Oral Manifestations of Wolf-Hirschhorn Syndrome: Genotype-Phenotype Correlation Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient Selection
2.2. Single Nucleotide Polymorphism (SNP) Array Analysis
2.3. Multiplex Ligation-Dependent Probe Amplification
2.4. Clinical and Radiological Oral Examination
2.5. Statistical Analysis
3. Results
3.1. Clinical and Radiological Oral Examination
3.2. Correlations between Systemic Findings, Genetic Variables, and Oral Manifestations
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Correction Statement
References
- Battaglia, A.; South, S.; Carey, J.C. Clinical utility gene card for: Wolf-Hirschhorn (4p-) syndrome. Eur. J. Hum. Genet. 2011, 19, 492. [Google Scholar] [CrossRef]
- Maas, N.M.; Van Vooren, S.; Hannes, F.; Van Buggenhout, G.; Mysliwiec, M.; Moreau, Y.; Fagan, K.; Midro, A.; Engiz, O.; Balci, S.; et al. The t(4;8) is mediated by homologous recombination between olfactory receptor gene clusters, but other 4p16 translocations occur at random. Genet. Couns. 2007, 18, 357–365. [Google Scholar] [PubMed]
- Zollino, M.; Murdolo, M.; Marangi, G.; Pecile, V.; Galasso, C.; Mazzanti, L.; Neri, G. On the nosology and pathogenesis of Wolf–Hirschhorn Syndrome: Genotype-phenotype correlation analysis of 80 patients and literature review. Am. J. Med. Genet. Part C 2008, 148C, 257–269. [Google Scholar] [CrossRef] [PubMed]
- Blanco-Lago, R.; Malaga-Dieguez, I.; Granizo-Martinez, J.J.; Carrera-Garcia, L.; Barruz-Galian, P.; Lapunzina, P.; Nevado-Blanco, J. En Representacion Del Grupo Colaborativo Para El Estudio Del Sindrome de Wolf-Hirschhorn ERDGCPEEDSW. Wolf-Hirschhorn syndrome. Description of a Spanish cohort of 51 cases and a literature review. Rev. Neurol. 2017, 64, 393–400. [Google Scholar] [PubMed]
- Nevado, J.; Ho, K.S.; Zollino, M.; Blanco, R.; Cobaleda, C.; Golzio, C.; Beaudry-Bellefeuille, I.; Berrocoso, S.; Limeres, J.; Barrúz, P.; et al. International meeting on Wolf-Hirschhorn syndrome: Update on the nosology and new insights on the pathogenic mechanisms for seizures and growth delay. Am. J. Med. Genet. A 2020, 182, 257–267. [Google Scholar] [CrossRef] [PubMed]
- Lurie, I.W.; Lazjuk, G.I.; Ussova, Y.I.; Presman, E.B.; Gurevich, D.B. TheWolf- Hirschhorn syndrome. I. Genetics. Clin. Genet. 1980, 17, 375–384. [Google Scholar] [CrossRef]
- Battaglia, A.; Carey, J.C.; South, S.T. Wolf-Hirschhorn syndrome: A review and update. Am. J. Med. Genet. C. Semin. Med. Gent. 2015, 169C, 216–223. [Google Scholar] [CrossRef]
- Hammond, P.; Hannes, F.; Suttie, M.; Devriendt, K.; Vermeesch, J.R.; Faravelli, F.; Forzano, F.; Parekh, S.; Williams, S.; McMullan, D.; et al. Fine-grained facial phenotype-genotype analysis in Wolf-Hirschhorn syndrome. Eur. J. Hum. Genet. 2012, 20, 33–40. [Google Scholar] [CrossRef]
- Zollino, M.; Di Stefano, C.; Zampino, G.; Mastroiacovo, P.; Wright, T.J.; Sorge, G.; Selicorni, A.; Tenconi, R.; Zappalà, A.; Battaglia, A.; et al. Genotype-phenotype correlations and clinical diagnostic criteria in Wolf-Hirschhorn syndrome. Am. J. Med. Genet. 2000, 94, 254–261. [Google Scholar] [CrossRef]
- Andersen, E.F.; Carey, J.C.; Earl, D.L.; Corzo, D.; Suttie, M.; Hammond, P.; South, S.T. Deletions involving genes WHSC1 and LETM1 may be necessary, but are not sufficient to cause Wolf–Hirschhorn Syndrome. Eur. J. Hum. Genet. 2013, 22, 464–470. [Google Scholar] [CrossRef]
- Maas, N.M.C.; Van Buggenhout, G.; Hannes, F.; Thienpont, B.; Sanlaville, D.; Kok, K.; Midro, A.; Andrieux, J.; Anderlid, B.M.; Schoumans, J.; et al. Genotype-phenotype correlation in 21 patients with Wolf-Hirschhorn syndrome using high resolution array comparative genome hybridisation (CGH). J. Med. Genet. 2008, 45, 71–80. [Google Scholar] [CrossRef] [PubMed]
- Catela, C.; Bilbao-Cortes, D.; Slonimsky, E.; Kratsios, P.; Rosenthal, N.; te Welscher, P. Multiple congenital malformations of Wolf–Hirschhorn syndrome are recapitulated in Fgfrl1 null mice. Dis. Model. Mech. 2009, 2, 283–294. [Google Scholar] [CrossRef] [PubMed]
- Boczek, N.J.; Lahner, C.A.; Nguyen, T.M.; Ferber, M.J.; Hasadsri, L.; Thorland, E.C.; Niu, Z.; Gavrilova, R.H. Developmental delay and failure to thrive associated with a loss-of-function variant in WHSC1 (NSD2). Am. J. Med. Genet. A 2018, 176, 2798–2802. [Google Scholar] [CrossRef] [PubMed]
- Barrie, E.S.; Alfaro, M.P.; Pfau, R.B.; Goff, M.J.; McBride, K.L.; Manickam, K.; Zmuda, E.J. De novo loss-of-function variants in NSD2 ( WHSC1) associate with a subset of Wolf-Hirschhorn syndrome. Cold Spring Harb. Mol. Case Stud. 2019, 5, a004044. [Google Scholar] [CrossRef]
- Yamamoto-Shimojima, K.; Kouwaki, M.; Kawashima, Y.; Itomi, K.; Momosaki, K.; Ozasa, S.; Okamoto, N.; Yokochi, K.; Yamamoto, T. Natural histories of patients with Wolf-Hirschhorn syndrome derived from variable chromosomal abnormalities. Congenit. Anom. (Kyoto). 2019, 59, 169–173. [Google Scholar] [CrossRef]
- Paradowska-Stolarz, A.M. Wolf-Hirschhorn Syndrome (WHS)—Literature review on the features of the syndrome. Adv. Clin. Exp. Med. 2014, 23, 485–489. [Google Scholar] [CrossRef]
- Battaglia, A.; Filippi, T.; Carey, J.C. Update on the clinical features and natural history of Wolf-Hirschhorn (4p-) syndrome: Experience with 87 patients and recommendations for routine health supervision. Am. J. Med. Genet. Part C Semin. Med. Genet. 2008, 148, 246–251. [Google Scholar] [CrossRef]
- Morishita, M.; Shiba, R.; Chiyo, H.; Furuyama, J.; Fujita, H.; Atsumi, Y. The oral manifestations of 4p- syndrome. J. Oral Maxillofac. Surg. 1983, 41, 601–605. [Google Scholar] [CrossRef]
- Nieminen, P.; Kotilainen, J.; Aalto, Y.; Knuutila, S.; Pirinen, S.; Thesleff, I. MSX1 gene is deleted in Wolf-Hirschhorn syndrome patients with oligodontia. J. Dent. Res. 2003, 82, 1013–1017. [Google Scholar] [CrossRef]
- Blanco-Lago, R.; Málaga, I.; García-Peñas, J.J.; García-Ron, A. Wolf-Hirschhorn syndrome. A series of 27 patients: Their epidemiological and clinical characteristics. The current situation of the patients and the opinions of their caregivers regarding the diagnostic process. Rev. Neurol. 2013, 57, 49–56. [Google Scholar]
- Chaudhry, C.; Kaur, A.; Panigrahi, I.; Kaur, A. Wolf–Hirschhorn syndrome: A case series from India. Am. J. Med. Genet. Part A. 2020, 1–4. [Google Scholar] [CrossRef]
- Stewart, D.R.; Kleefstra, T. The chromosome 9q subtelomere deletion syndrome. Am. J. Med. Genet. C. Semin. Med. Genet. 2007, 145C, 383–392. [Google Scholar] [CrossRef]
- Johnson, V.P.; Mulder, R.D.; Hosen, R. The Wolf-Hirschhorn (4p-) syndrome. Clin. Genet. 1976, 10, 104–112. [Google Scholar] [CrossRef]
- Mintz, S.M.; Siegel, M.A.; Seider, P.J. An overview of oral frena and their association with multiple syndromic and nonsyndromic conditions. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2005, 99, 321–324. [Google Scholar] [CrossRef]
- Lauritano, D.; Attuati, S.; Besana, M.; Rodilosso, G.; Quinzi, V.; Marzo, G.; Carinci, F. Oral and craniofacial manifestations of Ellis-Van Creveld syndrome: A systematic review. Eur. J. Paediatr. Dent. 2019, 20, 306–310. [Google Scholar] [PubMed]
- Wester, U.; Bondeson, M.L.; Edeby, C.; Annerén, G. Clinical and molecular characterization of individuals with 18p deletion: A genotype-phenotype correlation. Am. J. Med. Genet. A 2006, 140, 1164–1171. [Google Scholar] [CrossRef]
- Karempelis, P.; Hagen, M.; Morrell, N.; Roby, B.B. Associated syndromes in patients with Pierre Robin Sequence. Int. J. Pediatr. Otorhinolaryngol. 2020, 131, 109842. [Google Scholar] [CrossRef]
- Gomez-Ospina, N.; Bernstein, J.A. Clinical, cytogenetic, and molecular outcomes in a series of 66 patients with Pierre Robin sequence and literature review: 22q11.2 deletion is less common than other chromosomal anomalies. Am. J. Med. Genet. A 2016, 170A, 870–880. [Google Scholar] [CrossRef]
- Paradowska-Stolarz, A. MSX1 gene in the etiology of orofacial deformities. Postepy Hig. Med. Dosw. (Online). 2015, 69, 1499–1504. [Google Scholar]
- Miller, O.J.; Breg, W.R.; Warburton, D.; Miller, D.A.; DeCapoa, A.; Allderdice, P.W.; Davis, J.; Klinger, H.P.; McGilvray, E.; Allen, F.H., Jr. Partial deletion of the short arm of chromosome no. 4(4p-): Clinical studies in five unrelated patients. J. Pediatr. 1970, 77, 792–801. [Google Scholar] [CrossRef]
- Klingberg, G.; Oskarsdóttir, S.; Johannesson, E.L.; Norén, J.G. Oral manifestations in 22q11 deletion syndrome. Int. J. Paediatr. Dent. 2002, 12, 14–23. [Google Scholar] [PubMed]
- Breen, G.H. Taurodontism, an unreported dental finding in Wolf-Hirschhorn (4p-) syndrome. ASDC J. Dent. Child. 1998, 65, 344–355. [Google Scholar]
- Johnston, N.J.; Franklin, D.L. Dental findings of a child with Wolf-Hirschhorn syndrome. Int. J. Paediatr. Dent. 2006, 16, 139–142. [Google Scholar] [CrossRef] [PubMed]
- Dellavia, C.; Raiteri, S.; Ottolina, P.; Pregliasco, F. Oral features in five adult patients with Wolf-Hirschhorn syndrome. Minerva Stomatol. 2011, 60, 391–402. [Google Scholar] [PubMed]
- Castro, T.; de Paula Martins Saints, C.; de Oliveira Lira Ortega, A.; Gallottini, M. Oral characteristics and medical considered in the dental treatment of individual with Williams syndrome. Spec. Care Dentist. 2019, 39, 108–113. [Google Scholar] [CrossRef]
- Hanley-Lopez, J.; Estabrooks, L.L.; Stiehm, R. Antibody deficiency in Wolf-Hirschhorn syndrome. J. Pediatr. 1988, 133, 141–143. [Google Scholar] [CrossRef]
- Campos-Sanchez, E.; Deleyto-Seldas, N.; Dominguez, V.; Carrillo-de-Santa-Pau, E.; Ura, K.; Rocha, P.P.; Kim, J.H.; Aljoufi, A.; Esteve-Codina, A.; Dabad, M.; et al. Wolf-Hirschhorn syndrome candidate 1 is necessary for correct hematopoietic and B cell development. Cell. Rep. 2017, 19, 1586–1601. [Google Scholar] [CrossRef]
- Centerwall, W.R.; Thompson, W.P.; Allen, I.E.; Fobes, C.D. Translocation 4p—syndrome: A general review. Am. J. Dis. Child. 1975, 129, 366–370. [Google Scholar] [CrossRef]
- Guterman, S.; Beneteau, C.; Redon, S.; Dupont, C.; Missirian, C.; Jaeger, P.; Herve, B.; Jacquin, C.; Douet-Guilbert, N.; Till, M.; et al. Prenatal findings in 1p36 deletion syndrome: New cases and a literature review. Prenat. Diagn. 2019, 39, 871–882. [Google Scholar] [CrossRef]
- Corcuera-Flores, J.R.; Casttellanos-Cosano, L.; Torres-Lagares, D.; Serrera-Figallo, M.A.; Rodríguez-Caballero, A.; Machuca-Portillo, G. A systematic review of the oral and craniofacial manifestations of cri du chat syndrome. Clin. Anat. 2016, 29, 555–560. [Google Scholar] [CrossRef]
- Ivanoff, C.; Ivanoff, A. Deletion syndrome 22q13: What the dentist should know to manage children with Phelan-McDermid syndrome effectively. J. Tenn. Dent. Assoc. 2014, 94, 15–18. [Google Scholar]
- Guo, H.; Wang, T.; Niu, X.; Wang, H.; Yang, W.; Qiu, J.; Yang, L. The risk factor related to bruxism in children: A systematic review and meta-analysis. Arch. Oral. Biol. 2018, 86, 18–34. [Google Scholar] [CrossRef]
- Oporto, G.H., 5th; Bornhardt, T.; Iturriaga, V.; Salazar, L.A. Single nucleotide polymorphisms in genes of dopaminergic pathways are associated with bruxism. Clin. Oral. Investig. 2018, 22, 331–337. [Google Scholar] [CrossRef]
- Rezazadeh, A.; Uddin, M.; Snead, O.C., 3rd; Lira, V.; Silberberg, A.; Weiss, S.; Donner, E.J.; Zak, M.; Bradbury, L.; Scherer, S.W.; et al. STXBP1 encephalopathy is associated with awake bruxism. Epilepsy Behav. 2019, 92, 121–124. [Google Scholar] [CrossRef]
- Zollino, M.; Doronzio, P.N. Dissecting the Wolf-Hirschhorn syndrome phenotype: WHSC1 is a neurodevelopmental gene contributing to growth delay, intellectual disability, and to the facial dysmorphism. J. Hum. Genet. 2018, 63, 859–861. [Google Scholar] [CrossRef] [PubMed]
- Bergemann, A.D.; Cole, F.; Hirschhorn, K. The etiology of Wolf-Hirschhorn syndrome. Trends Genet. 2005, 21, 188–195. [Google Scholar] [CrossRef]
- Phan, M.; Conte, F.; Khandelwal, K.D.; Ockeloen, C.W.; Bartzela, T.; Kleefstra, T.; van Bokhoven, H.; Rubini, M.; Zhou, H.; Carels, C.E. Tooth agenesis and orofacial clefting: Genetic brothers in arms? Hum. Genet. 2016, 135, 1299–1327. [Google Scholar] [CrossRef]
- Li, F.F.; Han, Y.; Shi, S.; Li, X.; Zhu, X.D.; Zhou, J.; Shao, Q.L.; Li, X.Q.; Liu, S.L. Characterization of transcriptional repressor gene MSX1 variations for possible associations with congenital heart diseases. PLoS ONE 2015, 10, e0142666. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Yang, W.X.; Pan, H.; Li, L.; Wu, H.R.; Wang, S.T.; Bao, X.H.; Jiang, Y.W.; Qi, Y. Analyses of genotypes and phenotypes of ten Chinese patients with Wolf-Hirschhorn syndrome by multiplex ligation-dependent probe amplification and array comparative genomic hybridization. Chin. Med. J. (Engl.) 2016, 129, 672–678. [Google Scholar] [CrossRef]
- Wieczorek, D.; Krause, M.; Majewski, F.; Albrecht, B.; Horn, D.; Riess, O.; Gillessen-Kaesbach, G. Effect of the size of the deletion and clinical manifestation in Wolf-Hirschhorn syndrome: Analysis of 13 patients with a de novo deletion. Eur. J. Hum. Genet. 2000, 8, 519–526. [Google Scholar] [CrossRef]
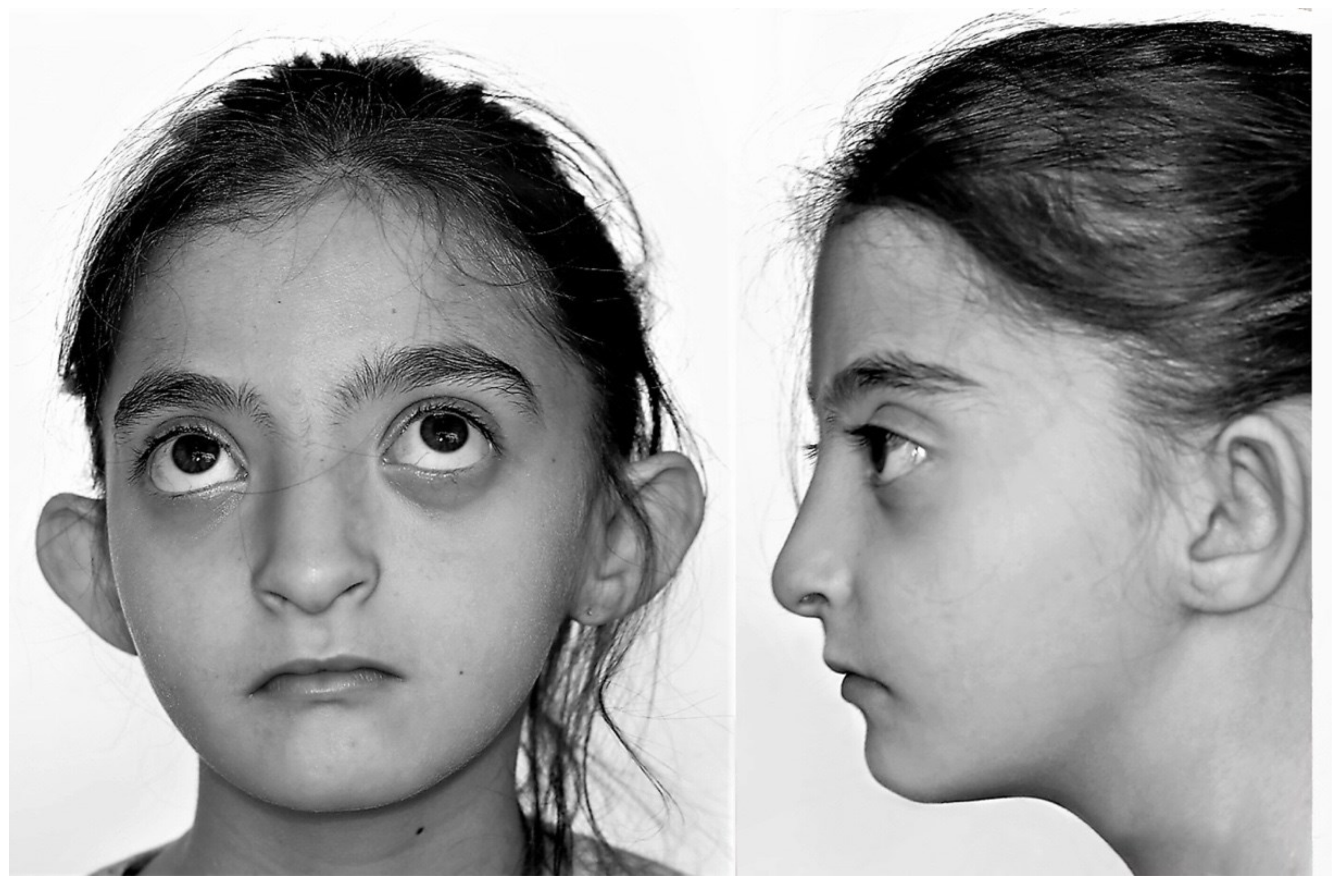
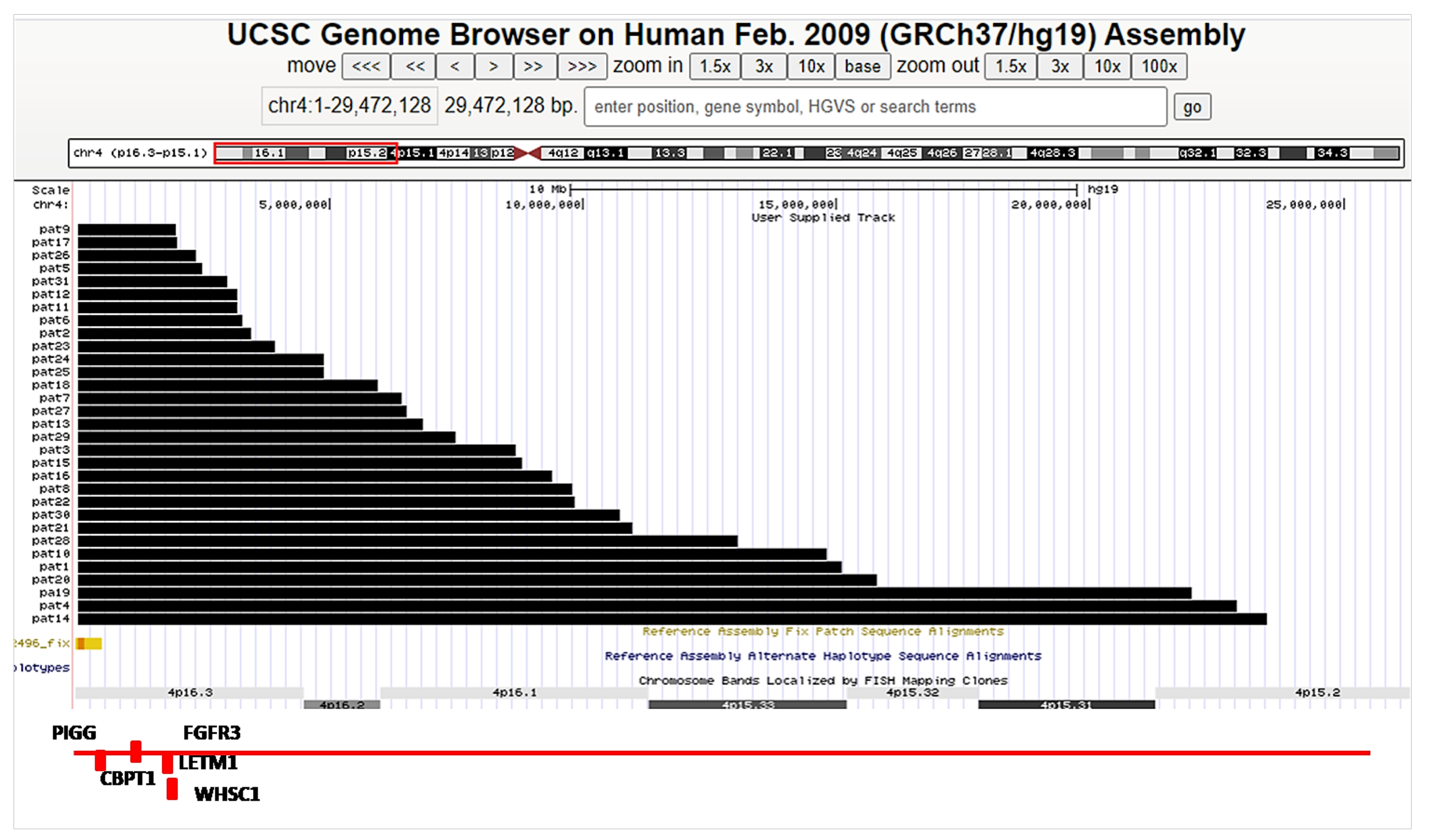
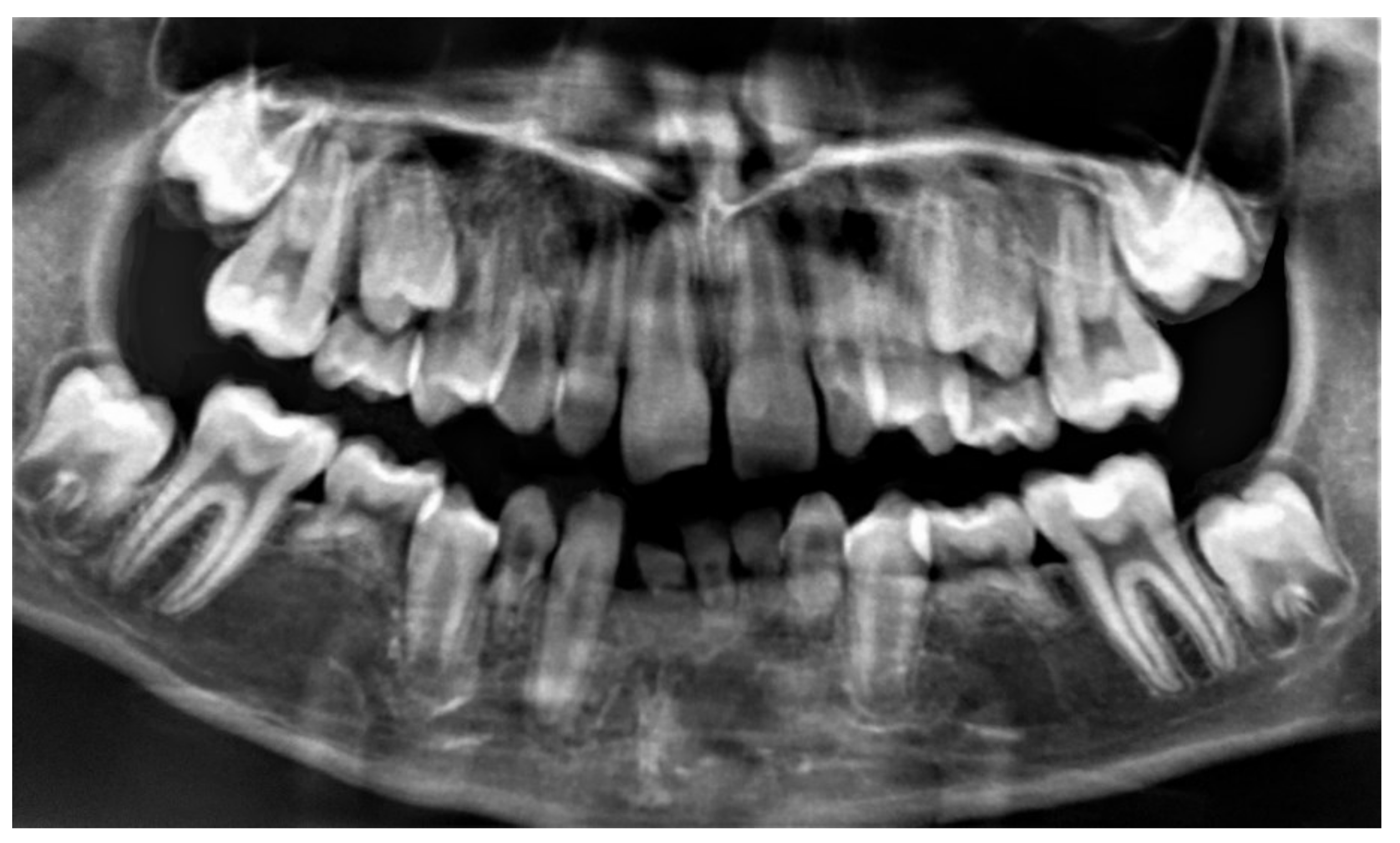

| Anatomical Variables | Comorbidities | |||
|---|---|---|---|---|
| Sex | Female | n = 21 (67.7%) | Cardiopathy | n = 13 (41.9%) |
| Male | n = 10 (23.2%) | Nephrologic–urologic abnormalities | n = 18 (58.1%) | |
| Mean age, years | 9.5 ± 3.6 (2.2–20.7) | Ophthalmologic manifestations | n = 19 (61.3%) | |
| Weeks of gestation | 30.8 ± 3.3 (range 23–39) | Otorhinolaryngologic manifestations | n = 15 (48.4%) | |
| Mean weight, g * | 1865.0 ± 527.8 (range 800–3440) | Recurrent respiratory infections | n = 14 (45.2%) | |
| Mean height, cm * | 43.3 ± 3.9 (range 34–52) | Central nervous system malformations | n = 15 (48.4%) | |
| Mean cranial circumference, cm * | 36.7 ± 2.5 (range 30–41) | Gastrostomy carrier | n = 3 (9.7%) | |
| Growth delay | n = 22 71.0% | Other surgical history | n = 21 (67.7%) | |
| Developmental Abnormalities | Epilepsy Characteristics | ||
|---|---|---|---|
| Head control | n = 30 (96.8%) | Diagnosis of epilepsy | n = 31 (100%) |
| Active sitting | n = 21 (67.7%) | Age at onset, months | 9.9 ± 5.0 (range 0–24) |
| Walking with support | n = 20 (64.5%) | Seizures triggered by fever | n = 12 (38.7.0%) |
| Independent walking | n = 11 (35.5%) | Seizures not triggered by fever | n = 7 (22.5%) |
| Seizures triggered by fever and other conditions | n = 12 (38.7%) | ||
| Autonomous feeding | n = 6 (19.4%) | Status epilepticus | n = 14 (45.2%) |
| episodes, 3.8 ± 11.0 (range 0–55) | |||
| Sphincter control | n = 25 (80.6%) | Admission to intensive care due to status epilepticus | n = 11 (35.5%) |
| Interaction with environment | n = 28 (90.3%) | Tonic-clonic seizures | n = 21 (67.7%) |
| Communication by gestures/pictograms | n = 19 (61.3%) | Atypical absences | n = 17 (54.8%) |
| Emits single words | n = 10 (32.3%) | Seizure-free period ≥ 2 years | n = 12 (38.7%) |
| Emits simple phrases | n = 4 (12.9%) | AED use | n = 26 (83.9%) |
| Psychomotor development level * | 139.1 ± 47.8 (range 36–230) | Uses AEDs in monotherapy | n = 17 (54.8%) |
| Psychomotor delay * | 18.4 ± 10.1 (range 1–33) | Valproic acid use | n = 16 (51.6%) |
| Number of comorbidities that affect development | 6.8 ± 3.1 (range 1–13) | Levetiracetam use | n = 14 (45.1%) |
| Genetic-Alteration Type | Number of Cases | |
|---|---|---|
| Terminal deletions | Simple 4p- terminal deletions | 16 (51.60%) |
| 4p- terminal deletions and additional terminal duplications in other chromosomes * | 13 (41.95%) | |
| 4p- terminal deletions and additional genomic interstitial duplications | 2 (6.45%) | |
| Interstitial deletions | 0 (0%) | |
| Upper Teeth n (%) | Lower Teeth n (%) | |||
|---|---|---|---|---|
| Temporary teeth n = 18 | Lateral incisor | 2 (11.1) | Lateral incisor | - |
| Second molar | 8 (44.4) | Second molar | 8 (44.4) | |
| Definitive teeth n = 47 | Central incisor | - | Central incisor | 2 (4.2) |
| Lateral incisor | 8 (17.0) | Lateral incisor | 4 (8.5) | |
| Canine | - | Canine | 1 (2.1) | |
| First premolar | 5 (10.6) | First premolar | - | |
| Second premolar | 12 (25.5) | Second premolar | 7 (14.9) | |
| First molar | 1 (2.1) | First molar | - | |
| Second molar | 2 (4.2) | Second molar | 5 (10.6) | |
| Sagittal Plane n = 15 | Transverse Plane n = 15 | Vertical Plane n = 15 | |||
|---|---|---|---|---|---|
| n (%) | n (%) | n (%) | |||
| Class I | 4 (26.6) | Normal occlusion | 5 (33.3) | Normal occlusion | 6 (40.0) |
| Class II | 9 (60.0) | Crossbite | 5 (33.3) | Overbite | 7 (46.6) |
| Class III | 2 (13.3) | Scissor bite | 3 (20.0) | Open bite | 2 (13.3) |
| Overjet | 4 (26.6) | ||||
| Anterior crossbite | 3 (20.0) | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Limeres, J.; Serrano, C.; De Nova, J.M.; Silvestre-Rangil, J.; Machuca, G.; Maura, I.; Cruz Ruiz-Villandiego, J.; Diz, P.; Blanco-Lago, R.; Nevado, J.; et al. Oral Manifestations of Wolf-Hirschhorn Syndrome: Genotype-Phenotype Correlation Analysis. J. Clin. Med. 2020, 9, 3556. https://doi.org/10.3390/jcm9113556
Limeres J, Serrano C, De Nova JM, Silvestre-Rangil J, Machuca G, Maura I, Cruz Ruiz-Villandiego J, Diz P, Blanco-Lago R, Nevado J, et al. Oral Manifestations of Wolf-Hirschhorn Syndrome: Genotype-Phenotype Correlation Analysis. Journal of Clinical Medicine. 2020; 9(11):3556. https://doi.org/10.3390/jcm9113556
Chicago/Turabian StyleLimeres, Jacobo, Candela Serrano, Joaquin Manuel De Nova, Javier Silvestre-Rangil, Guillermo Machuca, Isabel Maura, Jose Cruz Ruiz-Villandiego, Pedro Diz, Raquel Blanco-Lago, Julian Nevado, and et al. 2020. "Oral Manifestations of Wolf-Hirschhorn Syndrome: Genotype-Phenotype Correlation Analysis" Journal of Clinical Medicine 9, no. 11: 3556. https://doi.org/10.3390/jcm9113556
APA StyleLimeres, J., Serrano, C., De Nova, J. M., Silvestre-Rangil, J., Machuca, G., Maura, I., Cruz Ruiz-Villandiego, J., Diz, P., Blanco-Lago, R., Nevado, J., & Diniz-Freitas, M. (2020). Oral Manifestations of Wolf-Hirschhorn Syndrome: Genotype-Phenotype Correlation Analysis. Journal of Clinical Medicine, 9(11), 3556. https://doi.org/10.3390/jcm9113556







