Tadalafil 5 mg Alone or in Combination with Tamsulosin 0.4 mg for the Management of Men with Lower Urinary Tract Symptoms and Erectile Dysfunction: Results of a Prospective Observational Trial
Abstract
:1. Introduction
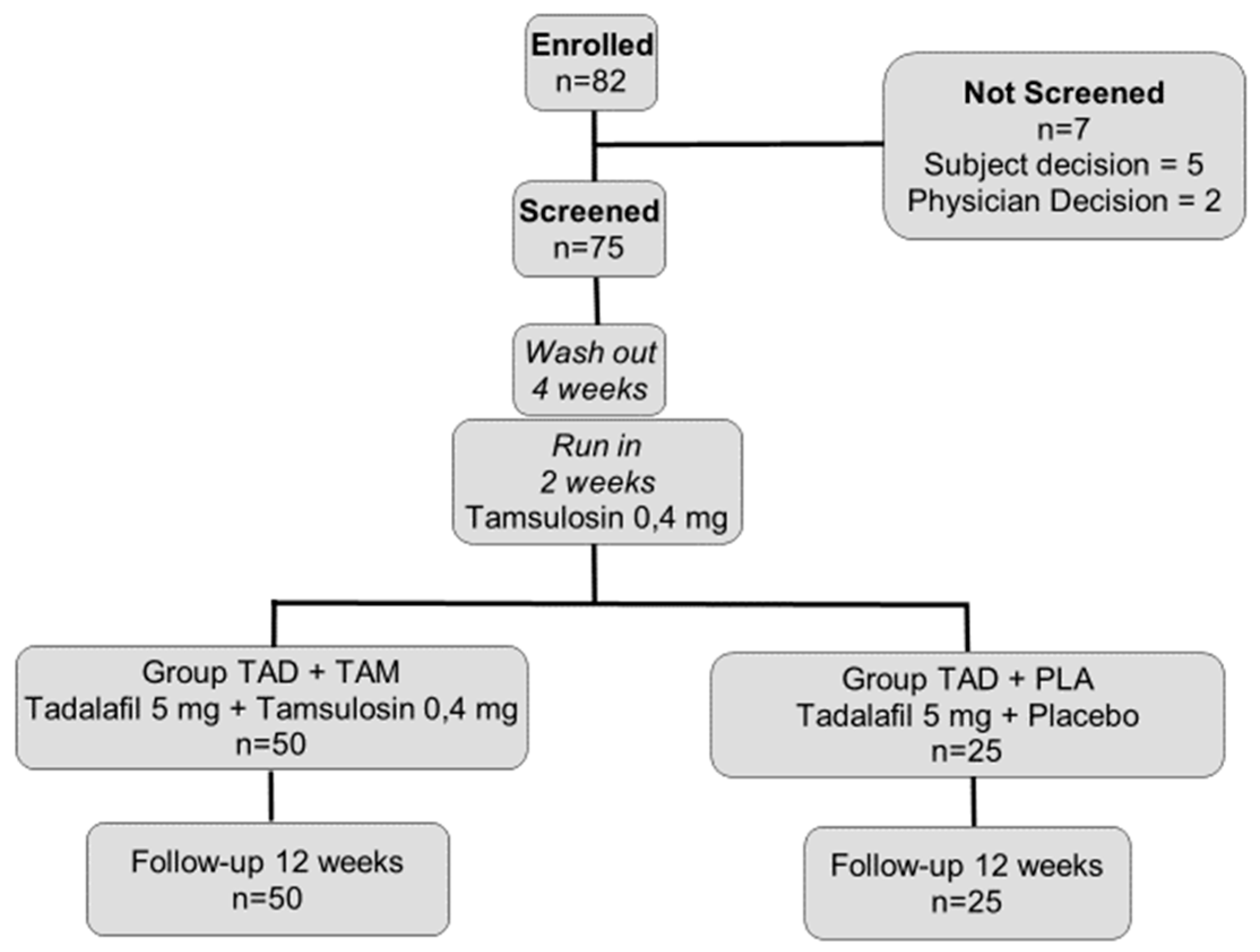
2. Materials and Methods
2.1. Study Population and Design
2.2. Statistical Analysis
3. Results
Experimental Results
4. Discussion
5. Conclusions
Author Contributions
Conflicts of Interest
References
- Gacci, M.; Eardley, I.; Giuliano, F.; Hatzichristou, D.; Kaplan, S.A.; Maggi, M.; McVary, K.T.; Mirone, V.; Porst, H.; Roehrborn, C.G. Critical analysis of the relationship between sexual dysfunctions and lower urinary tract symptoms due to benign prostatic hyperplasia. Eur. Urol. 2011, 60, 809–825. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, J.A.; Muracca, E.; Nakano, É.; Assalin, A.R.; Cordeiro, P.; Paranhos, M.; Cury, J.; Srougi, M.; Antunes, A.A. Interactions between lower urinary tract symptoms and cardiovascular risk factors determine distinct patterns of erectile dysfunction: A latent class analysis. J. Urol. 2013, 190, 2177–2182. [Google Scholar] [CrossRef] [PubMed]
- McVary, K.T. Erectile dysfunction and lower urinary tract symptoms secondary to BPH. Eur. Urol. 2005, 47, 838–845. [Google Scholar] [CrossRef] [PubMed]
- Gravas, S.; Bach, T.; Drake, M.; Gacci, M.; Gratzke, C.; Herrmann, T.R.W.; Madersbacher, S.; Mamoulakis, C.; Tikkinen, K.A.O. Treatment of Non-Neurogenic Male LUTS. 2017. Available online: http://uroweb.org/guideline/treatment-of-non-neurogenic-male-luts/ (accessed on 7 May 2019).
- Hedlund, H.; Andersson, K.E.; Larsson, B. Alpha-adrenoceptors and muscarinic receptors in the isolated human prostate. J. Urol. 1985, 134, 1291–1298. [Google Scholar] [CrossRef]
- Abrams, P.; Schulman, C.C.; Vaage, S. The European Tamsulosin Study Group. Tamsulosin, a selective alpha 1c-adrenoceptor antagonist: A randomized, controlled trial in patients with benign prostatic ‘obstruction’ (symptomatic BPH). Br. J. Urol. 1995, 76, 325–336. [Google Scholar] [CrossRef] [PubMed]
- Gacci, M.; Ficarra, V.; Sebastianelli, A.; Corona, G.; Serni, S.; Shariat, S.F.; Maggi, M.; Zattoni, F.; Carini, M.; Novara, G. Impact of medical treatments for male lower urinary tract symptoms due to benign prostatic hyperplasia on ejaculatory function: A systematic review and meta-analysis. J. Sex. Med. 2014, 11, 1554–1566. [Google Scholar] [CrossRef]
- Corona, G.; Tirabassi, G.; Santi, D.; Maseroli, E.; Gacci, M.; Dicuio, M.; Sforza, A.; Mannucci, E.; Maggi, M. Sexual dysfunction in subjects treated with inhibitors of 5α-reductase for benign prostatic hyperplasia: A comprehensive review and meta-analysis. Andrology 2017, 5, 671–678. [Google Scholar] [CrossRef] [PubMed]
- Mirone, V.; Sessa, A.; Giuliano, F.; Berges, R.; Kirby, M.; Moncada, I. Current benign prostatic hyperplasia treatment: Impact on sexual function and management of related sexual adverse events. Int. J. Clin. Pract. 2011, 65, 1005–1013. [Google Scholar] [CrossRef]
- McVary, K.T.; Roehrborn, C.G.; Kaminetsky, J.C.; Auerbach, S.; Wachs, B.; Young, J.; Esler, A.; Sides, G.; Denes, B. Tadalafil relieves lower urinary tract symptoms secondary to benign prostatic hyperplasia. J. Urol. 2007, 177, 1401–1407. [Google Scholar] [CrossRef]
- Roehrborn, C.G.; McVary, K.T.; Elion-Mboussa, A.; Viktrup, L. Tadalafil administered once daily for lower urinary tract symptoms secondary to benign prostatic hyperplasia: A dose finding study. J. Urol. 2008, 180, 1228–1234. [Google Scholar] [CrossRef]
- Broderick, G.A.; Brock, G.B.; Roehrborn, C.G.; Watts, S.D.; Elion-Mboussa, A.; Viktrup, L. Effects of tadalafil on lower urinary tract symptoms secondary to benign prostatic hyperplasia in men with or without erectile dysfunction. Urology 2010, 75, 1452–1458. [Google Scholar] [CrossRef] [PubMed]
- Porst, H.; Kim, E.D.; Casabé, A.R.; Mirone, V.; Secrest, R.J.; Xu, L.; Sundin, D.P.; Viktrup, L. LVHJ Study Team. Efficacy and safety of tadalafil once daily in the treatment of men with lower urinary tract symptoms suggestive of benign prostatic hyperplasia: Results of an international randomized, double-blind, placebo-controlled trial. Eur. Urol. 2011, 60, 1105–1113. [Google Scholar] [CrossRef] [PubMed]
- Gacci, M.; Andersson, K.E.; Chapple, C.; Maggi, M.; Mirone, V.; Oelke, M.; Porst, H.; Roehrborn, C.; Stief, C.; Giuliano, F. Latest Evidence on the Use of Phosphodiesterase Type 5 Inhibitors for the Treatment of Lower Urinary Tract Symptoms Secondary to Benign Prostatic Hyperplasia. Eur. Urol. 2016, 70, 124–133. [Google Scholar] [CrossRef] [PubMed]
- Filippi, S.; Morelli, A.; Sandner, P.; Fibbi, B.; Mancina, R.; Marini, M.; Gacci, M.; Vignozzi, L.; Vannelli, G.B.; Carini, M.; et al. Characterization and functional role of androgendependent PDE5 activity in the bladder. Endocrinology 2007, 148, 1019–1029. [Google Scholar] [CrossRef] [PubMed]
- Vignozzi, L.; Gacci, M.; Cellai, I.; Morelli, A.; Maneschi, E.; Comeglio, P.; Santi, R.; Filippi, S.; Sebastianelli, A.; Nesi, G.; et al. PDE5 inhibitors blunt inflammation in human BPH: A potential mechanism of action for PDE5 inhibitors in LUTS. Prostate 2013, 73, 1391–1402. [Google Scholar] [CrossRef] [PubMed]
- Morelli, A.; Comeglio, P.; Filippi, S.; Sarchielli, E.; Vignozzi, L.; Maneschi, E.; Cellai, I.; Gacci, M.; Lenzi, A.; Vannelli, G.B.; et al. Mechanism of action of phosphodiesterase type 5 inhibition in metabolic syndrome-associated prostate alterations: An experimental study in the rabbit. Prostate 2013, 73, 428–441. [Google Scholar] [CrossRef] [PubMed]
- Gacci, M.; Corona, G.; Salvi, M.; Vignozzi, L.; McVary, K.T.; Kaplan, S.A.; Roehrborn, C.G.; Serni, S.; Mirone, V.; Carini, M.; et al. A systematic review and meta-analysis on the use of phosphodiesterase 5 inhibitors alone or in combination with α-blockers for lower urinary tract symptoms due to benign prostatic hyperplasia. Eur. Urol. 2012, 61, 994–1003. [Google Scholar] [CrossRef] [PubMed]
- Barry, M.J.; Fowler, F.J.; O’Leary, M.P.; Bruskewitz, R.C.; Holtgrewe, H.L.; Mebust, W.K.; Cockett, A.T. Measurement Committee of the American Urological Association. The American Urological Association symptom index for benign prostatic hyperplasia. J. Urol. 1992, 148, 1549–1557. [Google Scholar] [CrossRef]
- Rosen, R.C.; Riley, A.; Wagner, G.; Osterloh, I.H.; Kirkpatrick, J.; Mishra, A. The International Index of Erectile Function (IIEF): A multidimensional scale for assessment of erectile dysfunction. Urology 1997, 49, 822–830. [Google Scholar] [CrossRef]
- Gacci, M.; Corona, G.; Vignozzi, L.; Salvi, M.; Serni, S.; De Nunzio, C.; Tubaro, A.; Oelke, M.; Carini, M.; Maggi, M. Metabolic syndrome and benign prostatic enlargement: A systematic review and meta-analysis. BJU Int. 2015, 115, 24–31. [Google Scholar] [CrossRef]
- Andersson, K.E. Storage and voiding symptoms: Pathophysiologic aspects. Urology 2003, 62 (Suppl. 2), 3–10. [Google Scholar] [CrossRef]
- Gacci, M.; Sebastianelli, A.; Spatafora, P.; Corona, G.; Serni, S.; De Ridder, D.; Gravas, S.; Abrams, P. Best practice in the management of storage symptoms in male lower urinary tract symptoms: A review of the evidence base. Ther. Adv. Urol. 2017, 10, 79–92. [Google Scholar] [CrossRef] [PubMed]
- Egerdie, R.B.; Auerbach, S.; Roehrborn, C.G.; Costa, P.; Garza, M.S.; Esler, A.L.; Wong, D.G.; Secrest, R.J. Tadalafil 2.5 or 5 mg administered once daily for 12 weeks in men with both erectile dysfunction and signs and symptoms of benign prostatic hyperplasia: Results of a randomized, placebo-controlled, double-blind study. J. Sex. Med. 2012, 9, 271. [Google Scholar] [CrossRef]
- Roehrborn, C.G.; Chapple, C.; Oelke, M.; Cox, D.; Esler, A.; Viktrup, L. Effects of tadalafil once daily on maximum urinary flow rate in men with lower urinary tract symptoms suggestive of benign prostatic hyperplasia. J. Urol. 2014, 191, 1045. [Google Scholar] [CrossRef]
- Yokoyama, O.; Yoshida, M.; Kim, S.C.; Wang, C.J.; Imaoka, T.; Morisaki, Y.; Viktrup, L. Tadalafil once daily for lower urinary tract symptoms suggestive of benign prostatic hyperplasia: A randomized placebo- and tamsulosin-controlled 12-week study in Asian men. Int. J. Urol. 2013, 20, 193. [Google Scholar] [CrossRef] [PubMed]
- Alan, W.; Shindel, M.D. 2009 update on phosphodiesterase type 5 inhibitor therapy part 1: Recent studies on routine dosing for penile rehabilitation, lower urinary tract symptoms, and other indications. J. Sex. Med. 2009, 6, 1794–1808. [Google Scholar]
- Laydner, H.K.; Oliveira, P.; Oliveira, C.R.; Makarawo, T.P.; Andrade, W.S.; Tannus, M.; Araújo, J.L. Phosphodiesterase 5 inhibitors for lower urinary tract symptoms secondary to benign prostatic hyperplasia: A systematic review. BJU Int. 2011, 107, 1104–1109. [Google Scholar] [CrossRef] [PubMed]
- Oelke, M.; Giuliano, F.; Mirone, V.; Xu, L.; Cox, D.; Viktrup, L. Monotherapy with tadalafil or tamsulosin similarly improved lower urinary tract symptoms suggestive of benign prostatic hyperplasia in an international, randomised, parallel, placebo-controlled clinical trial. Eur. Urol. 2012, 61, 917–925. [Google Scholar] [CrossRef]
- Oger, S.; Behr-Roussel, D.; Gorny, D.; Lecoz, O.; Lebret, T.; Denoux, Y.; Faix, A.; Leriche, A.; Wayman, C.; Alexandre, L.; et al. Combination of doxazosin and sildenafil exerts an additive relaxing effect compared to each compound alone on human cavernosal and prostatic tissue. J. Sex. Med. 2009, 6, 836–847. [Google Scholar] [CrossRef]
- Liguori, G.; Trombetta, C.; De Giorgi, G.; Pomara, G.; Maio, G.; Vecchio, D.; Ocello, G.; Ollandini, G.; Bucci, S.; Belgrano, E. Efficacy and safety of combined oral therapy with tadalafil and alfuzosin: An integrated approach to the preliminary report. J. Sex. Med. 2009, 6, 544–552. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.; Zong, H.; Cui, Y.; Li, N.; Zhang, Y. The efficacy of PDE5 inhibitors alone or in combination with alpha-blockers for the treatment of erectile dysfunction and lower urinary tract symptoms due to benign prostatic hyperplasia: A systematic review and meta-analysis. J. Sex. Med. 2014, 11, 1539–1545. [Google Scholar] [CrossRef]
- McVary, K.T.; Monnig, W.; Camps, J.L.; Young, J.M.; Tseng, L.J.; van den Ende, G. Sildenafil citrate improves erectile function and urinary symptoms in men with erectile dysfunction and lower urinary tract symptoms associated with benign prostatic hyperplasia: A randomized double-blind trial. J. Urol. 2007, 177, 1071–1077. [Google Scholar] [CrossRef]
- Tuncel, A.; Nalcacioglu, V.; Ener, K.; Aslan, Y.; Aydin, O.; Atan, A. Sildenafil citrate and tamsulosin combination is not superior to monotherapy in treating lower urinary tract symptoms and erectile dysfunction. World J. Urol. 2010, 28, 17–22. [Google Scholar] [CrossRef]
- Kaplan, S.; Gonzalez, R.; Te, A. Combination of alfuzosin and sildenafil is superior to monotherapy in treating lower urinary tract symptoms and erectile dysfunction. Eur. Urol. 2007, 51, 1717–1723. [Google Scholar] [CrossRef]
- Bechara, A.; Romano, S.; Casabé, A.; Haime, S.; Dedola, P.; Hernández, C.; Rey, H. Comparative efficacy assessment of tamsulosin vs. tamsulosin plus tadalafil in the treatment of LUTS/BPH. Pilot study. J. Sex. Med. 2008, 5, 2170–2178. [Google Scholar] [CrossRef]
- Chung, B.H.; Lee, J.Y.; Lee, S.H.; Yoo, S.J.; Lee, S.W.; Oh, C.Y. Safety and efficacy of the simultaneous administration of udenafil and an alpha-blocker in men with erectile dysfunction concomitant with BPH/LUTS. Int. J. Impot. Res. 2009, 21, 122–128. [Google Scholar] [CrossRef]
- Kloner, R.; Jackson, G.; Emmick, J.; Mitchell, M.; Bedding, A.; Warner, L.; Pereira, A. Interaction between the phosphodiesterase 5 inhibitor. Tadalafil and 2 alfa-blockers, doxazosin and tamsulosin in healthy normotensive men. J. Urol. 2004, 172, 1935–1940. [Google Scholar] [CrossRef]
| All Patients | TAD + TAM Group n = 50 | TAD + PLA Group n = 25 | p | ||
|---|---|---|---|---|---|
| Baseline Characteristics | Mean ± SD Deviation | (Minimum-Maximum) | Mean ± SD Deviation | (Minimum-Maximum) | |
| Age (years) | 65.7 ± 9.1 | 47–78 | 65.5 ± 6.3 | 51–74 | 0.238 |
| Weight (kg) | 78.2 ± 9.2 | 69–86 | 75.8 ± 10.4 | 67–83 | 0.216 |
| Body mass index (kg/m2) | 27 ± 3.1 | 25–31 | 26.7 ± 3.6 | 24–31 | 0.174 |
| Abdominal obesity: waist circumference (cm) | 108.8 ± 4.1 | 92–135 | 102.3 ± 5.4 | 76–120 | 0.136 |
| Triglycerides (mg/dL) | 156.7 ± 8.4 | 76–247 | 129.2 ± 7.2 | 83–185 | 0.117 |
| HDL cholesterol (mg/dL) | 49.4 ± 2.8 | 31–76 | 49.8 ± 2.3 | 32–60 | 0.259 |
| Glycemia (mg/dL) | 111.3 ± 3.5 | 76–211 | 102.7 ± 5.7 | 72–188 | 0.113 |
| IPSS base | 18.8 ± 5.9 | 8–32 | 17 ± 6.1 | 8–29 | 0.224 |
| IPSS voiding base | 8.6 ± 3.8 | 1–20 | 10 ± 4.1 | 3–18 | 0.146 |
| IPSS storage base | 8.3 ± 3.2 | 0–14 | 6.9 ± 4.2 | 1–15 | 0.118 |
| IPSS QoL base | 3.9 ± 1 | 2–6 | 3.5 ± 1.4 | 1–6 | 0.145 |
| IIEF-5 base | 12 ± 3.5 | 6–21 | 13.8 ± 5.2 | 1–21 | 0.09 |
| Q max base (mL/s) | 10.3 ± 3.5 | 3.4–17.8 | 9.6 ± 2.8 | 6–17 | 0.369 |
| Variables Assesed | TAD + TAM Group n = 50 | TAD + PLA Group n = 25 | p Value (Anova Analysis) |
|---|---|---|---|
| IPSS 12 week (Mean ± SD) | 11.5 ± 5.4 | 11.8 ± 6.3 | |
| Delta3 M (baseline - 12wks) | −7 | −5.2 | |
| p value (paired samples T-test) | <0.001 | <0.001 | 0.084 |
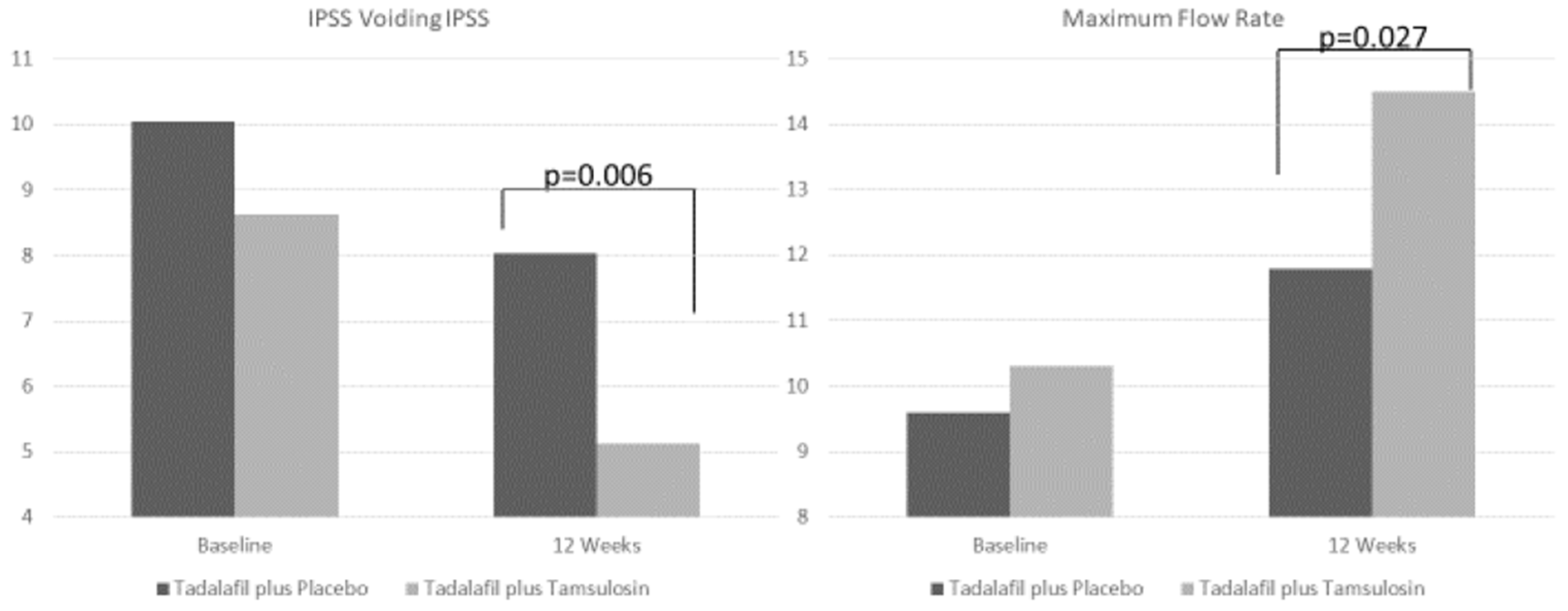
| IPSS voiding 12 week (Mean ± SD) | 5.1 ± 2.7 | 8 ± 4.7 | |
| Delta3 M (baseline - 12wks) | −3.5 | −2 | |
| p value (paired samples T-test) | <0.001 | 0.074 | 0.006 |
| IPSS storage 12 week (Mean ± SD) | 5.3 ± 2.7 | 3.8 ± 3.4 | |
| Delta3 M (baseline - 12wks) | −3 | −3.1 | |
| p value (paired samples T-test) | <0.001 | <0.001 | 0.08 |
| IPSS QoL 12 week (Mean ± SD) | 2.1 ± 1 | 2.1 ± 1.7 | |
| Delta3 M (baseline - 12wks) | −1.8 | −1.3 | |
| p value (paired samples T-test) | <0.001 | 0.009 | 0.321 |
| IIEF-5 12 week (Mean ± SD) | 17.7 ± 3.3 | 19.9 ± 5.1 | |
| Delta3 M (baseline - 12wks) | 5.7 | 6.1 | |
| p value (paired samples T-test) | <0.001 | <0.001 | 0.255 |
| Q max 12 week (Mean ± SD) | 14.5 ± 3.7 | 11.8 ± 4 | |
| Delta3 M (baseline - 12wks) | 4.2 | 2.2 | |
| p value (paired samples T-test) | <0.001 | <0.001 | 0.027 |
| Adverse Events | TAD + TAM Group n = 50, (n %) | TAD + PLA Group n = 25, (n %) | p Value |
|---|---|---|---|
| Any TEAEs | 11 (22%) | 4 (16%) | 0.075 |
| Serious AEs | 0 (0%) | 0 (0%) | 0.267 |
| Intensity | |||
| mild | 7 (14%) | 3 (12%) | 0.114 |
| moderate | 4 (8%) | 1 (4%) | 0.098 |
| severe | 0 (0%) | 0 (0%) | 0.286 |
| Headache | 4 (8%) | 2 (8%) | 0.163 |
| Nasopharyngitis | 0 (0%) | 1 (4%) | 0.196 |
| Back pain | 3 (6%) | 1(4%) | 0.087 |
| Dizziness | 1 (2%) | 0 (0%) | 0.173 |
| Dyspepsia | 1 (2%) | 0 (0%) | 0.185 |
| Ejaculatory dysfunction | 2 (4%) | 0 (0%) | 0.072 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sebastianelli, A.; Spatafora, P.; Frizzi, J.; Saleh, O.; Sessa, M.; De Nunzio, C.; Tubaro, A.; Vignozzi, L.; Maggi, M.; Serni, S.; et al. Tadalafil 5 mg Alone or in Combination with Tamsulosin 0.4 mg for the Management of Men with Lower Urinary Tract Symptoms and Erectile Dysfunction: Results of a Prospective Observational Trial. J. Clin. Med. 2019, 8, 1126. https://doi.org/10.3390/jcm8081126
Sebastianelli A, Spatafora P, Frizzi J, Saleh O, Sessa M, De Nunzio C, Tubaro A, Vignozzi L, Maggi M, Serni S, et al. Tadalafil 5 mg Alone or in Combination with Tamsulosin 0.4 mg for the Management of Men with Lower Urinary Tract Symptoms and Erectile Dysfunction: Results of a Prospective Observational Trial. Journal of Clinical Medicine. 2019; 8(8):1126. https://doi.org/10.3390/jcm8081126
Chicago/Turabian StyleSebastianelli, Arcangelo, Pietro Spatafora, Jacopo Frizzi, Omar Saleh, Maurizio Sessa, Cosimo De Nunzio, Andrea Tubaro, Linda Vignozzi, Mario Maggi, Sergio Serni, and et al. 2019. "Tadalafil 5 mg Alone or in Combination with Tamsulosin 0.4 mg for the Management of Men with Lower Urinary Tract Symptoms and Erectile Dysfunction: Results of a Prospective Observational Trial" Journal of Clinical Medicine 8, no. 8: 1126. https://doi.org/10.3390/jcm8081126





