Levamisole in Children with Idiopathic Nephrotic Syndrome: Clinical Efficacy and Pathophysiological Aspects
Abstract
1. Introduction
2. Side Effects of Traditional Immunosuppressive Agents in the Treatment of Nephrotic Syndrome
3. The Use of Levamisole in the Treatment of Nephrotic Syndrome
4. Known Side Effects of Levamisole Treatment
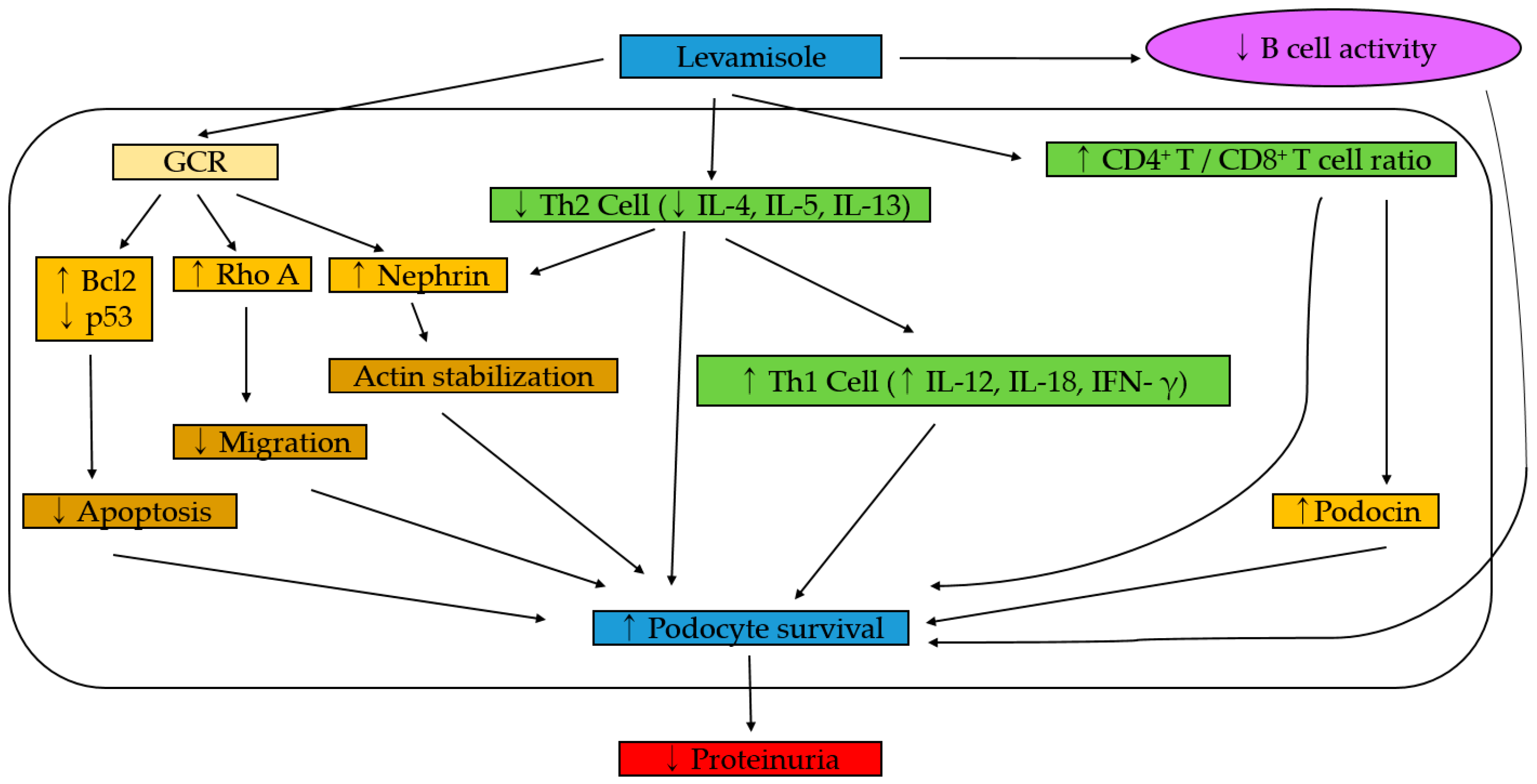
5. Mechanism Hypothesis
6. Summary and Conclusions
Author Contributions
Conflicts of Interest
References
- Eddy, A.A.; Symons, J.M. Nephrotic syndrome in childhood. Lancet 2003, 362, 629–639. [Google Scholar] [CrossRef]
- Kemper, M.J.; Zepf, K.; Klaassen, I.; Link, A.; Muller-Wiefel, D.E. Changes of lymphocyte populations in pediatric steroid-sensitive nephrotic syndrome are more pronounced in remission than in relapse. Am. J. Nephrol. 2005, 25, 132–137. [Google Scholar] [CrossRef] [PubMed]
- Shalaby, S.A.; Al-Edressi, H.M.; El-Tarhouny, S.A.; Fath El-Bab, M.; Zolaly, M.A. Type 1/type 2 cytokine serum levels and role of interleukin-18 in children with steroid-sensitive nephrotic syndrome. Arab. J. Nephrol. Transpl. 2013, 6, 83–88. [Google Scholar]
- Greka, A.; Mundel, P. Cell biology and pathology of podocytes. Annu. Rev. Physiol. 2012, 74, 299–323. [Google Scholar] [CrossRef] [PubMed]
- Van Husen, M.; Kemper, M.J. New therapies in steroid-sensitive and steroid-resistant idiopathic nephrotic syndrome. Pediatr. Nephrol. 2011, 26, 881–892. [Google Scholar] [CrossRef] [PubMed]
- Sureshkumar, P.; Hodson, E.M.; Willis, N.S.; Barzi, F.; Craig, J.C. Predictors of remission and relapse in idiopathic nephrotic syndrome: A prospective cohort study. Pediatr. Nephrol. 2014, 29, 1039–1046. [Google Scholar] [CrossRef] [PubMed]
- Ruth, E.M.; Kemper, M.J.; Leumann, E.P.; Laube, G.F.; Neuhaus, T.J. Children with steroid-sensitive nephrotic syndrome come of age: Long-term outcome. J. Pediatr. 2005, 147, 202–207. [Google Scholar] [CrossRef]
- The primary nephrotic syndrome in children. Identification of patients with minimal change nephrotic syndrome from initial response to prednisone. A report of the International Study of Kidney Disease in Children. J. Pediatr. 1981, 98, 561–564. [Google Scholar] [CrossRef]
- Arbeitsgemeinschaft für Pädiatrische Nephrologie. Short versus standard prednisone therapy for initial treatment of idiopathic nephrotic syndrome in children. Lancet 1988, 1, 380–383. [Google Scholar] [CrossRef]
- Ehrich, J.H.; Brodehl, J. Long versus standard prednisone therapy for initial treatment of idiopathic nephrotic syndrome in children. Eur. J. Pediatr. 1993, 152, 357–361. [Google Scholar] [CrossRef]
- Tarshish, P.; Tobin, J.N.; Bernstein, J.; Edelmann, C.M., Jr. Prognostic significance of the early course of minimal change nephrotic syndrome: Report of the International Study of Kidney Disease in Children. J. Am. Soc. Nephrol. 1997, 8, 769–776. [Google Scholar] [PubMed]
- Kyrieleis, H.A.; Lowik, M.M.; Pronk, I.; Cruysberg, H.R.; Kremer, J.A.; Oyen, W.J.; van den Heuvel, B.L.; Wetzels, J.F.; Levtchenko, E.N. Long-term outcome of biopsy-proven, frequently relapsing minimal-change nephrotic syndrome in children. Clin. J. Am. Soc. Nephrol. 2009, 4, 1593–1600. [Google Scholar] [CrossRef] [PubMed]
- Poetker, D.M.; Reh, D.D. A comprehensive review of the adverse effects of systemic corticosteroids. Otolaryngol. Clin. N. Am. 2010, 43, 753–768. [Google Scholar] [CrossRef] [PubMed]
- Fardet, L.; Cabane, J.; Lebbe, C.; Morel, P.; Flahault, A. Incidence and risk factors for corticosteroid-induced lipodystrophy: A prospective study. J. Am. Acad. Derm. 2007, 57, 604–609. [Google Scholar] [CrossRef] [PubMed]
- Larkins, N.; Kim, S.; Craig, J.; Hodson, E. Steroid-sensitive nephrotic syndrome: An evidence-based update of immunosuppressive treatment in children. Arch. Dis. Child. 2016, 101, 404–408. [Google Scholar] [CrossRef] [PubMed]
- Martin, F.; Lauwerys, B.; Lefebvre, C.; Devogelaer, J.P.; Houssiau, F.A. Side-effects of intravenous cyclophosphamide pulse therapy. Lupus 1997, 6, 254–257. [Google Scholar] [CrossRef] [PubMed]
- Tedesco, D.; Haragsim, L. Cyclosporine: A review. J. Transpl. 2012, 2012, 230386. [Google Scholar] [CrossRef] [PubMed]
- Kitchin, J.E.; Pomeranz, M.K.; Pak, G.; Washenik, K.; Shupack, J.L. Rediscovering mycophenolic acid: A review of its mechanism, side effects, and potential uses. J. Am. Acad. Derm. 1997, 37, 445–449. [Google Scholar] [CrossRef]
- Mihatsch, M.J.; Kyo, M.; Morozumi, K.; Yamaguchi, Y.; Nickeleit, V.; Ryffel, B. The side-effects of ciclosporine-A and tacrolimus. Clin. Nephrol. 1998, 49, 356–363. [Google Scholar]
- Kar, H.K.; Bhatia, V.N.; Kumar, C.H.; Sirumban, P.; Roy, R.G. Evaluation of levamisole, an immunopotentiator, in the treatment of lepromatous leprosy. Indian J. Lepr. 1986, 58, 592–600. [Google Scholar]
- Mutch, R.S.; Hutson, P.R. Levamisole in the adjuvant treatment of colon cancer. Clin. Pharm. 1991, 10, 95–109. [Google Scholar] [CrossRef] [PubMed]
- Tanphaichitr, P.; Tanphaichitr, D.; Sureeratanan, J.; Chatasingh, S. Treatment of nephrotic syndrome with levamisole. J. Pediatr. 1980, 96, 490–493. [Google Scholar] [CrossRef]
- Bagga, A.; Sharma, A.; Srivastava, R.N. Levamisole therapy in corticosteroid-dependent nephrotic syndrome. Pediatr. Nephrol. 1997, 11, 415–417. [Google Scholar] [CrossRef] [PubMed]
- Neuhaus, T.J.; Fay, J.; Dillon, M.J.; Trompeter, R.S.; Barratt, T.M. Alternative treatment to corticosteroids in steroid sensitive idiopathic nephrotic syndrome. Arch. Dis. Child. 1994, 71, 522–526. [Google Scholar] [CrossRef] [PubMed]
- British Association for Paediatric Nephrology. Levamisole for corticosteroid-dependent nephrotic syndrome in childhood. Lancet 1991, 337, 1555–1557. [Google Scholar] [CrossRef]
- Kemper, M.J.; Amon, O.; Timmermann, K.; Altrogge, H.; Muller-Wiefel, D.E. The treatment with levamisole of frequently recurring steroid-sensitive idiopathic nephrotic syndrome in children. Dtsch. Med. Wochenschr. 1998, 123, 239–243. [Google Scholar] [CrossRef] [PubMed]
- Bagga, A.; Ali, U.; Banerjee, S.; Kanitkar, M.; Phadke, K.D.; Senguttuvan, P.; Sethi, S.; Shah, M. Management of steroid sensitive nephrotic syndrome: Revised guidelines. Indian Pediatr. 2008, 45, 203–214. [Google Scholar] [CrossRef]
- Gruppen, M.P.; Bouts, A.H.; Jansen-van der Weide, M.C.; Merkus, M.P.; Zurowska, A.; Maternik, M.; Massella, L.; Emma, F.; Niaudet, P.; Cornelissen, E.A.M.; et al. A randomized clinical trial indicates that levamisole increases the time to relapse in children with steroid-sensitive idiopathic nephrotic syndrome. Kidney Int. 2018, 93, 510–518. [Google Scholar] [CrossRef]
- Kemper, M.J.; Neuhaus, T.J. Levamisole in relapsing steroid-sensitive nephrotic syndrome: Where do we stand? Kidney Int. 2018, 93, 310–313. [Google Scholar] [CrossRef]
- Sinha, A.; Puraswani, M.; Kalaivani, M.; Goyal, P.; Hari, P.; Bagga, A. Efficacy and safety of mycophenolate mofetil versus levamisole in frequently relapsing nephrotic syndrome: An open-label randomized controlled trial. Kidney Int. 2019, 95, 210–218. [Google Scholar] [CrossRef]
- Pravitsitthikul, N.; Willis, N.S.; Hodson, E.M.; Craig, J.C. Non-corticosteroid immunosuppressive medications for steroid-sensitive nephrotic syndrome in children. Cochrane Database Syst. Rev. 2013, Cd002290. [Google Scholar] [CrossRef] [PubMed]
- Hodson, E.M.; Craig, J.C.; Willis, N.S. Evidence-based management of steroid-sensitive nephrotic syndrome. Pediatr. Nephrol. 2005, 20, 1523–1530. [Google Scholar] [CrossRef] [PubMed]
- Davin, J.C.; Merkus, M.P. Levamisole in steroid-sensitive nephrotic syndrome of childhood: The lost paradise? Pediatr. Nephrol. 2005, 20, 10–14. [Google Scholar] [CrossRef] [PubMed]
- Donia, A.F.; Ammar, H.M.; El-Agroudy Ael, B.; Moustafa Fel, H.; Sobh, M.A. Long-term results of two unconventional agents in steroid-dependent nephrotic children. Pediatr. Nephrol. 2005, 20, 1420–1425. [Google Scholar] [CrossRef] [PubMed]
- Ekambaram, S.; Mahalingam, V.; Nageswaran, P.; Udani, A.; Geminiganesan, S.; Priyadarshini, S. Efficacy of levamisole in children with frequently relapsing and steroid-dependent nephrotic syndrome. Indian Pediatr. 2014, 51, 371–373. [Google Scholar] [CrossRef] [PubMed]
- Kuzma-Mroczkowska, E.; Skrzypczyk, P.; Panczyk-Tomaszewska, M. Levamisole therapy in children with frequently relapsing and steroid-dependent nephrotic syndrome: A single-center experience. Cent. Eur. J. Immunol. 2016, 41, 243–247. [Google Scholar] [CrossRef] [PubMed]
- Boyer, O.; Moulder, J.K.; Grandin, L.; Somers, M.J. Short- and long-term efficacy of levamisole as adjunctive therapy in childhood nephrotic syndrome. Pediatr. Nephrol. 2008, 23, 575–580. [Google Scholar] [CrossRef]
- Madani, A.; Isfahani, S.T.; Rahimzadeh, N.; Fereshtehnejad, S.M.; Hoseini, R.; Moghtaderi, M.; Mohseni, P.; Ataiee, N. Effect of levamisole in steroid-dependent nephrotic syndrome. Iran. J. Kidney Dis. 2010, 4, 292–296. [Google Scholar]
- Basu, B.; Babu, B.G.; Mahapatra, T.K. Long-term efficacy and safety of common steroid-sparing agents in idiopathic nephrotic children. Clin. Exp. Nephrol. 2017, 21, 143–151. [Google Scholar] [CrossRef]
- Elmas, A.T.; Tabel, Y.; Elmas, O.N. Short- and long-term efficacy of levamisole in children with steroid-sensitive nephrotic syndrome. Int. Urol. Nephrol. 2013, 45, 1047–1055. [Google Scholar] [CrossRef]
- Kreeftmeijer-Vegter, A.R.; Dorlo, T.P.; Gruppen, M.P.; de Boer, A.; de Vries, P.J. Population pharmacokinetics of levamisole in children with steroid-sensitive nephrotic syndrome. Br. J. Clin. Pharm. 2015, 80, 242–252. [Google Scholar] [CrossRef] [PubMed]
- Niaudet, P.; Drachman, R.; Gagnadoux, M.F.; Broyer, M. Treatment of idiopathic nephrotic syndrome with levamisole. Acta Paediatr. Scand. 1984, 73, 637–641. [Google Scholar] [CrossRef] [PubMed]
- Mehta, K.P.; Ali, U.; Kutty, M.; Kolhatkar, U. Immunoregulatory treatment for minimal change nephrotic syndrome. Arch. Dis. Child. 1986, 61, 153–158. [Google Scholar] [CrossRef] [PubMed]
- La Manna, A.; Polito, C.; Del Gado, R.; Foglia, A.C. Levamisole in children’s idiopathic nephrotic syndrome. Child. Nephrol. Urol. 1988, 9, 200–202. [Google Scholar] [PubMed]
- Srivastava, R.N.; Vasudev, A.S.; Bagga, A. Levamisole in nephrotic syndrome. Lancet 1991, 338, 1275. [Google Scholar] [CrossRef]
- Meregalli, P.; Bianchetti, M.G.; Imoberdorf, G.; Lutschg, J.; Reymond, D.; Oetliker, O.H. Levamisole in children with frequently recurring idiopathic nephrotic syndrome. Schweiz. Med. Wochenschr. 1994, 124, 801–805. [Google Scholar]
- Ksiazek, J.; Krynski, J. Evaluation of the efficacy of levamisole in corticosteroid-dependent nephrotic syndrome in children. Pediatr. Pol. 1995, 70, 1037–1042. [Google Scholar]
- Ginevri, F.; Trivelli, A.; Ciardi, M.R.; Ghiggeri, G.M.; Parfumo, F.; Gusmano, R. Protracted levamisole in children with frequent-relapse nephrotic syndrome. Pediatr. Nephrol. 1996, 10, 550. [Google Scholar]
- Fu, L.S.; Chi, C.S. Levamisole in steroid-sensitive nephrotic syndrome children with steroid-dependency and/or frequent relapses. Acta Paediatr. Taiwan 2000, 41, 80–84. [Google Scholar]
- Alsaran, K.; Grisaru, S.; Stephens, D.; Arbus, G. Levamisole vs. cyclophosphamide for frequently-relapsing steroid-dependent nephrotic syndrome. Clin. Nephrol. 2001, 56, 289–294. [Google Scholar]
- Donia, A.F.; Amer, G.M.; Ahmed, H.A.; Gazareen, S.H.; Moustafa, F.E.; Shoeib, A.A.; Ismail, A.M.; Khamis, S.; Sobh, M.A. Levamisole: Adjunctive therapy in steroid dependent minimal change nephrotic children. Pediatr. Nephrol. 2002, 17, 355–358. [Google Scholar] [CrossRef] [PubMed]
- Al-Ibrahim, A.A.; Al-Kharraz, S.M.; Al-Sadoon, D.M.; Al-Madani, A.J.; Al-Musallam, S.A. Levamisole Therapy as a Second-line Immunosuppressive Agent in Corticosteroid-sensitive Nephrotic Syndrome in Children. Saudi J. Kidney Dis. Transpl. 2003, 14, 153–157. [Google Scholar] [PubMed]
- Sumegi, V.; Haszon, I.; Ivanyi, B.; Bereczki, C.; Papp, F.; Turi, S. Long-term effects of levamisole treatment in childhood nephrotic syndrome. Pediatr. Nephrol. 2004, 19, 1354–1360. [Google Scholar] [CrossRef] [PubMed]
- Fu, L.S.; Shien, C.Y.; Chi, C.S. Levamisole in steroid-sensitive nephrotic syndrome children with frequent relapses and/or steroid dependency: Comparison of daily and every-other-day usage. Nephron Clin. Pract. 2004, 97, c137–c141. [Google Scholar] [CrossRef] [PubMed]
- Hafeez, F.; Ahmed, T.M.; Samina, U. Levamisole in steroid dependent and frequently relapsing nephrotic syndrome. J. Coll. Physicians Surg. Pak. 2006, 16, 35–37. [Google Scholar] [PubMed]
- Chen, S.Y.; Wu, C.Y.; Tsai, I.J.; Tsau, Y.K. Treatment course of steroid-dependent nephrotic syndrome: Emphasized on treatment effect. Nephrology 2010, 15, 336–339. [Google Scholar] [CrossRef] [PubMed]
- Skrzypczyk, P.; Panczyk-Tomaszewska, M.; Roszkowska-Blaim, M.; Wawer, Z.; Bienias, B.; Zajgzkowska, M.; Kilis-Pstrusinska, K.; Jakubowska, A.; Szczepaniak, M.; Pawlak-Bratkowska, M.; et al. Long-term outcomes in idiopathic nephrotic syndrome: From childhood to adulthood. Clin. Nephrol. 2014, 81, 166–173. [Google Scholar] [CrossRef]
- Jiang, L.; Dasgupta, I.; Hurcombe, J.A.; Colyer, H.F.; Mathieson, P.W.; Welsh, G.I. Levamisole in steroid-sensitive nephrotic syndrome: Usefulness in adult patients and laboratory insights into mechanisms of action via direct action on the kidney podocyte. Clin. Sci. 2015, 128, 883–893. [Google Scholar] [CrossRef] [PubMed]
- Alsaran, K.; Mirza, K.; Al-Talhi, A.; Al-Kanani, E. Experience with second line drugs in frequently relapsing and steroid dependent childhood nephrotic syndrome in a large Saudi center. Int. J. Pediatr. Adolesc. Med. 2017, 4, 66–70. [Google Scholar] [CrossRef]
- Abeyagunawardena, A.S.; Karunadasa, U.; Jayaweera, H.; Thalgahagoda, S.; Tennakoon, S.; Abeyagunawardena, S. Efficacy of higher-dose levamisole in maintaining remission in steroid-dependant nephrotic syndrome. Pediatr. Nephrol. 2017, 32, 1363–1367. [Google Scholar] [CrossRef]
- Weiss, R. Randomized double-blind placebo controlled, multi-center trial of levamisole for children with frequently relapsing/steroid dependent nephrotic syndrome. J. Am. Soc. Nephrol. 1993, 4, 289. [Google Scholar]
- Dayal, U.; Dayal, A.K.; Shastry, J.C.; Raghupathy, P. Use of levamisole in maintaining remission in steroid-sensitive nephrotic syndrome in children. Nephron 1994, 66, 408–412. [Google Scholar] [CrossRef] [PubMed]
- Rashid, H.U.; Ahmed, S.; Fatima, N.; Khanam, A. Levamisole in the treatment of steroid dependent or frequent relapsing nephrotic syndrome in children. Bangladesh Ren. J. 1996, 15, 6–8. [Google Scholar]
- Sural, S.; Pahari, D.K.; Mitra, K.; Bhattacharya, S.; Mondal, S.; Taraphder, A. Efficacy of levamisole compared to cyclophosphamide and steroid in frequently relapsing minimal change nephrotic syndrome. J. Am. Soc. Nephrol. 2001, 12, 126. [Google Scholar]
- Al-Saran, K.; Mirza, K.; Al-Ghanam, G.; Abdelkarim, M. Experience with levamisole in frequently relapsing, steroid-dependent nephrotic syndrome. Pediatr. Nephrol. 2006, 21, 201–205. [Google Scholar] [CrossRef] [PubMed]
- Abeyagunawardena, A.; Trompeter, R.S. Efficacy of levamisole as a single agent in maintaining remission in steroid dependent nephrotic syndrome. Pediatr. Nephrol. 2006, 21, 1503. [Google Scholar]
- Durkan, A.M.; Hodson, E.M.; Willis, N.S.; Craig, J.C. Immunosuppressive agents in childhood nephrotic syndrome: A meta-analysis of randomized controlled trials. Kidney Int. 2001, 59, 1919–1927. [Google Scholar] [CrossRef][Green Version]
- Durkan, A.; Hodson, E.M.; Willis, N.S.; Craig, J.C. Non-corticosteroid treatment for nephrotic syndrome in children. Cochrane Database Syst. Rev. 2005, Cd002290. [Google Scholar] [CrossRef]
- Hodson, E.M.; Willis, N.S.; Craig, J.C. Non-corticosteroid treatment for nephrotic syndrome in children. Cochrane Database Syst. Rev. 2008, Cd002290. [Google Scholar] [CrossRef]
- Cascio, M.J.; Jen, K.Y. Cocaine/levamisole-associated autoimmune syndrome: A disease of neutrophil-mediated autoimmunity. Curr. Opin. Hematol. 2018, 25, 29–36. [Google Scholar] [CrossRef]
- Quirt, I.C.; Shelley, W.E.; Pater, J.L.; Bodurtha, A.J.; McCulloch, P.B.; McPherson, T.A.; Paterson, A.H.; Prentice, R.; Silver, H.K.; Willan, A.R.; et al. Improved survival in patients with poor-prognosis malignant melanoma treated with adjuvant levamisole: A phase III study by the National Cancer Institute of Canada Clinical Trials Group. J. Clin. Oncol. 1991, 9, 729–735. [Google Scholar] [CrossRef] [PubMed]
- Scheinfeld, N.; Rosenberg, J.D.; Weinberg, J.M. Levamisole in dermatology: A review. Am. J. Clin. Derm. 2004, 5, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Brunt, T.M.; van den Berg, J.; Pennings, E.; Venhuis, B. Adverse effects of levamisole in cocaine user: A reveiw and risk assessment. Arch. Toxicol. 2017, 91, 2303–2313. [Google Scholar] [CrossRef] [PubMed]
- Carmona-Rivera, C.; Purmalek, M.M.; Moore, E.; Waldman, M.; Walter, P.J.; Garrafo, H.M.; Phillips, K.A.; Preston, K.L.; Graf, J.; Kaplan, M.J.; et al. A role for muscarinic receptors in neutrophil extracellular trap formation and levamisole-induced autoimmunity. JCI Insight 2017, 2, e89780. [Google Scholar] [CrossRef]
- Prandota, J.; Kobylinska, J.; Lipowska, K. Administration of levamisole to children with kidney disease exacerbation caused by recurrent respiratory infections. Pol. Tyg. Lek. 1989, 44, 673–676. [Google Scholar] [PubMed]
- Youssef, D.M.; Abd Al-Atif, A.M.; El-Khateeb, S.S.H.; Elshal, A.S. Evaluation of interleukin-18 in children with steroid-sensitive nephrotic syndrome before and after using levamisole. Saudi J. Kidney Dis. Transpl. 2018, 29, 591–597. [Google Scholar] [CrossRef]
- Lombel, R.M.; Gipson, D.S.; Hodson, E.M. Treatment of steroid-sensitive nephrotic syndrome: New guidelines from KDIGO. Pediatr. Nephrol. 2013, 28, 415–426. [Google Scholar] [CrossRef]
- Yoo, T.H.; Fornoni, A. Nonimmunologic targets of immunosuppressive agents in podocytes. Kidney Res. Clin. Pract. 2015, 34, 69–75. [Google Scholar] [CrossRef][Green Version]
- Yap, H.K.; Cheung, W.; Murugasu, B.; Sim, S.K.; Seah, C.C.; Jordan, S.C. Th1 and Th2 cytokine mRNA profiles in childhood nephrotic syndrome: Evidence for increased IL-13 mRNA expression in relapse. J. Am. Soc. Nephrol. 1999, 10, 529–537. [Google Scholar]
- Kim, J.E.; Park, S.J.; Ha, T.S.; Shin, J.I. Effect of rituximab in MCNS: A role for IL-13 suppression? Nat. Rev. Nephrol. 2013, 9, 551. [Google Scholar] [CrossRef]
- Kim, A.H.; Chung, J.J.; Akilesh, S.; Koziell, A.; Jain, S.; Hodgin, J.B.; Miller, M.J.; Stappenbeck, T.S.; Miner, J.H.; Shaw, A.S. B cell-derived IL-4 acts on podocytes to induce proteinuria and foot process effacement. JCI Insight 2017, 2, e81836. [Google Scholar] [CrossRef] [PubMed]
- Lai, K.W.; Wei, C.L.; Tan, L.K.; Tan, P.H.; Chiang, G.S.; Lee, C.G.; Jordan, S.C.; Yap, H.K. Overexpression of interleukin-13 induces minimal-change-like nephropathy in rats. J. Am. Soc. Nephrol. 2007, 18, 1476–1485. [Google Scholar] [CrossRef] [PubMed]
- Faul, C.; Donnelly, M.; Merscher-Gomez, S.; Chang, Y.H.; Franz, S.; Delfgaauw, J.; Chang, J.M.; Choi, H.Y.; Campbell, K.N.; Kim, K.; et al. The actin cytoskeleton of kidney podocytes is a direct target of the antiproteinuric effect of cyclosporine A. Nat. Med. 2008, 14, 931–938. [Google Scholar] [CrossRef] [PubMed]
- Faul, C.; Asanuma, K.; Yanagida-Asanuma, E.; Kim, K.; Mundel, P. Actin up: Regulation of podocyte structure and function by components of the actin cytoskeleton. Trends Cell Biol. 2007, 17, 428–437. [Google Scholar] [CrossRef] [PubMed]
- Funauchi, M.; Ikoma, S.; Enomoto, H.; Horiuchi, A. Decreased Th1-like and increased Th2-like cells in systemic lupus erythematosus. Scand. J. Rheumatol. 1998, 27, 219–224. [Google Scholar] [CrossRef] [PubMed]
- Robinson, D.S.; Hamid, Q.; Ying, S.; Tsicopoulos, A.; Barkans, J.; Bentley, A.M.; Corrigan, C.; Durham, S.R.; Kay, A.B. Predominant TH2-like bronchoalveolar T-lymphocyte population in atopic asthma. N. Engl. J. Med. 1992, 326, 298–304. [Google Scholar] [CrossRef] [PubMed]
- Kocabas, C.N.; Sekerel, B.E.; Firat, P.A.; Okur, H.; Adahoglu, G. Levamisole: Might it be used in treatment and prevention of atopic diseases? J. Asthma 2004, 41, 547–551. [Google Scholar] [CrossRef]
- Heurtier, A.H.; Boitard, C. T-cell regulation in murine and human autoimmune diabetes: The role of TH1 and TH2 cells. Diabetes Metab. 1997, 23, 377–385. [Google Scholar]
- Szeto, C.; Gillespie, K.M.; Mathieson, P.W. Levamisole induces interleukin-18 and shifts type 1/type 2 cytokine balance. Immunology 2000, 100, 217–224. [Google Scholar] [CrossRef]
- Qian, H.; Jiao, L.; Fan, Z.; Wang, L.; Liu, B.; Miao, G. Analysis of Immunologic Function Changes in Lichen Planus After Clinical Treatment. Med. Sci. Monit. 2018, 24, 8716–8721. [Google Scholar] [CrossRef]
- Park, S.J.; Saleem, M.A.; Nam, J.A.; Ha, T.S.; Shin, J.I. Effects of interleukin-13 and montelukast on the expression of zonula occludens-1 in human podocytes. Yonsei Med. J. 2015, 56, 426–432. [Google Scholar] [CrossRef] [PubMed]
- Vivarelli, M.; Emma, F. Levamisole for children with nephrotic syndrome: New evidence for the use of an “old” drug. Kidney Int. 2019, 95, 25–28. [Google Scholar] [CrossRef] [PubMed]
- Van Wauwe, J.; Janssen, P.A. On the biochemical mode of action of levamisole: An update. Int. J. Immunopharmacol. 1991, 13, 3–9. [Google Scholar] [CrossRef]
- Colucci, M.; Carsetti, R.; Cassioli, S.; Serafinelli, J.; Emma, F.; Vivarelli, M. B cell phenotype in pediatric idiopathic nephrotic syndrome. Pediatr. Nephrol. 2019, 34, 177–181. [Google Scholar] [CrossRef] [PubMed]
- Wada, T.; Pippin, J.W.; Marshall, C.B.; Griffin, S.V.; Shankland, S.J. Dexamethasone prevents podocyte apoptosis induced by puromycin aminonucleoside: Role of p53 and Bcl-2-related family proteins. J. Am. Soc. Nephrol. 2005, 16, 2615–2625. [Google Scholar] [CrossRef] [PubMed]
- Xing, C.Y.; Saleem, M.A.; Coward, R.J.; Ni, L.; Witherden, I.R.; Mathieson, P.W. Direct effects of dexamethasone on human podocytes. Kidney Int. 2006, 70, 1038–1045. [Google Scholar] [CrossRef] [PubMed]
- Ransom, R.F.; Lam, N.G.; Hallett, M.A.; Atkinson, S.J.; Smoyer, W.E. Glucocorticoids protect and enhance recovery of cultured murine podocytes via actin filament stabilization. Kidney Int. 2005, 68, 2473–2483. [Google Scholar] [CrossRef] [PubMed]
- Tenbrock, K.; Muller-Berghaus, J.; Fuchshuber, A.; Michalk, D.; Querfeld, U. Levamisole treatment in steroid-sensitive and steroid-resistant nephrotic syndrome. Pediatr. Nephrol. 1998, 12, 459–462. [Google Scholar] [CrossRef]
| Author, Year | Study Design | Comparison (Dose of Levamisole) | Total Patients | Relapse (%) | FU | Other Effect |
|---|---|---|---|---|---|---|
| Tanphaichitr P, 1980 [22] | Obs | (1.5–3.9 mg/kg twice per week) | 7 | - | 1–6 mo | (33.5 ± 9.5 % vs. 69.3 ± 3.9%) a |
| Niaudet P, 1984 [42] | Obs | (2.5 mg/kg twice per week) | 30 | - | 9.9 mo | - |
| Mehta KP, 1986 [43] | Obs | (2.5 mg/kg qod or qd) | 14 | - | 6–24 mo | 6 Pts CR, 6 Pts PR |
| La Manna A, 1988 [44] | Obs | (2.5 mg/kg twice a week or qd) | 13 | - | 7–29 mo | b |
| Srivastava RN, 1991 [45] | Obs | (5 mg/kg qod) | 12 | Before L (3.3/yr), After L (2.3/yr) | 3 mo | - |
| Meregalli P, 1994 [46] | Obs | (5 mg/kg weekly) | 10 | Reduction SD 62–75% | - | - |
| Ksiazek J, 1995 [47] | Obs | - | 22 | 18.2% relapse/36.4% not response | - | 45.5% remission maintained |
| Ginevri F, 1996 [48] | Obs | (2.5 mg/kg qod) | 20 | 45.5% remission maintained after L, 36.4% not respond to L | - | - |
| Bagga A, 1997 [23] | Obs | (2.5 mg/kg qod) | 43 | Before L (5.2/yr), After 2 years L (0.69/yr) | 6–31 mo | - |
| Kemper MJ, 1998 [26] | Obs | (2.0 mg/kg qod) | 25 | Prior to L 0.5/month, During L 0.3/month | 3–24 mo | - |
| Fu LS, 2000 [49] | Obs | (2–3 mg/kg qod) | 27 | Prior to L (5.74 ± 3.24/yr), During L (1.91 ± 2.0/yr) | 6–24 mo | - |
| Alsaran K, 2001 [50] | Obs | L (2.5 mg/kg) vs. CPA | 24 | L (0.28/month), CPA (0.32/month) | - | c |
| Donia AF, 2002 [51] | Obs | (2.5 mg/kg qod) | 20 | 25% in remission 12 mo, 75% relapses during Tx | 12 mo | - |
| Al-Ibrahim AA, 2003 [52] | Obs | (2.5 mg/kg qod) | 24 | Prior to L (4/yr), During L (1.3/yr) | 12–24 mo | - |
| Sumegi V, 2004 [53] | Obs | (2 mg/kg qd) | 34 | Prior to L (4.41/yr), During L (0.41/yr) | 24 mo | Cumulative steroid dose 7564 mg/yr vs. 1472 mg/yr |
| Fu LS, 2004 [54] | Obs | (2–3 mg/kg qod or qd) | 36 | qod 2.01 ± 2.5/yr, qd 1.34 ± 2.1/yr | 4–36 mo | - |
| Hafeez F, 2006 [55] | Obs | (2.5 mg/kg qod) | 70 | 19 (27.14%) pts did not relapse on Tx | 12 mo | d |
| Boyer O, 2008 [37] | Obs | (2.5 mg/kg 3 days/week) | 10 | Prior to L (6.0/yr), During L (0.0/yr) | 48 mo | e |
| Madani A, 2010 [38] | Obs | (2.5 mg/kg qod) | 304 | Prior to L (2.0/yr), During L (1.1/yr) | 1–22 yrs | Steroid dose was significantly reduced |
| Chen SY, 2010 [56] | Obs | (2–3.3 mg/kg qd) | 15 | 1 pt CR, 10 pts No effect | 3–20 mo | - |
| Elmas AT, 2013 [40] | Obs | (2.5 mg/kg 3 days/week) | 29 | Prior to L (4.0/yr), During L (0.0/yr) | 12 mo | Proteinuria, annual SD reduced |
| Ekambaram S, 2014 [35] | Obs | (2 mg/kg qd) | 97 | Prior to L (2.4/yr), During L (1.3/yr) | 6–24 mo | Steroid dose (4.1 g/m2 to 2.5 g/m2) |
| Skrzypczyk P, 2014 [57] | Obs | (2.5 mg/kg/qd or qod or twice a week) | 21 | - | - | f |
| Jiang L, 2015 [58] | Obs | (1.25 mg/kg qd) | 15 | Adult 14 pts reduction or stopping other medications | 1–14 yrs | |
| Kuzma-Mroczkowska E, 2016 [36] | Obs | (2.5 mg/kg qod) | 53 | Prior to L (2.7/yr), During L (1.8/yr) | 36 mo | 8.8 mo to relapse on levamisole Tx |
| Alsaran, K, 2017 [59] | Obs | (2.5 mg/kg qod) | 20 | Prior to L (3.6/yr), During L (1.6/yr) | 12 mo | Cumulative steroid dose was reduced |
| Abeyagunawardena, 2017 [60] | Obs | (2.5 mg/kg qod vs. qd) | 58 | L qod (163/yr), L qd (77/yr) | 12 mo | M annual SD (mg/kg/yr) qod vs. qd (254 vs. 154) |
| Basu B, 2017 [39] | Obs | (2.5 mg/kg qod) | 129 | During L (1.7/yr) After L (2.8/yr) | 30 mo | g |
| Author, Year | Inclusion | Exclusion | Comparison | Total Patients/Control | Relapse (%) | FU Weeks | Effect Size (RR, 95% CI) |
|---|---|---|---|---|---|---|---|
| BAPN, 1991 [25] | SSNS | NR | L (2.5 mg/kg qod) + steroid vs. Placebo + steroid (16 weeks) | 61/30 | L 57% (17/31) Placebo 87% (26/30) | 16 | 0.63 (0.45–0.90) |
| Weiss, 1993 * [61] | FRNS, SDNS | Prior Tx with other IS within 6mo | L (2.5 mg/kg twice a week) + steroid vs. Placebo + steroid (6 mo) | 48/26 | L 95% (21/22) Placebo 81% (21/26) | 24 | 1.18 (0.96–1.46) |
| Dayal, 1994 [62] | SSNS | SRNS | L (2–3 mg/kg twice a week) + steroid vs. steroid alone (48 weeks) | 36/14 | L 41% (9/22) Steroid alone 71% (10/14) | 48 | 0.57 (0.31–1.05) |
| Rashid, 1996 * [63] | FRNS, SDNS | NR | L (2.5 mg/kg qod) + steroid vs. steroid alone (6 mo) | 40/20 | L 30% (6/20) Steroid alone 60% (12/20) | 44 | 0.50 (0.23–1.07) |
| Sural, 2001 * [64] | FRNS, SDNS | NR | L (2.5 mg/kg qod) + steroid vs. steroid alone (6 mo) | 58/28 | L 27% (8/30) Steroid alone 82% (23/28) | 48 | 0.32 (0.17–0.60) |
| Donia, 2005 [34] | SSNS | NR | L (2.5 mg/kg qod) + steroid vs. IV CPA + steroid | 40/20 | L 50% (10/20) CPA 55% (11/20) | 96 | 0.91 (0.50–1.64) |
| Al–Saran, 2006 [65] | FRNS, SDNS | Prior Tx with IS | L (2.5 mg/kg qod) + steroid vs. steroid alone (1 yr) | 56/24 | Levamisole 9% (3/32) Steroid alone 50% (12/24) | 48 | 0.19 (0.06–0.59) |
| Abeyagunawardena AS, 2006 * [66] | SSNS | NR | L (2.5 mg/kg qod) + steroid vs. No Tx | 76/34 | L 19% (8/42) Control 76% (26/34) | 48 | 0.25 (0.13–0.48) |
| Gruppen, 2018 [28] | FRNS, SDNS | Unresponsiveness to cyclosporine or MMF | L (2.5 mg/kg qod) + steroid vs. Placebo (maximum 21 days) | 99/49 | L 66% (33/50) Placebo 86% (42/49) | 48 | 0.30 (0.11–0.82) |
| Sinha, 2019 [30] | FRNS, SDNS | Prior Tx with other IS | L (2–2.5 mg/kg qod) vs. MMF | 149/76 | L 66% (48/73) MMF 59% (45/76) | 48 | 0.79 (0.58–1.07) |
| Author, Year | Comparison | Outcome | No. of Studies | No. of Cases/Controls | Type of Metrics | Reported Summary Effect (95% CI) | Reported p-Value | Largest Effect (95% CI) | I2 (p Value) | No. of Significant Study/Total Study |
|---|---|---|---|---|---|---|---|---|---|---|
| Durkan AM, et al. 2001 [67] | L vs. placebo | Relapse (4–12 mo) | 3 | 137/64 | RR | 0.60 (0.45–0.79) | 0.0004 | 0.63 (0.45–0.90) | 0.0% (0.84) | 1/3 |
| Durkan AM, et al. 2005 [68] | L vs. placebo/no treatment | Relapse (4–12 mo) | 4 | 185/90 | RR | 0.71 (0.41–1.23) | 0.23 | 0.63 (0.45–0.90) | 86% (<0.001) | 1/4 |
| Hodson EM, et al. 2008. [69] | L vs. placebo | Relapse (4–12 mo) | 6 | 317/148 | RR | 0.50 (0.25–0.99) | 0.046 | 0.25 (0.13–0.48) | 92% (0.046) | 3/6 |
| Hodson EM, et al. 2008 [69] | L vs. IV CPA | Relapse (E) | 1 | 40/20 | RR | 0.91 (0.50–1.64) | NA | - | - | 0/1 |
| Pravitsitthikul N, et al. 2013 [31] | L vs. steroids or placebo or both, or no treatment | Relapse (4–12 mo) | 7 | 375/176 | RR | 0.47 (0.24–0.89) | 0.021 | 0.25 (0.13–0.48) | 92% (<0.001) | 4/7 |
| Pravitsitthikul N, et al. 2013 [31] | L vs. IV CPA | Relapse (E) | 2 | 97/47 | RR | 2.14 (0.22–20.95) | 0.51 | 7.20 (0.96–53.89) | 79% (0.03) | 0/2 |
| Author, Year | Levamisole Dose | Number | Leukopenia | GI Upset | Skin Rash | Arthritis | Other |
|---|---|---|---|---|---|---|---|
| BAPN, 1991 [25] | 2.5 mg/kg qod | 31 | 0 | 1 | 0 | 0 | 0 |
| Weiss, 1993 * [61] | 2.5 mg/kg twice a week | 22 | 0 | 0 | 0 | 0 | 0 |
| Sural, 2001 * [64] | 2.5 mg/kg qod | 30 | 1 | 0 | 0 | 0 | 0 |
| Donia, 2005 [34] | 2.5 mg/kg qod | 20 | 0 | 0 | 0 | 0 | 14 a |
| Al-Saran, 2006 [65] | 2.5 mg/kg qod | 32 | 0 | 1 | 0 | 0 | 0 |
| Gruppen, 2018 [28] | 2.5 mg/kg qod | 50 | 8 | 1 | 0 | 1 | 26 b |
| Sinha, 2019 [30] | 2–2.5 mg/kg qod | 73 | 0 | 18 | 2 | 0 | 0 |
| Tanphaichitr P, 1980 [22] | 1.5–3.9 mg/kg twice a week | 7 | 0 | 0 | 0 | 0 | 0 |
| Niaudet P, 1984 [42] | 2.5 mg/kg twice a week | 30 | 7 | 0 | 0 | 0 | 0 |
| Metha KP, 1986 [43] | 2.5 mg/kg qod | 14 | 0 | 3 | 1 | 0 | 2 c |
| La Manna A, 1988 [44] | 2.5 mg/kg qd | 13 | 0 | 1 | 1 | 0 | 1 d |
| Prandota J, 1989 [75] | 2.1–3.1 mg/kg qd | 6 | 0 | 0 | 0 | 0 | 6 e |
| Srivastava RN, 1991 [45] | 5 mg/kg qod | 12 | 1 | 0 | 1 | 0 | 0 |
| Meregalli P, 1994 [46] | 5 mg/kg weekly | 10 | 1 | 0 | 1 | 0 | 0 |
| Bagga A, 1997 [23] | 2.5 mg/kg qod | 43 | 0 | 0 | 0 | 0 | 0 |
| Kemper MJ, 1998 [26] | 2.0 mg/kg qod | 25 | 2 | 1 | 1 | 0 | 0 |
| Fu LS, 2000 [49] | 2-3 mg/kg qod | 27 | 7 | 0 | 0 | 0 | 0 |
| Alsaran K, 2001 [50] | 2.5 mg/kg qod | 24 | 0 | 0 | 1 | 0 | 0 |
| Donia AF, 2002 [51] | 2.5 mg/kg qod | 20 | 0 | 0 | 0 | 0 | 0 |
| Al-Ibrahim AA, 2003 [52] | 2.5 mg/kg qod | 24 | 1 | 2 | 2 | 0 | 0 |
| Sumegi V, 2004 [53] | 2 mg /kg qd | 34 | 5 | 0 | 0 | 0 | 0 |
| Fu LS, 2004 [54] | 2–3 mg/kg qd or qod | 36 | 9 | 0 | 0 | 0 | 0 |
| Hafeez F, 2006 [55] | 2.5 mg/kg qod | 70 | 0 | 0 | 0 | 0 | 0 |
| Boyer O, 2008 [37] | 2.5 mg/kg twice a week | 10 | 0 | 0 | 0 | 0 | 0 |
| Madani A, 2010 [38] | 2.5 mg/kg qod | 304 | 1 | 0 | 0 | 0 | 1 f |
| Elmas AT, 2013 [40] | 2.5 mg/kg twice a week | 29 | 0 | 0 | 0 | 0 | 0 |
| Ekambaram S, 2014 [35] | 2 mg/kg qd | 97 | 0 | 0 | 0 | 0 | 0 |
| Jiang L, 2015 [58] | 1.25 mg/kg qod | 15 | 3 | 2 | 2 | 0 | 0 |
| Kuzma-Mroczkowska,2016 [36] | 2.5 mg/kg qod | 53 | 1 | 3 | 9 | 0 | 6 g |
| Alsaran K, 2017 [59] | 2.5 mg/kg qod | 20 | 4 | 0 | 0 | 0 | 0 |
| Abeyagunawardena AS, 2017 [60] | 2.5 mg/kg qd and qod | 58 | 0 | 0 | 0 | 0 | 0 |
| Basu B, 2017 [39] | 2.5 mg/kg qod | 129 | 0 | 0 | 0 | 0 | 3 h |
| Youssef DM, 2018 [76] | 2.5 mg/kg qod | 23 | 0 | 0 | 0 | 0 | 0 |
| Total | 1391 | 51 (3.7%) | 33 (2.4%) | 21 (1.5%) | 1 (0%) | 59 (4.2%) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mühlig, A.K.; Lee, J.Y.; Kemper, M.J.; Kronbichler, A.; Yang, J.W.; Lee, J.M.; Shin, J.I.; Oh, J. Levamisole in Children with Idiopathic Nephrotic Syndrome: Clinical Efficacy and Pathophysiological Aspects. J. Clin. Med. 2019, 8, 860. https://doi.org/10.3390/jcm8060860
Mühlig AK, Lee JY, Kemper MJ, Kronbichler A, Yang JW, Lee JM, Shin JI, Oh J. Levamisole in Children with Idiopathic Nephrotic Syndrome: Clinical Efficacy and Pathophysiological Aspects. Journal of Clinical Medicine. 2019; 8(6):860. https://doi.org/10.3390/jcm8060860
Chicago/Turabian StyleMühlig, Anne K., Jun Young Lee, Markus J. Kemper, Andreas Kronbichler, Jae Won Yang, Jiwon M. Lee, Jae Il Shin, and Jun Oh. 2019. "Levamisole in Children with Idiopathic Nephrotic Syndrome: Clinical Efficacy and Pathophysiological Aspects" Journal of Clinical Medicine 8, no. 6: 860. https://doi.org/10.3390/jcm8060860
APA StyleMühlig, A. K., Lee, J. Y., Kemper, M. J., Kronbichler, A., Yang, J. W., Lee, J. M., Shin, J. I., & Oh, J. (2019). Levamisole in Children with Idiopathic Nephrotic Syndrome: Clinical Efficacy and Pathophysiological Aspects. Journal of Clinical Medicine, 8(6), 860. https://doi.org/10.3390/jcm8060860







