Correlations of the Gastric and Duodenal Microbiota with Histological, Endoscopic, and Symptomatic Gastritis
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Subjects
2.2. Upper Gastrointestinal Endoscopy and Biopsy Procedures
2.3. Histopathological Assessment
2.4. Library Preparation for 16S rRNA Gene Sequencing
2.5. Template Sequencing and Data Availability
2.6. Quality Control of Sequence Data
2.7. Bioinformatics Processing and Sequence Analysis
2.8. Screening and Assessment for Organisms
2.9. Assessment of Alpha and Beta Diversity Indices
2.10. Statistical Analysis
3. Results
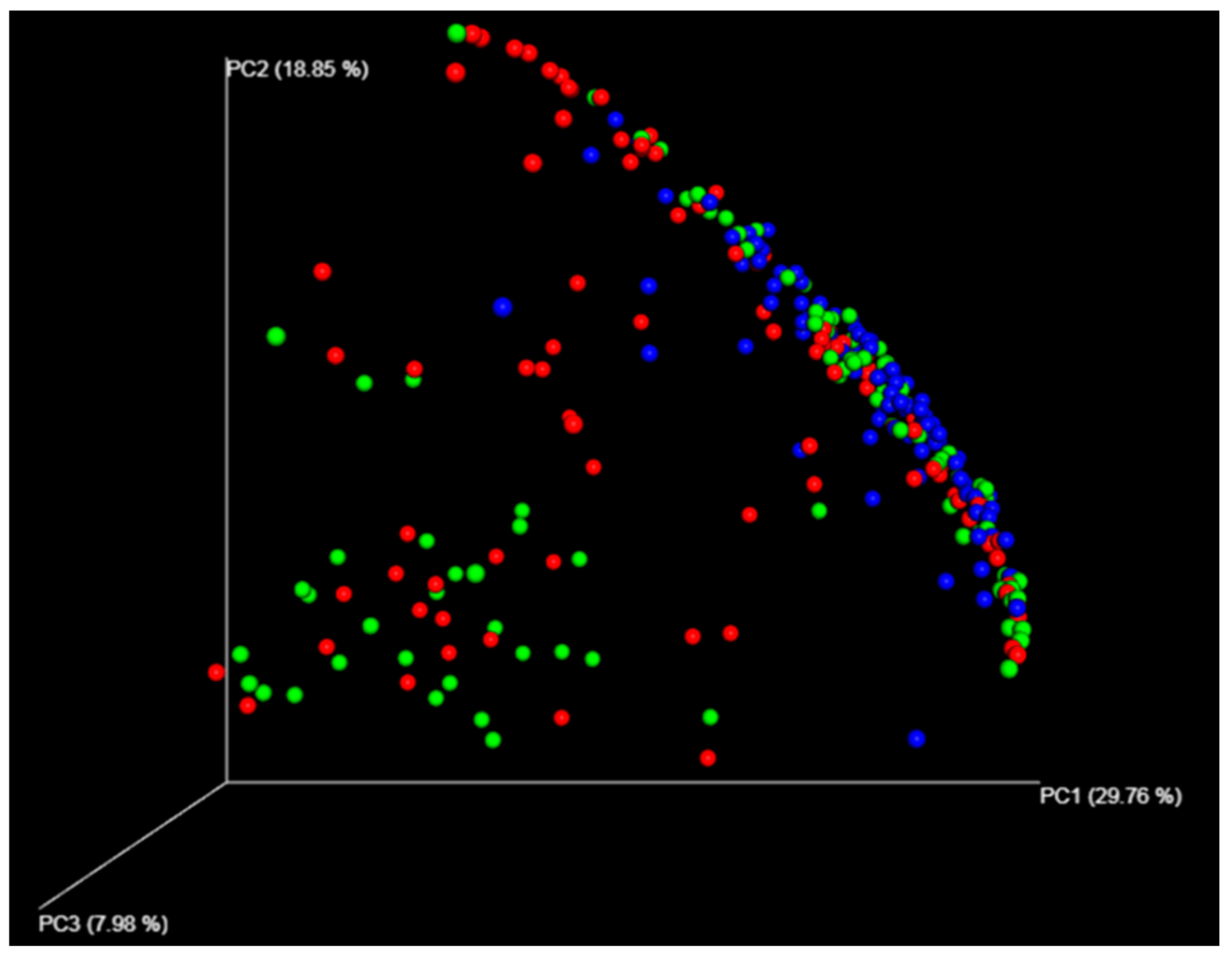
3.1. Different Compositions Between the Gastric and Duodenal Microbiota
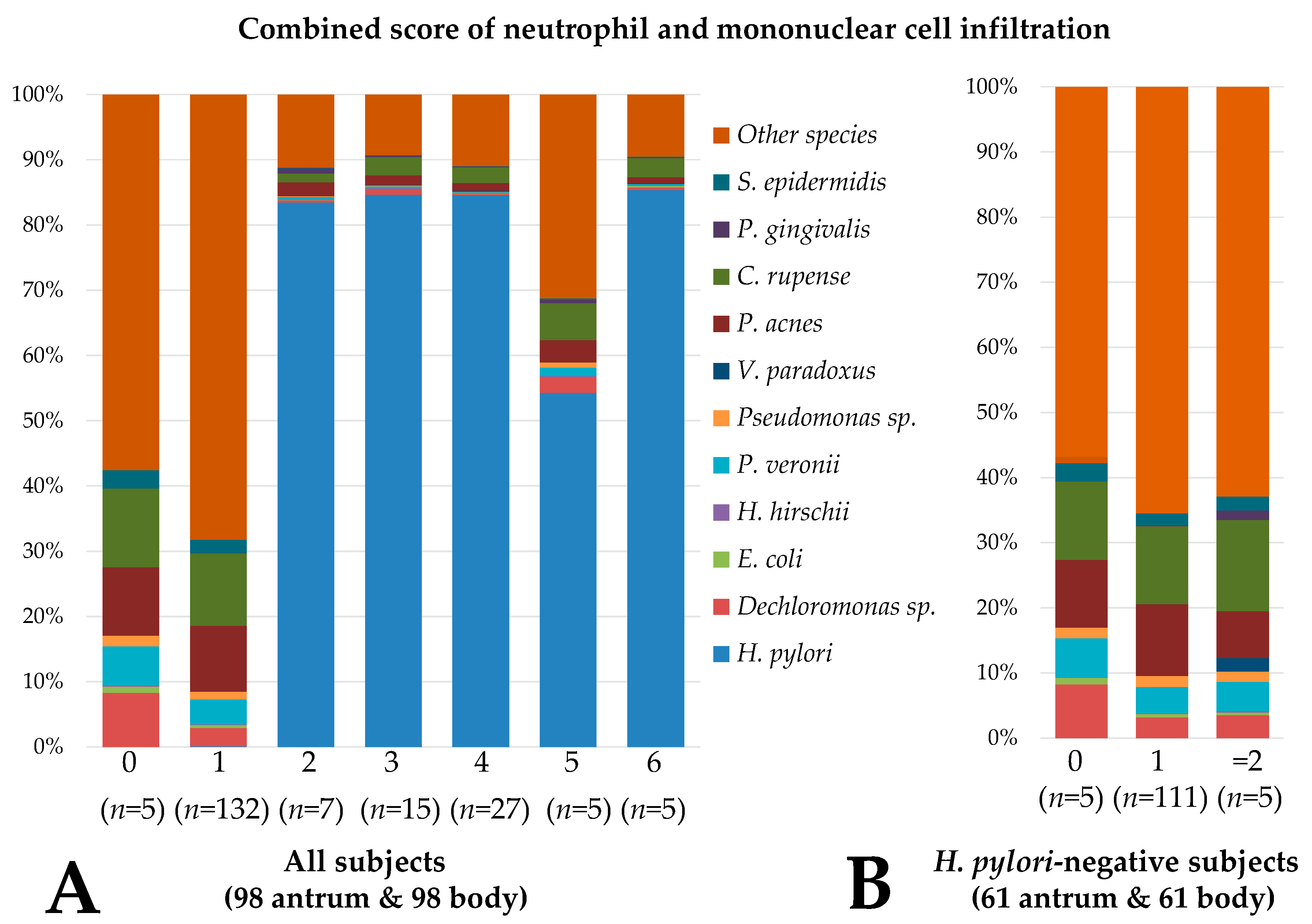
3.2. Gastric and Duodenal Microbiota Related to Mucosal Inflammation
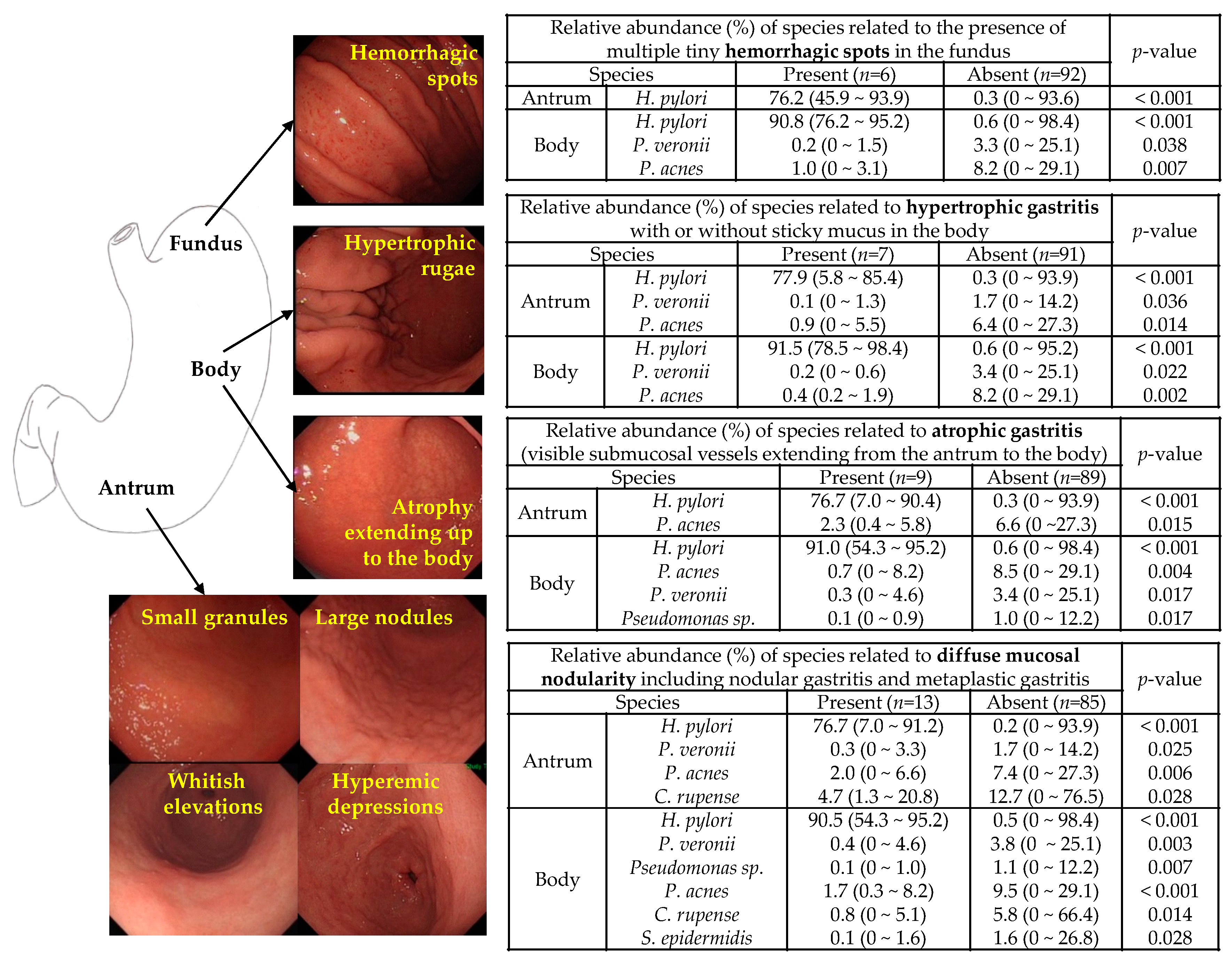
3.3. Microbiota Associated with Abnormal Endoscopic Findings
3.4. Microbiota Associated with the PAGI-SYM Score
3.5. Association Between the Microbiota Correlated with Histological, Endoscopic, and Symptomatic Gastritis
3.6. Microbiota Associated with Gastritis in H. pylori-Negative Subjects
3.7. Differences in Microbial Composition between Corpus- and Antrum-Dominant Atrophic Gastritis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kim, S.E.; Park, H.K.; Kim, N.; Joo, Y.E.; Baik, G.H.; Shin, J.E.; Seo, G.S.; Kim, G.H.; Kim, H.U.; Kim, H.Y.; et al. Prevalence and risk factors of functional dyspepsia: A nationwide multicenter prospective study in Korea. J. Clin. Gastroenterol. 2014, 48, e12–e18. [Google Scholar] [CrossRef] [PubMed]
- Dixon, M.F.; Genta, R.M.; Yardley, J.H.; Correa, P. Classification and grading of gastritis. The updated Sydney System. International Workshop on the Histopathology of Gastritis, Houston 1994. Am. J. Surg. Pathol. 1996, 20, 1161–1181. [Google Scholar] [CrossRef] [PubMed]
- Venerito, M.; Varbanova, M.; Röhl, F.W.; Reinhold, D.; Frauenschläger, K.; Jechorek, D.; Weigt, J.; Link, A.; Malfertheiner, P. Oxyntic gastric atrophy in Helicobacter pylori gastritis is distinct from autoimmune gastritis. J. Clin. Pathol. 2016, 69, 677–685. [Google Scholar] [CrossRef] [PubMed]
- Sugimoto, M.; Ban, H.; Ichikawa, H.; Sahara, S.; Otsuka, T.; Inatomi, O.; Bamba, S.; Furuta, T.; Andoh, A. Efficacy of the Kyoto classification of gastritis in identifying patients at high risk for gastric cancer. Intern. Med. 2017, 56, 579–586. [Google Scholar] [CrossRef] [PubMed]
- Eun, C.S.; Kim, B.K.; Han, D.S.; Kim, S.Y.; Kim, K.M.; Choi, B.Y.; Song, K.S.; Kim, Y.S.; Kim, J.F. Differences in gastric mucosal microbiota profiling in patients with chronic gastritis, intestinal metaplasia, and gastric cancer using pyrosequencing methods. Helicobacter 2014, 19, 407–416. [Google Scholar] [CrossRef] [PubMed]
- Thorell, K.; Bengtsson-Palme, J.; Liu, O.H.; Palacios Gonzales, R.V.; Nookaew, I.; Rabeneck, L.; Paszat, L.; Graham, D.Y.; Nielsen, J.; Lundin, S.B.; et al. Analysis of the viable microbiota and Helicobacter pylori transcriptome in gastric infection and early stages of carcinogenesis. Infect. Immun. 2017, 85. [Google Scholar] [CrossRef] [PubMed]
- Rugge, M.; Genta, R.M. Staging and grading of chronic gastritis. Hum. Pathol. 2005, 36, 228–233. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, R.M.; Pereira-Marques, J.; Pinto-Ribeiro, I.; Costa, J.L.; Carneiro, F.; Machado, J.C.; Figueiredo, C. Gastric microbial community profiling reveals a dysbiotic cancer-associated microbiota. Gut 2018, 67, 226–236. [Google Scholar] [CrossRef] [PubMed]
- Li, T.H.; Qin, Y.; Sham, P.C.; Lau, K.S.; Chu, K.M.; Leung, W.K. Alterations in gastric microbiota after H. pylori eradication and in different histological stages of gastric carcinogenesis. Sci. Rep. 2017, 7, 44935. [Google Scholar] [CrossRef] [PubMed]
- Aviles-Jimenez, F.; Vazquez-Jimenez, F.; Medrano-Guzman, R.; Mantilla, A.; Torres, J. Stomach microbiota composition varies between patients with non-atrophic gastritis and patients with intestinal type of gastric cancer. Sci. Rep. 2014, 4, 4202. [Google Scholar] [CrossRef] [PubMed]
- Yang, I.; Woltemate, S.; Piazuelo, M.B.; Bravo, L.E.; Yepez, M.C.; Romero-Gallo, J.; Delgado, A.G.; Wilson, K.T.; Peek, R.M.; Correa, P.; et al. Different gastric microbiota compositions in two human populations with high and low gastric cancer risk in Colombia. Sci. Rep. 2016, 6, 18594. [Google Scholar] [CrossRef] [PubMed]
- Sohn, S.H.; Kim, N.; Jo, H.J.; Kim, J.; Park, J.H.; Nam, R.H.; Seok, Y.J.; Kim, Y.R.; Lee, D.H. Analysis of gastric body microbiota by pyrosequencing: Possible role of bacteria other than Helicobacter pylori. J. Cancer Prev. 2017, 22, 115–125. [Google Scholar] [CrossRef] [PubMed]
- Jo, H.J.; Kim, J.; Kim, N.; Park, J.H.; Nam, R.H.; Seok, Y.J.; Kim, Y.R.; Kim, J.S.; Kim, J.M.; Kim, J.M.; et al. Analysis of gastric microbiota by pyrosequencing: Minor role of bacteria other than Helicobacter pylori in the gastric carcinogenesis. Helicobacter 2016, 21, 364–374. [Google Scholar] [CrossRef] [PubMed]
- Schulz, C.; Schütte, K.; Koch, N.; Vilchez-Vargas, R.; Wos-Oxley, M.L.; Oxley, A.P.A.; Vital, M.; Malfertheiner, P.; Pieper, D. The active bacterial assemblages of the upper GI tract in individuals with and without Helicobacter infection. Gut 2018, 67, 216–225. [Google Scholar] [CrossRef] [PubMed]
- Walker, M.M.; Talley, N.J. The role of duodenal inflammation in functional dyspepsia. J. Clin. Gastroenterol. 2017, 51, 12–18. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.Y.; Masaoka, T.; Han, H.S.; Matsuzaki, J.; Hong, M.J.; Fukuhara, S.; Choi, H.S.; Suzuki, H. A prospective study on symptom generation according to spicy food intake and TRPV1 genotypes in functional dyspepsia patients. Neurogastroenterol. Motil. 2016, 28, 1401–1408. [Google Scholar] [CrossRef] [PubMed]
- Kato, T.; Yagi, N.; Kamada, T.; Shimbo, T.; Watanabe, H.; Ida, K.; Study Group for Establishing Endoscopic Diagnosis of Chronic Gastritis. Diagnosis of Helicobacter pylori infection in gastric mucosa by endoscopic features: A multicenter prospective study. Dig. Endosc. 2013, 25, 508–518. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.Y. Endoscopic gastritis, serum pepsinogen assay, and Helicobacter pylori infection. Korean J. Intern. Med. 2016, 31, 835–844. [Google Scholar] [CrossRef] [PubMed]
- Dong, Q.; Xin, Y.; Wang, L.; Meng, X.; Yu, X.; Lu, L.; Xuan, S. Characterization of gastric microbiota in twins. Curr. Microbiol. 2017, 74, 224–229. [Google Scholar] [CrossRef] [PubMed]
- Von Rosenvinge, E.C.; Song, Y.; White, J.R.; Maddox, C.; Blanchard, T.; Fricke, W.F. Immune status, antibiotic medication and pH are associated with changes in the stomach fluid microbiota. ISME J. 2013, 7, 1354–1366. [Google Scholar] [CrossRef] [PubMed]
- Oh, S.Y.; Kim, W.Y.; Hwang, T.S.; Han, H.S.; Lim, S.D.; Kim, W.S. Development of an ammonium sulfate DNA extraction method for obtaining amplifiable DNA in a small number of cells and its application to clinical specimens. Biomed. Res. Int. 2013, 2013, 546727. [Google Scholar] [CrossRef] [PubMed]
- Mao, D.P.; Zhou, Q.; Chen, C.Y.; Quan, Z.X. Coverage evaluation of universal bacterial primers using the metagenomic datasets. BMC Microbiol. 2012, 12, 66. [Google Scholar] [CrossRef] [PubMed]
- Dubey, A.K.; Uppadhyaya, N.; Nilawe, P.; Chauhan, N.; Kumar, S.; Gupta, U.A.; Bhaduri, A. LogMPIE, pan-India profiling of the human gut microbiome using 16S rRNA sequencing. Sci. Data 2018, 5, 180232. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Kim, N.; Jo, H.J.; Park, J.H.; Nam, R.H.; Seok, Y.J.; Kim, Y.R.; Kim, J.S.; Kim, J.M.; Kim, J.M.; et al. An appropriate cutoff value for determining the colonization of Helicobacter pylori by the pyrosequencing method: Comparison with conventional methods. Helicobacter 2015, 20, 370–380. [Google Scholar] [CrossRef] [PubMed]
- Shichijo, S.; Hirata, Y.; Niikura, R.; Hayakawa, Y.; Yamada, A.; Koike, K. Association between gastric cancer and the Kyoto classification of gastritis. J. Gastroenterol. Hepatol. 2017, 32, 1581–1586. [Google Scholar] [CrossRef] [PubMed]
- Kamada, T.; Inoue, K.; Haruma, K. Kyoto classification of gastritis. Jpn. J. Helicobacter. Res. 2015, 16, 80–85. [Google Scholar]
- Watanabe, K.; Nagata, N.; Nakashima, R.; Furuhata, E.; Shimbo, T.; Kobayakawa, M.; Sakurai, T.; Imbe, K.; Niikura, R.; Yokoi, C.; et al. Predictive findings for Helicobacter pylori-uninfected, -infected and -eradicated gastric mucosa: Validation study. World J. Gastroenterol. 2013, 19, 4374–4379. [Google Scholar] [CrossRef] [PubMed]
- Yuan, X.; Liu, Y.; Kong, J.; Gu, B.; Qi, Y.; Wang, X.; Sun, M.; Chen, P.; Sun, W.; Wang, H.; et al. Different frequencies of Porphyromonas gingivalis infection in cancers of the upper digestive tract. Cancer Lett. 2017, 404, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Shiota, S.; Thrift, A.P.; Green, L.; Shah, R.; Verstovsek, G.; Rugge, M.; Graham, D.Y.; El-Serag, H.B. Clinical manifestations of Helicobacter pylori-negative gastritis. Clin. Gastroenterol. Hepatol. 2017, 15, 1037–1046. [Google Scholar] [CrossRef] [PubMed]
- Yoshioka, T.; Takeshita, E.; Sakata, Y.; Hara, M.; Akutagawa, K.; Sakata, N.; Endo, H.; Ohyama, T.; Matsunaga, K.; Tanaka, Y.; et al. Helicobacter pylori infection status had no influence on upper gastrointestinal symptoms: A cross-sectional analysis of 3005 Japanese subjects without upper gastrointestinal lesions undergoing medical health checkups. Esophagus 2017, 14, 249–253. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.P.; Lee, S.Y.; Kim, J.H.; Sung, I.K.; Park, H.S.; Shim, C.S. Factors related to upper gastrointestinal symptom generation in 2275 Helicobacter pylori seroprevalent adults. Dig. Dis. Sci. 2017, 62, 1561–1570. [Google Scholar] [CrossRef] [PubMed]
- Kawakubo, H.; Tanaka, Y.; Tsuruoka, N.; Hara, M.; Yamamoto, K.; Hidaka, H.; Sakata, Y.; Shimoda, R.; Iwakiri, R.; Kusano, M.; et al. Upper gastrointestinal symptoms are more frequent in female than male young healthy Japanese volunteers as evaluated by questionnaire. J. Neurogastroenterol. Motil. 2016, 22, 248–253. [Google Scholar] [CrossRef] [PubMed]
- Konturek, P.C.; Brzozowski, T.; Konturek, S.J. Stress and the gut: Pathophysiology, clinical consequences, diagnostic approach and treatment options. J. Physiol. Pharmacol. 2011, 62, 591–599. [Google Scholar] [PubMed]
- Budzyński, J.; Kłopocka, M. Brain-gut axis in the pathogenesis of Helicobacter pylori infection. World J. Gastroenterol. 2014, 20, 5212–5225. [Google Scholar] [CrossRef] [PubMed]
- Ghoshal, U.C.; Gwee, K.A.; Chen, M.; Gong, X.R.; Pratap, N.; Hou, X.; Syam, A.F.; Abdullah, M.; Bak, Y.T.; Choi, M.G.; et al. Development, translation and validation of enhanced Asian Rome III questionnaires for diagnosis of functional bowel diseases in major Asian languages: A Rome foundation-Asian neurogastroenterology and motility association working team report. J. Neurogastroenterol. Motil. 2015, 21, 83–92. [Google Scholar] [CrossRef] [PubMed]
| Findings | With no Helicobacter pylori Infection (n = 61) | With H. pylori Infection (n = 31) | With past H. pylori Infection (n = 6) |
|---|---|---|---|
| Age (year-old) | 35.4 ± 11.9 | 40.5 ± 9.9 | 49.8 ± 13.9 *,** |
| Sex (male:female) | 16:45 | 3:28 | 2:4 |
| Body mass index (kg/m2) | 21.9 ± 3.4 | 22.4 ± 3.8 | 21.7 ± 2.2 |
| Antrum: 16S ribosomal RNA (rRNA) sequencing analysis findings | |||
| Target reads | 20,759 ± 18373 | 18,744 ± 11,508 | 26,175 ± 9597 |
| Chao 1 index | 46.9 ± 16.6 | 46.7 ± 12.6 | 60.2 ± 9.4 |
| Shannon diversity index | 3.7 ± 0.5 | 3.4 ± 0.7 * | 3.9 ± 0.3 ** |
| Simpson diversity index | 0.85 ± 0.05 | 0.78 ± 0.12 * | 0.86 ± 0.04 ** |
| Body: 16S rRNA sequencing analysis findings | |||
| Target reads | 14,998 ± 9734 | 13,041 ± 7148 | 12,271 ± 9245 |
| Chao 1 index | 39.2 ± 7.4 | 32.0 ± 8.9 * | 33.8 ± 12.1 |
| Shannon diversity index | 3.4 ± 0.4 | 2.6 ± 0.6 * | 3.3 ± 0.7 ** |
| Simpson diversity index | 0.82 ± 0.04 | 0.68 ± 0.13 * | 0.82 ± 0.06 ** |
| Duodenum: 16S rRNA sequencing analysis findings | |||
| Target reads | 9329 ± 4067 | 11,440 ± 6949 | 9834 ± 2278 |
| Chao 1 index | 31.5 ± 6.2 | 37.0 ± 7.2 * | 32.8 ± 2.7 |
| Shannon diversity index | 3.2 ± 0.3 | 3.5 ± 0.4 * | 3.3 ± 0.2 |
| Simpson diversity index | 0.81 ± 0.05 | 0.83 ± 0.05 | 0.83 ± 0.02 |
| Antrum: updated Sydney system (no:mild:moderate:marked) | |||
| Neutrophil | 4:53:4:0 | 0:5:20:6 * | 0:6:0:0 ** |
| Mononuclear cell | 58:2:0:1 | 6:10:12:3 * | 6:0:0:0 ** |
| Atrophy | 37:24:0:0 | 15:16:0:0 | 3:3:0:0 |
| Intestinal metaplasia | 60:1:0:0 | 28:2:1:0 | 5:1:0:0 |
| Body: updated Sydney system (no:mild:moderate:marked) | |||
| Neutrophil | 1:58:1:1 | 0:8:20:3 * | 0:6:0:0 ** |
| Mononuclear cell | 59:0:1:1 | 6:6:18:1 * | 6:0:0:0 ** |
| Atrophy | 35:26:0:0 | 23:8:0:0 | 4:2:0:0 |
| Intestinal metaplasia | 67:0:0:0 | 30:1:0:0 | 6:0:0:0 |
| Duodenum: inflammatory cell infiltration (no:mild) | |||
| Neutrophil | 55:6 | 22:8 | 4:2 |
| Mononuclear cell | 9:52 | 4:27 | 0:6 |
| Microbiota | Combined Inflammation Score | ||
|---|---|---|---|
| Site | Level | Positive Correlation | Negative Correlation |
| Antrum | Genus | Helicobacter 0.794 | Dechloromonas − 0.354 |
| Staphylococcus − 0.326 | |||
| Pseudomonas − 0.294 | |||
| Species | Helicobacter pylori 0.800 | Propionibacterium acnes − 0.445 | |
| Pseudomonas veronii − 0.360 | |||
| Dechloromonas sp. − 0.323 | |||
| Cloacibacterium rupense − 0.320 | |||
| Pseudomonas sp. − 0.320 | |||
| Staphylococcus epidermidis − 0.280 | |||
| Hydrogenophilus hirschii − 0.250 | |||
| Streptococcus sp. − 0.231 | |||
| Body | Genus | Helicobacter 0.713 Variovorax 0.324 | Propionibacterium − 0.469 |
| Corynebacterium − 0.457 | |||
| Methylobacterium − 0.454 | |||
| Pseudomonas − 0.402 | |||
| Escherichia − 0.342 | |||
| Hydrogenophilus − 0.307 | |||
| Prevotella − 0.307 | |||
| Staphylococcus − 0.280 | |||
| Sphingomonas − 0.258 | |||
| Dechloromonas sp. − 0.256 | |||
| Species | Helicobacter pylori 0.719 Variovorax paradoxus 0.325 Porphyromonas gingivalis 0.249 | Propionibacterium acnes − 0.514 | |
| Pseudomonas sp. − 0.377 | |||
| Pseudomonas veronii − 0.371 | |||
| Escherichia coli − 0.331 | |||
| Dechloromonas sp. − 0.279 | |||
| Bacillus sp. − 0.274 | |||
| Staphylococcus epidermidis − 0.265 | |||
| Duodenum | Genus | Helicobacter 0.215 | – |
| Species | Leptotrichia genomosp. 0.218 Helicobacter pylori 0.201 | Moraxella osloensis − 0.279 | |
| Staphylococcus epidermidis − 0.245 | |||
| Actinomyces odontolyticus − 0.217 | |||
| PAGI-SYM Questionnaires | Site | Correlation with the Relative Abundance of Species | |
|---|---|---|---|
| Positive Correlation | Negative Correlation | ||
| Heartburn and regurgitation | Stomach | Corynebacterium segmentosum 0.189 | Prevotella pallens − 0.193 |
| Bloating | Duodenum | Prevotella nanceiensis 0.283 | Propionibacterium acnes − 0.220 Prevotella pallens − 0.215 |
| Stomach | Helicobacter pylori 0.239 | - | |
| Nausea and vomiting | Duodenum | Prevotella nanceiensis 0.302 Actinomyces odontolyticus 0.265 | - |
| Stomach | H. pylori 0.194 | Propionibacterium acnes − 0.234 Staphylococcus epidermidis − 0.145 | |
| Upper abdominal pain | Stomach | - | - |
| Duodenum | - | - | |
| Fullness and early satiety | Stomach | - | Dechloromonas sp. − 0.189 |
| Lower abdominal pain | Duodenum | Prevotella nanceiensis 0.313 | - |
| Stomach | H. pylori 0.228 | ||
| PAGI-SYM Questionnaires | Site | Correlations Found in 61 H. pylori-Negative Subjects | |
| Positive Correlation | Negative Correlation | ||
| Heartburn & regurgitation | Stomach | Corynebacterium segmentosum 0.274 | - |
| Bloating | Duodenum | Pseudomonas grimontii 0.308 Cloacibacterium normanense 0.281 | - |
| Nausea and vomiting | Duodenum | Actinomyces odontolyticus 0.348 Prevotella nanceiensis 0.258 | - |
| Stomach | Neisseria enlongata 0.245 | - | |
| Upper abdominal pain | Duodenum | Actinobacillus parahaemolyticus 0.364 Rothia mucilaginosa 0.246 Pseudomonas grimontii 0.242 | Paracoccus sp. − 0.242 Neisseria perflava − 0.242 |
| Fullness and early satiety | Duodenum | Porphyromonas catoniae 0.332 | - |
| Stomach | Pantoea sp. 0.278 | - | |
| Lower abdominal pain | Stomach | Neisseria enlongata 0.249 | - |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Han, H.S.; Lee, S.-Y.; Oh, S.Y.; Moon, H.W.; Cho, H.; Kim, J.-H. Correlations of the Gastric and Duodenal Microbiota with Histological, Endoscopic, and Symptomatic Gastritis. J. Clin. Med. 2019, 8, 312. https://doi.org/10.3390/jcm8030312
Han HS, Lee S-Y, Oh SY, Moon HW, Cho H, Kim J-H. Correlations of the Gastric and Duodenal Microbiota with Histological, Endoscopic, and Symptomatic Gastritis. Journal of Clinical Medicine. 2019; 8(3):312. https://doi.org/10.3390/jcm8030312
Chicago/Turabian StyleHan, Hye Seung, Sun-Young Lee, Seo Young Oh, Hee Won Moon, Hyunseok Cho, and Ji-Hoon Kim. 2019. "Correlations of the Gastric and Duodenal Microbiota with Histological, Endoscopic, and Symptomatic Gastritis" Journal of Clinical Medicine 8, no. 3: 312. https://doi.org/10.3390/jcm8030312
APA StyleHan, H. S., Lee, S.-Y., Oh, S. Y., Moon, H. W., Cho, H., & Kim, J.-H. (2019). Correlations of the Gastric and Duodenal Microbiota with Histological, Endoscopic, and Symptomatic Gastritis. Journal of Clinical Medicine, 8(3), 312. https://doi.org/10.3390/jcm8030312





