Sequential Circulating Tumor Cell Counts in Patients with Locally Advanced or Metastatic Hepatocellular Carcinoma: Monitoring the Treatment Response
Abstract
1. Introduction
2. Experimental Section
2.1. Patients
2.2. Enrichment and Isolation of CTCs
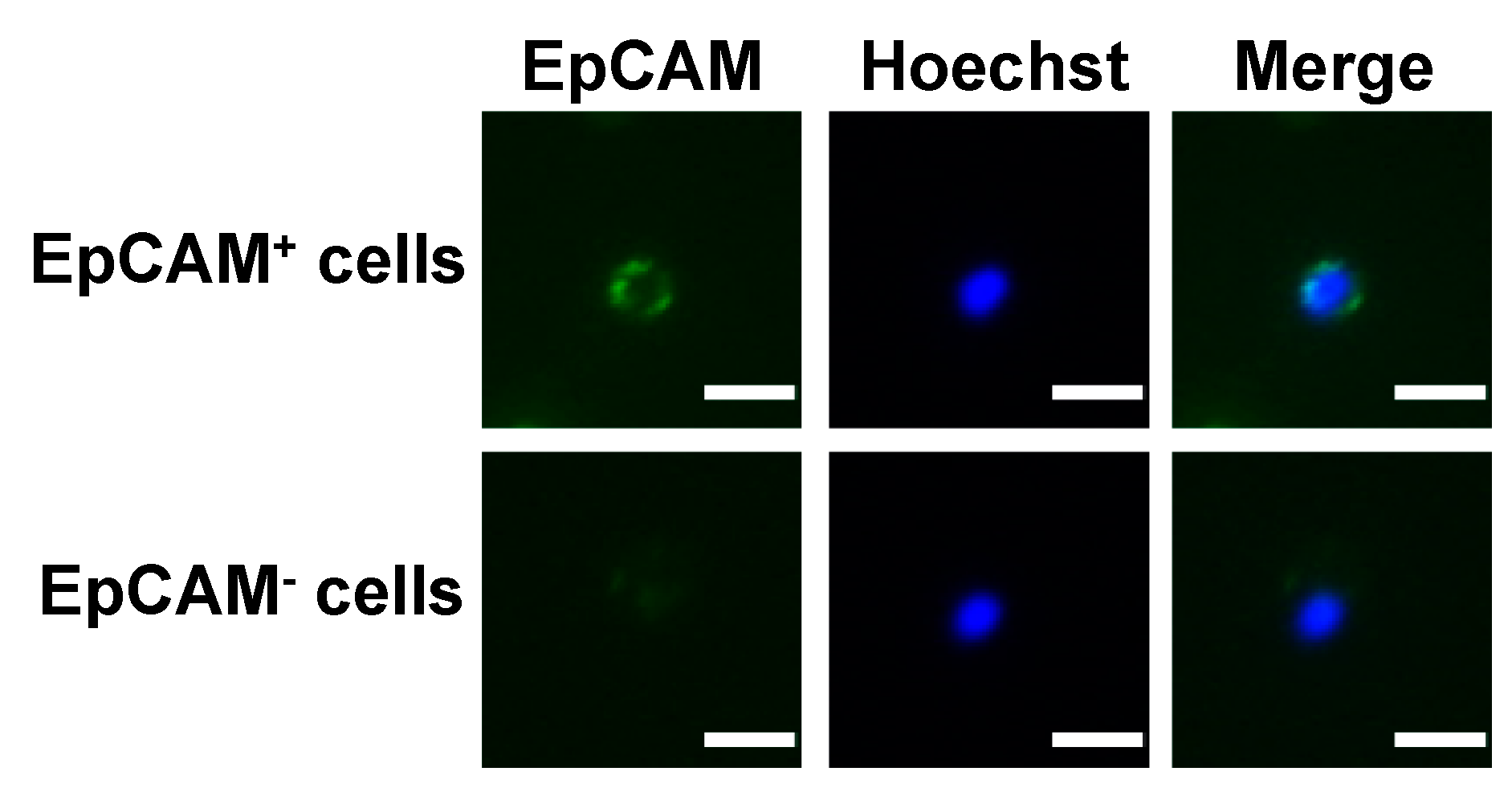
2.3. Immunofluorescence Staining and CTC Counting
2.4. Measurement of Serum AFP
2.5. Statistical Analysis
3. Results
3.1. Basic Characteristics of Enrolled Patients
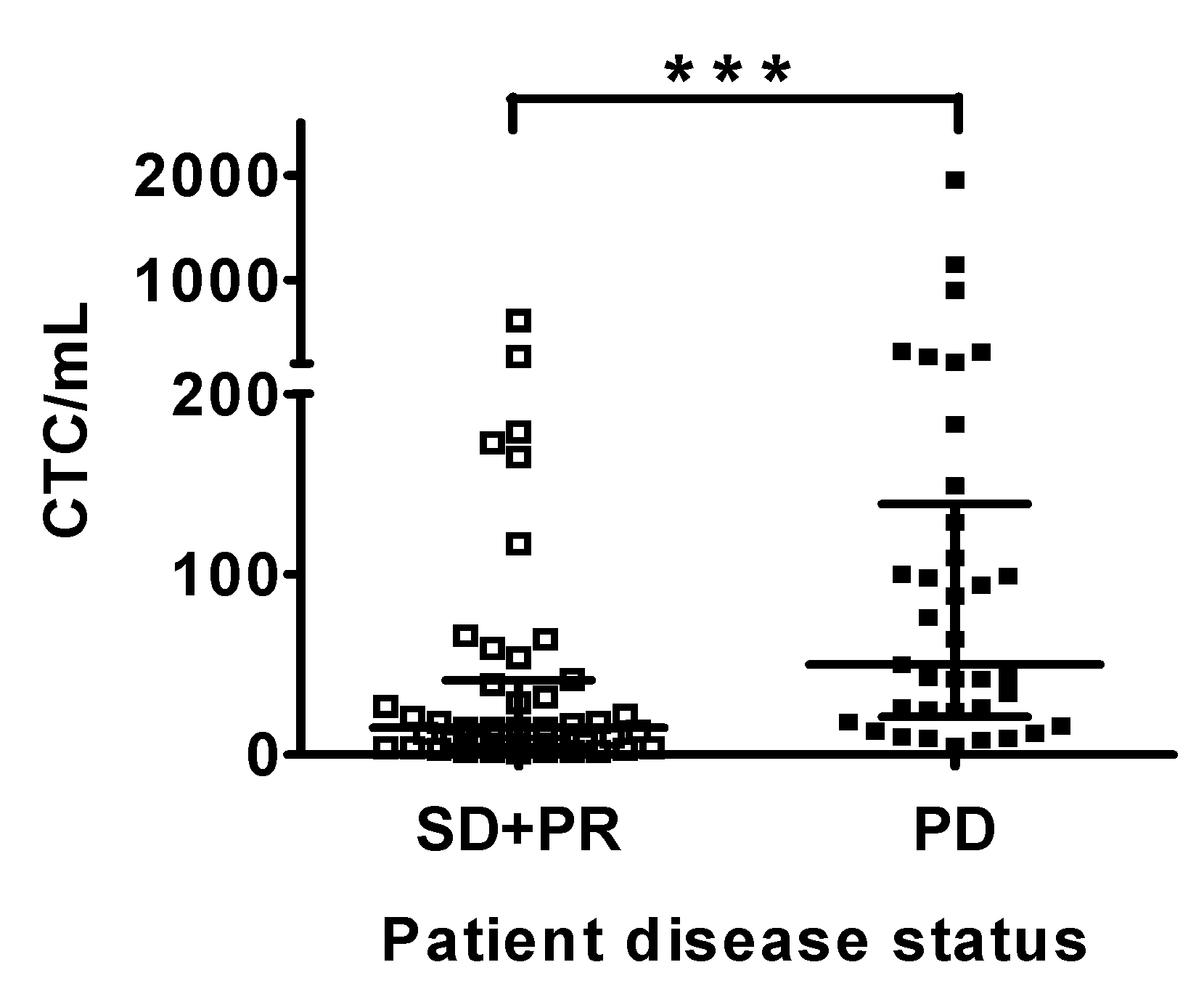
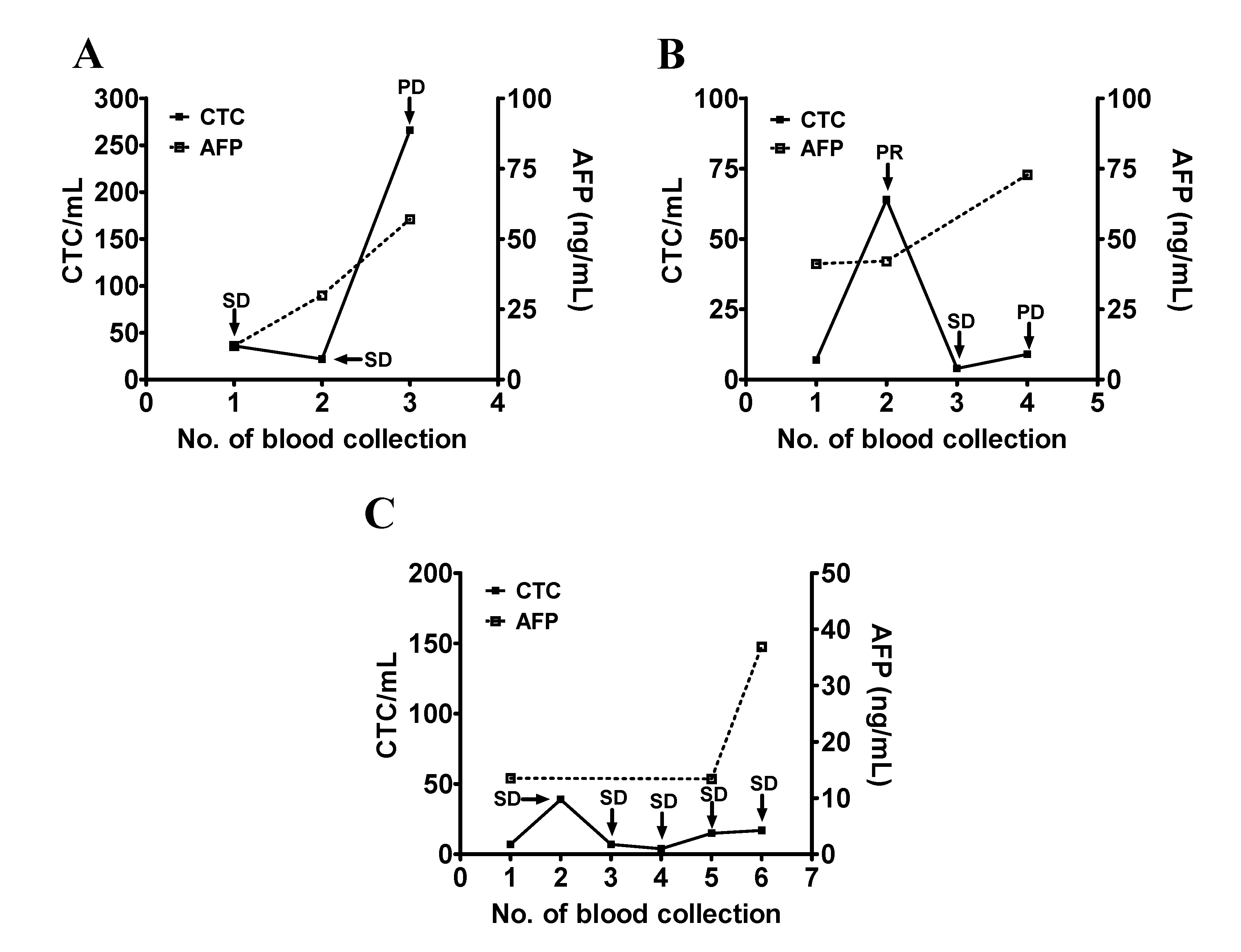
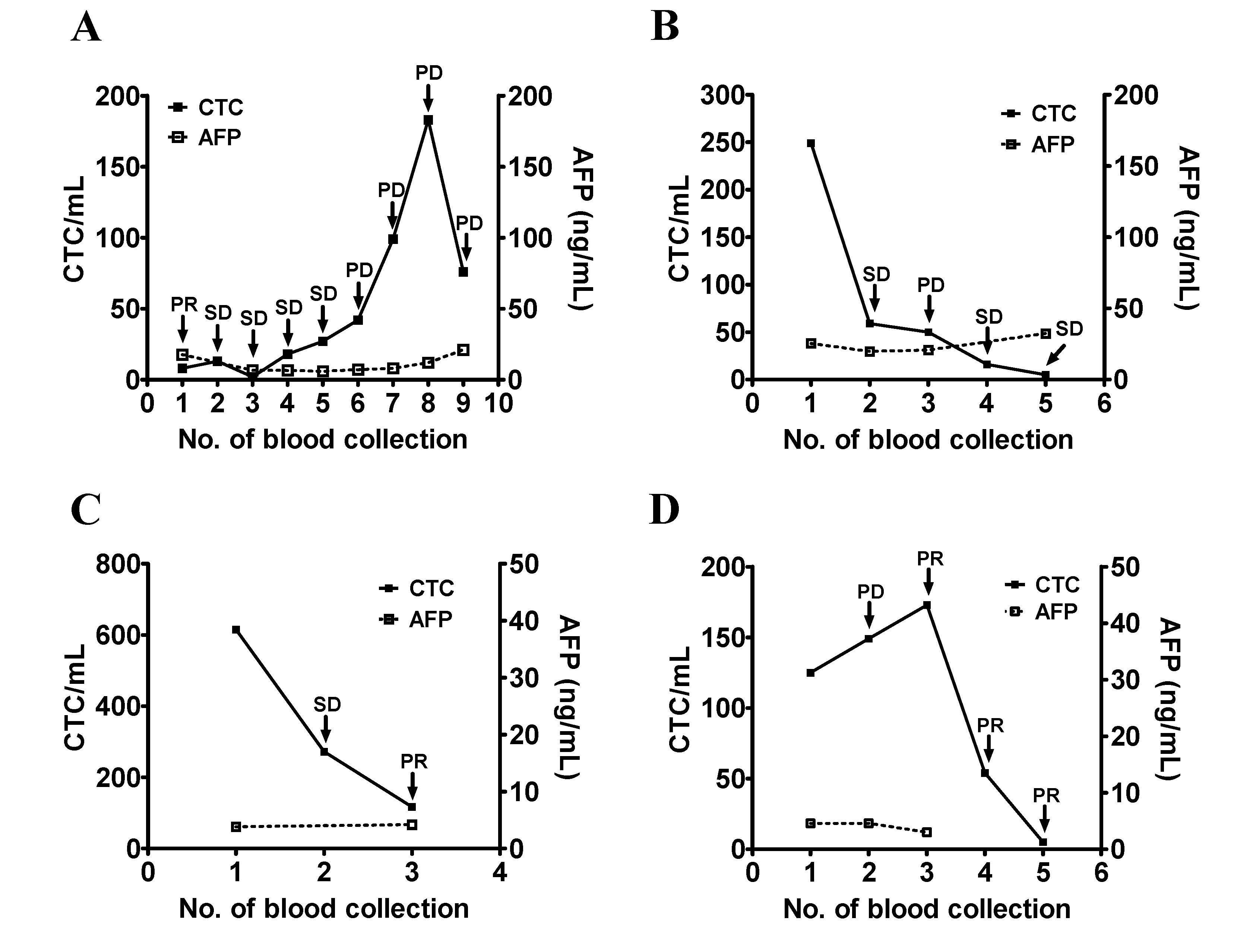
3.2. CTC Counts are Correlated with Disease Status of Patients with HCC
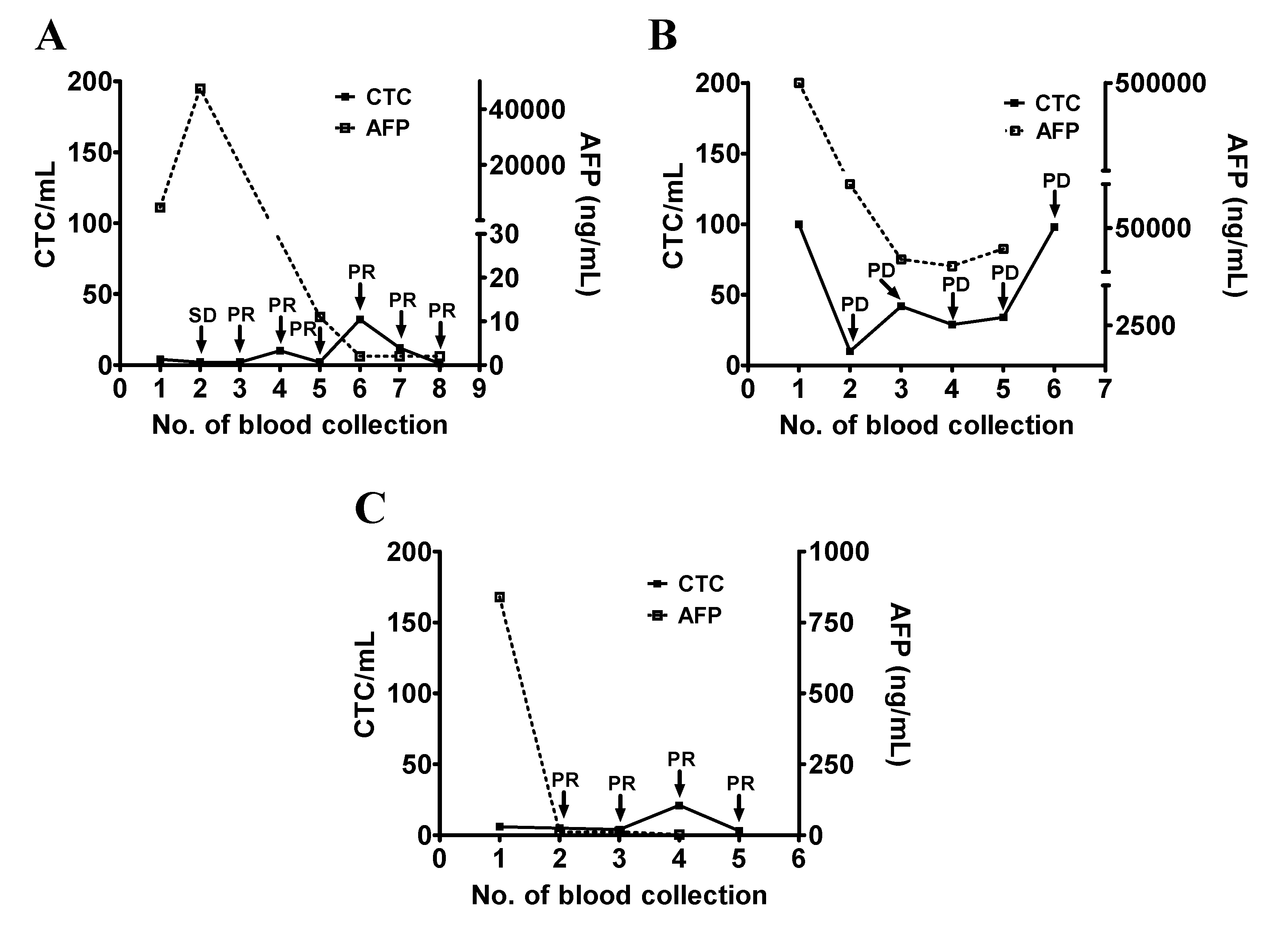
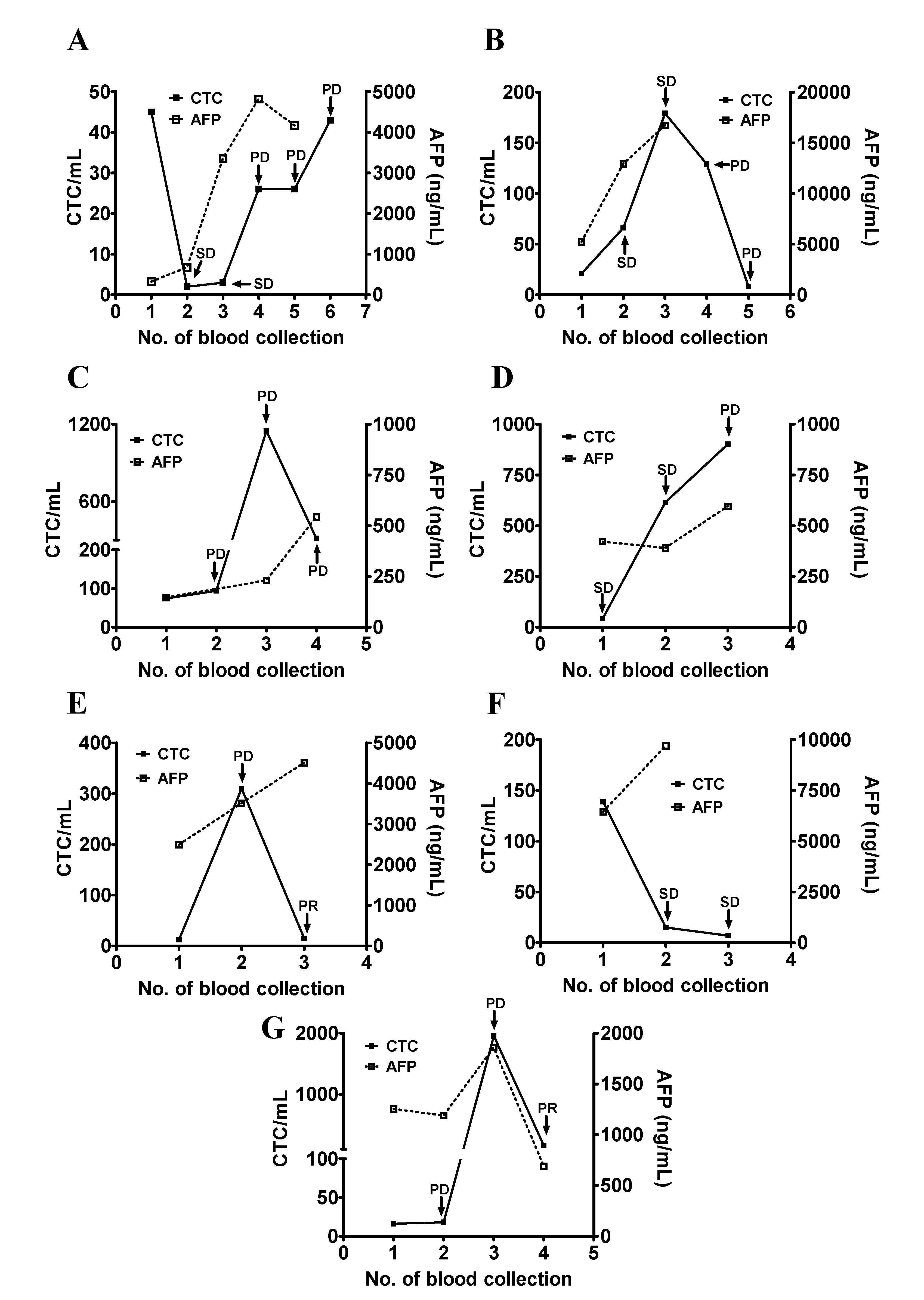
3.3. Use of CTC Counts for Longitudinal Follow-Up and Disease Monitoring of Patients with HCC
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Rebecca, L.; Siegel, R.L.; Lindsey, A.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA A Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed]
- Chang, M.H. Hepatitis B vaccination: Disease and cancer prevention-a Taiwanese experience. Clin. Liver Dis. 2010, 14, 521–530. [Google Scholar] [CrossRef] [PubMed]
- Sulkowski, M.S.; Gardiner, D.F.; Rodriguez-Torres, M.; Reddy, K.R.; Hassanein, T.; Jacobson, I.; Lawitz, E.; Lok, A.S.; Hinestrosa, F.; Thuluvath, P.J.; et al. Daclatasvir plus sofosbuvir for previously treated or untreated chronic HCV infection. N. Engl. J. Med. 2014, 370, 211–221. [Google Scholar] [CrossRef] [PubMed]
- White, D.L.; Kanwal, F.; El-Serag, H.B. Association between nonalcoholic fatty liver disease and risk for hepatocellular cancer, based on systematic review. Clin. Gastroenterol. Hepatol. 2012, 10, 1342–1359. [Google Scholar] [CrossRef]
- Marrero, J.A.; Kulik, L.M.; Sirlin, C.B.; Zhu, A.X.; Finn, R.S.; Abecassis, M.M.; Roberts, L.R.; Heimbach, J.K. Diagnosis, staging, and management of hepatocellular carcinoma: 2018 practice guidance by the American Association for the Study of Liver Diseases. Hepatology 2018, 68, 723–750. [Google Scholar] [CrossRef]
- European Association for the Study of the Liver. EASL clinical practice guidelines: Management of hepatocellular carcinoma. J. Hepatol. 2018, 69, 182–236. [Google Scholar] [CrossRef]
- Daher, S.; Massarwa, M.; Benson, A.A.; Khoury, T. Current and future treatment of hepatocellular carcinoma: An updated comprehensive review. J. Clin. Transl. Hepatol. 2018, 6, 69–78. [Google Scholar] [CrossRef]
- Llovet, J.M.; Ricci, S.; Mazzaferro, V.; Hilgard, P.; Gane, E.; Blanc, J.F.; de Oliveira, A.C.; Santoro, A.; Raoul, J.L.; Forner, A.; et al. Sorafenib in advanced hepatocellular carcinoma. N. Engl. J. Med. 2008, 359, 378–390. [Google Scholar] [CrossRef]
- Cheng, A.L.; Kang, Y.K.; Chen, Z.; Tsao, C.J.; Qin, S.; Kim, J.S.; Luo, R.; Feng, J.; Ye, S.; Yang, T.S.; et al. Efficacy and safety of sorafenib in patients in the Asia-Pacific region with advanced hepatocellular carcinoma: A phase III randomised, double-blind, placebo-controlled trial. Lancet Oncol. 2009, 10, 25–34. [Google Scholar] [CrossRef]
- Hu, J.; Wang, N.; Yang, Y.; Ma, L.; Han, R.; Zhang, W.; Yan, C.; Zheng, Y.; Wang, X. Diagnostic value of alpha-fetoprotein combined with neutrophil-to-lymphocyte ratio for hepatocellular carcinoma. BMC Gastroenterol. 2018, 18, 186. [Google Scholar] [CrossRef]
- Cabel, L.; Proudhon, C.; Gortais, H.; Loirat, D.; Coussy, F.; Pierga, J.Y.; Bidard, F.C. Circulating tumor cells: Clinical validity and utility. Int. J. Clin. Oncol. 2017, 22, 421–430. [Google Scholar] [CrossRef] [PubMed]
- Olmos, D.; Baird, R.D.; Yap, T.A.; Massard, C.; Pope, L.; Sandhu, S.K.; Attard, G.; Dukes, J.; Papadatos-Pastos, D.; Grainger, P.; et al. Baseline circulating tumor cell counts significantly enhance a prognostic score for patients participating in phase I oncology trials. Clin. Cancer Res. 2011, 17, 5188–5196. [Google Scholar] [CrossRef] [PubMed]
- Cristofanilli, M.; Budd, G.T.; Ellis, M.J.; Stopeck, A.; Matera, J.; Miller, M.C.; Reuben, J.M.; Doyle, G.V.; Allard, W.J.; Terstappen, L.W.; et al. Circulating tumor cells, disease progression, and survival in metastatic breast cancer. N. Engl. J. Med. 2004, 351, 781–791. [Google Scholar] [CrossRef] [PubMed]
- Yap, K.; Cohen, E.N.; Reuben, J.M.; Khoury, J.D. Circulating tumor cells: State-of-the-art update on technologies and clinical applications. Curr. Hematol. Malig. Rep. 2019, 14, 353–357. [Google Scholar] [CrossRef] [PubMed]
- von Felden, J.; Schulze, K.; Krech, T.; Ewald, F.; Nashan, B.; Pantel, K.; Lohse, A.W.; Riethdorf, S.; Wege, H. Circulating tumor cells as liquid biomarker for high HCC recurrence risk after curative liver resection. Oncotarget 2017, 8, 89978–89987. [Google Scholar] [CrossRef]
- Zhou, J.; Huang, A.; Yang, X.R. Liquid biopsy and its potential for management of hepatocellular carcinoma. J. Gastrointest. Cancer 2016, 47, 157–167. [Google Scholar] [CrossRef]
- Ogle, L.F.; Orr, J.G.; Willoughby, C.E.; Hutton, C.; McPherson, S.; Plummer, R.; Boddy, A.V.; Curtin, N.J.; Jamieson, D.; Reeves, H.L. Imagestream detection and characterisation of circulating tumour cells-a liquid biopsy for hepatocellular carcinoma? J. Hepatol. 2016, 65, 305–313. [Google Scholar] [CrossRef]
- Lin, H.C.; Hsu, H.C.; Hsieh, C.H.; Wang, H.M.; Huang, C.Y.; Wu, M.H.; Tseng, C.P. A negative selection system PowerMag for effective leukocyte depletion and enhanced detection of EpCAM positive and negative circulating tumor cells. Clin. Chim. Acta 2013, 419, 77–84. [Google Scholar] [CrossRef]
- Lin, J.D.; Liou, M.J.; Hsu, H.L.; Leong, K.K.; Chen, Y.T.; Wang, Y.R.; Hung, W.S.; Lee, H.Y.; Tsai, H.J.; Tseng, C.P. Circulating epithelial cell characterization and correlation with remission and survival in patients with thyroid cancer. Thyroid 2018, 28, 1479–1489. [Google Scholar] [CrossRef]
- Ma, M.C.; Chen, Y.Y.; Li, S.H.; Cheng, Y.F.; Wang, C.C.; Chiu, T.J.; Pei, S.N.; Liu, C.T.; Huang, T.L.; Huang, C.H.; et al. Intra-arterial chemotherapy with doxorubicin and cisplatin is effective for advanced hepatocellular cell carcinoma. Sci. World J. 2014, 2014, 160138. [Google Scholar] [CrossRef]
- Qin, S.; Cheng, Y.; Liang, J.; Shen, L.; Bai, Y.; Li, J.; Fan, J.; Liang, L.; Zhang, Y.; Wu, G.; et al. Efficacy and safety of the FOLFOX4 regimen versus doxorubicin in Chinese patients with advanced hepatocellular carcinoma: A subgroup analysis of the EACH study. Oncologist 2014, 19, 1169–1178. [Google Scholar] [CrossRef]
- Schwartz, L.H.; Seymour, L.; Litière, S.; Ford, R.; Gwyther, S.; Mandrekar, S.; Shankar, L.; Bogaerts, J.; Chen, A.; Dancey, J.; et al. RECIST 1.1-standardisation and disease-specific adaptations: Perspectives from the RECIST Working Group. Eur. J. Cancer 2016, 62, 138–145. [Google Scholar] [CrossRef]
- Hiraoka, A.; Michitaka, K.; Kumada, T.; Izumi, N.; Kadoya, M.; Kokudo, N.; Kubo, S.; Matsuyama, Y.; Nakashima, O.; Sakamoto, M.; et al. Prediction of prognosis of intermediate-stage HCC patients: Validation of the tumor marker score in a nationwide database in Japan. Liver Cancer 2019, 8, 403–411. [Google Scholar] [CrossRef]
- Ma, Y.; Wang, Q.; Dong, Q.; Zhan, L.; Zhang, J. How to differentiate pseudoprogression from true progression in cancer patients treated with immunotherapy. Am. J. Cancer Res. 2019, 9, 1546–1553. [Google Scholar]
- Gomaa, A.I.; Khan, S.A.; Leen, E.L.S.; Waked, I.; Taylor-Robinson, S.D. Diagnosis of hepatocellular carcinoma. World J. Gastroenterol. 2009, 15, 1301–1314. [Google Scholar] [CrossRef]
- Ahmed Mohammed, H.F.; Roberts, L.R. Should AFP (or any biomarkers) be used for HCC surveillance? Curr. Hepatol. Rep. 2017, 16, 137–145. [Google Scholar] [CrossRef]
- Marrero, J.A.; Feng, Z.; Wang, Y.; Nguyen, M.H.; Befeler, A.S.; Roberts, L.R.; Reddy, K.R.; Harnois, D.; Llovet, J.M.; Normolle, D.; et al. Alpha-fetoprotein, des-gamma carboxyprothrombin, and lectin-bound alpha-fetoprotein in early hepatocellular carcinoma. Gastroenterology 2009, 137, 110–118. [Google Scholar] [CrossRef]
- Lou, J.; Zhang, L.; Lv, S.; Zhang, C.; Jiang, S. Biomarkers for hepatocellular carcinoma. Biomark. Cancer 2017, 9, 1–9. [Google Scholar] [CrossRef]
- Ye, Q.; Ling, S.; Zheng, S.; Xu, X. Liquid biopsy in hepatocellular carcinoma: Circulating tumor cells and circulating tumor DNA. Mol. Cancer 2019, 18, 114. [Google Scholar] [CrossRef]
- Zhou, Y.; Wang, B.; Wu, J.; Zhang, C.; Zhou, Y.; Yang, X.; Zhou, J.; Guo, W.; Fan, J. Association of preoperative EpCAM circulating tumor cells and peripheral Treg cell levels with early recurrence of hepatocellular carcinoma following radical hepatic resection. BMC Cancer 2016, 16, 506. [Google Scholar] [CrossRef]
- Kelley, R.K.; Magbanua, M.J.; Butler, T.M.; Collisson, E.A.; Hwang, J.; Sidiropoulos, N.; Evason, K.; McWhirter, R.M.; Hameed, B.; Wayne, E.M.; et al. Circulating tumor cells in hepatocellular carcinoma: A pilot study of detection, enumeration, and next-generation sequencing in cases and controls. BMC Cancer 2015, 15, 206. [Google Scholar] [CrossRef]
- Chen, Y.; Li, S.; Li, W.; Yang, R.; Zhang, X.; Ye, Y.; Yu, J.; Ye, L.; Tang, W. Circulating tumor cells undergoing EMT are poorly correlated with clinical stages or predictive of recurrence in hepatocellular carcinoma. Sci. Rep. 2019, 9, 7084. [Google Scholar] [CrossRef]
- Wang, Z.; Luo, L.; Cheng, Y.; He, G.; Peng, B.; Gao, Y.; Jiang, Z.S.; Pan, M. Correlation between postoperative early recurrence of hepatocellular carcinoma and mesenchymal circulating tumor cells in peripheral blood. J. Gastrointest. Surg. 2018, 22, 633–639. [Google Scholar] [CrossRef]
- Sun, C.; Liao, W.; Deng, Z.; Li, E.; Feng, Q.; Lei, J.; Yuan, R.; Zou, S.; Mao, Y.; Shao, J.; et al. The diagnostic value of assays for circulating tumor cells in hepatocellular carcinoma: A meta-analysis. Medicine (Baltimore) 2017, 96, e7513. [Google Scholar] [CrossRef]
- Yu, J.; Xiao, W.; Dong, S.; Liang, H.; Zhang, Z.; Zhang, B.; Huang, Z.; Chen, Y.; Zhang, W.; Lu, H.; et al. Effect of surgical liver resection on circulating tumor cells in patients with hepatocellular carcinoma. BMC Cancer 2018, 18, 835. [Google Scholar] [CrossRef]
- Lin, H.C.; Liou, M.J.; Hsu, H.L.; Hsieh, J.C.; Chen, Y.A.; Tseng, C.P.; Lin, J.D. Combined analysis of circulating epithelial cells and serum thyroglobulin for distinguishing disease status of the patients with papillary thyroid carcinoma. Oncotarget 2016, 7, 17242–17253. [Google Scholar] [CrossRef]
- Hsieh, J.C.; Lin, H.C.; Huang, C.Y.; Hsu, H.L.; Wu, T.M.; Lee, C.L.; Chen, M.C.; Wang, H.M.; Tseng, C.P. Prognostic value of circulating tumor cells with podoplanin expression in patients with locally advanced or metastatic head and neck squamous cell carcinoma. Head Neck 2015, 37, 1448–1455. [Google Scholar] [CrossRef]
- Hsieh, C.H.; Lin, H.C.; Huang, S.B.; Hsueh, C.; Hsu, H.L.; Wang, H.M.; Wu, M.H.; Tseng, C.P.; Lin, J.D. Circulating epithelial cell enumeration facilitates the identification and follow-up of a patient with early stage papillary thyroid microcarcinoma: A case report. Clin. Chim. Acta. 2016, 454, 107–111. [Google Scholar] [CrossRef]
- Li, Y.R.; Tseng, C.P.; Hsu, H.L.; Lin, H.C.; Chen, Y.A.; Chen, S.T.; Liou, M.J.; Lin, J.D. Circulating epithelial cells as potential biomarkers for detection of recurrence in patients of papillary thyroid carcinoma with positive serum anti-thyroglobulin antibody. Clin. Chim. Acta. 2018, 477, 74–80. [Google Scholar] [CrossRef]
- Li, Y.M.; Xu, S.C.; Li, J.; Han, K.Q.; Pi, H.F.; Zheng, L.; Zuo, G.H.; Huang, X.B.; Li, H.Y.; Zhao, H.Z.; et al. Epithelial-mesenchymal transition markers expressed in circulating tumor cells in hepatocellular carcinoma patients with different stages of disease. Cell Death Dis. 2013, 4, e831. [Google Scholar] [CrossRef]
- Lee, H.Y.; Yu, N.Y.; Lee, S.H.; Tsai, H.J.; Wu, C.C.; Cheng, J.C.; Chen, D.P.; Wang, Y.R.; Tseng, C.P. Podoplanin promotes cancer-associated thrombosis and contributes to the unfavorable overall survival in an ectopic xenograft mouse model of oral cancer. Biomed. J. 2019. [Google Scholar] [CrossRef]
- Manion, K.; Frey, T. Apoptosis of cells in aged samples as detected by the ProCOUNT reagent. Cytometry 1996, 26, 317–322. [Google Scholar] [CrossRef]
- Yu, H.K.; Lee, H.J.; Choi, H.N.; Ahn, J.H.; Choi, J.Y.; Song, H.S.; Lee, K.H.; Yoon, Y.; Yi, L.S.; Kim, J.S.; et al. Characterization of CD45−/CD31+/CD105+ circulating cells in the peripheral blood of patients with gynecologic malignancies. Clin. Cancer Res. 2013, 19, 5340–5350. [Google Scholar] [CrossRef]
- Fang, C.; Fan, C.; Wang, C.; Huang, Q.; Meng, W.; Yu, Y.; Yang, L.; Peng, Z.; Hu, J.; Li, Y.; et al. CD133+CD54+CD44+ circulating tumor cells as a biomarker of treatment selection and liver metastasis in patients with colorectal cancer. Oncotarget 2016, 7, 77389–77403. [Google Scholar] [CrossRef]
- Kalinich, M.; Bhan, I.; Kwan, T.T.; Miyamoto, D.T.; Javaid, S.; LiCausi, J.A.; Milner, J.D.; Hong, X.; Goyal, L.; Sil, S.; et al. An RNA-based signature enables high specificity detection of circulating tumor cells in hepatocellular carcinoma. Proc. Natl. Acad. Sci. USA 2017, 114, 1123–1128. [Google Scholar] [CrossRef]
| Characteristics | No. of Patients |
|---|---|
| Total enrollment | 30 |
| Stage at the time of diagnosis | |
| I | 8 |
| II | 4 |
| III | 17 |
| IV | 1 |
| Age at enrollment: median (interquartile range) | 64 (56–69) |
| Sex (M/F) | 20/10 |
| Hepatitis history | |
| 1 HBV+ only | 11 |
| HCV+ only | 13 |
| HBV+ HCV+ | 2 |
| Non-B, non-C | 4 |
| Tumor status at enrollment | |
| Locally advanced | 7 |
| Extrahepatic metastasis | 9 |
| Both | 14 |
| Treatment at enrollment | |
| Sorafenib | 21 |
| Ramucirumab | 2 |
| Nivolumab | 1 |
| Thalidomide | 2 |
| IA-CT | 3 |
| IV-CT | 1 |
| Patient ID | Clinical Parameters | Treatment | No. of Blood Collection | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | |||
| 1 | CTCs (cells/mL) | Sorafenib; thalidomide after the 2nd blood collection | 4 | 2 | 2 | 10 | 2 | 32 | 12 | 1 | |
| AFP (ng/mL) | 4737 | 47,360 | − | − | 11 | <2 | <2 | <2 | |||
| Disease status | − | 1 SD | PR | PR | PR | PR | PR | PR | |||
| 2 | CTCs (cells/mL) | Sorafenib | 45 | 2 | 3 | 26 | 26 | 43 | |||
| AFP (ng/mL) | 323 | 675 | 3356 | 4822 | 4171 | − | |||||
| Disease status | − | SD | SD | PD | PD | PD | |||||
| 3 | CTCs (cells/mL) | Sorafenib; ramucirumab after the 7th blood collection | 8 | 13 | 2 | 18 | 27 | 42 | 99 | 183 | 76 |
| AFP (ng/mL) | 18 | − | 7 | 7 | 6 | 7 | 8 | 12 | 21 | ||
| Disease status | PR | SD | SD | SD | SD | PD | PD | PD | PD | ||
| 4 | CTCs (cells/mL) | Sorafenib | 21 | 66 | 179 | 129 | 8 | ||||
| AFP (ng/mL) | 5222 | 12,929 | 16,727 | − | − | ||||||
| Disease status | − | SD | SD | PD | PD | ||||||
| 5 | CTCs (cells/mL) | Sorafenib | 20 | 211 | |||||||
| AFP (ng/mL) | 59,233 | − | |||||||||
| Disease status | − | PD | |||||||||
| 6 | CTCs (cells/mL) | Sorafenib; thalidomide after the 2nd blood collection | 73 | 25 | |||||||
| AFP (ng/mL) | 35 | − | |||||||||
| Disease status | − | PD | |||||||||
| 7 | CTCs (cells/mL) | Sorafenib | 64 | ||||||||
| AFP (ng/mL) | 542 | ||||||||||
| Disease status | PD | ||||||||||
| 8 | CTCs (cells/mL) | Sorafenib | 88 | ||||||||
| AFP (ng/mL) | 8535 | ||||||||||
| Disease status | PD | ||||||||||
| 9 | CTCs (cells/mL) | Sorafenib | 43 | ||||||||
| AFP (ng/mL) | 656 | ||||||||||
| Disease status | PD | ||||||||||
| 10 | CTCs (cells/mL) | Sorafenib | 74 | 94 | 1148 | 315 | |||||
| AFP (ng/mL) | 146 | − | 231 | 543 | |||||||
| Disease status | − | PD | PD | PD | |||||||
| 11 | CTCs (cells/mL) | Sorafenib | 9 | 19 | |||||||
| AFP (ng/mL) | 4 | 2 | |||||||||
| Disease status | PD | − | |||||||||
| 12 | CTCs (cells/mL) | Sorafenib | 198 | 18 | |||||||
| AFP (ng/mL) | 12,222 | 26,771 | |||||||||
| Disease status | − | SD | |||||||||
| 13 | CTCs (cells/mL) | Sorafenib | 42 | 615 | 902 | ||||||
| AFP (ng/mL) | 421 | 390 | 596 | ||||||||
| Disease status | SD | SD | PD | ||||||||
| 14 | CTCs (cells/mL) | Sorafenib | 109 | ||||||||
| AFP (ng/mL) | 54 | ||||||||||
| Disease status | PD | ||||||||||
| 15 | CTCs (cells/mL) | Sorafenib | 36 | 22 | 266 | ||||||
| AFP (ng/mL) | 12 | 30 | 57 | ||||||||
| Disease status | − | SD | PD | ||||||||
| 16 | CTCs (cells/mL) | IA-CT | 13 | 16 | 5 | ||||||
| AFP (ng/mL) | 27,621 | − | − | ||||||||
| Disease status | PD | − | PD | ||||||||
| 17 | CTCs (cells/mL) | IA-CT | 100 | 10 | 42 | 29 | 34 | 98 | |||
| AFP (ng/mL) | >500,000 | >80,000 | 28,614 | 23,990 | 35,835 | − | |||||
| Disease status | PD | PD | PD | PD | PD | PD | |||||
| 18 | CTCs (cells/mL) | IV-CT | 2 | 12 | |||||||
| AFP (ng/mL) | 6633 | 11,677 | |||||||||
| Disease status | − | PD | |||||||||
| 19 | CTCs (cells/mL) | Ramucirumab | 6 | 5 | 4 | 21 | 3 | ||||
| AFP (ng/mL) | 840 | 11 | 11 | 3 | − | ||||||
| Disease status | − | PR | PR | PR | PR | ||||||
| 20 | CTCs (cells/mL) | Nivolumab; sorafenib after the 2nd blood collection | 12 | 310 | 15 | ||||||
| AFP (ng/mL) | 2491 | 3513 | 4508 | ||||||||
| Disease status | − | PD | PR | ||||||||
| 21 | CTCs (cells/mL) | Ramucirumab | 6 | 16 | |||||||
| AFP (ng/mL) | 14 | 14 | |||||||||
| Disease status | − | PD | |||||||||
| 22 | CTCs (cells/mL) | IA-CT | 139 | 15 | 7 | ||||||
| AFP (ng/mL) | 6453 | 9695 | − | ||||||||
| Disease status | − | SD | SD | ||||||||
| 23 | CTCs (cells/mL) | Sorafenib; IA-VT after the 3rd blood collection | 249 | 59 | 50 | 16 | 5 | ||||
| AFP (ng/mL) | 25 | 20 | 21 | − | 32 | ||||||
| Disease status | SD | PD | − | SD | |||||||
| 24 | CTCs (cells/mL) | Sorafenib | 7 | 64 | 4 | 9 | |||||
| AFP (ng/mL) | 41 | 42 | - | 73 | |||||||
| Disease status | − | PR | SD | PD | |||||||
| 25 | CTCs (cells/mL) | Sorafenib | 7 | 39 | 7 | 4 | 15 | 17 | |||
| AFP (ng/mL) | 14 | − | − | − | 13 | 37 | |||||
| Disease status | − | SD | SD | SD | SD | SD | |||||
| 26 | CTCs (cells/mL) | Sorafenib | 615 | 272 | 117 | ||||||
| AFP (ng/mL) | 4 | − | 4 | ||||||||
| Disease status | − | SD | PR | ||||||||
| 27 | CTCs (cells/mL) | Sorafenib; IA-CT after the 2nd blood collection | 125 | 149 | 173 | 54 | 5 | ||||
| AFP (ng/mL) | 5 | 4 | 3 | − | − | ||||||
| Disease status | − | PD | PR | PR | PR | ||||||
| 28 | CTCs (cells/mL) | Sorafenib | 16 | 18 | 1955 | 165 | |||||
| AFP (ng/mL) | 1254 | 1188 | 1859 | 689 | |||||||
| Disease status | − | PD | PD | PR | |||||||
| 29 | CTCs (cells/mL) | Thalidomide | 134 | 24 | |||||||
| AFP (ng/mL) | 16 | 29 | |||||||||
| Disease status | − | PD | |||||||||
| 30 | CTCs (cells/mL) | Thalidomide | 15 | ||||||||
| AFP (ng/mL) | 67,658 | ||||||||||
| Disease status | SD | ||||||||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rau, K.-M.; Liu, C.-T.; Hsiao, Y.-C.; Hsiao, K.-Y.; Wang, T.-M.; Hung, W.-S.; Su, Y.-L.; Liu, W.-C.; Wang, C.-H.; Hsu, H.-L.; et al. Sequential Circulating Tumor Cell Counts in Patients with Locally Advanced or Metastatic Hepatocellular Carcinoma: Monitoring the Treatment Response. J. Clin. Med. 2020, 9, 188. https://doi.org/10.3390/jcm9010188
Rau K-M, Liu C-T, Hsiao Y-C, Hsiao K-Y, Wang T-M, Hung W-S, Su Y-L, Liu W-C, Wang C-H, Hsu H-L, et al. Sequential Circulating Tumor Cell Counts in Patients with Locally Advanced or Metastatic Hepatocellular Carcinoma: Monitoring the Treatment Response. Journal of Clinical Medicine. 2020; 9(1):188. https://doi.org/10.3390/jcm9010188
Chicago/Turabian StyleRau, Kun-Ming, Chien-Ting Liu, Yu-Chiao Hsiao, Kai-Yin Hsiao, Tzu-Min Wang, Wei-Shan Hung, Yu-Li Su, Wei-Ching Liu, Cheng-Hsu Wang, Hsueh-Ling Hsu, and et al. 2020. "Sequential Circulating Tumor Cell Counts in Patients with Locally Advanced or Metastatic Hepatocellular Carcinoma: Monitoring the Treatment Response" Journal of Clinical Medicine 9, no. 1: 188. https://doi.org/10.3390/jcm9010188
APA StyleRau, K.-M., Liu, C.-T., Hsiao, Y.-C., Hsiao, K.-Y., Wang, T.-M., Hung, W.-S., Su, Y.-L., Liu, W.-C., Wang, C.-H., Hsu, H.-L., Chuang, P.-H., Cheng, J.-C., & Tseng, C.-P. (2020). Sequential Circulating Tumor Cell Counts in Patients with Locally Advanced or Metastatic Hepatocellular Carcinoma: Monitoring the Treatment Response. Journal of Clinical Medicine, 9(1), 188. https://doi.org/10.3390/jcm9010188






