, R.H. Multiple biomarker expression on circulating tumor cells in comparison to tumor tissues from primary and metastatic sites in patients with locally advanced/inflammatory, and stage IV breast cancer, using a novel detection technology. Breast Cancer Res. Treat. 2011, 128, 155–163. [Google Scholar] [CrossRef]
- Shim, S.; Stemke-Hale, K.; Tsimberidou, A.M.; Noshari, J.; Anderson, T.E.; Gascoyne, P.R. Antibody-independent isolation of circulating tumor cells by continuous-flow dielectrophoresis. Biomicrofluidics 2013, 7, 011807. [Google Scholar] [CrossRef]
- Negishi, R.; Hosokawa, M.; Nakamura, S.; Kanbara, H.; Kanetomo, M.; Kikuhara, Y.; Tanaka, T.; Matsunaga, T.; Yoshino, T. Development of the automated circulating tumor cell recovery system with microcavity array. Biosens. Bioelectron. 2015, 67, 438–442. [Google Scholar] [CrossRef]
- Adams, D.L.; Zhu, P.; Makarova, O.V.; Martin, S.S.; Charpentier, M.; Chumsri, S.; Li, S.; Amstutz, P.; Tang, C.-M. The systematic study of circulating tumor cell isolation using lithographic microfilters. RSC Adv. 2014, 4, 4334–4342. [Google Scholar] [CrossRef]
- Kang, Y.-T.; Kim, Y.J.; Lee, T.H.; Cho, Y.-H.; Chang, H.J.; Lee, H.-M. Cytopathological study of the circulating tumor cells filtered from the cancer patients’ blood using hydrogel-based cell block formation. Sci. Rep. 2018, 8, 15218. [Google Scholar] [CrossRef] [PubMed]
- Park, E.S.; Yan, J.P.; Ang, R.A.; Lee, J.H.; Deng, X.; Duffy, S.P.; Beja, K.; Annala, M.; Black, P.C.; Chi, K.N. Isolation and genome sequencing of individual circulating tumor cells using hydrogel encapsulation and laser capture microdissection. Lab Chip 2018, 18, 1736–1749. [Google Scholar] [CrossRef] [PubMed]
- Yoshino, T.; Tanaka, T.; Nakamura, S.; Negishi, R.; Hosokawa, M.; Matsunaga, T. Manipulation of a single circulating tumor cell using visualization of hydrogel encapsulation toward single-cell whole-genome amplification. Anal. Chem. 2016, 88, 7230–7237. [Google Scholar] [CrossRef] [PubMed]
- Ozkumur, E.; Shah, A.M.; Ciciliano, J.C.; Emmink, B.L.; Miyamoto, D.T.; Brachtel, E.; Yu, M.; Chen, P.-i.; Morgan, B.; Trautwein, J. Inertial focusing for tumor antigen-dependent and -independent sorting of rare circulating tumor cells. Sci. Transl. Med. 2013, 5, 179ra147. [Google Scholar] [CrossRef]
- Sheng, W.; Ogunwobi, O.O.; Chen, T.; Zhang, J.; George, T.J.; Liu, C.; Fan, Z.H. Capture, release and culture of circulating tumor cells from pancreatic cancer patients using an enhanced mixing chip. Lab Chip 2014, 14, 89–98. [Google Scholar] [CrossRef]
- Winer-Jones, J.P.; Vahidi, B.; Arquilevich, N.; Fang, C.; Ferguson, S.; Harkins, D.; Hill, C.; Klem, E.; Pagano, P.C.; Peasley, C. Circulating tumor cells: Clinically relevant molecular access based on a novel CTC flow cell. PLoS ONE 2014, 9, e86717. [Google Scholar] [CrossRef]
- Cho, D.; Yoo, H.; Lee, H.; Choi, Y.; Lee, M.; Ahn, D.; Hong, S. High-speed lateral flow strategy for a fast biosensing with an improved selectivity and binding affinity. Sensors 2018, 18, 1507. [Google Scholar] [CrossRef]
- Song, P.; Ye, D.; Zuo, X.; Li, J.; Wang, J.; Liu, H.; Hwang, M.T.; Chao, J.; Su, S.; Wang, L. DNA hydrogel with aptamer-toehold-based recognition, cloaking, and decloaking of circulating tumor cells for live cell analysis. Nano Lett. 2017, 17, 5193–5198. [Google Scholar] [CrossRef]
- Cheng, S.-B.; Xie, M.; Chen, Y.; Xiong, J.; Liu, Y.; Chen, Z.; Guo, S.; Shu, Y.; Wang, M.; Yuan, B.-F. Three-dimensional scaffold chip with thermosensitive coating for capture and reversible release of individual and cluster of circulating tumor cells. Anal. Chem. 2017, 89, 7924–7932. [Google Scholar] [CrossRef]
- Chen, L.; An, H.Z.; Haghgooie, R.; Shank, A.T.; Martel, J.M.; Toner, M.; Doyle, P.S. Flexible octopus-shaped hydrogel particles for specific cell capture. Small 2016, 12, 2001–2008. [Google Scholar] [CrossRef]
- LeValley, P.J.; Tibbitt, M.W.; Noren, B.; Kharkar, P.; Kloxin, A.M.; Anseth, K.S.; Toner, M.; Oakey, J. Immunofunctional photodegradable poly (ethylene glycol) hydrogel surfaces for the capture and release of rare cells. Colloids Surf. B Biointerfaces 2019, 174, 483–492. [Google Scholar] [CrossRef] [PubMed]
- Cho, I.-H.; Paek, E.-H.; Lee, H.; Choi, J.-W.; Paek, S.-H. Site-directed immobilization of antibody onto solid surfaces for the construction of immunochip. Biotechnol. Bioprocess Eng. 2004, 9, 112–117. [Google Scholar] [CrossRef]
- Kent, S.P.; Ryan, K.H.; Siegel, A.L. Steric hindrance as a factor in the reaction of labeled antibody with cell surface antigenic determinants. J. Histochem. Cytochem. 1978, 26, 618–621. [Google Scholar] [CrossRef]
- Lee, J.; Lee, S.Y.; Lim, D.-K.; Ahn, D.J.; Lee, S. Antifreezing Gold Colloids. J. Am. Chem. Soc. 2019, 141, 18682–18693. [Google Scholar] [CrossRef] [PubMed]
- Choi, N.W.; Kim, J.; Chapin, S.C.; Duong, T.; Donohue, E.; Pandey, P.; Broom, W.; Hill, W.A.; Doyle, P.S. Multiplexed detection of mRNA using porosity-tuned hydrogel microparticles. Anal. Chem. 2012, 84, 9370–9378. [Google Scholar] [CrossRef] [PubMed]
- Jung, S.; Tang, Y.; Shim, G.; Lee, C.-S.; Choi, C.-H.; Yi, H. Controlled network structures of chitosan-poly (ethylene glycol) hydrogel microspheres and their impact on protein conjugation. Biochem. Eng. 2018, 135, 123–132. [Google Scholar] [CrossRef]
- Kang, E.; Jung, S.; Abel, J.H.; Pine, A.; Yi, H. Shape-encoded chitosan—Polyacrylamide hybrid hydrogel microparticles with controlled macroporous structures via replica molding for programmable biomacromolecular conjugation. Langmuir 2016, 32, 5394–5402. [Google Scholar] [CrossRef]
- Lee, A.G.; Arena, C.P.; Beebe, D.J.; Palecek, S.P. Development of macroporous poly (ethylene glycol) hydrogel arrays within microfluidic channels. Biomacromolecules 2010, 11, 3316–3324. [Google Scholar] [CrossRef]
- Appleyard, D.C.; Chapin, S.C.; Srinivas, R.L.; Doyle, P.S. Bar-coded hydrogel microparticles for protein detection: Synthesis, assay and scanning. Nat. Protoc. 2011, 6, 1761–1774. [Google Scholar] [CrossRef]
- Baah, D.; Floyd-Smith, T. Microfluidics for particle synthesis from photocrosslinkable materials. Microfluid. Nanofluidics 2014, 17, 431–455. [Google Scholar] [CrossRef]
- Kim, H.U.; Lim, Y.J.; Lee, H.J.; Lee, N.J.; Bong, K.W. Degassed micromolding lithography for rapid fabrication of anisotropic hydrogel microparticles with high-resolution and high uniformity. Lab Chip 2020, 20, 74–83. [Google Scholar] [CrossRef] [PubMed]
- Chun, M.J.; Choi, Y.K.; Ahn, D.J. Formation of nanopores in DiynePC–DPPC complex lipid bilayers triggered by on-demand photo-polymerization. RSC Adv. 2018, 8, 27988–27994. [Google Scholar] [CrossRef]
- Choi, Y.K.; Lee, S.Y.; Ahn, D.J. Hyperconjugation-induced chromism in linear responsive polymers. J. Mater. Chem. C 2019, 7, 13130–13138. [Google Scholar] [CrossRef]
- Kim, T.; Moon, D.; Park, J.H.; Yang, H.; Cho, S.; Park, T.H.; Ahn, D.J. Visual detection of odorant geraniol enabled by integration of a human olfactory receptor into polydiacetylene/lipid nano-assembly. Nanoscale 2019, 11, 7582–7587. [Google Scholar] [CrossRef]
- Huang, J.; Park, J.H.; Back, S.H.; Feng, Y.; Cui, C.; Jin, L.Y.; Ahn, D.J. Mercury ion–DNA specificity triggers a distinctive photoluminescence depression in organic semiconductor probes guided with a thymine-rich oligonucleotide sequence. Nanoscale 2018, 10, 17540–17545. [Google Scholar] [CrossRef]
- Dendukuri, D.; Panda, P.; Haghgooie, R.; Kim, J.M.; Hatton, T.A.; Doyle, P.S. Modeling of oxygen-inhibited free radical photopolymerization in a PDMS microfluidic device. Macromolecules 2008, 41, 8547–8556. [Google Scholar] [CrossRef]
- Andrzejewska, E. Photopolymerization kinetics of multifunctional monomers. Prog. Polym. Sci. 2001, 26, 605–665. [Google Scholar] [CrossRef]
- Moon, H.J.; Ku, M.; Lee, H.; Yoon, N.; Yang, J.; Bong, K.W. Implantable photothermal agents based on gold nanorods-encapsulated microcube. Sci. Rep. 2018, 8, 13683. [Google Scholar] [CrossRef]
| Group | Composition | |||
|---|---|---|---|---|
| PEGDA 700 | PEG 200 | Acrylic Acid | PI | |
| A | 2.5% | 62.5% | 30.0% | 5.0% |
| B | 5.0% | 60.0% | 30.0% | 5.0% |
| C | 10.0% | 55.0% | 30.0% | 5.0% |
| D | 20.0% | 45.0% | 30.0% | 5.0% |
| E | 40.0% | 25.0% | 30.0% | 5.0% |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, N.J.; Maeng, S.; Kim, H.U.; Roh, Y.H.; Hwang, C.; Kim, J.; Hwang, K.-T.; Bong, K.W. Affinity-Enhanced CTC-Capturing Hydrogel Microparticles Fabricated by Degassed Mold Lithography. J. Clin. Med. 2020, 9, 301. https://doi.org/10.3390/jcm9020301
Lee NJ, Maeng S, Kim HU, Roh YH, Hwang C, Kim J, Hwang K-T, Bong KW. Affinity-Enhanced CTC-Capturing Hydrogel Microparticles Fabricated by Degassed Mold Lithography. Journal of Clinical Medicine. 2020; 9(2):301. https://doi.org/10.3390/jcm9020301
Chicago/Turabian StyleLee, Nak Jun, Sejung Maeng, Hyeon Ung Kim, Yoon Ho Roh, Changhyun Hwang, Jongjin Kim, Ki-Tae Hwang, and Ki Wan Bong. 2020. "Affinity-Enhanced CTC-Capturing Hydrogel Microparticles Fabricated by Degassed Mold Lithography" Journal of Clinical Medicine 9, no. 2: 301. https://doi.org/10.3390/jcm9020301
APA StyleLee, N. J., Maeng, S., Kim, H. U., Roh, Y. H., Hwang, C., Kim, J., Hwang, K.-T., & Bong, K. W. (2020). Affinity-Enhanced CTC-Capturing Hydrogel Microparticles Fabricated by Degassed Mold Lithography. Journal of Clinical Medicine, 9(2), 301. https://doi.org/10.3390/jcm9020301






