The Effects of Helmet Therapy Relative to the Size of the Anterior Fontanelle in Nonsynostotic Plagiocephaly: A Retrospective Study
Abstract
:1. Introduction
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kane, A.A.; Mitchell, L.E.; Craven, K.P.; Marsh, J.L. Observations on a recent increase in plagiocephaly without synostosis. Pediatrics 1996, 97, 877–885. [Google Scholar] [PubMed]
- Peitsch, W.K.; Keefer, C.H.; LaBrie, R.A.; Mulliken, J.B. Incidence of cranial asymmetry in healthy newborns. Pediatrics 2002, 110, e72. [Google Scholar] [CrossRef] [PubMed]
- Bruneteau, R.J.; Mulliken, J.B. Frontal plagiocephaly: Synostotic, compensational, or deformational. Plast. Reconstr. Surg. 1992, 889, 21–31. [Google Scholar] [CrossRef]
- Littlefield, T.R.; Kelly, K.M.; Pomatto, J.K.; Beals, S.P. Multiple-birth infants at higher risk for development of deformational plagiocephaly. Pediatrics 1999, 103, 565–569. [Google Scholar] [CrossRef]
- Clarren, S.K. Plagiocephaly and torticollis: Etiology, natural history, and helmet treatment. J. Pediatr. 1981, 98, 92–95. [Google Scholar] [CrossRef]
- Stellwagen, L.; Hubbard, E.; Chambers, C.; Jones, K.L. Torticollis, facial asymmetry and plagiocephaly in normal newborns. Arch. Dis. Child. 2008, 93, 827–831. [Google Scholar] [CrossRef]
- Clarren, S.K.; Smith, D.W.; Hanson, J.W. Helmet treatment for plagiocephaly and congenital muscular torticollis. J. Pediatr. 1979, 94, 43–46. [Google Scholar] [CrossRef]
- Morrison, C.S.; Chariker, M. Positional plagiocephaly: Pathogenesis, diagnosis, and management. J. Ky. Med. Assoc. 2006, 104, 136–140. [Google Scholar]
- American Academy of Pediatrics. AAP task force on infant positioning and SIDS: Positioning and SIDS. Pediatrics 1992, 89, 1120–1126. [Google Scholar]
- Pedroso, F.S.; Rotta, N.; Quintal, A.; Giordani, G. Evolution of anterior fontanel size in normal infants in the first year of life. J. Child. Neurol. 2008, 23, 1419–1423. [Google Scholar] [CrossRef]
- Seruya, M.; Oh, A.K.; Taylor, J.H. Helmet treatment of deformational plagiocephaly: The relationship between age at initiation and rate of correction. Plastic. Reconstr. Surg. 2013, 131, 55e–61e. [Google Scholar] [CrossRef] [PubMed]
- Captier, G.; Leboucq, N.; Bigorre, M.; Canovas, F.; Bonnel, F.; Bonnafe, A.; Montoya, P. Plagiocephaly: Morphometry of skull base asymmetry. Surg. Radiol. Anat. 2003, 25, 226–233. [Google Scholar] [CrossRef] [PubMed]
- Kelly, K.M.; Littlefield, T.R.; Pomatto, J.K.; Ripley, C.E.; Beals, S.P.; Joganic, E.F. Importance of early recognition and treatment of deformational plagiocephaly with orthotic cranioplasty. Cleft Palate Craniofac. J. 1999, 36, 127–130. [Google Scholar] [CrossRef] [PubMed]
- St John, D.; Mulliken, J.B.; Kaban, L.B.; Padwa, B.L. Anthropometric analysis of mandibular asymmetry in infants with deformational posterior plagiocephaly. J. Oral Maxillofac. Surg. 2002, 60, 873–877. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.Y.; Chung, Y.K.; Kim, Y.O. Effectiveness of helmet cranial remodeling in older infants with positional plagiocephaly. Arch. Craniofac. Surg. 2014, 15, 47–52. [Google Scholar] [CrossRef] [PubMed]
- Moon, I.Y.; Lim, S.Y.; Oh, K.S. Analysis of facial asymmetry in deformational plagiocephaly using three dimensional computed tomographic review. Arch. Craniofac. Surg. 2014, 15, 109–116. [Google Scholar] [CrossRef] [PubMed]
- Cho, I.K.; Eom, J.R.; Lee, J.W.; Yang, J.D.; Chung, H.Y.; Cho, B.C.; Choi, K.Y. A clinical photographic method to evaluate the need for helmet therapy in deformational plagiocephaly. J. Craniofac. Surg. 2018, 29, 149–152. [Google Scholar] [CrossRef]
- Yoo, H.-S.; Rah, D.K.; Kim, Y.O. Outcome analysis of cranial molding therapy in nonsynostotic plagiocephaly. Arch Plast. Surg. 2012, 39, 338–344. [Google Scholar] [CrossRef]
- Silva, B.P.; Jimenez-Castellanos, E.; Martinez-de-Fuentes, R.; Fernandez, A.A.V.; Chu, S. Perception of maxillary dental midline shift in asymmetric faces. Int. J. Esthet. Dent. 2015, 10, 588–596. [Google Scholar] [PubMed]
- Meyer-Marcotty, P.; Bohm, H.; Linz, C.; Kunz, F.; Keil, N.; Stellzig-Eisenhauer, A.; Schweitzer, T. Head orthesis therapy in infants with unilateral positional plagiocephaly: An interdisciplinary approach to broadening the range of orthodontic treatment. J. Orofac. Orthop. 2012, 73, 151–165. [Google Scholar] [CrossRef]
- Meyer-Marcotty, P.; Stellzig-Eisenhauer, A. Interdisziplin are kieferorthopadisch asthetische therapie. Der Freie Zahnarzt 2013, 57, 78–85. [Google Scholar] [CrossRef]
- Xia, J.J.; Kennedy, K.A.; Teichgraeber, J.F.; Wu, K.Q.; Baumgartner, J.B.; Gateno, J. Nonsurgical treatment of deformational plagiocephaly: A systematic review. Arch. Pediatr. Adolesc. Med. 2008, 162, 719–727. [Google Scholar] [CrossRef] [PubMed]
- Kluba, S.; Kraut, W.; Reinert, S.; Krimmel, M. What is the optimal time to start helmet therapy in positional plagiocephaly. Plast. Reconstr. Surg. 2011, 128, 492–498. [Google Scholar] [CrossRef] [PubMed]
- Aihara, Y.; Komatsu, K.; Dairoku, H.; Kubo, O.; Hori, T.; Okada, Y. Cranial molding helmet therapy and establishment of practical criteria for management in Asian infant positional head deformity. Childs Nerv. Syst. 2014, 30, 1499–1509. [Google Scholar] [CrossRef] [PubMed]
- Kreutz, M.; Fitze, B.; Blecher, C.; Marcello, A.; Simon, R.; Cremer, R.; Zeilhofer, H.F.; Kunz, C.; Mayr, J. Facial asymmetry correction with moulded helmet therapy in infants with deformational skull base plagiocephaly. J. Craniomaxillofac. Surg. 2018, 46, 28–34. [Google Scholar] [CrossRef] [PubMed]
- Wendling-Keim, D.S.; Mace, Y.; Lochbihler, H.; Dietz, H.G.; Lehner, M. A new parameter for the management of positional plagiocephaly: The size of the anterior fontanelle matters. Childs Nerv. Syst. 2019, 35, 1–9. [Google Scholar] [CrossRef]
- Hutchison, B.L.; Stewart, A.W.; De Chalaain, T.B.; Mitchell, E.A. A randomized controlled trial of positioning treatments in infants with positional head shape deformities. Acta Paediatr. 2010, 99, 1556–1560. [Google Scholar] [CrossRef]
- van Wijk, R.M.; van Vlimmeren, L.A.; Groothuis-Oudshoorn, C.G.; Van der Ploeg, C.P.; Ijzerman, M.J.; Boere-Boonekamp, M.M. Helmet therapy in infants with positional skull deformation: Randomized controlled trial. Br. Med. J. 2014, 348, 2741. [Google Scholar] [CrossRef] [Green Version]
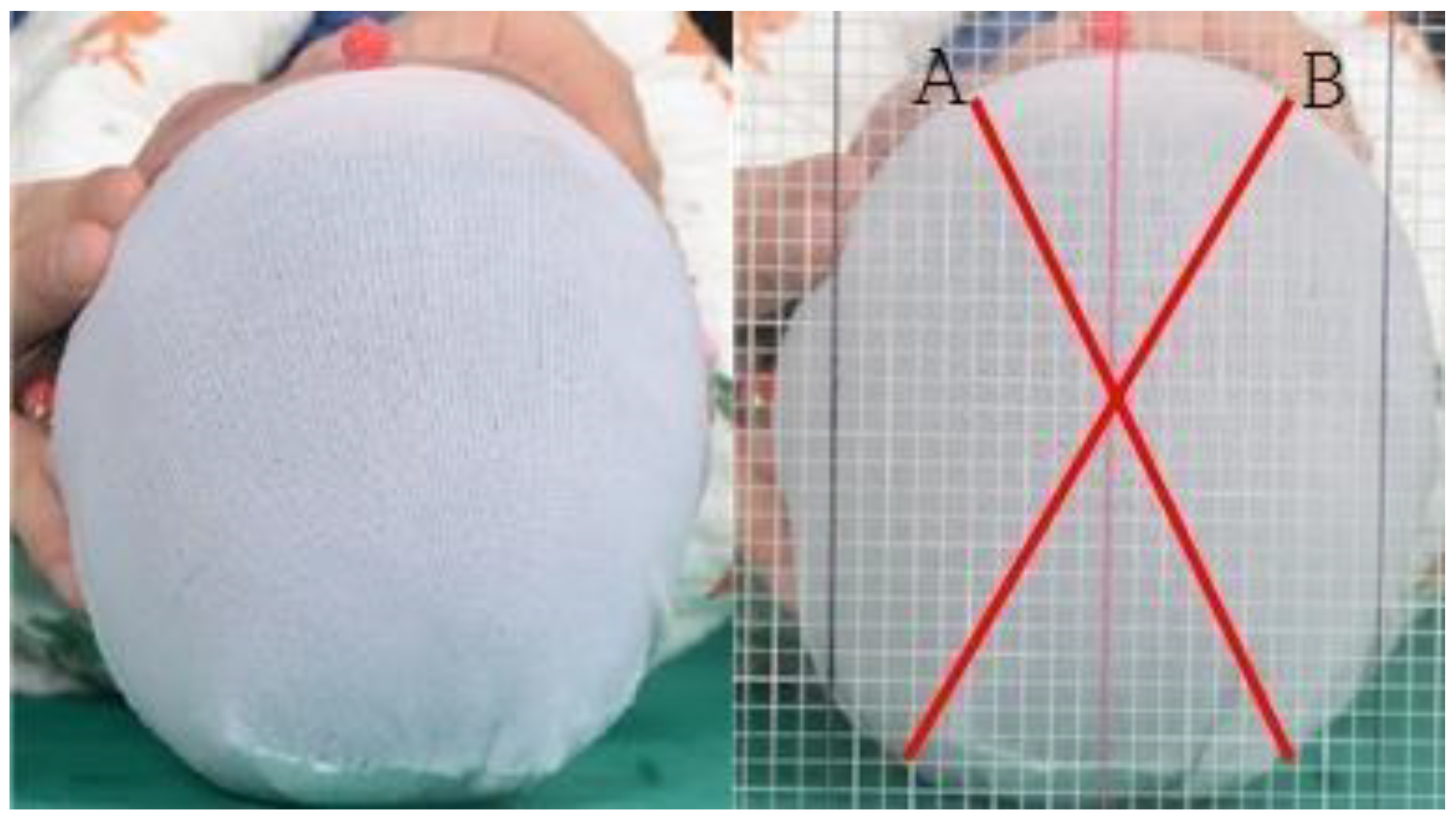

| 12 wk–23 wk (n = 152) | 24 wk– (n = 48) | |||||
|---|---|---|---|---|---|---|
| Pre | Post | Duration | Pre | Post | Duration | |
| CVA (mm) | 11.4 | 7.36 | 12.5 wk | 11.11 | 7.6 | 16.3 wk * (p < 0.05) |
| CVAI (%) | 8.73 | 5.4 | 8.5 | 5.5 | ||
| Patient Demographics | ||
|---|---|---|
| Total | 200 | |
| M | 105 | |
| F | 95 | |
| Age | 12 wk–23 wk | 152 |
| 24 wk–27 wk | 35 | |
| 28 wk– | 13 | |
| Grouped by anterior fontanelle size | ||
| A (0–25%) | 53 | |
| B (25–75%) | 102 | |
| C (75–100%) | 45 | |
| Grouped by anterior fontanelle size (20wk–23wk infants) | ||
| I (0–25%) | 21 | |
| II (25–75%) | 41 | |
| III (75–100%) | 20 | |
| Grouped by treatment initiation | ||
| 12 wk–23 wk | 152 | |
| 24 wk– | 48 | |
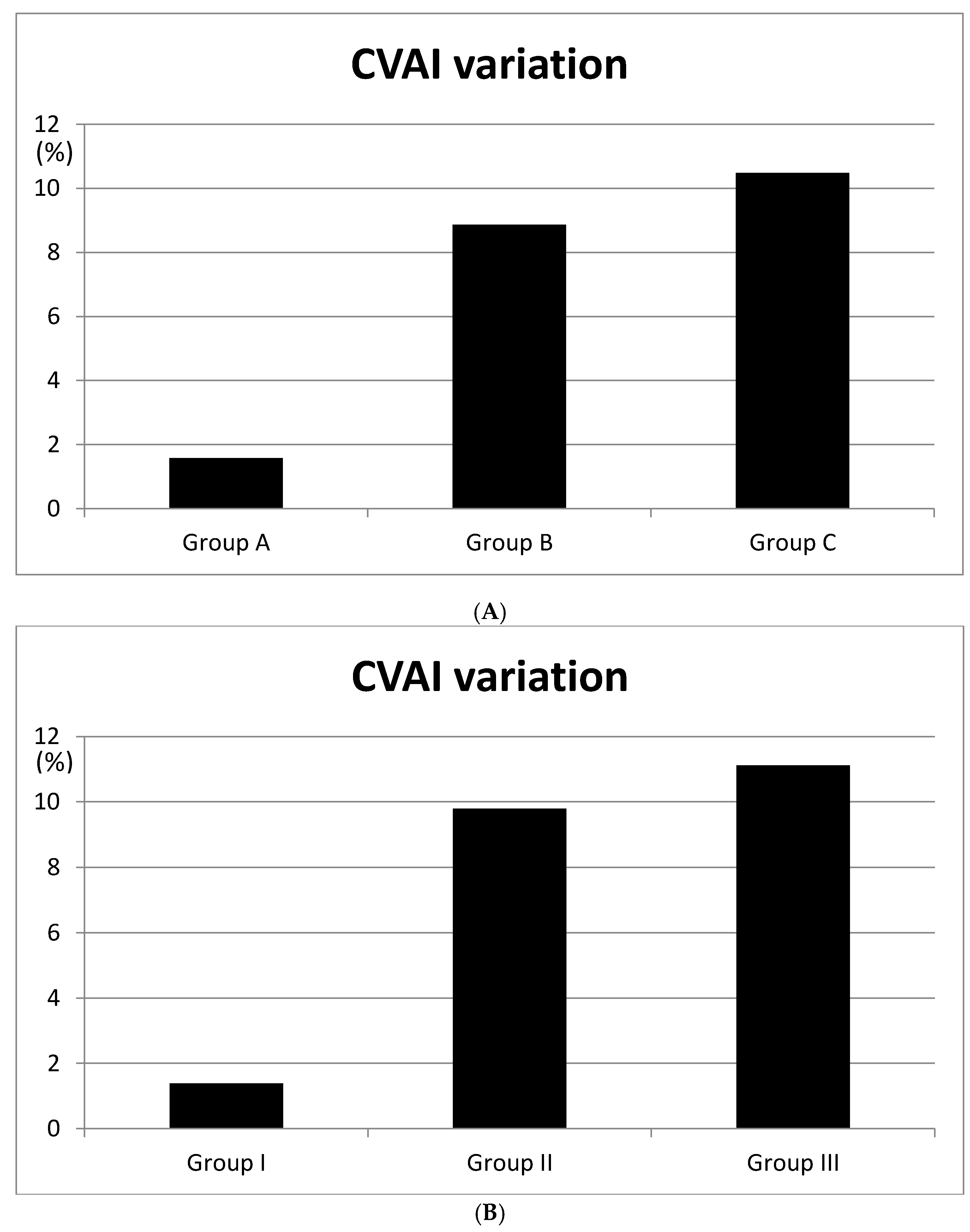
| All | Group A | Group B | Group C | |||||
|---|---|---|---|---|---|---|---|---|
| Pre | Post | Pre | Post | Pre | Post | Pre | Post | |
| CVA (mm) | 11.4 | 5.36 | 7.5 | 6.23 | 13.21 | 5.14 | 15.4 | 4.63 |
| CVAI (%) | 12.73 | 4.91 | 6.5 | 4.92 | 13.81 | 4.95 | 15.31 | 4.83 |
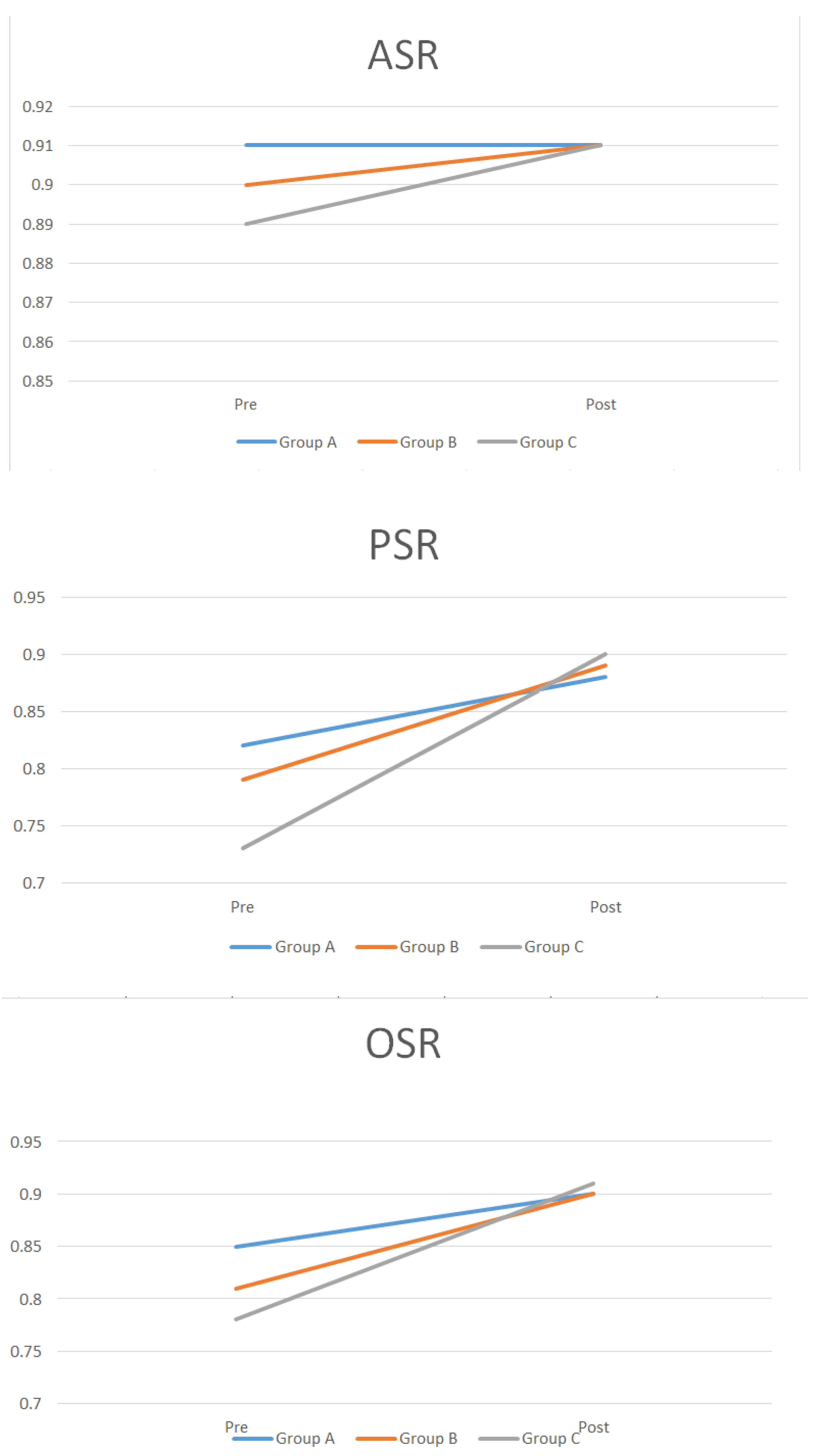
| ASR | 0.9 | 0.91 | 0.91 | 0.91 | 0. 9 | 0.91 | 0.89 | 0.91 |
| PSR | 0.78 | 0.88 | 0.82 | 0.88 | 0.79 | 0.89 | 0.73 | 0.9 |
| OSR | 0.83 | 0.92 | 0.85 | 0.9 | 0.81 | 0.9 | 0.78 | 0.91 |
| All | Group I | Group II | Group III | |||||
|---|---|---|---|---|---|---|---|---|
| Pre | Post | Pre | Post | Pre | Post | Pre | Post | |
| CVA (mm) | 11.4 | 5.36 | 6.5 | 6.21 | 14.15 | 5.66 | 16.49 | 6.1 |
| CVAI (%) | 12.73 | 4.91 | 6.3 | 4.92 | 13.81 | 4.02 | 15.81 | 4.69 |
| ASR | 0.9 | 0.91 | 0.91 | 0.91 | 0.9 | 0.91 | 0.9 | 0.91 |
| PSR | 0.78 | 0.88 | 0.84 | 0.86 | 0.82 | 0.88 | 0.78 | 0.91 |
| OSR | 0.83 | 0.92 | 0.88 | 0.91 | 0.86 | 0.92 | 0.86 | 0.93 |
| Group X | Group Y |
|---|---|
| b = 2.073 + 0.12 × a (p = 0.003) * | b = 4.184 + 0.09 × a (p = 0.006) * |
| * a = anterior fontanelle size (mm), b = CVAI difference (%) | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, D.G.; Lee, J.S.; Lee, J.W.; Yang, J.D.; Chung, H.Y.; Cho, B.C.; Choi, K.Y. The Effects of Helmet Therapy Relative to the Size of the Anterior Fontanelle in Nonsynostotic Plagiocephaly: A Retrospective Study. J. Clin. Med. 2019, 8, 1977. https://doi.org/10.3390/jcm8111977
Kim DG, Lee JS, Lee JW, Yang JD, Chung HY, Cho BC, Choi KY. The Effects of Helmet Therapy Relative to the Size of the Anterior Fontanelle in Nonsynostotic Plagiocephaly: A Retrospective Study. Journal of Clinical Medicine. 2019; 8(11):1977. https://doi.org/10.3390/jcm8111977
Chicago/Turabian StyleKim, Do Gon, Joon Seok Lee, Jeong Woo Lee, Jung Dug Yang, Ho Yun Chung, Byung Chae Cho, and Kang Young Choi. 2019. "The Effects of Helmet Therapy Relative to the Size of the Anterior Fontanelle in Nonsynostotic Plagiocephaly: A Retrospective Study" Journal of Clinical Medicine 8, no. 11: 1977. https://doi.org/10.3390/jcm8111977
APA StyleKim, D. G., Lee, J. S., Lee, J. W., Yang, J. D., Chung, H. Y., Cho, B. C., & Choi, K. Y. (2019). The Effects of Helmet Therapy Relative to the Size of the Anterior Fontanelle in Nonsynostotic Plagiocephaly: A Retrospective Study. Journal of Clinical Medicine, 8(11), 1977. https://doi.org/10.3390/jcm8111977





