Association of Primary Sjögren’s Syndrome and Vitamin B12 Deficiency: A Cross-Sectional Case-Control Study
Abstract
1. Introduction
2. Methods
2.1. Ethics
2.2. Study Population
2.3. Definitions of Primary Sjögren’s Syndrome and Nutrient Deficiencies
2.4. Data Collection
2.5. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Green, R.; Allen, L.H.; Bjørke-Monsen, A.-L.; Brito, A.; Guéant, J.-L.; Miller, J.W.; Molloy, A.M.; Nexo, E.; Stabler, S.; Toh, B.-H.; et al. Vitamin B12 deficiency. Nat. Rev. Dis. Primers 2017, 3. [Google Scholar] [CrossRef] [PubMed]
- Meziere, A.; Audureau, E.; Vairelles, S.; Krypciak, S.; Dicko, M.; Monie, M.; Giraudier, S. B12 Deficiency Increases With Age in Hospitalized Patients: A Study on 14,904 Samples. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2014, 69, 1576–1585. [Google Scholar] [CrossRef] [PubMed]
- Dali-Youcef, N.; Andres, E. An update on cobalamin deficiency in adults. QJM 2009, 102, 17–28. [Google Scholar] [CrossRef] [PubMed]
- Serraj, K.; Vogel, T.; Federici, L.; Ciobanu, E.; Mecili, M.; Kaltenbach, G.; Andrès, E. Food-cobalamin syndrome. La Presse Médicale 2009, 38, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Andrès, E.; Perrin, A.E.; Kraemer, J.P.; Goichot, B.; Demengeat, C.; Ruellan, A.; Grunenberger, F.; Constantinesco, A.; Schlienger, J.L. Anemia caused by vitamin B 12 deficiency in subjects aged over 75 years: New hypotheses. A study of 20 cases. La Revue de Médecine Interne 2000, 21, 946–954. [Google Scholar] [CrossRef]
- Andrès, E. Sjogren’s syndrome: A potential new aetiology of mild cobalamin deficiency. Rheumatology 2001, 40, 1196–1197. [Google Scholar] [CrossRef]
- Segal, B.; Thomas, W.; Rogers, T.; Leon, J.M.; Hughes, P.; Patel, D.; Patel, K.; Novitzke, J.; Rohrer, M.; Gopalakrishnan, R.; et al. Prevalence, severity, and predictors of fatigue in subjects with primary Sjögren’s syndrome. Arthritis Rheum. 2008, 59, 1780–1787. [Google Scholar] [CrossRef]
- Lacout, C.; Cassereau, J.; Lozac’h, P.; Gury, A.; Ghali, A.; Lavigne, C.; Letournel, F.; Urbanski, G. Differences in clinical features between small fiber and sensitive large fiber neuropathies in Sjögren’s syndrome. Eur. J. Intern. Med. 2020, 79, 58–62. [Google Scholar] [CrossRef]
- Lundström, I.; Lindström, F. Iron and vitamin deficiencies, endocrine and immune status in patients with primary Sjögren’s syndrome. Oral Dis. 2001, 7, 144–149. [Google Scholar] [CrossRef]
- Andrès, E.; Blicklé, F.; Sordet, C.; Cohen-Solal, J.; Sibilia, J.; Sapin, R. Primary Sjögren’s Syndrome and Vitamin B12 Deficiency: Preliminary Results in 80 Patients. Am. J. Med. 2006, 119, e9–e10. [Google Scholar] [CrossRef]
- Stabler, S.P. Vitamin B 12 Deficiency. N. Engl. J. Med. 2013, 368, 149–160. [Google Scholar] [CrossRef] [PubMed]
- Hannibal, L.; Lysne, V.; Bjørke-Monsen, A.-L.; Behringer, S.; Grünert, S.C.; Spiekerkoetter, U.; Jacobsen, D.W.; Blom, H.J. Biomarkers and Algorithms for the Diagnosis of Vitamin B12 Deficiency. Front. Mol. Biosci. 2016, 3. [Google Scholar] [CrossRef] [PubMed]
- Fedosov, S.N. Biochemical markers of vitamin B12 deficiency combined in one diagnostic parameter: The age-dependence and association with cognitive function and blood hemoglobin. Clin. Chim. Acta 2013, 422, 47–53. [Google Scholar] [CrossRef]
- Shiboski, C.H.; Shiboski, S.C.; Seror, R.; Criswell, L.A.; Labetoulle, M.; Lietman, T.M.; Rasmussen, A.; Scofield, H.; Vitali, C.; Bowman, S.J.; et al. 2016 American College of Rheumatology/European League Against Rheumatism Classification Criteria for Primary Sjögren’s Syndrome: A Consensus and Data-Driven Methodology Involving Three International Patient Cohorts: ACR/EULAR CLASSIFICATION CRITERIA FOR PRIMARY SS. Arthritis Rheumatol. 2017, 69, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Carmel, R. How I treat cobalamin (vitamin B12) deficiency. Blood 2008, 112, 2214–2221. [Google Scholar] [CrossRef]
- Clarke, R.; Refsum, H.; Birks, J.; Evans, J.G.; Johnston, C.; Sherliker, P.; Ueland, P.M.; Schneede, J.; McPartlin, J.; Nexo, E.; et al. Screening for vitamin B-12 and folate deficiency in older persons. Am. J. Clin. Nutr. 2003, 77, 1241–1247. [Google Scholar] [CrossRef] [PubMed]
- Miller, J.W.; Garrod, M.G.; Rockwood, A.L.; Kushnir, M.M.; Allen, L.H.; Haan, M.N.; Green, R. Measurement of Total Vitamin B12 and Holotranscobalamin, Singly and in Combination, in Screening for Metabolic Vitamin B12 Deficiency. Clin. Chem. 2006, 52, 278–285. [Google Scholar] [CrossRef] [PubMed]
- de Benoist, B. Conclusions of a WHO Technical Consultation on Folate and Vitamin B 12 Deficiencies. Food Nutr. Bull. 2008, 29, S238–S244. [Google Scholar] [CrossRef]
- Dignass, A.U.; Gasche, C.; Bettenworth, D.; Birgegård, G.; Danese, S.; Gisbert, J.P.; Gomollon, F.; Iqbal, T.; Katsanos, K.; Koutroubakis, I.; et al. European Consensus on the Diagnosis and Management of Iron Deficiency and Anaemia in Inflammatory Bowel Diseases. J. Crohn’s Colitis 2015, 9, 211–222. [Google Scholar] [CrossRef]
- Supakul, S.; Chabrun, F.; Genebrier, S.; N’Guyen, M.; Valarche, G.; Derieppe, A.; Villoteau, A.; Lacombe, V.; Urbanski, G. Diagnostic Performances of Urinary Methylmalonic Acid/Creatinine Ratio in Vitamin B12 Deficiency. J. Clin. Med. 2020, 9, 2335. [Google Scholar] [CrossRef]
- Olson, S.R.; Deloughery, T.G.; Taylor, J.A. Time to Abandon the Serum Cobalamin Level for Diagnosing Vitamin B12 Deficiency. Blood 2016, 128, 2447. [Google Scholar] [CrossRef]
- Ebert, E.C. Gastrointestinal and Hepatic Manifestations of Sjogren Syndrome. J. Clin. Gastroenterol. 2012, 46, 25–30. [Google Scholar] [CrossRef]
- Rodriguez-Cuartero, A.; Perez-Blanco, F.J.; Urbano-Jimenez, F. Sjögren’s syndrome and pernicious anaemia: Case report. Scand. J. Rheumatol. 1998, 27, 83–85. [Google Scholar] [CrossRef]
- Bowman, S.J.; Everett, C.C.; O’Dwyer, J.L.; Emery, P.; Pitzalis, C.; Ng, W.-F.; Pease, C.T.; Price, E.J.; Sutcliffe, N.; Gendi, N.S.T.; et al. Randomized Controlled Trial of Rituximab and Cost-Effectiveness Analysis in Treating Fatigue and Oral Dryness in Primary Sjögren’s Syndrome: RITUXIMAB FOR SYMPTOMATIC FATIGUE AND ORAL DRYNESS IN PRIMARY SS. Arthritis Rheumatol. 2017, 69, 1440–1450. [Google Scholar] [CrossRef]
- Luo, J.; Xu, S.; Lv, Y.; Huang, X.; Zhang, H.; Zhu, X.; Wang, X. Clinical features and potential relevant factors of renal involvement in primary Sjögren’s syndrome. Int. J. Rheum. Dis. 2019, 22, 182–190. [Google Scholar] [CrossRef]
- Brito-Zerón, P.; Acar-Denizli, N.; Ng, W.-F.; Zeher, M.; Rasmussen, A.; Mandl, T.; Seror, R.; Li, X.; Baldini, C.; Gottenberg, J.-E.; et al. How immunological profile drives clinical phenotype of primary Sjögren’s syndrome at diagnosis: Analysis of 10,500 patients (Sjögren Big Data Project). Clin. Exp. Rheumatol. 2018, 36 (Suppl. 112), 102–112. [Google Scholar]
| pSS Patients | Controls | |
|---|---|---|
| Number of patients | 21 | 105 |
| Age (years) | 70 (51–75) | 70 (51–77) |
| Women | 20 (95.2%) | 100 (95.2%) |
| Body mass index (kg/m²) | 25.5 (22.3–29.4) | 25.9 (21.8–30.4) |
| pSS duration at time of B12 measurement (months) | 12 (2–46) | |
| Schirmer’s test ≤ 5 mm/5 min | 13 (61.9%) | - |
| Unstimulated whole saliva flow ≤1.5 mL/15 min | 12 (57.1%) | - |
| Lymphocytic sialadenitis with focus score ≥ 1 | 20 (95.2%) | - |
| Anti-SSA antibodies | 7 (33.3%) | - |
| Anti-SSB antibodies | 5 (23.8%) | - |
| ANA positivity | 19 (90.5%) | - |
| ANA ≥ 1/320 | 14 (66.7%) | - |
| Rheumatoid factor | 12 (57.1%) | - |
| Age at pSS diagnosis (years) | 64 (49–71) | - |
| Extra-glandular manifestations | 15 (71.4%) | - |
| Inflammatory arthralgia | 7 (33.3%) | - |
| Polyarthritis | 1 (4.8%) | - |
| Parotidomegaly | 4 (19.0%) | - |
| Serositis | 1 (4.8%) | - |
| Large fiber neuropathy | 3 (14.3%) | - |
| Small fiber neuropathy | 2 (9.5%) | - |
| CNS involvement * | 2 (9.5%) | - |
| Pulmonary interstitial disease | 2 (9.5%) | - |
| Lymphoma | 1 (4.8%) | - |
| pSS | Controls | p Value | |
|---|---|---|---|
| Number of patients | 21 | 105 | |
| Vitamin B12 status | |||
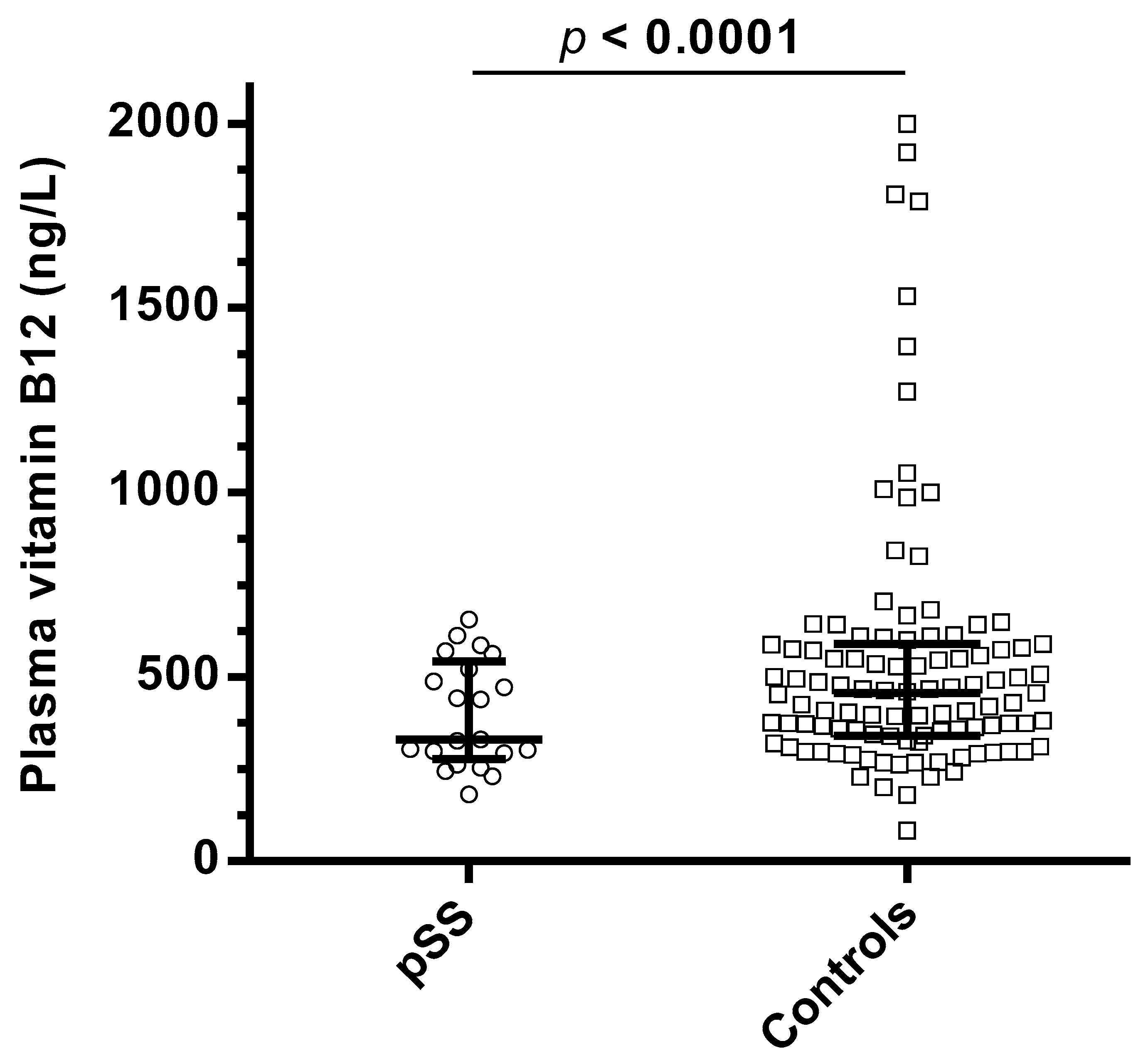
| Plasma vitamin B12 (ng/L, NR 350–1000) | 329 (293–521) | 456 (341–587) | <0.0001 |
| Plasma homocysteine (µmol/L *) | 17.6 (13.9–19.4) | 14.3 (10.3–17.9) | 0.07 |
| Plasma methylmalonic acid (µmol/L, NR ≤ 0.35) | 0.48 (0.20–0.56) | 0.31 (0.20–0.45) | 0.66 |
| Number of patients with B12 deficiency | 9 (42.9%) | 12 (11.4%) | <0.0001 |
| Other biological measurements | |||
| Serum folate (µg/L, NR ≥ 4) | 5.2 (4.3–7.5) | 6.2 (4.4–8.3) | 0.24 |
| Folate deficiency (<4 µg/L) | 3 (14.3%) | 16 (15.2%) | 0.86 |
| Serum ferritin level (µg/L, NR > 30) | 99 (47–151) | 142 (83–252) | <0.0001 |
| Iron deficiency | 5 (23.8%) | 13 (12.4%) | 0.02 |
| Creatinine clearance (mL/min/1.73 m²) MDRD (NR ≥ 90) | 89.5 (69.2–100.6) | 85.3 (65.2–102.8) | 0.77 |
| Serum gastrin (pg/mL, NR 28–115) | 46.3 (38.1–66.1) | 55.1 (44.2–71.3) | 0.36 |
| Other causes of B12 deficiency among patients with B12 deficiency | |||
| Metformin use | 0/9 (0%) | 2/12 (16.7%) | 0.04 |
| Chronic antacid use | 5/9 (55.6%) | 3/12 (25.0%) | 0.71 |
| Nutritional deficiency | 0/9 (0%) | 0/12 (0%) | >0.99 |
| Fundic gastric or ileum resection | 1/9 (11.1%) | 1/12 (8.3%) | 0.10 |
| Helicobacter pylori gastric infection | 0/9 (0%) | 2/12 (16.7%) | 0.04 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Urbanski, G.; Chabrun, F.; Schaepelynck, B.; May, M.; Loiseau, M.; Schlumberger, E.; Delattre, E.; Lavigne, C.; Lacombe, V. Association of Primary Sjögren’s Syndrome and Vitamin B12 Deficiency: A Cross-Sectional Case-Control Study. J. Clin. Med. 2020, 9, 4063. https://doi.org/10.3390/jcm9124063
Urbanski G, Chabrun F, Schaepelynck B, May M, Loiseau M, Schlumberger E, Delattre E, Lavigne C, Lacombe V. Association of Primary Sjögren’s Syndrome and Vitamin B12 Deficiency: A Cross-Sectional Case-Control Study. Journal of Clinical Medicine. 2020; 9(12):4063. https://doi.org/10.3390/jcm9124063
Chicago/Turabian StyleUrbanski, Geoffrey, Floris Chabrun, Baudouin Schaepelynck, Morgane May, Marianne Loiseau, Esther Schlumberger, Estelle Delattre, Christian Lavigne, and Valentin Lacombe. 2020. "Association of Primary Sjögren’s Syndrome and Vitamin B12 Deficiency: A Cross-Sectional Case-Control Study" Journal of Clinical Medicine 9, no. 12: 4063. https://doi.org/10.3390/jcm9124063
APA StyleUrbanski, G., Chabrun, F., Schaepelynck, B., May, M., Loiseau, M., Schlumberger, E., Delattre, E., Lavigne, C., & Lacombe, V. (2020). Association of Primary Sjögren’s Syndrome and Vitamin B12 Deficiency: A Cross-Sectional Case-Control Study. Journal of Clinical Medicine, 9(12), 4063. https://doi.org/10.3390/jcm9124063







