A Real-World Comparative Analysis of Lenvatinib and Sorafenib as a Salvage Therapy for Transarterial Treatments in Unresectable HCC
Abstract
1. Introduction
2. Patients and Methods
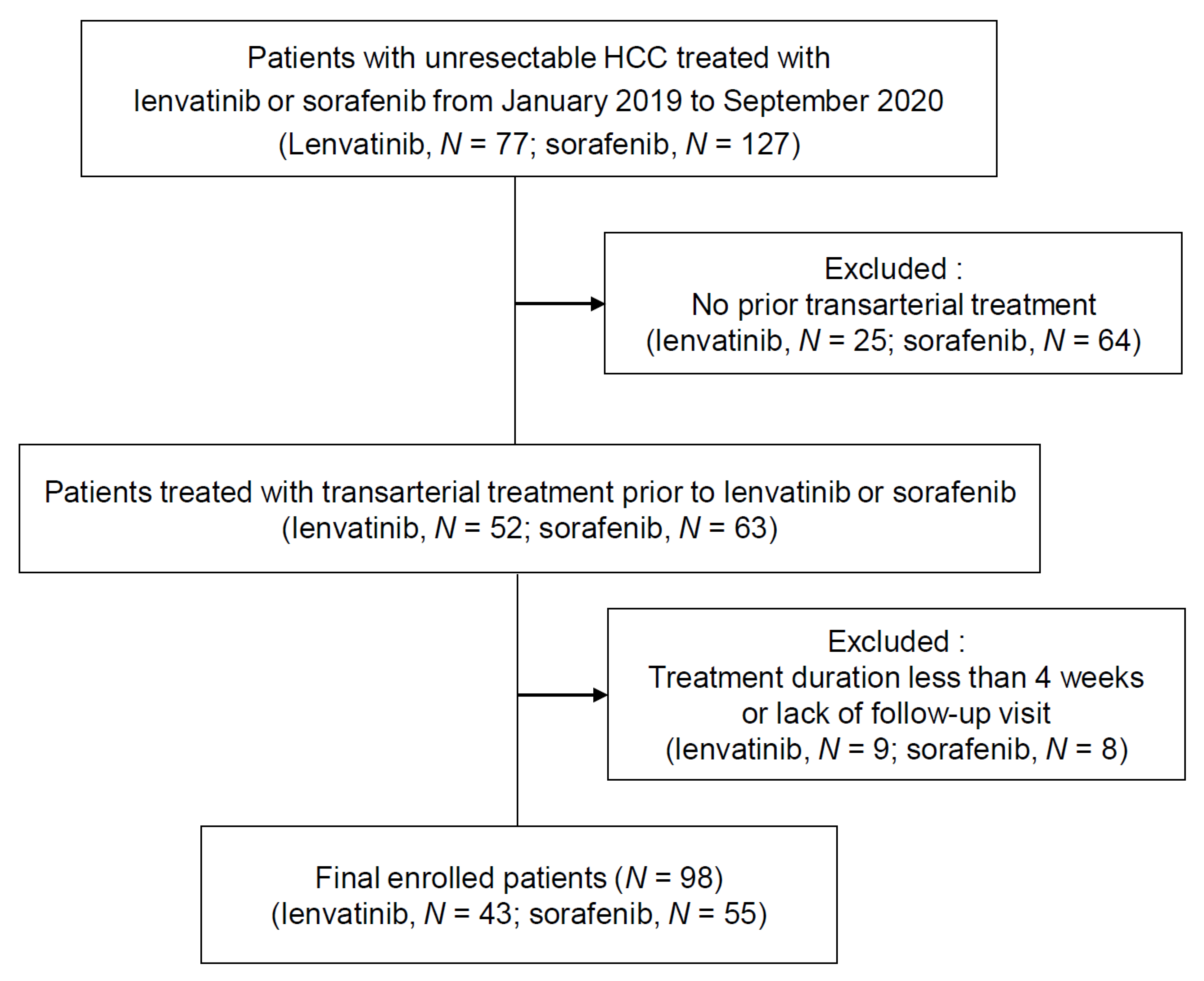
2.1. Patients
2.2. Lenvatinib and Sorafenib Treatment
2.3. Evaluation of Treatment Responses and Assessment of Adverse Events
2.4. Statistical Analysis
3. Results
3.1. Baseline Characteristics
3.2. Treatment Responses
3.3. Survival Outcomes
3.4. Adverse Events
3.5. Factors Contributing to Survival Outcomes
3.6. A representative Patient Case
4. Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
References
- Yang, H.; Sung, P.S.; Lee, J.; Yoon, S.K.; Jung, E.S.; Park, C.S.; Bae, S.H. Heterogenous responses to nivolumab in a single metastatic nodule in hepatocellular carcinoma: Role of salvage surgery. Hepatobiliary Surg. Nutr. 2019, 8, 569–571. [Google Scholar] [CrossRef] [PubMed]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.H.; Kim, S.Y.; Tang, A.; Lee, J.M. Comparison of international guidelines for noninvasive diagnosis of hepatocellular carcinoma: 2018 update. Clin. Mol. Hepatol. 2019, 25, 245–263. [Google Scholar] [CrossRef] [PubMed]
- Song, M.J. Hepatic artery infusion chemotherapy for advanced hepatocellular carcinoma. World J. Gastroenterol. 2015, 21, 3843–3849. [Google Scholar] [CrossRef] [PubMed]
- Sung, P.S.; Yang, K.; Bae, S.H.; Oh, J.S.; Chun, H.J.; Nam, H.C.; Jang, J.W.; Choi, J.Y.; Yoon, S.K. Reduction of Intrahepatic Tumour by Hepatic Arterial Infusion Chemotherapy Prolongs Survival in Hepatocellular Carcinoma. Anticancer Res. 2019, 39, 3909–3916. [Google Scholar] [CrossRef]
- Ogasawara, S.; Ooka, Y.; Koroki, K.; Maruta, S.; Kanzaki, H.; Kanayama, K.; Kobayashi, K.; Kiyono, S.; Nakamura, M.; Kanogawa, N.; et al. Switching to systemic therapy after locoregional treatment failure: Definition and best timing. Clin. Mol. Hepatol. 2020, 26, 155–162. [Google Scholar] [CrossRef]
- Llovet, J.M.; Ricci, S.; Mazzaferro, V.; Hilgard, P.; Gane, E.; Blanc, J.F.; de Oliveira, A.C.; Santoro, A.; Raoul, J.L.; Forner, A.; et al. Sorafenib in advanced hepatocellular carcinoma. N. Engl. J. Med. 2008, 359, 378–390. [Google Scholar] [CrossRef]
- Liu, Z.; Lin, Y.; Zhang, J.; Zhang, Y.; Li, Y.; Liu, Z.; Li, Q.; Luo, M.; Liang, R.; Ye, J. Molecular targeted and immune checkpoint therapy for advanced hepatocellular carcinoma. J. Exp. Clin. Cancer Res. 2019, 38, 447. [Google Scholar] [CrossRef]
- Cheng, A.L.; Kang, Y.K.; Lin, D.Y.; Park, J.W.; Kudo, M.; Qin, S.; Chung, H.C.; Song, X.; Xu, J.; Poggi, G.; et al. Sunitinib versus sorafenib in advanced hepatocellular cancer: Results of a randomized phase III trial. J. Clin. Oncol. 2013, 31, 4067–4075. [Google Scholar] [CrossRef]
- Cainap, C.; Qin, S.; Huang, W.T.; Chung, I.J.; Pan, H.; Cheng, Y.; Kudo, M.; Kang, Y.K.; Chen, P.J.; Toh, H.C.; et al. Linifanib versus Sorafenib in patients with advanced hepatocellular carcinoma: Results of a randomized phase III trial. J. Clin. Oncol. 2015, 33, 172–179. [Google Scholar] [CrossRef]
- Kudo, M.; Finn, R.S.; Qin, S.; Han, K.-H.; Ikeda, K.; Piscaglia, F.; Baron, A.; Park, J.-W.; Han, G.; Jassem, J.; et al. Lenvatinib versus sorafenib in first-line treatment of patients with unresectable hepatocellular carcinoma: A randomised phase 3 non-inferiority trial. Lancet 2018, 391, 1163–1173. [Google Scholar] [CrossRef]
- Obi, S.; Sato, T.; Sato, S.; Kanda, M.; Tokudome, Y.; Kojima, Y.; Suzuki, Y.; Hosoda, K.; Kawai, T.; Kondo, Y.; et al. The efficacy and safety of lenvatinib for advanced hepatocellular carcinoma in a real-world setting. Hepatol. Int. 2019, 13, 199–204. [Google Scholar] [CrossRef] [PubMed]
- Kudo, M. A New Treatment Option for Intermediate-Stage Hepatocellular Carcinoma with High Tumor Burden: Initial Lenvatinib Therapy with Subsequent Selective TACE. Liver Cancer 2019, 8, 299–311. [Google Scholar] [CrossRef] [PubMed]
- Cheon, J.; Chon, H.J.; Bang, Y.; Park, N.H.; Shin, J.W.; Kim, K.M.; Lee, H.C.; Lee, J.; Yoo, C.; Ryoo, B.-Y. Real-World Efficacy and Safety of Lenvatinib in Korean Patients with Advanced Hepatocellular Carcinoma: A Multicenter Retrospective Analysis. Liver Cancer 2020, 1–12. [Google Scholar] [CrossRef]
- Hiraoka, A.; Kumada, T.; Atsukawa, M.; Hirooka, M.; Tsuji, K.; Ishikawa, T.; Takaguchi, K.; Kariyama, K.; Itobayashi, E.; Tajiri, K.; et al. Prognostic factor of lenvatinib for unresectable hepatocellular carcinoma in real-world conditions-Multicenter analysis. Cancer Med. 2019, 8, 3719–3728. [Google Scholar] [CrossRef]
- Ohki, T.; Sato, K.; Kondo, M.; Goto, E.; Sato, T.; Kondo, Y.; Akamatsu, M.; Sato, S.; Yoshida, H.; Koike, Y.; et al. Impact of Adverse Events on the Progression-Free Survival of Patients with Advanced Hepatocellular Carcinoma Treated with Lenvatinib: A Multicenter Retrospective Study. Drugs Real World Outcomes 2020, 7, 141–149. [Google Scholar] [CrossRef]
- Kuzuya, T.; Ishigami, M.; Ito, T.; Ishizu, Y.; Honda, T.; Ishikawa, T.; Fujishiro, M. Sorafenib vs. Lenvatinib as First-line Therapy for Advanced Hepatocellular Carcinoma with Portal Vein Tumor Thrombosis. Anticancer Res. 2020, 40, 2283–2290. [Google Scholar] [CrossRef]
- Maruta, S.; Ogasawara, S.; Ooka, Y.; Obu, M.; Inoue, M.; Itokawa, N.; Haga, Y.; Seki, A.; Okabe, S.; Azemoto, R.; et al. Potential of Lenvatinib for an Expanded Indication from the REFLECT Trial in Patients with Advanced Hepatocellular Carcinoma. Liver Cancer 2020, 9, 382–396. [Google Scholar] [CrossRef]
- Kudo, M.; Ueshima, K.; Chan, S.; Minami, T.; Chishina, H.; Aoki, T.; Takita, M.; Hagiwara, S.; Minami, Y.; Ida, H.; et al. Lenvatinib as an Initial Treatment in Patients with Intermediate-Stage Hepatocellular Carcinoma Beyond Up-To-Seven Criteria and Child-Pugh a Liver Function: A Proof-of-Concept Study. Cancers 2019, 11, 1084. [Google Scholar] [CrossRef]
- Kudo, M.; Han, K.-H.; Ye, S.-L.; Zhou, J.; Huang, Y.-H.; Lin, S.-M.; Wang, C.-K.; Ikeda, M.; Chan, S.L.; Choo, S.P.; et al. A Changing Paradigm for the Treatment of Intermediate-Stage Hepatocellular Carcinoma: Asia-Pacific Primary Liver Cancer Expert Consensus Statements. Liver Cancer 2020, 9, 245–260. [Google Scholar] [CrossRef]
- Korean Liver Cancer Association; National Cancer Center. 2018 Korean Liver Cancer Association-National Cancer Center Korea Practice Guidelines for the Management of Hepatocellular Carcinoma. Gut Liver 2019, 13, 227–299. [Google Scholar] [CrossRef] [PubMed]
- European Association for the Study of the Liver. EASL Clinical Practice Guidelines: Management of hepatocellular carcinoma. J. Hepatol. 2018, 69, 182–236. [Google Scholar] [CrossRef] [PubMed]
- Kudo, M.; Matsui, O.; Izumi, N.; Kadoya, M.; Okusaka, T.; Miyayama, S.; Yamakado, K.; Tsuchiya, K.; Ueshima, K.; Hiraoka, A.; et al. Transarterial chemoembolization failure/refractoriness: JSH-LCSGJ criteria 2014 update. Oncology 2014, 87 (Suppl. 1), 22–31. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.S.; Kim, B.K.; Kim, S.U.; Park, J.Y.; Ahn, S.H.; Seong, J.S.; Han, K.H.; Kim, D.Y. A survey on transarterial chemoembolization refractoriness and a real-world treatment pattern for hepatocellular carcinoma in Korea. Clin. Mol. Hepatol. 2020, 26, 24–32. [Google Scholar] [CrossRef] [PubMed]
- Miyaki, D.; Aikata, H.; Kan, H.; Fujino, H.; Urabe, A.; Masaki, K.; Fukuhara, T.; Kobayashi, T.; Naeshiro, N.; Nakahara, T.; et al. Clinical outcome of sorafenib treatment in patients with advanced hepatocellular carcinoma refractory to hepatic arterial infusion chemotherapy. J. Gastroenterol. Hepatol. 2013, 28, 1834–1841. [Google Scholar] [CrossRef] [PubMed]
- Lencioni, R.; Llovet, J.M. Modified RECIST (mRECIST) assessment for hepatocellular carcinoma. Semin. Liver Dis. 2010, 30, 52–60. [Google Scholar] [CrossRef] [PubMed]
- Sung, P.S.; Park, H.L.; Yang, K.; Hwang, S.; Song, M.J.; Jang, J.W.; Choi, J.Y.; Yoon, S.K.; Yoo, I.R.; Bae, S.H. (18)F-fluorodeoxyglucose uptake of hepatocellular carcinoma as a prognostic predictor in patients with sorafenib treatment. Eur. J. Nucl. Med. Mol. Imaging 2018, 45, 384–391. [Google Scholar] [CrossRef]
- Sung, P.S.; Jang, J.W.; Lee, J.; Lee, S.K.; Lee, H.L.; Yang, H.; Nam, H.C.; Lee, S.W.; Bae, S.H.; Choi, J.Y.; et al. Real-World Outcomes of Nivolumab in Patients with Unresectable Hepatocellular Carcinoma in an Endemic Area of Hepatitis B Virus Infection. Front. Oncol. 2020, 10, 1043. [Google Scholar] [CrossRef]
- National Cancer Institute. Common Terminology Criteria for Adverse Events (CTCAE) v5.0. Updated May 27, 2017. Available online: https://ctep.cancer.gov/protocolDevelopment/electronic_applications/ctc.htm#ctc_50 (accessed on 1 October 2020).
- Lencioni, R.; Montal, R.; Torres, F.; Park, J.W.; Decaens, T.; Raoul, J.L.; Kudo, M.; Chang, C.; Rios, J.; Boige, V.; et al. Objective response by mRECIST as a predictor and potential surrogate end-point of overall survival in advanced HCC. J. Hepatol. 2017, 66, 1166–1172. [Google Scholar] [CrossRef]
- Kudo, M.; Finn, R.S.; Qin, S.; Han, K.-H.; Ikeda, K.; Cheng, A.-L.; Piscaglia, F.; Ueshima, K.; Aikata, H.; Vogel, A.; et al. Analysis of survival and objective response (OR) in patients with hepatocellular carcinoma in a phase III study of lenvatinib (REFLECT). J. Clin. Oncol. 2019, 37, 186. [Google Scholar] [CrossRef]
- Ueshima, K.; Nishida, N.; Hagiwara, S.; Aoki, T.; Minami, T.; Chishina, H.; Takita, M.; Minami, Y.; Ida, H.; Takenaka, M.; et al. Impact of Baseline ALBI Grade on the Outcomes of Hepatocellular Carcinoma Patients Treated with Lenvatinib: A Multicenter Study. Cancers 2019, 11, 952. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, T.; Kudo, M.; Ikeda, K.; Izumi, N.; Tateishi, R.; Ikeda, M.; Aikata, H.; Kawaguchi, Y.; Wada, Y.; Numata, K.; et al. REFLECT-a phase 3 trial comparing efficacy and safety of lenvatinib to sorafenib for the treatment of unresectable hepatocellular carcinoma: An analysis of Japanese subset. J. Gastroenterol. 2020, 55, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Matsui, J.; Funahashi, Y.; Uenaka, T.; Watanabe, T.; Tsuruoka, A.; Asada, M. Multi-kinase inhibitor E7080 suppresses lymph node and lung metastases of human mammary breast tumor MDA-MB-231 via inhibition of vascular endothelial growth factor-receptor (VEGF-R) 2 and VEGF-R3 kinase. Clin. Cancer Res. 2008, 14, 5459–5465. [Google Scholar] [CrossRef] [PubMed]
- Matsui, J.; Yamamoto, Y.; Funahashi, Y.; Tsuruoka, A.; Watanabe, T.; Wakabayashi, T.; Uenaka, T.; Asada, M. E7080, a novel inhibitor that targets multiple kinases, has potent antitumor activities against stem cell factor producing human small cell lung cancer H146, based on angiogenesis inhibition. Int. J. Cancer 2008, 122, 664–671. [Google Scholar] [CrossRef]
- Hoshi, T.; Watanabe Miyano, S.; Watanabe, H.; Sonobe, R.M.K.; Seki, Y.; Ohta, E.; Nomoto, K.; Matsui, J.; Funahashi, Y. Lenvatinib induces death of human hepatocellular carcinoma cells harboring an activated FGF signaling pathway through inhibition of FGFR-MAPK cascades. Biochem. Biophys. Res. Commun. 2019, 513, 1–7. [Google Scholar] [CrossRef]
- Shimose, S.; Kawaguchi, T.; Tanaka, M.; Iwamoto, H.; Miyazaki, K.; Moriyama, E.; Suzuki, H.; Niizeki, T.; Shirono, T.; Nakano, M.; et al. Lenvatinib prolongs the progression-free survival time of patients with intermediate-stage hepatocellular carcinoma refractory to transarterial chemoembolization: A multicenter cohort study using data mining analysis. Oncol. Lett. 2020, 20, 2257–2265. [Google Scholar] [CrossRef]
- Finn, R.S.; Ikeda, M.; Zhu, A.X.; Sung, M.W.; Baron, A.D.; Kudo, M.; Okusaka, T.; Kobayashi, M.; Kumada, H.; Kaneko, S.; et al. Phase 1b study of lenvatinib plus pembrolizumab in patients with unresectable hepatocellular carcinoma. J. Clin. Oncol. 2020. [Google Scholar] [CrossRef]
- Cheng, A.L.; Hsu, C.; Chan, S.L.; Choo, S.P.; Kudo, M. Challenges of combination therapy with immune checkpoint inhibitors for hepatocellular carcinoma. J. Hepatol. 2020, 72, 307–319. [Google Scholar] [CrossRef]
- Finn, R.S.; Qin, S.; Ikeda, M.; Galle, P.R.; Ducreux, M.; Kim, T.Y.; Kudo, M.; Breder, V.; Merle, P.; Kaseb, A.O.; et al. Atezolizumab plus Bevacizumab in Unresectable Hepatocellular Carcinoma. N. Engl. J. Med. 2020, 382, 1894–1905. [Google Scholar] [CrossRef]
- Casadei Gardini, A.; Puzzoni, M.; Montagnani, F.; Marisi, G.; Tamburini, E.; Cucchetti, A.; Solaini, L.; Foschi, F.G.; Conti, F.; Ercolani, G.; et al. Profile of lenvatinib in the treatment of hepatocellular carcinoma: Design, development, potential place in therapy and network meta-analysis of hepatitis B and hepatitis C in all Phase III trials. Oncol. Targets Ther. 2019, 12, 2981–2988. [Google Scholar] [CrossRef]
- Kawamura, Y.; Kobayashi, M.; Shindoh, J.; Kobayashi, Y.; Kasuya, K.; Sano, T.; Fujiyama, S.; Hosaka, T.; Saitoh, S.; Sezaki, H.; et al. 18 F-Fluorodeoxyglucose Uptake in Hepatocellular Carcinoma as a Useful Predictor of an Extremely Rapid Response to Lenvatinib. Liver Cancer 2020, 9, 84–92. [Google Scholar] [CrossRef] [PubMed]

| Parameters | Lenvatinib (N = 43) | Sorafenib (N = 55) | Total (N = 98) | p |
|---|---|---|---|---|
| Epidemiology | ||||
| Sex, M/F (%) | 35 (81.4)/8 (18.6) | 42 (76.4)/13 (23.6) | 77 (78.6)/21 (21.4) | 0.723 |
| Age, median (range) | 60 (32–85) | 63 (43–86) | 62.5 (32–86) | 0.451 |
| Etiology | 0.881 | |||
| HBV, n (%) | 31 (72.1) | 42 (76.4) | 73 (74.5) | |
| HCV, n (%) | 3 (7.0) | 2 (3.6) | 5 (5.1) | |
| Alcohol, n (%) | 7 (16.3) | 8 (14.5) | 15 (15.3) | |
| Others, n (%) | 2 (4.7) | 3 (5.5) | 5 (5.1) | |
| Child-Pugh score | 0.289 | |||
| 5, n (%) | 24 (55.8) | 37 (67.3) | 61 (62.2) | |
| 6, n (%) | 13 (30.2) | 15 (27.3) | 28 (28.6) | |
| 7, n (%) | 6 (14.0) | 3 (5.5) | 9 (9.2) | |
| BCLC stage | 0.792 | |||
| A, n (%) | 0 (0) | 0 (0) | 0 (0) | |
| B, n (%) | 8 (18.6) | 8 (14.5) | 16 (16.3) | |
| C, n (%) | 35 (81.4) | 47 (85.5) | 82 (83.7) | |
| D, n (%) | 0 (0) | 0 (0) | 0 (0) | |
| ECOG | 0.942 | |||
| 0, n (%) | 16 (37.2) | 22 (40.0) | 38 (38.8) | |
| 1, n (%) | 27 (62.8) | 33 (60.0) | 60 (61.2) | |
| AFP, median (range), ng/mL | 278.9 (1.4–115807) | 708.8 (1.3–512682) | 647.4 (1.3–512682) | 0.463 |
| PIVKA-II, median(range), mAU/mL | 806 (11–300000) | 532.3 (14–52576) | 638.3 (11–300000) | 0.020 |
| Macrovascular invasion, n (%) | 19 (44.2) | 25 (45.5) | 44 (44.9) | 1.000 |
| Extrahepatic metastasis, n (%) | 24 (55.8) | 39 (70.9) | 63 (64.3) | 0.182 |
| Previous treatments | ||||
| TACE, n (%) | 40 (93.0) | 54 (98.2) | 94 (95.9) | |
| HAIC, n (%) | 8 (20.9) | 4 (7.3) | 12 (12.2) | |
| Radiation therapy, n (%) | 13 (30.2) | 14 (25.5) | 27 (27.6) | |
| Surgical resection, n (%) | 5 (11.6) | 9 (16.4) | 14 (14.3) | |
| Radiofrequency ablation, n (%) | 5 (11.6) | 8 (14.5) | 13 (13.3) | |
| Systemic chemotherapy, n (%) | 5 (11.6) | 1 (1.8) | 6 (6.1) |
| Treatment Response | Lenvatinib, n (%) | Sorafenib, n (%) | p |
|---|---|---|---|
| CR | 1 (2.3) | 0 (0) | |
| PR | 10 (23.2) | 2 (3.6) | |
| SD | 14 (32.6) | 11 (20.0) | |
| PD | 14 (32.6) | 35 (63.6) | |
| NA (Not Assessed) | 4 (9.3) | 7 (12.7) | |
| ORR | 25.6 | 3.6 | 0.002 |
| DCR | 58.1 | 23.6 | 0.001 |
| Adverse Event | Lenvatinib, n (%) | Sorafenib, n (%) | p |
|---|---|---|---|
| AE grade ≥3 | 17 | 18 | 0.529 |
| HFSR | 4 (9.3) | 8 (14.5) | |
| Proteinuria | 3 (7.0) | 0 (0) | |
| Hyperbilirubinemia | 1 (2.3) | 1 (1.8) | |
| Hepatic encephalopathy | 2 (4.7) | 2 (3.6) | |
| Diarrhea | 2 (4.7) | 2 (3.6) | |
| Hypertension | 4 (9.3) | 0 (0) | |
| Decreased appetite | 1 (2.3) | 3 (5.5) | |
| Elevated aspartate aminotransferase level | 0 (0) | 1 (1.8) |
| Univariate Analysis | Multivariate Analysis | |||||
|---|---|---|---|---|---|---|
| HR | 95% CI | p | HR | 95% CI | p | |
| LEN vs SOR | 0.399 | 0.230–0.692 | 0.001 | 0.359 | 0.203–0.635 | <0.001 |
| Age (< 60 vs ≥ 60) | 1.186 | 0.714–1.969 | 0.509 | |||
| ECOG (0 vs 1) | 0.839 | 0.500–1.407 | 0.506 | |||
| HBsAg positivity | 1.573 | 0.850–2.911 | 0.149 | 1.445 | 0.770–2.710 | 0.252 |
| Tumor size ( | 0.928 | 0.557–1.547 | 0.928 | |||
| ALBI grade 1 | 0.576 | 0.336–0.987 | 0.045 | 0.612 | 0.353–1.061 | 0.080 |
| 0.594 | 0.361–0.979 | 0.041 | 0.719 | 0.406–1.272 | 0.257 | |
| PIVKA-II (mAU/mL) | 0.592 | 0.319–1.098 | 0.097 | 0.702 | 0.346–1.426 | 0.328 |
| Child Pugh score (5) | 0.751 | 0.452–1.249 | 0.270 | |||
| Macrovascular invasion | 1.664 | 1.005–2.755 | 0.048 | 1.302 | 0.729–2.327 | 0.372 |
| Extrahepatic metastasis | 0.932 | 0.577–1.655 | 0.977 | |||
| Univariate Analysis | Multivariate Analysis | |||||
|---|---|---|---|---|---|---|
| Model 1 | Model 2 | |||||
| HR (95% CI) | p | HR (95% CI) | p | HR (95% CI) | p | |
| Objective Response | 0.488 (0.192–1.238) | 0.131 | 0.356 (0.132–0.957) | 0.041 | ||
| LEN vs SOR | 1.166 (0.629–2.160) | 0.625 | 0.771(0.390–1.524) | 0.454 | ||
| Age (<60 vs ≥60) | 1.416 (0.782–2.552) | 0.247 | ||||
| ECOG (0 vs 1) | 0.570 (0.305–1.065) | 0.078 | 1.488 (0.678–3.264) | 0.322 | 1.327 (0.599–2.938) | 0.486 |
| HBsAg positivity | 0.776 (0.412–1.462) | 0.432 | ||||
| 0.423 (0.232–0.771) | 0.005 | 0.557(0.292–1.065) | 0.077 | 0.562 (0.289–1.091) | 0.088 | |
| ALBI grade 1 | 0.425 (0.215–0.838) | 0.013 | 0.422 (0.186–0.959) | 0.039 | 0.408 (0.179–0.930) | 0.033 |
| AFP (ng/mL) | 0.447 (0.248–0.808) | 0.008 | 0.668 (0.342–1.305) | 0.238 | 0.629 (0.318–1.246) | 0.184 |
| PIVKA-II (mAU/mL) | 0.228 (0.088–0.592) | 0.002 | 0.336 (0.113–1.003) | 0.051 | 0.336 (0.113–0.998) | 0.050 |
| Child Pugh score (5) | 0.553 (0.310–0.988) | 0.045 | 0.877 (0.412–1.867) | 0.877 | 0.940 (0.432–2.046) | 0.877 |
| Macrovascular invasion | 2.365 (1.299–4.304) | 0.005 | 1.950 (0.960–3.963) | 0.065 | 1.546 (0.788–3.034) | 0.205 |
| Extrahepatic metastasis | 0.949 (0.517–1.742) | 0.865 | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, J.; Sung, P.S.; Yang, H.; Lee, S.K.; Nam, H.C.; Yoo, S.H.; Lee, H.L.; Kim, H.Y.; Lee, S.W.; Kwon, J.H.; et al. A Real-World Comparative Analysis of Lenvatinib and Sorafenib as a Salvage Therapy for Transarterial Treatments in Unresectable HCC. J. Clin. Med. 2020, 9, 4121. https://doi.org/10.3390/jcm9124121
Lee J, Sung PS, Yang H, Lee SK, Nam HC, Yoo SH, Lee HL, Kim HY, Lee SW, Kwon JH, et al. A Real-World Comparative Analysis of Lenvatinib and Sorafenib as a Salvage Therapy for Transarterial Treatments in Unresectable HCC. Journal of Clinical Medicine. 2020; 9(12):4121. https://doi.org/10.3390/jcm9124121
Chicago/Turabian StyleLee, Jaejun, Pil Soo Sung, Hyun Yang, Soon Kyu Lee, Hee Chul Nam, Sun Hong Yoo, Hae Lim Lee, Hee Yeon Kim, Sung Won Lee, Jung Hyun Kwon, and et al. 2020. "A Real-World Comparative Analysis of Lenvatinib and Sorafenib as a Salvage Therapy for Transarterial Treatments in Unresectable HCC" Journal of Clinical Medicine 9, no. 12: 4121. https://doi.org/10.3390/jcm9124121
APA StyleLee, J., Sung, P. S., Yang, H., Lee, S. K., Nam, H. C., Yoo, S. H., Lee, H. L., Kim, H. Y., Lee, S. W., Kwon, J. H., Jang, J. W., Kim, C. W., Nam, S. W., Bae, S. H., Choi, J. Y., & Yoon, S. K. (2020). A Real-World Comparative Analysis of Lenvatinib and Sorafenib as a Salvage Therapy for Transarterial Treatments in Unresectable HCC. Journal of Clinical Medicine, 9(12), 4121. https://doi.org/10.3390/jcm9124121






