Metabolic Syndrome Including Glycated Hemoglobin A1c in Adults: Is It Time to Change?
Abstract
1. Introduction
2. Experimental Section
2.1. Variables
2.2. Statistical Analysis
2.2.1. MetS as Quantitative Scale
2.2.2. MetS as a Dichotomic Scale
3. Results
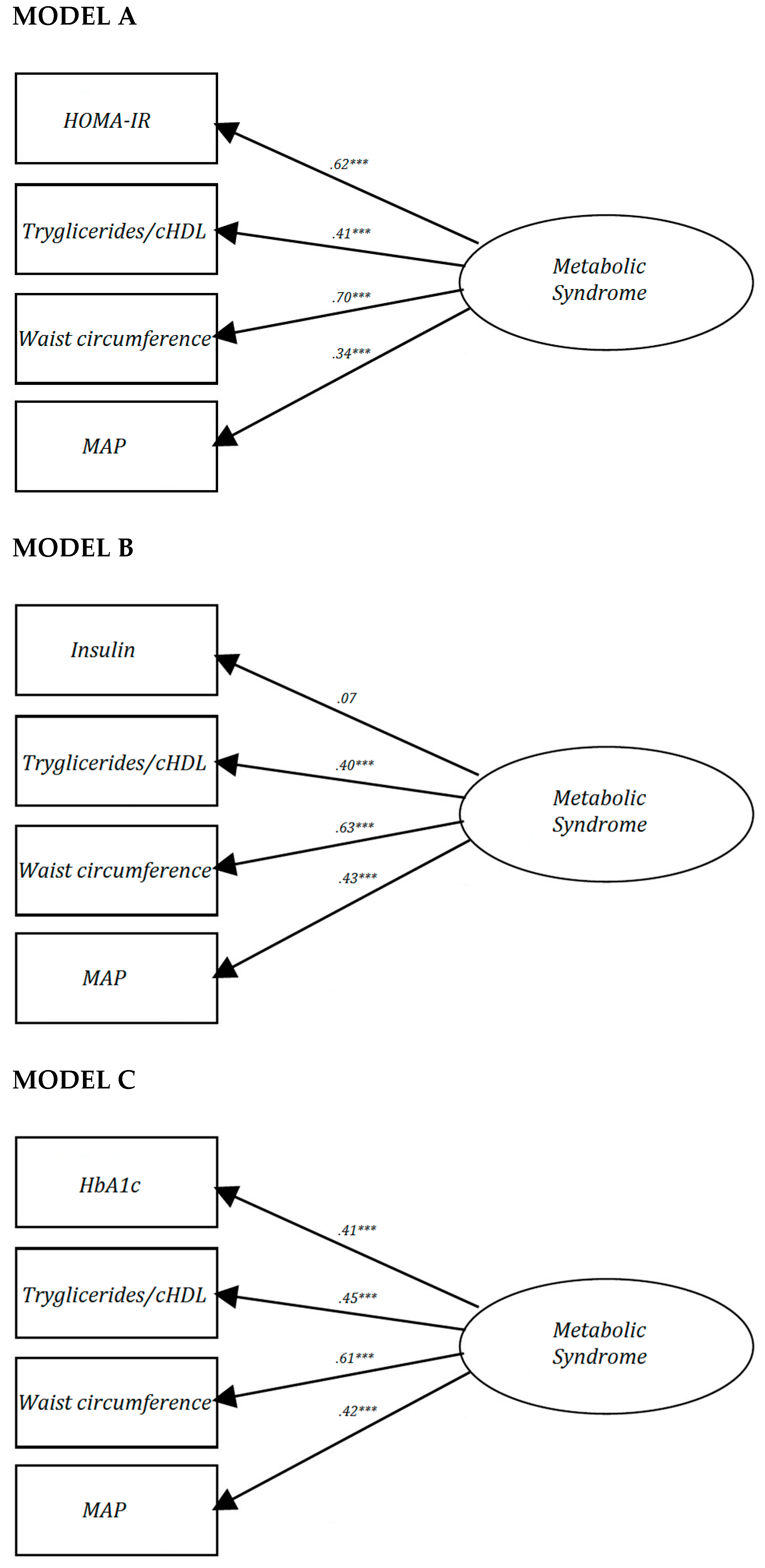
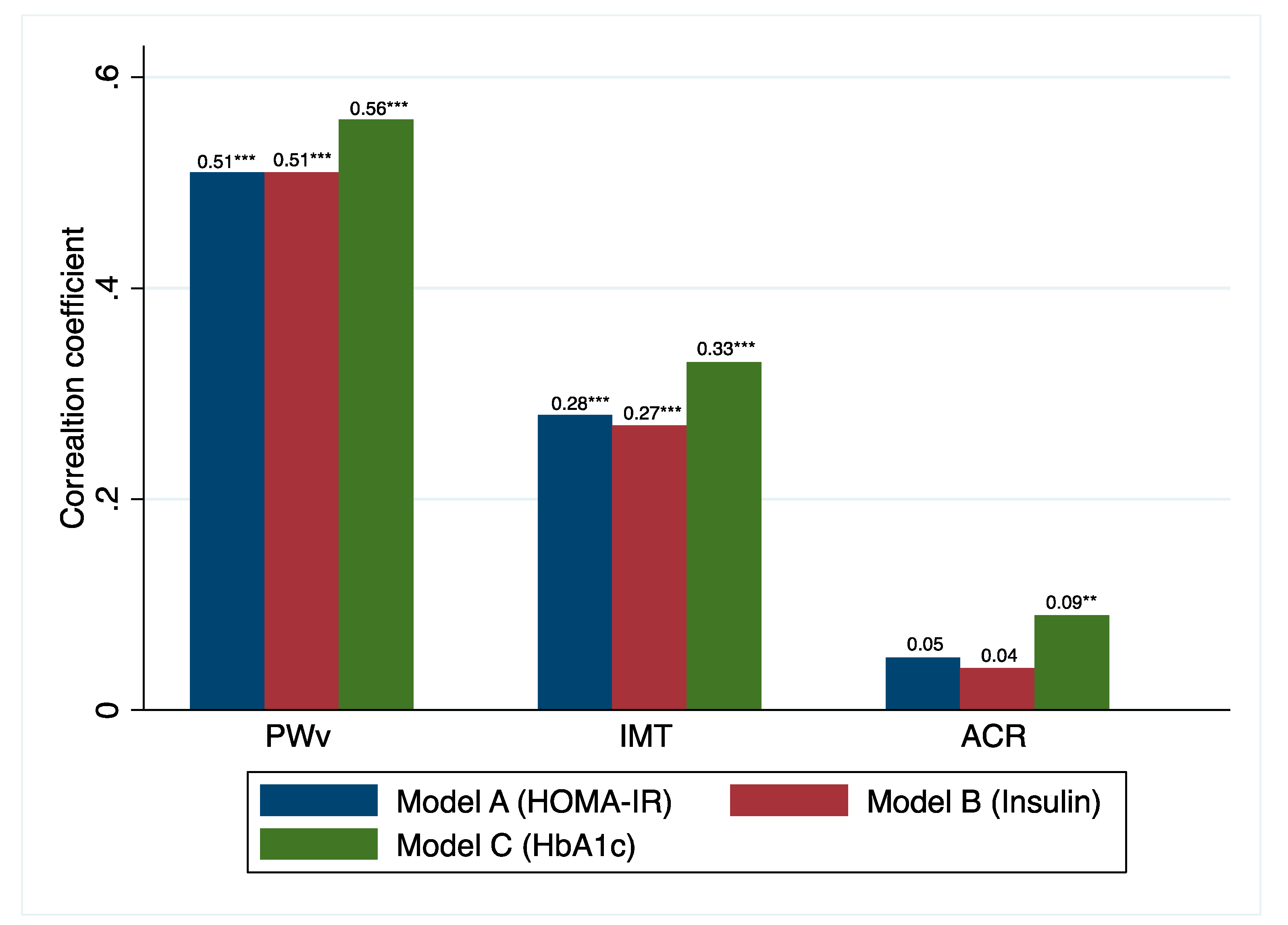
3.1. MetS as a Quantitative Scale
3.2. MetS as a Quantitative Scale
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Alberti, K.G.; Eckel, R.H.; Grundy, S.M.; Zimmet, P.Z.; Cleeman, J.I.; Donato, K.A.; Fruchart, J.C.; James, W.P.; Loria, C.M.; Smith, S.C., Jr. International Diabetes Federation Task Force on Epidemiology and Prevention; Hational Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; International Association for the Study of Obesity. Harmonizing the metabolic syndrome: A joint interim statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation 2009, 120, 1640–1645. [Google Scholar]
- Grundy, S.M. Metabolic syndrome update. Trends Cardiovasc. Med. 2016, 26, 364–373. [Google Scholar] [CrossRef]
- Mottillo, S.; Filion, K.B.; Genest, J.; Joseph, L.; Pilote, L.; Poirier, P.; Rinfret, S.; Schiffrin, E.L.; Eisenberg, M.J. The metabolic syndrome and cardiovascular risk: A systematic review and meta-analysis. J. Am. Coll. Cardiol. 2010, 56, 1113–1132. [Google Scholar] [CrossRef] [PubMed]
- Rachas, A.; Raffaitin, C.; Barberger-Gateau, P.; Helmer, C.; Ritchier, K.; Tzourio, C.; Amouyel, P.; Ducimetière, P.; Empana, J.P. Clinical usefulness of the metabolic syndrome for the risk of coronary heart disease does not exceed the sum of its individual components in older men and women. The Three-City (3C) Study Heart 2012, 98, 650–655. [Google Scholar] [CrossRef] [PubMed]
- Kahn, R.; Buse, J.; Ferrannini, E.; Stern, M. The metabolic syndrome: Time for a critical appraisal. Joint statement from the American Diabetes Association and the European Association for the Study of Diabetes. Diabetologia 2005, 48, 1684–1699. [Google Scholar] [CrossRef] [PubMed]
- Shen, B.J.; Goldberg, R.B.; Llabre, M.M.; Schneiderman, N. Is the factor structure of the metabolic syndrome comparable between men and women and across three ethnic groups: The Miami Community Health Study. Ann. Epidemiol. 2006, 16, 131–137. [Google Scholar] [CrossRef]
- Novak, S.; Stapleton, L.M.; Litaker, J.R.; Lawson, K.A. A confirmatory factor analysis evaluation of the coronary heart disease risk factors of metabolic syndrome with emphasis on the insulin resistance factor. Diabetes Obes. Metab. 2003, 5, 388–396. [Google Scholar] [CrossRef]
- Shah, S.; Novak, S.; Stapleton, L.M. Evaluation and comparison of models of metabolic syndrome using confirmatory factor analysis. Eur. J. Epidemiol. 2006, 21, 343–349. [Google Scholar] [CrossRef]
- Pladevall, M.; Singal, B.; Williams, L.K.; Brotons, C.; Guyer, H.; Sadurni, J.; Falces, C.; Serrano-Rios, M.; Gabriel, R.; Shaw, J.E.; et al. A single factor underlies the metabolic syndrome: A confirmatory factor analysis. Diabetes Care 2006, 29, 113–122. [Google Scholar] [CrossRef][Green Version]
- Solera-Martínez, M.; López-Martínez, S.; Sánchez-López, M.; Moya-Martinez, P.; Notario-Pacheco, B.; Arias-Palencia, N.; Franquelo-Morales, P.; Martinez-Vizcaino, V. Validity of a single-factor model underlying the metabolic syndrome in young adults: Confirmatory factor analysis. Rev. Esp. Cardiol. 2011, 64, 379–384. [Google Scholar] [CrossRef]
- Cavero-Redondo, I.; Peleteiro, B.; Álvarez-Bueno, C.; Rodriguez-Artalejo, F.; Martínez-Vizcaíno, V. Glycated haemoglobin A1c as a risk factor of cardiovascular outcomes and all-cause mortality in diabetic and non-diabetic populations: A systematic review and meta-analysis. BMJ Open 2017, 7, e015949. [Google Scholar] [CrossRef] [PubMed]
- Cavero-Redondo, I.; Martínez-Vizcaíno, V.; Álvarez-Bueno, C.; Recio-Rodríguez, J.I.; Gómez-Marcos, M.A.; García-Ortiz, L. Relationship between glycaemic levels and arterial stiffness in non-diabetic adults. Med. Clin. (Engl. Ed.) 2018, 150, 56–60. [Google Scholar] [CrossRef]
- Scicali, R.; Giral, P.; Gallo, A.; Di Pino, A.; Rabuazzo, A.M.; Purrello, F.; Cluzel, P.; Redheuil, A.; Bruckert, E.; Rosenbaum, D. HbA1c increase is associated with higher coronary and peripheral atherosclerotic burden in non diabetic patients. Atherosclerosis 2016, 255, 102–108. [Google Scholar] [CrossRef] [PubMed]
- Low, S.; Zhang, X.; Wang, J.; Yeoh, L.Y.; Liu, Y.L.; Ang, S.F.; Subramaniam, T.; Sum, C.F.; Lim, S.C. Impact of HbA1c trajectories on chronic kidney disease progression in type 2 diabetes. Nephrology 2018, 24, 1026–1032. [Google Scholar] [CrossRef]
- American Diabetes Association. 2. Classification and diagnosis of diabetes. Diabetes Care 2017, 40, S11–S24. [Google Scholar] [CrossRef]
- World Health Organization. Use of Glycated Haemoglobin (HbA1c) in the Diagnosis of Diabetes Mellitus: Abbreviated Report of a WHO Consultation; WHO: Geneva, Switzerland, 2011. [Google Scholar]
- Martinez-Vizcaino, V.; Cavero-Redondo, I.; Álvarez-Bueno, C.; Rodríguez-Artalejo, F. The accuracy of diagnostic methods for diabetic retinopathy: A systematic review and meta-analysis. PLoS ONE 2016, 11, e0154411. [Google Scholar] [CrossRef]
- Recio-Rodríguez, J.I.; Martín-Cantera, C.; González-Viejo, N.; Gómez-Arranz, A.; Ariataleannizbeascoa, M.S.; Schmolling-Guinovart, Y.; Maderuelo-Fernandez, J.; Perez-Arechaederra, D.; Rodriguez-Sanchez, E.; Gomez-Marcos, M.A.; et al. Effectiveness of a smartphone application for improving healthy lifestyles, a randomized clinical trial (EVIDENT II): Study protocol. BMC Public Health 2014, 14, 254. [Google Scholar] [CrossRef]
- World Medical Association. World Medical Association Declaration of Helsinki. Ethical principles for medical research involving human subjects. Bull. World Health Organ. 2001, 79, 373. [Google Scholar]
- O’brien, E.; Asmar, R.; Beilin, L.; Imai, Y.; Mancia, G.; Mengden, T.; Myers, T.; Padfield, M.; Palatini, P.; Parati, P.; et al. Practice guidelines of the European Society of Hypertension for clinic, ambulatory and self blood pressure measurement. J. Hypertens. 2005, 23, 697–701. [Google Scholar] [CrossRef]
- Van Bortel, L.M.; Laurent, S.; Boutouyrie, P.; Chowienczyk, P.; Cruickshank, J.K.; De Backer, T.; Filipovsky, J.; Huybrechts, S.; Mattace-Raso, F.; Protogerou, A.D.; et al. Expert consensus document on the measurement of aortic stiffness in daily practice using carotid-femoral pulse wave velocity. J. Hypertens. 2012, 30, 445–448. [Google Scholar] [CrossRef]
- Gomez-Marcos, M.A.; Recio-Rodriguez, J.I.; Patino-Alonso, M.C.; Agudo-Conde, C.; Gomez-Sanchez, L.; Gomez-Sanchez, M.; Rodriguez-Sanchez, E.; Garcia-Ortiz, L. Protocol for measuring carotid intima-media thickness that best correlates with cardiovascular risk and target organ damage. Am. J. Hypertens. 2012, 25, 955–961. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Vizcaíno, V.; Martínez, M.S.; Aguilar, F.S.; Martinez, S.S.; Gutierrez, R.F.; Lopez, M.S.; Martinez, P.M.; Rodriguez-Artalejo, F. Validity of a single-factor model underlying the metabolic syndrome in children: A confirmatory factor analysis. Diabetes Care 2010, 33, 1370–1372. [Google Scholar] [CrossRef] [PubMed]
- Hillier, T.A.; Rousseau, A.; Lange, C.; Lepinay, P.; Cailleau, M.; Novak, M.; Calliez, E.; Ducimetière, P.; Balkau, B. Practical way to assess metabolic syndrome using a continuous score obtained from principal components analysis. Diabetologia 2006, 49, 1528–1535. [Google Scholar] [CrossRef] [PubMed]
- Hu, L.; Bentler, P.M. Cutoff criteria for fit indexes in covariance structure analysis: Conventional criteria versus new alternatives. Struct. Equ. Model. Multidiscip. J. 1999, 6, 1–55. [Google Scholar] [CrossRef]
- Alberti, K.G.; Zimmet, P.; Shaw, J. Metabolic syndrome-a new world-wide definition. A Consensus Statement from the International Diabetes Federation. Diabet. Med. 2006, 23, 469–480. [Google Scholar] [CrossRef]
- De Groot, E.; Hovingh, G.K.; Wiegman, A.; Duriez, P.; Smit, A.J.; Fruchart, J.C.; Kastelein, J.J.P. Measurement of arterial wall thickness as a surrogate marker for atherosclerosis. Circulation 2004, 109, III33-8. [Google Scholar] [CrossRef]
- KDIGO. Chapter 1: Definition and classification of CKD. Kidney Int. Suppl. 2013, 3, 19–62. [Google Scholar] [CrossRef]
- Glas, A.S.; Lijmer, J.G.; Prins, M.H.; Bonsel, G.J.; Bossuyt, P.M. The diagnostic odds ratio: A single indicator of test performance. J. Clin. Epidemiol. 2003, 56, 1129–1135. [Google Scholar] [CrossRef]
- Vihinen, M. How to evaluate performance of prediction methods? Measures and their interpretation in variation effect analysis. BMC Genom. 2012, 13, S2. [Google Scholar] [CrossRef]
- Osei, K.; Rhinesmith, S.; Gaillard, T.; Schuster, D. Is glycosylated hemoglobin A1c a surrogate for metabolic syndrome in nondiabetic, first-degree relatives of African-American patients with type 2 diabetes? J. Clin. Endocrinol. Metab. 2003, 88, 4596–4601. [Google Scholar] [CrossRef]
- Wallace, T.M.; Levy, J.C.; Matthews, D.R. Use and abuse of HOMA modeling. Diabetes Care 2004, 27, 1487–1495. [Google Scholar] [CrossRef] [PubMed]
- Brun, J.F.; Raynaud, E.; Mercier, J. Homeostasis model assessment and related simplified evaluations of insulin sensitivity from fasting insulin and glucose. Diabetes Care 2000, 23, 1037–1038. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Nathan, D.M.; Turgeon, H.; Regan, S. Relationship between glycated haemoglobin levels and mean glucose levels over time. Diabetologia 2007, 50, 2239–2244. [Google Scholar] [CrossRef] [PubMed]
- American Diabetes Association. Diagnosis and classification of diabetes mellitus. Diabetes Care 2013, 36, S67–S74. [Google Scholar] [CrossRef]
- Lee, Y.; Fluckey, J.D.; Chakraborty, S.; Muthuchamy, M. Hyperglycemia-and hyperinsulinemia-induced insulin resistance causes alterations in cellular bioenergetics and activation of inflammatory signaling in lymphatic muscle. FASEB J. 2017, 31, 2744–2759. [Google Scholar] [CrossRef]
- Mels, C.M.; Huisman, H.W.; Smith, W.; Schutte, R.; Schwedhelm, E.; Atzler, D.; Böger, R.H.; Ware, L.J.; Schutte, A.E. The relationship of nitric oxide synthesis capacity, oxidative stress, and albumin-to-creatinine ratio in black and white men: The SABPA study. Age 2016, 38, 9. [Google Scholar] [CrossRef]
- Sung, K.C.; Rhee, E.J. Glycated haemoglobin as a predictor for metabolic syndrome in non-diabetic Korean adults. Diabet. Med. 2007, 24, 848–854. [Google Scholar] [CrossRef]
- Park, S.H.; Yoon, J.S.; Won, K.C.; Lee, H.W. Usefulness of glycated hemoglobin as diagnostic criteria for metabolic syndrome. J. Korean Med. Sci. 2012, 27, 1057–1061. [Google Scholar] [CrossRef]
- Lyons, T.J.; Basu, A. Biomarkers in diabetes: Hemoglobin A1c, vascular and tissue markers. Transl. Res. 2012, 159, 303–312. [Google Scholar] [CrossRef]
- Osawa, S.; Katakami, N.; Kuroda, A.; Takahara, M.; Sakamoto, F.; Kawamori, D.; Matsuoka, T.; Matsuhisa, M.; Shimomura, I. Skin autofluorescence is associated with early-stage atherosclerosis in patients with type 1 diabetes. J. Atheroscler. Thromb. 2017, 24, 312–326. [Google Scholar] [CrossRef]
- Cavero-Redondo, I.; Soriano-Cano, A.; Álvarez-Bueno, C.; Cunha, P.G.; Martinez-Hortelano, J.A.; Garrido-Miguel, M.; Berlanga-Macias, C.; Martinez-Vizcaino, V. Skin Autofluorescence–Indicated Advanced Glycation End Products as Predictors of Cardiovascular and All-Cause Mortality in High-Risk Subjects: A Systematic Review and Meta-analysis. J. Am. Heart Assoc. 2018, 7, e009833. [Google Scholar] [CrossRef] [PubMed]
- Graham, I.D.; Logan, J.; Harrison, M.B.; Straus, S.E.; Tetroe, J.; Caswell, W.; Robinson, N. Lost in knowledge translation: Time for a map? J. Contin. Edu. Health Prof. 2006, 26, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Lang, E.S.; Wyer, P.C.; Haynes, R.B. Knowledge translation: Closing the evidence-to-practice gap. Ann. Emerg. Med. 2007, 49, 355–363. [Google Scholar] [CrossRef] [PubMed]
- UCLM, C. (2019). CalcuMovi: Para calcular su riesgo de síndrome metabólico. Movidavida.org. Available online: http://www.movidavida.org/calcumovi (accessed on 27 November 2019).
- Leyva, F.; Godsland, I.F.; Worthington, M.; Walton, C.; Stevenson, J.C. Factors of the metabolic syndrome: Baseline interrelationships in the first follow-up cohort of the HDDRISC Study (HDDRISC-1). Heart disease and diabetes risk indicators in a screened cohort. Arterioscler. Thromb. Vasc. Biol. 1998, 18, 208–214. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sakkinen, P.A.; Wahl, P.; Cushman, M.; Lewis, M.R.; Tracy, R.P. Clustering of procoagulation, inflammation, and fibrinolysis variables with metabolic factors in insulin resistance syndrome. Am. J. Epidemiol. 2000, 152, 897–907. [Google Scholar] [CrossRef] [PubMed]
| Variables | Total n = 1243 | Men n = 504 | Women n = 739 |
|---|---|---|---|
| MetS prevalence (%) 1 | 611 (49.2) | 262 (52.0) | 349 (47.2) |
| Age (years) | 55.94 ± 13.69 | 56.48 ± 13.65 | 54.11 ± 13.65 |
| Waist circumference (cm) | 93.40 ± 12.60 | 98.68 ± 11.22 | 89.82 ± 12.24 |
| SBP (mmHg) | 125.06 ± 16.94 | 129.70 ± 14.83 | 122.31 ± 17.78 |
| DBP (mmHg) | 77.43 ± 10.43 | 79.04 ± 10.45 | 75.65 ± 11.03 |
| MAP (mmHg) | 51.43 ± 13.97 | 55.80 ± 13.33 | 48.46 ± 13.63 |
| HbA1c (%) (n = 1231) | 5.65 ± 0.75 | 5.73 ± 0.80 | 5.60 ± 0.71 |
| FPG (mg/dL) | 93.81 ± 23.10 | 97.32 ± 25.81 | 91.44 ± 20.76 |
| Fasting insulin (µIU/mL) (n = 1153) | 8.09 ± 6.09 | 8.39 ± 6.16 | 7.91 ± 6.03 |
| HOMA-IR (n = 1150) | 1.93 ± 1.70 | 2.07 ± 1.76 | 1.84 ± 1.66 |
| cHDL (mg/dL) | 58.83 ± 15.58 | 51.94 ± 12.47 | 63.51 ± 15.74 |
| Tryglicerides (mg/dL) | 123.54 ± 118.47 | 143.36 ± 143.36 | 110.08 ± 95.89 |
| PWv (m/s) (n = 243) | 7.65 ± 2.01 | 8.24 ± 2.29 | 7.23 ± 1.68 |
| IMT (mm) (n =247) | 0.68 ± 0.11 | 0.71 ± 1.20 | 0.66 ± 0.09 |
| ACR (mg/g) (n = 1041) | 10.99 ± 36.43 | 10.44 ± 28.96 | 11.38 ± 40.85 |
| Without MetS Mean ± SD | MetS Mean ± SD | Mean difference | p | |
|---|---|---|---|---|
| IDF criteria | ||||
| PWv (m/s) | 7.10 ± 1.67 (n = 133) | 8.33 ± 2.20 (n = 109) | −1.23 | <0.001 |
| IMT (mm) | 0.67 ± 0.11 (n = 134) | 0.70 ± 0.10 (n = 112) | −0.03 | 0.053 |
| ACR (mg/g) | 9.37 ± 34.10 (n = 520) | 12.69 ± 38.76 (n = 516) | −3.32 | 0.143 |
| IDF with HbA1c criteria | ||||
| PWv (m/s) | 7.17 ± 1.68 (n = 178) | 8.96 ± 2.01 (n = 65) | −1.80 | <0.001 |
| IMT (mm) | 0.67 ± 0.11 (n = 180) | 0.71 ± 0.10 (n = 67) | −0.03 | 0.028 |
| ACR (mg/g) | 9.27 ± 33.29 (n = 746) | 15.36 ± 43.12 (n = 295) | −6.09 | 0.015 |
| n | Sensitivity (%) | Specificity (%) | PLR | NLR | dOR | Accuracy a | |
|---|---|---|---|---|---|---|---|
| PWv (>10 m/s) | |||||||
| IDF criteria | 242 | 79.3 (52.7–100.0) | 59.6 (50.1–71.0) | 1.96 (0.16–24.63) | 0.35 (0.02–5.11) | 5.66 (2.21–14.48) | 0.62 (0.14–2.76) |
| IDF with HbA1c criteria | 243 | 65.5 (41.8–100) | 78.5 (67.5–91.3) | 3.05 (0.23–40.48) | 0.44 (0.04–5.32) | 6.94 (3.02–15.95) | 0.77 (0.14–4.20) |
| IMT (>0.8mm) | |||||||
| IDF criteria | 246 | 50.0 (31.1–80.4) | 54.9 (45.8–65.8) | 1.11 (0.05–23.86) | 0.91 (0.04–18.02) | 1.22 (0.59–2.51) | 0.54 (0.13–2.20) |
| IDF with HbA1c criteria | 247 | 32.4 (17.9–58.4) | 73.7 (63.0–86.2) | 1.23 (0.03–44.56) | 0.92 (0.06–15.01) | 1.34 (0.61–2.93) | 0.68 (0.13–3.37) |
| ACR (>30mg/g) | |||||||
| IDF criteria | 1036 | 56.9 (41.9–77.3) | 50.7 (46.4–55.4) | 1.16 (0.06–21.84) | 0.85 (0.04–17.70) | 1.36 (0.84–2.21) | 0.51 (0.13–2.02) |
| IDF with HbA1c criteria | 1041 | 41.7 (29.1–59.6) | 72.7 (67.5–91.3) | 1.52 (0.06–37.55) | 0.80 (0.05–12.48) | 1.90 (1.16–3.10) | 0.71 (0.14–3.62) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cavero-Redondo, I.; Martínez-Vizcaíno, V.; Álvarez-Bueno, C.; Agudo-Conde, C.; Lugones-Sánchez, C.; García-Ortiz, L. Metabolic Syndrome Including Glycated Hemoglobin A1c in Adults: Is It Time to Change? J. Clin. Med. 2019, 8, 2090. https://doi.org/10.3390/jcm8122090
Cavero-Redondo I, Martínez-Vizcaíno V, Álvarez-Bueno C, Agudo-Conde C, Lugones-Sánchez C, García-Ortiz L. Metabolic Syndrome Including Glycated Hemoglobin A1c in Adults: Is It Time to Change? Journal of Clinical Medicine. 2019; 8(12):2090. https://doi.org/10.3390/jcm8122090
Chicago/Turabian StyleCavero-Redondo, Iván, Vicente Martínez-Vizcaíno, Celia Álvarez-Bueno, Cristina Agudo-Conde, Cristina Lugones-Sánchez, and Luis García-Ortiz. 2019. "Metabolic Syndrome Including Glycated Hemoglobin A1c in Adults: Is It Time to Change?" Journal of Clinical Medicine 8, no. 12: 2090. https://doi.org/10.3390/jcm8122090
APA StyleCavero-Redondo, I., Martínez-Vizcaíno, V., Álvarez-Bueno, C., Agudo-Conde, C., Lugones-Sánchez, C., & García-Ortiz, L. (2019). Metabolic Syndrome Including Glycated Hemoglobin A1c in Adults: Is It Time to Change? Journal of Clinical Medicine, 8(12), 2090. https://doi.org/10.3390/jcm8122090







