Opportunistic Infections and Efficacy Following Conversion to Belatacept-Based Therapy after Kidney Transplantation: A French Multicenter Cohort
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Efficacy and Safety
2.3. Statistical Analyses
3. Results
3.1. Population Study
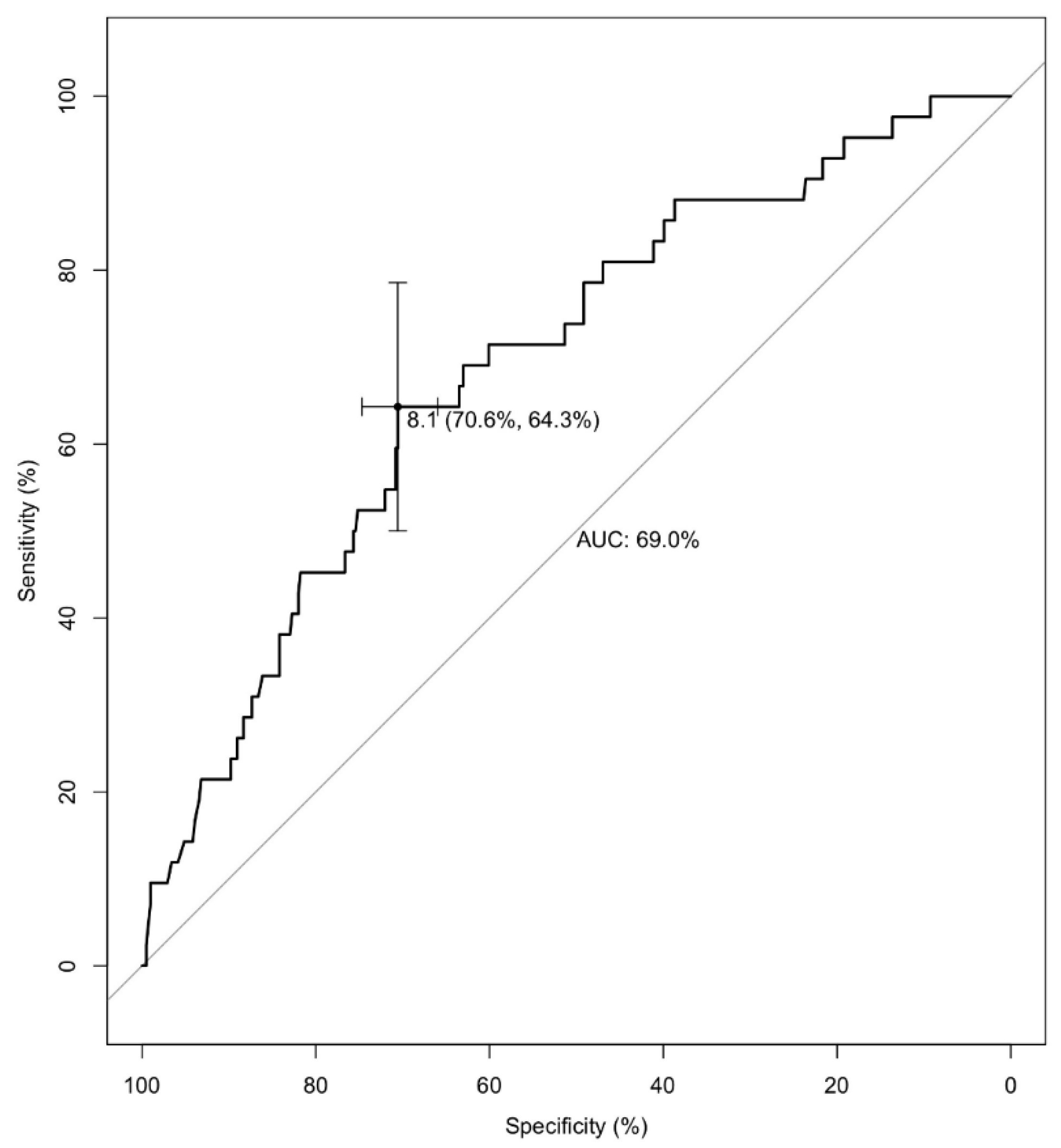
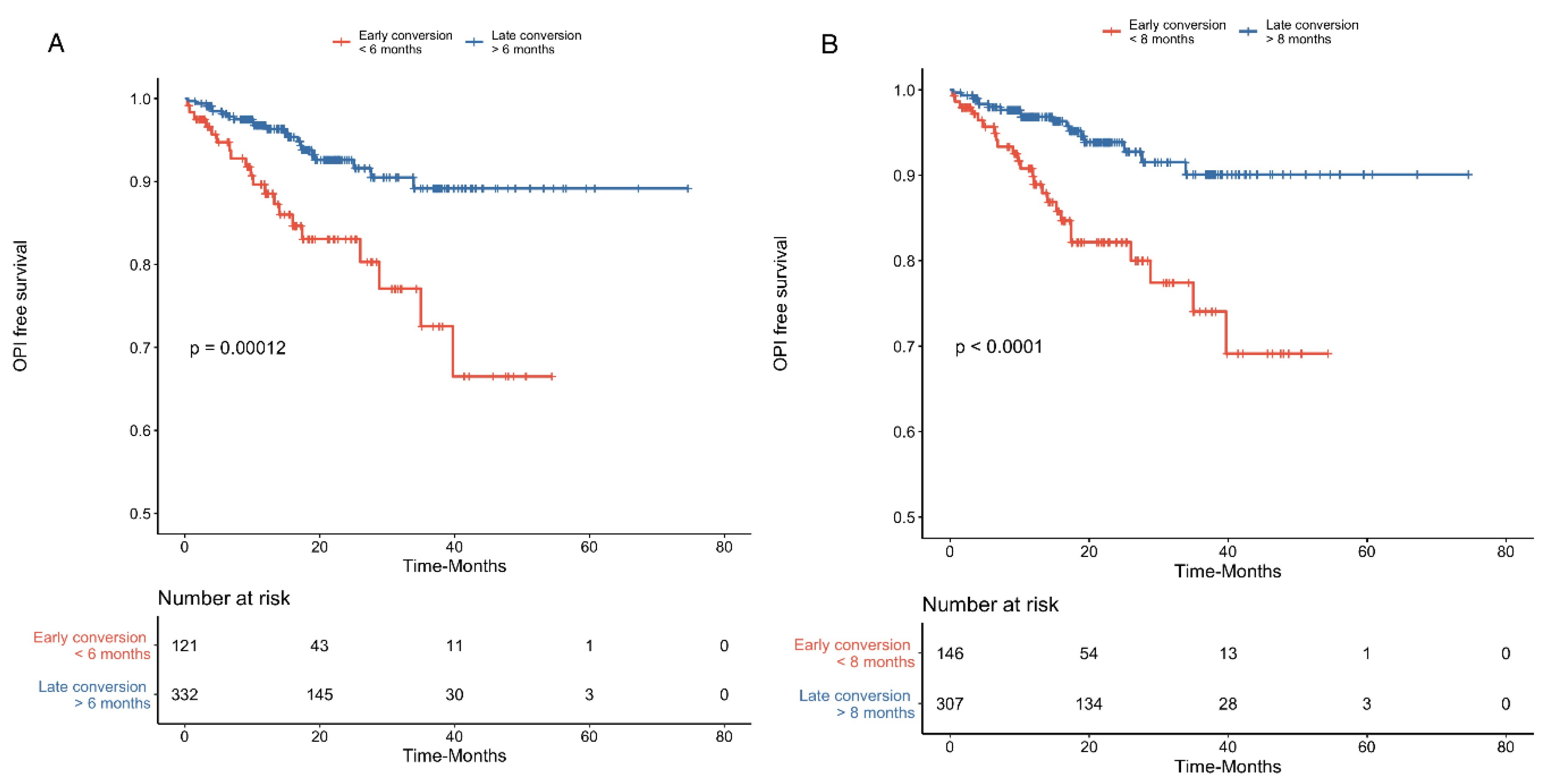
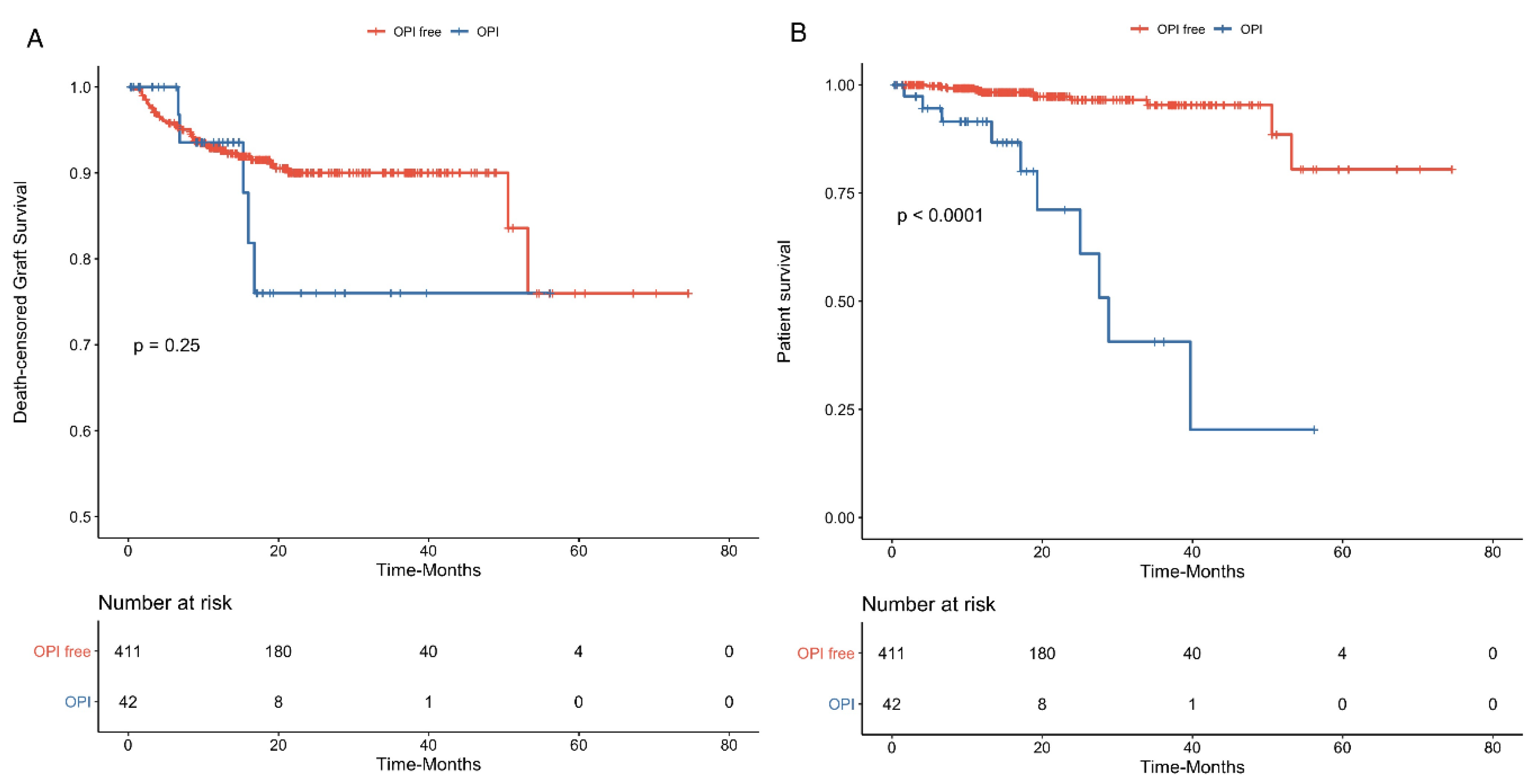
3.2. Opportunistic Infections
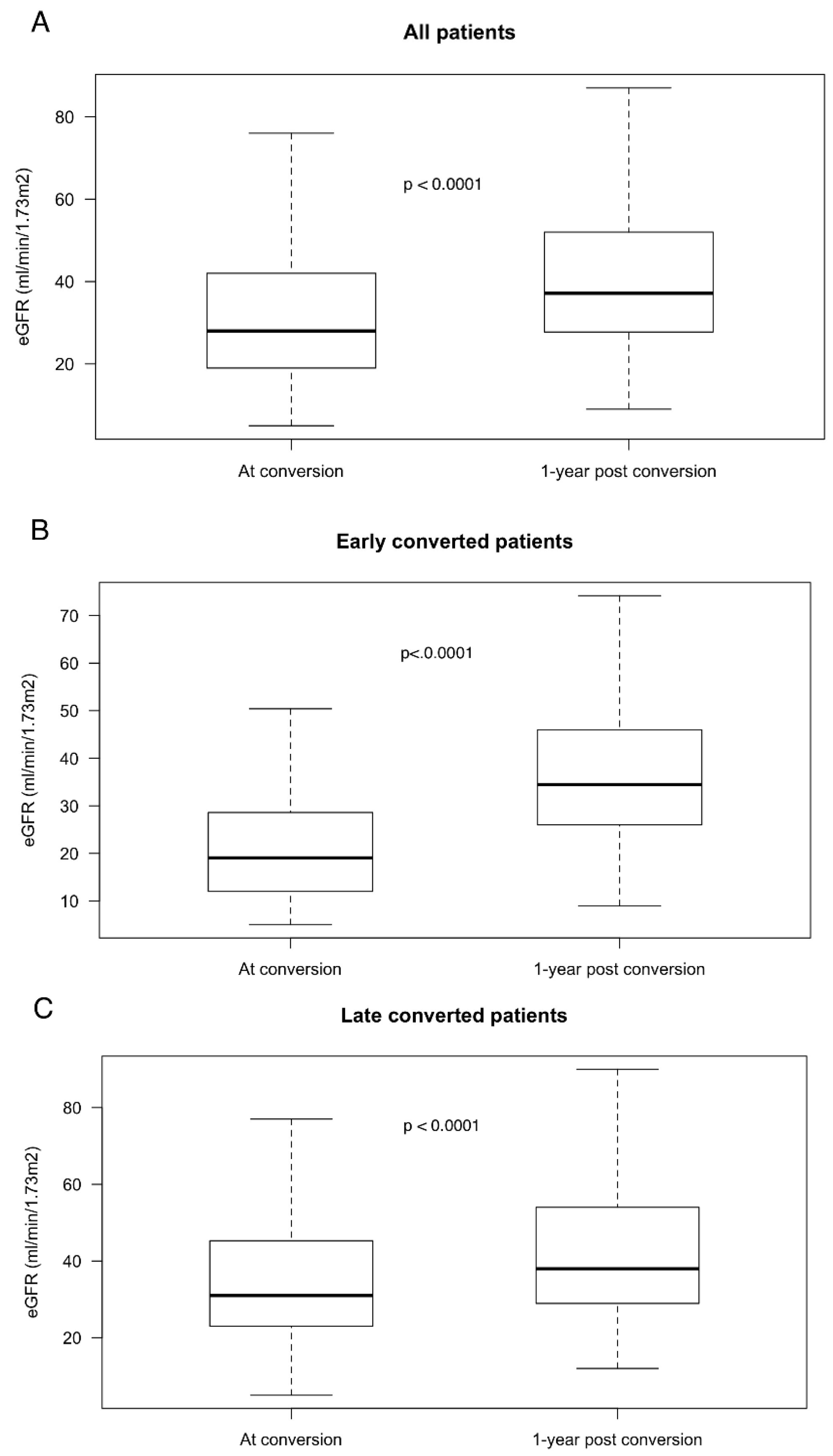
3.3. Renal Function
4. Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Ekberg, H.; Tedesco-Silva, H.; Demirbas, A.; Vítko, Š.; Nashan, B.; Gürkan, A.; Margreiter, R.; Hugo, C.; Grinyó, J.M.; Frei, U.; et al. Reduced exposure to calcineurin inhibitors in renal transplantation. N. Engl. J. Med. 2007, 357, 2562–2575. [Google Scholar] [CrossRef]
- Malvezzi, P.; Rostaing, L. The safety of calcineurin inhibitors for kidney-transplant patients. Expert Opin. Drug Saf. 2015, 14, 1531–1546. [Google Scholar] [CrossRef]
- Nankivell, B.J.; Borrows, R.J.; Fung, C.L.-S.; O’Connell, P.J.; Allen, R.D.M.; Chapman, J.R. The natural history of chronic allograft nephropathy. N. Engl. J. Med. 2003, 349, 2326–2333. [Google Scholar] [CrossRef]
- Nankivell, B.J.; P’Ng, C.H.; O’Connell, P.J.; Chapman, J.R. Calcineurin Inhibitor Nephrotoxicity through the Lens of Longitudinal Histology: Comparison of Cyclosporine and Tacrolimus Eras. Transplantation 2016, 100, 1723–1731. [Google Scholar] [CrossRef]
- Malvezzi, P.; Jouve, T.; Rostaing, L. Costimulation Blockade in Kidney Transplantation: An Update. Transplantation 2016, 100, 2315–2323. [Google Scholar] [CrossRef]
- Vincenti, F.; Larsen, C.; Durrbach, A.; Wekerle, T.; Nashan, B.; Blancho, G.; Lang, P.; Grinyo, J.; Halloran, P.F.; Solez, K.; et al. Costimulation blockade with belatacept in renal transplantation. N. Engl. J. Med. 2005, 353, 770–781. [Google Scholar] [CrossRef]
- Vincenti, F.; Charpentier, B.; Vanrenterghem, Y.; Rostaing, L.; Bresnahan, B.; Darji, P.; Massari, P.; Mondragon-Ramirez, G.A.; Agarwal, M.; Di Russo, G.; et al. A phase III study of belatacept-based immunosuppression regimens versus cyclosporine in renal transplant recipients (BENEFIT study). Am. J. Transplant. 2010, 10, 535–546. [Google Scholar] [CrossRef]
- Vincenti, F. Belatacept and Long-Term Outcomes in Kidney Transplantation. N. Engl. J. Med. 2016, 374, 2600–2601. [Google Scholar] [CrossRef]
- Durrbach, A.; Pestana, J.M.; Florman, S.; Del Carmen Rial, M.; Rostaing, L.; Kuypers, D.; Matas, A.; Wekerle, T.; Polinsky, M.; Meier-Kriesche, H.U.; et al. Long-Term Outcomes in Belatacept- Versus Cyclosporine-Treated Recipients of Extended Criteria Donor Kidneys: Final Results From BENEFIT-EXT, a Phase III Randomized Study. Am. J. Transplant. 2016, 16, 3192–3201. [Google Scholar] [CrossRef]
- Hošková, L.; Málek, I.; Kopkan, L.; Kautzner, J. Pathophysiological mechanisms of calcineurin inhibitor-induced nephrotoxicity and arterial hypertension. Physiol. Res. 2017, 66, 167–180. [Google Scholar]
- Rodrigo, E.; Fernández-Fresnedo, G.; Valero, R.; Ruiz, J.C.; Pinera, C.; Palomar, R.; González-Cotorruelo, J.; Gómez-Alamillo, C.; Arias, M. New-onset diabetes after kidney transplantation: Risk factors. J. Am. Soc. Nephrol. 2006, 17, S291–S295. [Google Scholar] [CrossRef] [PubMed]
- Terrec, F.; Jouve, T.; Naciri-Bennani, H.; Benhamou, P.Y.; Malvezzi, P.; Janbon, B.; Giovannini, D.; Rostaing, L.; Noble, J. Late Conversion from Calcineurin Inhibitors to Belatacept in Kidney-Transplant Recipients Has a Significant Beneficial Impact on Glycemic Parameters. Transplant. Direct 2020, 6, e517. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6964931/ (accessed on 4 June 2020). [CrossRef] [PubMed]
- Bray, R.A.; Gebel, H.M.; Townsend, R.; Roberts, M.E.; Polinsky, M.; Yang, L.; Meier-Kriesche, H.U.; Larsen, C.P. De novo donor-specific antibodies in belatacept-treated vs cyclosporine-treated kidney-transplant recipients: Post hoc analyses of the randomized phase III BENEFIT and BENEFIT-EXT studies. Am. J. Transplant. 2018, 18, 1783–1789. [Google Scholar] [CrossRef]
- Bray, R.A.; Gebel, H.M.; Townsend, R.; Roberts, M.E.; Polinsky, M.; Yang, L.; Meier-Kriesche, H.U.; Larsen, C.P. Posttransplant reduction in preexisting donor-specific antibody levels after belatacept- versus cyclosporine-based immunosuppression: Post hoc analyses of BENEFIT and BENEFIT-EXT. Am. J. Transplant. 2018, 18, 1774–1782. [Google Scholar] [CrossRef] [PubMed]
- Rostaing, L.; Massari, P.; Garcia, V.D.; Mancilla-Urrea, E.; Nainan, G.; del Carmen Rial, M.; Steinberg, S.; Vincenti, F.; Shi, R.; Di Russo, G.; et al. Switching from calcineurin inhibitor-based regimens to a belatacept-based regimen in renal transplant recipients: A randomized phase II study. Clin. J. Am. Soc. Nephrol. 2011, 6, 430–439. [Google Scholar] [CrossRef] [PubMed]
- Bertrand, D.; Chavarot, N.; Gatault, P.; Garrouste, C.; Bouvier, N.; Grall-Jezequel, A.; Jaureguy, M.; Caillard, S.; Lemoine, M.; Colosio, C.; et al. Opportunistic infections after conversion to belatacept in kidney transplantation. Nephrol. Dial. Transplant. 2020, 35, 336–345. [Google Scholar] [CrossRef] [PubMed]
- Levey, A.S.; Greene, T.; Schluchter, M.D.; Cleary, P.A.; Teschan, P.E.; Lorenz, R.A.; Molitch, M.E.; Mitch, W.E.; Siebert, C.; Hall, P.M. Glomerular Filtration Rate Measurements in Clinical Trials. J. Am. Soc. Nephrol. 1993, 4, 1159–1171. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2866096/ (accessed on 7 June 2020). [PubMed]
- Kotton, C.N.; Kumar, D.; Caliendo, A.M.; Huprikar, S.; Chou, S.; Danziger-Isakov, L.; Humar, A. The Third International Consensus Guidelines on the Management of Cytomegalovirus in Solid-organ Transplantation. Transplantation 2018, 102, 900–931. [Google Scholar] [CrossRef]
- Fishman, J.A. Infection in Organ Transplantation. Am. J. Transplant. 2017, 17, 856–879. [Google Scholar] [CrossRef]
- Ljungman, P.; Boeckh, M.; Hirsch, H.H.; Josephson, F.; Lundgren, J.; Nichols, G.; Pikis, A.; Razonable, R.R.; Miller, V.; Griffiths, P.D.; et al. Definitions of Cytomegalovirus Infection and Disease in Transplant Patients for Use in Clinical Trials. Clin. Infect. Dis. 2017, 64, 87–91. [Google Scholar]
- Bamoulid, J.; Courivaud, C.; Coaquette, A.; Chalopin, J.M.; Gaiffe, E.; Saas, P.; Ducloux, D. Subclinical Epstein-Barr virus viremia among adult renal transplant recipients: Incidence and consequences. Am. J. Transplant. 2013, 13, 656–662. [Google Scholar]
- Blazquez-Navarro, A.; Dang-Heine, C.; Wittenbrink, N.; Bauer, C.; Wolk, K.; Sabat, R.; Westhoff, T.H.; Sawitzki, B.; Reinke, P.; Thomusch, O.; et al. BKV, CMV, and EBV Interactions and their Effect on Graft Function One Year Post-Renal Transplantation: Results from a Large Multi-Centre Study. EBioMedicine 2018, 34, 113–121. [Google Scholar]
- Cippà, P.E.; Schiesser, M.; Ekberg, H.; van Gelder, T.; Mueller, N.J.; Cao, C.A.; Fehr, T.; Bernasconi, C. Risk Stratification for Rejection and Infection after Kidney Transplantation. Clin. J. Am. Soc. Nephrol. 2015, 10, 2213–2220. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4670759/ (accessed on 4 June 2020).
- Darres, A.; Ulloa, C.; Brakemeier, S.; Garrouste, C.; Bestard, O.; Del Bello, A.; Soussan, R.S.; Dürr, M.; Budde, K.; Legendre, C.; et al. Conversion to Belatacept in Maintenance Kidney Transplant Patients: A Retrospective Multicenter European Study. Transplantation 2018, 102, 1545–1552. [Google Scholar]
- Martin, S.T.; Powell, J.T.; Patel, M.; Tsapepas, D. Risk of posttransplant lymphoproliferative disorder associated with use of belatacept. Am. J. Health Syst. Pharm. 2013, 70, 1977–1983. [Google Scholar] [PubMed]
- Nafar, M.; Roshan, A.; Pour-Reza-Gholi, F.; Samadian, F.; Ahmadpoor, P.; Samavat, S.; Abbasi, M.A. Prevalence and risk factors of recurrent cytomegalovirus infection in kidney transplant recipients. Iran J. Kidney Dis. 2014, 8, 231–235. [Google Scholar]
- Kizilbash, S.J.; Rheault, M.N.; Bangdiwala, A.; Matas, A.; Chinnakotla, S.; Chavers, B.M. Infection Rates in Tacrolimus versus Cyclosporine Treated Pediatric Kidney Transplant Recipients on a Rapid Discontinuation of Prednisone Protocol: 1-year Analysis. Pediatr. Transplant. 2017, 21, e12919. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5423828/ (accessed on 4 June 2020).
- Grinyó, J.M.; Del Carmen Rial, M.; Alberu, J.; Steinberg, S.M.; Manfro, R.C.; Nainan, G.; Vincenti, F.; Jones-Burton, C.; Kamar, N. Safety and Efficacy Outcomes 3 Years After Switching to Belatacept from a Calcineurin Inhibitor in Kidney Transplant Recipients: Results from a Phase 2 Randomized Trial. Am. J. Kidney Dis. 2017, 69, 587–594. [Google Scholar]
- Gupta, S.; Rosales, I.; Wojciechowski, D. Pilot Analysis of Late Conversion to Belatacept in Kidney Transplant Recipients for Biopsy-Proven Chronic Tacrolimus Toxicity. J. Transplant. 2018, 2018, 1968029. [Google Scholar]
- Brakemeier, S.; Kannenkeril, D.; Dürr, M.; Braun, T.; Bachmann, F.; Schmidt, D.; Wiesener, M.; Budde, K. Experience with belatacept rescue therapy in kidney transplant recipients. Transpl. Int. 2016, 29, 1184–1195. [Google Scholar]
- Attias, P.; Melica, G.; Boutboul, D.; De Castro, N.; Audard, V.; Stehlé, T.; Gaube, G.; Fourati, S.; Botterel, F.; Fihman, V.; et al. Epidemiology, Risk Factors, and Outcomes of Opportunistic Infections after Kidney Allograft Transplantation in the Era of Modern Immunosuppression: A Monocentric Cohort Study. J. Clin. Med. 2019, 8, 594. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6572426/ (accessed on 1 June 2020). [CrossRef] [PubMed]
- Helfrich, M.; Dorschner, P.; Thomas, K.; Stosor, V.; Ison, M.G. A retrospective study to describe the epidemiology and outcomes of opportunistic infections after abdominal organ transplantation. Transpl. Infect. Dis. 2017, 19, e12691. [Google Scholar] [CrossRef]
| Total Patients N = 453 | Early Conversion a N = 121 | Late Conversion a N = 332 | p-Value | |
|---|---|---|---|---|
| Recipients’ characteristics | ||||
| Age at transplantation—years | 52.6 ± 15 | 59.1 ± 13 | 50.2 ± 16 | <0.001 |
| Age at conversion—years | 56.7 ± 14 | 59.3 ± 13 | 55.7 ± 15 | 0.012 |
| Gender: male n (%) | 285 (62.9) | 80 (66.1) | 205 (61.7) | 0.458 |
| CMV R+ n (%) | 243 (58.2) | 70 (57.8) | 173 (58.2) | 1.000 |
| First transplantation n (%) | 364 (80.3) | 103 (85.1) | 261 (78.6) | 0.158 |
| Nephropathy | 0.007 | |||
| Glomerulopathy n (%) | 86 (19.7) | 29 (23.9) | 57 (17.9) | - |
| Diabetes and/or hypertension n (%) | 87 (19.9) | 27 (22.3) | 60 (18.9) | - |
| Polycystic kidney n (%) | 66 (15.1) | 14 (11.7) | 52 (16.4) | - |
| IgA nephropathy n (%) | 21 (4.8) | 0 | 21 (6.6) | - |
| Malformation n (%) | 51 (11.7) | 10 (0.8) | 41 (12.9) | - |
| Undetermined n (%) | 70 (16.0) | 20 (16.7) | 50 (15.7) | - |
| Donors’ characteristics | ||||
| Age—years | 54.2 ± 15 | 59.7 ± 19 | 53.6 ± 15 | 0.397 |
| Donation after death n (%) | 383 (87.4) | 115 (96.7) | 268 (84.0) | <0.001 |
| Living donor n (%) | 55 (12.5) | 4 (3.3) | 51 (16.0) | <0.001 |
| Characteristics at conversion | ||||
| Time between KT and belatacept conversion—months | 49.8 ± 65 | 2.7 ± 1.7 | 66.9 ± 69 | <0.001 |
| eGFR at conversion—MDRD-mL/min/1.73 m2 | 32.0 ± 18 | 22.1 ± 14 | 35.6 ± 18 | <0.001 |
| Diabetes at conversion n (%) | 133 (29.3) | 36 (29.7) | 97 (29.2) | 1.000 |
| Lymphopenia at conversion n (%) | 296 (65.3) | 100 (82.6) | 196 (59.0) | <0.001 |
| Immunosuppression | ||||
| Induction therapy | ||||
| Antithymoglobulin n (%) | 254 (56.7) | 51 (42.1) | 203 (62.0) | <0.001 |
| Basiliximab n (%) | 175 (39.0) | 70 (57.8) | 105 (32.1) | <0.001 |
| Maintenance therapy at conversion | ||||
| Steroids n (%) | 276 (60.9) | 108 (89.2) | 168 (50.7) | <0.001 |
| Tacrolimus n (%) | 363 (80.3) | 96 (79.3) | 267 (80.7) | 0.856 |
| Tacrolimus trough concentration—ng/mL | 6.5 ± 2.6 | 7.0 ± 3.0 | 6.4 ± 2.5 | 0.061 |
| Cyclosporine n (%) | 77 (17.0) | 23 (19) | 54 (16.3) | 0.593 |
| Mycophenolate n (%) | 396 (87.6) | 108 (89.2) | 288 (87.0) | 0.630 |
| mTOR inhibitors n (%) | 44 (9.7) | 12 (9.9) | 32 (9.6) | 1.000 |
| Azathioprine n (%) | 17 (3.8) | 3 (2.4) | 14 (4.2) | 0.557 |
| Total Patients N = 453 | Early Conversion a N = 121 | Late Conversion a N = 332 | p-Value | |
|---|---|---|---|---|
| Renal complications | ||||
| Kidney allograft failure n (%) | 42 (9.3) | 23 (19.0) | 19 (5.7) | <0.001 |
| Rejection post-conversion n (%) | 24 (5.3) | 10 (8.3) | 14 (4.2) | 0.143 |
| Viral complications | ||||
| CMV DNAemia n (%) | 74 (16.9) | 37 (31.6) | 37 (11.5) | <0.001 |
| CMV disease n (%) | 22 (4.8) | 14 (11.6) | 8 (2.4) | <0.001 |
| Time between belatacept conversion to CMV disease—months | 10.7 ± 9 | 9.6 ± 10 | 12.7 ± 8 | 0.188 |
| EBV DNAemia n (%) | 133 (36.0) | 13 (14.8) | 120 (42.7) | <0.001 |
| BK virus DNAemia n (%) | 6 (2.0) | 4 (7.2) | 2 (0.8) | 1 |
| BK nephropathy n (%) | 0 | 0 | 0 | - |
| JC Virus n (%) | 2 (0.4) | 2 (1.6) | 0 | 0.121 |
| VZV n (%) | 5 (1.1) | 2 (1.6) | 3 (0.9) | 0.610 |
| Bacterial complications | ||||
| Tuberculosis n (%) | 2 (0.4) | 1 (0.8) | 1 (0.3) | 0.458 |
| Fungal complications | ||||
| Pneumocystis pneumonia n (%) | 13 (2.8) | 6 (4.9) | 7 (2.1) | 0.557 |
| Aspergillosis pneumonia n (%) | 2 (0.4) | 2 (1.6) | 0 | 0.121 |
| Other complications | ||||
| Death n (%) | 22 (4.8) | 10 (8.3) | 12 (3.6) | 0.073 |
| HR [95%CI] | p-Value Univariate Analyses | HR [95%CI] | p-Value Multivariate Analyses a | |
|---|---|---|---|---|
| Age at KT, median | 2.37 [1.2–4.5] | 0.009 | - | - |
| Early belatacept conversion b | 3.1 [1.7–5.6] | <0.001 | - | - |
| Lymphopenia at conversion | 2.5 [1.17–5.5] | 0.018 | - | - |
| eGFR < 25 mL/min/1.73 m2 at conversion | 6.8 [3.3–14.4] | <0.001 | 4.7 [2.2–10.3] | <0.001 |
| Basiliximab | 2.2 [1.2–4.0] | 0.012 | - | - |
| Steroids c | 3.9 [1.7–8.8] | 0.001 | 2.1 [0.8–5.2] | 0.121 |
| Tacrolimus c | 0.43 [0.2–0.8] | 0.009 | - | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bertrand, D.; Terrec, F.; Etienne, I.; Chavarot, N.; Sberro, R.; Gatault, P.; Garrouste, C.; Bouvier, N.; Grall-Jezequel, A.; Jaureguy, M.; et al. Opportunistic Infections and Efficacy Following Conversion to Belatacept-Based Therapy after Kidney Transplantation: A French Multicenter Cohort. J. Clin. Med. 2020, 9, 3479. https://doi.org/10.3390/jcm9113479
Bertrand D, Terrec F, Etienne I, Chavarot N, Sberro R, Gatault P, Garrouste C, Bouvier N, Grall-Jezequel A, Jaureguy M, et al. Opportunistic Infections and Efficacy Following Conversion to Belatacept-Based Therapy after Kidney Transplantation: A French Multicenter Cohort. Journal of Clinical Medicine. 2020; 9(11):3479. https://doi.org/10.3390/jcm9113479
Chicago/Turabian StyleBertrand, Dominique, Florian Terrec, Isabelle Etienne, Nathalie Chavarot, Rebecca Sberro, Philippe Gatault, Cyril Garrouste, Nicolas Bouvier, Anne Grall-Jezequel, Maïté Jaureguy, and et al. 2020. "Opportunistic Infections and Efficacy Following Conversion to Belatacept-Based Therapy after Kidney Transplantation: A French Multicenter Cohort" Journal of Clinical Medicine 9, no. 11: 3479. https://doi.org/10.3390/jcm9113479
APA StyleBertrand, D., Terrec, F., Etienne, I., Chavarot, N., Sberro, R., Gatault, P., Garrouste, C., Bouvier, N., Grall-Jezequel, A., Jaureguy, M., Caillard, S., Thervet, E., Colosio, C., Golbin, L., Rerolle, J.-P., Thierry, A., Sayegh, J., Janbon, B., Malvezzi, P., ... Noble, J. (2020). Opportunistic Infections and Efficacy Following Conversion to Belatacept-Based Therapy after Kidney Transplantation: A French Multicenter Cohort. Journal of Clinical Medicine, 9(11), 3479. https://doi.org/10.3390/jcm9113479







