Celiac Disease Prevalence Is Increased in Primary Sjögren’s Syndrome and Diffuse Systemic Sclerosis: Lessons from a Large Multi-Center Study
Abstract
1. Introduction
2. Experimental Section
2.1. Study Population
2.2. Laboratory Methods
2.3. Study Protocol to Confirm CD Diagnosis
2.4. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Ethics Approval and Consent to Participate
Acknowledgments
Conflicts of Interest
References
- Catassi, C.; Gatti, S.; Lionetti, E. World perspective and celiac disease epidemiology. Dig. Dis. 2015, 33, 141–146. [Google Scholar] [CrossRef] [PubMed]
- Lebwohl, B.; Sanders, D.S.; Green, P. Coeliac disease. Lancet 2018, 391, 70–81. [Google Scholar] [CrossRef]
- Lindfors, K.; Ciacci, C.; Kurppa, K.; Lundin, K.E.A.; Makharia, G.K.; Mearin, M.L.; Murray, J.A.; Verdu, E.F.; Kaukinen, K. Coeliac Disease. Nat. Rev. Dis. Primers 2019, 5, 3. [Google Scholar] [CrossRef] [PubMed]
- Lauret, E.; Rodrigo, L. Celiac disease and autoimmune-associated conditions. BioMed Res. Int. 2013, 2013, 127589. [Google Scholar] [CrossRef] [PubMed]
- National Institute for Health and Care Excellence. Recognition, Management, and Assessment of Coeliac Disease. (Clinical Guideline NG20) 2015. Available online: www.nice.org.uk/guidance/NG20/evidence (accessed on 15 November 2017).
- Ludvigsson, J.F.; Rubio-Tapia, A.; Chowdhary, V.; Murray, J.A.; Simard, J.F. Increased risk of systemic lupus erythematosus in 29,000 patients with biopsy-verified celiac disease. J. Rheumatol. 2012, 39, 1964–1970. [Google Scholar] [CrossRef] [PubMed]
- Dahan, S.; Shor, D.B.; Comaneshter, D.; Takes-Manova, D.; Shovman, O.; Amital, H.; Cohen, A.D. All disease begins in the gut: Celiac disease co-existence with SLE. Autoimmun. Rev. 2016, 15, 848–853. [Google Scholar] [CrossRef] [PubMed]
- Bibbò, S.; Pes, G.M.; Usai-Satta, P.; Salis, R.; Soro, S.; Quarta Colosso, B.M.; Dore, M.P. Chronic autoimmune disorders are increased in coeliac disease. A case-control study. Medicine 2017, 96, e8562. [Google Scholar] [CrossRef] [PubMed]
- Catassi, C.; Fasano, A. Celiac disease diagnosis: Simple rules are better than complicated algorithms. Am. J. Med. 2010, 123, 691–693. [Google Scholar] [CrossRef] [PubMed]
- Bai, J.C.; Fried, M.; Corazza, G.R.; Schuppan, D.; Farthing, M.; Catassi, C.; Greco, L.; Cohen, H.; Ciacci, C.; Eliakim, R.; et al. World Gastroenterology Organisation global guidelines on celiac disease. J. Clin. Gastroenterol. 2013, 47, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Tan, E.M.; Cohen, A.S.; Fries, J.F.; Masi, A.T.; McShane, D.J.; Rothfield, N.F.; Schaller, J.G.; Talal, N.; Winchester, R.J. The 1982 revised criteria for the classification of systemic lupus erythematosus. Arthritis Rheum. 1982, 25, 1271–1277. [Google Scholar] [CrossRef] [PubMed]
- Vitali, C.; Bombardieri, S.; Jonsson, R.; Moutsopoulos, H.M.; Alexander, E.L.; Carsons, S.E.; Daniels, T.E.; Fox, P.C.; Fox, R.I.; Kassan, S.S.; et al. Classification criteria for the European Study Group on Classification Criteria for Sjögren’s syndrome. Classification criteria for Sjögren’s syndrome: A revised version of the European criteria proposed by the American-European Consensus Group. Ann. Rheum. Dis. 2002, 61, 554–558. [Google Scholar] [CrossRef] [PubMed]
- Van den Hoogen, F.; Khanna, D.; Fransen, J.; Johnson, S.R.; Baron, M.; Tyndall, A.; Matucci-Cerinic, M.; Naden, R.P.; Medsger, T.A., Jr.; Carreira, P.E.; et al. 2013 classification criteria for systemic sclerosis: An American College of Rheumatology/European League against Rheumatism collaborative initiative. Arthritis Rheum. 2013, 65, 2737–2747. [Google Scholar] [CrossRef] [PubMed]
- LeRoy, E.C.; Medsger, T.A., Jr. Criteria for the classification of early systemic sclerosis. J. Rheumatol. 2001, 28, 1573–1576. [Google Scholar] [PubMed]
- Valentini, G.; Marcoccia, A.; Cuomo, G.; Iudici, M.; Vettori, S. The concept of early systemic sclerosis following 2013 ACR\EULAR criteria for the classification of systemic sclerosis. Curr. Rheumatol. Rev. 2014, 10, 38–44. [Google Scholar] [CrossRef] [PubMed]
- Bartoloni, E.; Baldini, C.; Schillaci, G.; Quartuccio, L.; Priori, R.; Carubbi, F.; Bini, V.; Alunno, A.; Bombardieri, S.; De Vita, S.; et al. Cardiovascular disease risk burden in primary Sjögren’s syndrome: Results of a population-based multicentre cohort study. J. Intern. Med. 2015, 278, 185–192. [Google Scholar] [CrossRef] [PubMed]
- Cervera, R.; Doria, A.; Amoura, Z.; Khamashta, M.; Schneider, M.; Guillemin, F.; Maurel, F.; Garofano, A.; Roset, M.; Perna, A.; et al. Patterns of systemic lupus erythematosus expression in Europe. Autoimmun. Rev. 2014, 13, 621–629. [Google Scholar] [CrossRef] [PubMed]
- Bartoloni, E.; Pucci, G.; Cannarile, F.; Battista, F.; Alunno, A.; Giuliani, M.; Cafaro, G.; Gerli, R.; Schillaci, G. Central hemodynamics and arterial stiffness in systemic sclerosis. Hypertension 2016, 68, 1504–1511. [Google Scholar] [CrossRef] [PubMed]
- Rensch, M.J.; Szyjkowski, R.; Shaffer, R.T.; Fink, S.; Kopecky, C.; Grissmer, L.; Enzenhauer, R.; Kadakia, S. The prevalence of celiac disease autoantibodies in patients with systemic lupus erythematosus. Am. J. Gastroenterol. 2001, 96, 1113–1115. [Google Scholar] [CrossRef] [PubMed]
- Luft, L.; Barr, S.G.; Martin, L.O.; Chan, E.K.; Fritzler, M.J. Autoantibodies to tissue transglutaminase in Sjögren’s syndrome and related rheumatic diseases. J. Rheumatol. 2003, 30, 2613–2619. [Google Scholar] [PubMed]
- Marai, I.; Shoenfeld, Y.; Bizzaro, N.; Villalta, D.; Doria, A.; Tonutti, E.; Tozzoli, R. IgA and IgG tissue transglutaminase antibodies in systemic lupus erythematosus. Lupus 2004, 13, 241–244. [Google Scholar] [CrossRef] [PubMed]
- Koehne, V.; Bahia, M.; Lanna, C.C.; Pinto, M.R.; Bambirra, E.A.; Cunha, A.S. Prevalence of serological markers for celiac disease (IgA and IgG class antigliadin antibodies and IgA class antiendomysium antibodies) in patients with autoimmune rheumatologic diseases in Belo Horizonte, MG, Brazil. Arq. Gastroenterol. 2010, 47, 250–256. [Google Scholar] [CrossRef] [PubMed]
- Ben Abdelghani, K.; Mouelhi, L.; Hriz, A.; Hajri, S.; Najjar, T.; Mahfoudhi, M.; Turki, S.; Khedher, A. Systemic lupus erythematosus and celiac disease. Joint Bone Spine 2012, 79, 199–206. [Google Scholar] [CrossRef] [PubMed]
- Iltanen, S.; Collin, P.; Korpela, M.; Holm, K.; Partanen, J.; Polvi, A.; Mäki, M. Celiac disease and markers of celiac disease latency in patients with primary Sjögren’s syndrome. Am. J. Gastroenterol. 1999, 94, 1042–1046. [Google Scholar] [CrossRef] [PubMed]
- Bizzaro, N.; Villalta, D.; Tonutti, E.; Doria, A.; Tampoia, M.; Bassetti, D.; Tozzoli, R. IgA and IgG tissue transglutaminase antibody prevalence and clinical significance in connective tissue diseases, inflammatory bowel disease, and primary biliary cirrhosis. Dig. Dis. Sci. 2003, 48, 2360–2365. [Google Scholar] [CrossRef] [PubMed]
- Szodoray, P.; Barta, Z.; Lakos, G.; Szakàll, S.; Zeher, M. Coeliac disease in Sjögren’s syndrome—A study of 111 Hungarian patients. Rheumatol. Int. 2004, 24, 278–282. [Google Scholar] [CrossRef] [PubMed]
- Rosato, E.; De Nitto, D.; Rossi, C.; Libanori, V.; Donato, G.; Di Tola, M.; Pisarri, S.; Salsano, F.; Picarelli, A. High incidence of celiac disease in patients with systemic sclerosis. J. Rheumatol. 2009, 36, 965–969. [Google Scholar] [CrossRef] [PubMed]
- Nisihara, R.; Utiyama, S.R.; Azevedo, P.M.; Skare, T.L. Celiac disease screening in patients with scleroderma. Arq. Gastroenterol. 2011, 48, 163–164. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Forbess, L.J.; Gordon, J.K.; Doobay, K.; Bosworth, B.; Lyman, S.; Davids, M.; Spiera, R.F. Low prevalence of coeliac disease in patients with systemic sclerosis: A cross-sectional study of a registry cohort. Rheumatology 2013, 52, 939–943. [Google Scholar] [CrossRef] [PubMed]
- Oberhuber, G.; Granditsch, G.; Vogelsang, H. The histopathology of coeliac disease: Time for a standardized report scheme for pathologists. Eur. J. Gastroenterol. Pathol. 1999, 11, 1185–1194. [Google Scholar] [CrossRef] [PubMed]
- Alaibac, M.; Berti, E.; Chizzolini, C.; Fineschi, S.; Marzano, A.V.; Pigozzi, B.; Riboldi, E.; Sozzani, S.; Kuhn, A. Role of cellular immunity in the pathogenesis of autoimmune skin diseases. Clin. Exp. Rheumatol 2006, 24 (Suppl. 40), S14–S19. [Google Scholar] [PubMed]
- Mustalahti, K.; Catassi, C.; Reunanen, A.; Fabiani, E.; Heier, M.; McMillan, S.; Murray, L.; Metzger, M.H.; Gasparin, M.; Bravi, E.; et al. The prevalence of celiac disease in Europe: Results of a centralized, international mass screening project. Ann. Med. 2010, 42, 587–595. [Google Scholar] [CrossRef] [PubMed]
- Dieli-Crimi, R.; Cènit, M.C.; Núñez, C. The genetics of celiac disease: A comprehensive review of clinical implications. J. Autoimmun. 2015, 64, 26–41. [Google Scholar] [CrossRef] [PubMed]
- McLean, M.H.; Dieguez, D., Jr.; Miller, L.M.; Young, H.A. Does the microbiota play a role in the pathogenesis of autoimmune diseases? Gut 2015, 64, 332–341. [Google Scholar] [CrossRef] [PubMed]
- Volkmann, E.R.; Chang, Y.L.; Barroso, N.; Furst, D.E.; Clements, P.J.; Gorn, A.H.; Roth, B.E.; Conklin, J.L.; Getzug, T.; Borneman, J.; et al. Association of systemic sclerosis with a unique colonic microbial consortium. Arthritis Rheumatol. 2016, 68, 1483–1492. [Google Scholar] [CrossRef] [PubMed]
- Mandl, T.; Marsal, J.; Olsson, P.; Ohlsson, B.; Andréasson, K. Severe intestinal dysbiosis is prevalent in primary Sjögren’s syndrome and is associated with systemic disease activity. Arthritis Res. Ther. 2017, 19, 237. [Google Scholar] [CrossRef] [PubMed]
- Rosenbaum, J.T. Celiac Disease and Autoimmunity—The Missing Ingredient. N. Engl. J. Med. 2017, 377, 1489–1490. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, H.; Kawakami, A. What is the evidence for Sjögren’s syndrome being triggered by viral infection? Subplot: Infections that cause clinical features of Sjögren’s syndrome. Curr. Opin. Rheumatol. 2016, 28, 390–397. [Google Scholar] [CrossRef] [PubMed]
- Argyropoulou, O.D.; Valentini, E.; Ferro, F.; Leone, M.C.; Cafaro, G.; Bartoloni, E.; Baldini, C. One year in review 2018: Primary Sjögren’s syndrome. Clin. Exp. Rheumatol. 2018, 112, 14–26. [Google Scholar] [PubMed]
- Carroccio, A.; D’Alcamo, A.; Cavataio, F.; Soresi, M.; Seidita, A.; Sciumè, C.; Geraci, G.; Iacono, G.; Mansueto, P. High proportions of people with nonceliac wheat sensitivity have autoimmune disease or antinuclear antibodies. Gastroenterology 2015, 149, 596–603. [Google Scholar] [CrossRef] [PubMed]
- Lerner, A.; Blank, M.; Lahat, N.; Shoenfeld, Y. Increased prevalence of autoantibodies in celiac disease. Dig. Dis. Sci. 1998, 43, 723–726. [Google Scholar] [CrossRef] [PubMed]
- Cosnes, J.; Cellier, C.; Viola, S.; Colombel, J.F.; Michaud, L.; Sarles, J.; Hugot, J.P.; Ginies, J.L.; Dabadie, A.; Mouterde, O.; et al. Incidence of autoimmune diseases in celiac disease: Protective effect of the gluten-free diet. Clin. Gastroenterol. Hepatol. 2008, 6, 753–758. [Google Scholar] [CrossRef] [PubMed]
- Cappello, M.; Morreale, G.C.; Licata, A. Elderly onset Celiac disease: A narrative review. Clin. Med. Insights Gastroenterol. 2016, 9, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Volta, U.; Caio, G.; Stanghellini, V.; De Giorgio, R. The changing clinical profile of celiac disease: A 15-year experience (1998-2012) in an Italian referral center. BMC Gastroenterol. 2014, 14, 194. [Google Scholar] [CrossRef] [PubMed]
- Shannahan, S.; Leffler, D.A. Diagnosis and updates in Celiac disease. Gastrointest. Endoscopy Clin. N. Am. 2017, 27, 79–92. [Google Scholar] [CrossRef] [PubMed]
- Ventura, A.; Magazzù, G.; Greco, L. Duration of exposure to gluten and risk for autoimmune disorders in patients with celiac disease. SIGEP Study Group for Autoimmune Disorders in Celiac Disease. Gastroenterology 1999, 117, 297–303. [Google Scholar] [CrossRef] [PubMed]
- Snyder, M.R.; Murray, J.A. Celiac Disease: Advances in diagnosis. Expert Rev. Clin. Immunol. 2016, 12, 449–463. [Google Scholar] [CrossRef] [PubMed]
- Rubio-Tapia, A.; Kyle, R.A.; Kaplan, E.L.; Johnson, D.R.; Page, W.; Erdtmann, F.; Brantner, T.L.; Kim, W.R.; Phelps, T.K.; Lahr, B.D.; et al. Increased prevalence and mortality in undiagnosed celiac disease. Gastroenterology 2009, 137, 88–93. [Google Scholar] [CrossRef] [PubMed]
- Tio, M.; Cox, M.R.; Eslick, G.D. Meta-analysis: Coeliac disease and the risk of all-cause mortality, any malignancy and lymphoid malignancy. Aliment Pharmacol. Ther. 2012, 35, 540–551. [Google Scholar] [CrossRef] [PubMed]
- Gandolfo, S.; Quartuccio, L.; De Vita, S. Sjögren’s Syndrome-associated lymphoma. In Sjögren’s Syndrome. Novel Insights in Pathogenic, Clinical and Therapeutic Aspects, 1st ed.; Alunno, A., Bartoloni, E., Gerli, R., Eds.; Elsevier: Amsterdam, The Netherlands, 2018; Chapter 6; pp. 73–98. ISBN 9780128036327. [Google Scholar]
| Author Year [ref] | Pts, n Country | Screening for CD | Ab Result (Prevalence) | Adopted Criteria to Perform SBB | SBB Positive Results (Prevalence) |
|---|---|---|---|---|---|
| Rensh M 2001 [19] | 103 USA | IgA/IgG AGA and EMA | 24 pts AGA+ (all pts EMA−)(23%) | AGA+ and/or EMA+ | 0/24 (0%) |
| Luft LM 2003 [20] | 50 USA | IgA tTG IgA EMA in tTG+ | 3 pts tTG+/EMA– (6%) | Retrospective data | ND |
| Marai I 2004 [21] | 100 Italy/ Israel | IgA/IgG tTG IgA/IgG EMA in tTG+ or HLA-DQ2/8 in EMA– | 3 pts tTG+ (3%) (1 EMA+ and 2 EMA–/DQ2/8–) | tTG+ and EMA+ or EMA–/DQ2/8+ | 1/1 (1%) |
| Koehne V 2010 [22] | 69 Brazil | IgA/IgG AGA and IgA EMA IgA-tTG in EMA+ | 2 pts AGA+/EMA– (3%) 3 pts EMA+/tTG– (4%) | AGA+ or EMA+ | 0/5 (0%) |
| Ben Abdelghani K 2012 [23] | 24 Tunisia | AGA and tTG | 5 pts AGA+/tTG– (21%) 2 pts AGA+/tTG+ (8%) | All pts | 1/24 (AGA+/tTG+) (4%) |
| Author Year [ref] | Pts, n Country | Screening for CD | Ab Results (Prevalence) | Adopted Criteria to Perform SBB | SBB Positive Results (Prevalence) |
|---|---|---|---|---|---|
| Iltanen S 1999 [24] | 34 Finland | ND | 3 pts EMA+ (9%) 13 pts AGA+ (38%) 19 pts DQ2+ (56%) | All pts | 5 (15%) |
| Bizzaro N 2003 [25] | 100 Italy/ Israel | IgA/IgG tTG IgA/IgG EMA in tTG+ or HLA-DQ2/8 in EMA− | 1 pt tTG+/ EMA−/DQ2/8− (1%) | tTG+ and EMA+ or EMA−/DQ2/8+ | ND |
| Luft LM 2003 [20] | 50 USA | IgA tTG IgA EMA in tTG+ | 5 pts tTG+/EMA+ (10%) 1 pt tTG+/EMA– (2%) | Retrospective data | 5 (tTG+/EMA+) (10%) |
| Szodoray P 2004 [26] | 111 Hungary | IgA tTG, IgA EMA, IgG/IgA AGA | 6 pts with serology+ (not specified) (5%) | tTG+ and/or EMA+ and/or AGA+ | 5 (4.5%) |
| Author Year [ref] | Pts, n Country | Screening for CD | Ab Results (Prevalence) | Adopted Criteria to Perform SBB | SBB Positive Results (Prevalence) |
|---|---|---|---|---|---|
| Luft LM 2003 [20] | 30 USA | IgA tTG IgA EMA in tTG+ | 2 pts tTG+/EMA+ (7%) | Retrospective data | ND |
| Bizzaro N 2003 [25] | 100 Italy/ Israel | IgA/IgG tTG IgA/IgG EMA in tTG+ or HLA-DQ2/8 in EMA− | 1 pt tTG+/ EMA−/DQ2/8– (1%) | tTG+ and EMA+ or EMA−/DQ2/8+ | ND |
| Rosato E 2009 [27] | 50 Italy | IgA/IgG tTG IgA/IgG EMA in tTG+ | 2 pts tTG+/EMA+ 3 pts tTG+/EMA− (10%) | tTG+/EMA+/– | 4/5 (1 pt refused) (8%) |
| Nisihara R 2011 [28] | 105 Brazil | IgA EMA | All pts EMA– | NA | ND |
| Forbess LJ 2013 [29] | 72 USA | IgA/IgG tTG and IgA/IgG DGP EMA in tTG+ and/or DGP+ | 1 tTG+/EMA− 2 DGP+/EMA− (4%) | tTG+ and/or DGP+ | 0/3 (1 pt died) (0%) |
| Normal Controls | SLE | pSS | SSc | |
|---|---|---|---|---|
| Subjects, n | 14,298 | 580 | 354 | 524 |
| Age, mean ± SD (range) | 53 ± 22 (15–90) | 46 ± 13 (19–83) | 55 ± 12 (21–90) | 61 ± 14 (15–87) |
| Female (%) | 91 | 89 | 97 | 90 |
| Previously diagnosed CD, n | 91 | 8 | 24 | 7 |
| Previously diagnosed CD prevalence, % (95% CI) | 0.64 (0.5–0.8) | 1.38 (0.7–2.7) | 6.78 (4.6–9.9) | 1.34 (0.7–2.7) |
| Subclinical CD, n | ND | 2 | 1 | 0 |
| Overall CD prevalence, % (95% CI) | NA | 1.72 (0.9–3.1) | 7.06 (4.8–10.2) | 1.34 (0.7–2.7) |
| Age at AD diagnosis, mean ± SD (range) | NA | 33 ± 14 (3–79) | 48 ± 12 (20–77) | 51 ± 14 (13–81) |
| SSc Subset | Diffuse | Limited | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Auto-Abs | All | ATA+ * | ACA+ § | Other Auto-Abs+ ^ | None | All | ATA+ | ACA+ | Other Auto-Abs+ | None |
| Subjects, n | 134 | 94 | 8 | 26 | 21 | 390 | 77 | 209 | 90 | 51 |
| % with respect to all subjects | - | 70.1 | 5.9 | 19.4 | 19.4 | - | 19.7 | 53.6 | 23.1 | 13.1 |
| Age, mean ± SD | 57 ± 14 | 57 ± 14 | 63 ± 9 | 58 ± 15 | 53 ± 14 | 62 ± 13 | 60 ± 15 | 64 ± 12 | 63 ± 13 | 59 ± 13 |
| Previously diagnosed CD, n | 6 | 4 | 0 | 2 | 0 | 1 | 0 | 0 | 1 | 0 |
| Previously diagnosed CD prevalence, % (95% CI) | 4.5 (2.1–9.4) | 4.3 (1.7–10.4) | 0 | 7.7 (2.1–24.1) | 0 | 0.3 (0.1–1.4) | 0 | 0 | 1.1 (0.5–1.9) | 0 |
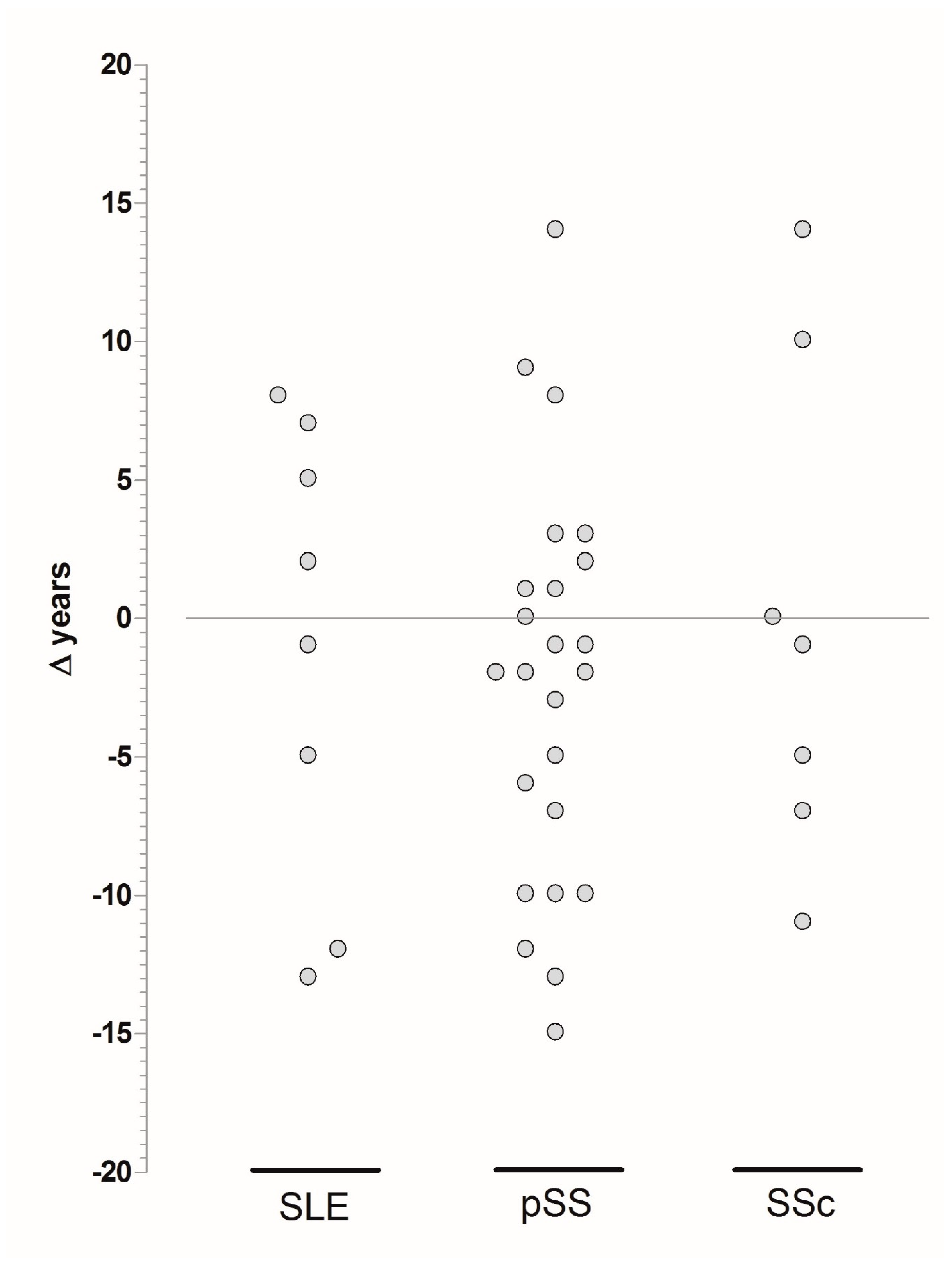
| Disease | Age, Mean ± SD (Range) | Age at Diagnosis, Mean ± SD (Range) | Age at Symptom Onset, Mean ± SD (Range) |
|---|---|---|---|
| SLE–CD (n = 10) | 39 ± 8 (25–50) | 31 ± 11 (14–47) | 26 ± 12 (14–47) |
| SLE–non-CD (n = 570) | 46 ± 13 (19–83) | 33 ± 14 (3–79) | 31 ± 14 (2–72) |
| pSS–CD | 49 ± 9 (34–76) | 43 ± 10 (27–73) | 40 ± 10 (27–74) |
| pSS–non-CD | 55 ± 12 (21–90) * | 48 ± 12 (20–77) ^ | 44 ± 12 (18–76) |
| SSc–CD | 46 ± 16 (15–63) | 40 ± 14 (13–61) | 41 ± 13 (28–60) |
| SSc–non-CD | 61 ± 13 (22–87) * | 51 ± 14 (0–81) ^ | 46 ± 15 (0–81) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bartoloni, E.; Bistoni, O.; Alunno, A.; Cavagna, L.; Nalotto, L.; Baldini, C.; Priori, R.; Fischetti, C.; Fredi, M.; Quartuccio, L.; et al. Celiac Disease Prevalence Is Increased in Primary Sjögren’s Syndrome and Diffuse Systemic Sclerosis: Lessons from a Large Multi-Center Study. J. Clin. Med. 2019, 8, 540. https://doi.org/10.3390/jcm8040540
Bartoloni E, Bistoni O, Alunno A, Cavagna L, Nalotto L, Baldini C, Priori R, Fischetti C, Fredi M, Quartuccio L, et al. Celiac Disease Prevalence Is Increased in Primary Sjögren’s Syndrome and Diffuse Systemic Sclerosis: Lessons from a Large Multi-Center Study. Journal of Clinical Medicine. 2019; 8(4):540. https://doi.org/10.3390/jcm8040540
Chicago/Turabian StyleBartoloni, Elena, Onelia Bistoni, Alessia Alunno, Lorenzo Cavagna, Linda Nalotto, Chiara Baldini, Roberta Priori, Colomba Fischetti, Micaela Fredi, Luca Quartuccio, and et al. 2019. "Celiac Disease Prevalence Is Increased in Primary Sjögren’s Syndrome and Diffuse Systemic Sclerosis: Lessons from a Large Multi-Center Study" Journal of Clinical Medicine 8, no. 4: 540. https://doi.org/10.3390/jcm8040540
APA StyleBartoloni, E., Bistoni, O., Alunno, A., Cavagna, L., Nalotto, L., Baldini, C., Priori, R., Fischetti, C., Fredi, M., Quartuccio, L., Carubbi, F., Montecucco, C., Doria, A., Mosca, M., Valesini, G., Franceschini, F., De Vita, S., Giacomelli, R., Mirabelli, G., ... Gerli, R. (2019). Celiac Disease Prevalence Is Increased in Primary Sjögren’s Syndrome and Diffuse Systemic Sclerosis: Lessons from a Large Multi-Center Study. Journal of Clinical Medicine, 8(4), 540. https://doi.org/10.3390/jcm8040540








