Clinical Factors Associated with a Shorter or Longer Course of Antibiotic Treatment in Patients with Exacerbations of Bronchiectasis: A Prospective Cohort Study
Abstract
1. Background
2. Materials and Methods
2.1. Study Design
2.2. Patients
2.3. Definitions
2.4. Data Collection and Evaluation
2.5. Follow-Up
2.6. Statistical Analysis
3. Results
3.1. Participants
3.2. Comparison of Patients by Treatment Duration Group
3.3. Outcomes
3.4. Factors Associated with Longer Courses of Antibiotic Treatment
3.5. Supplementary Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AUC | Area under the curve |
| BSI | Bronchiectasis Severity Index |
| CF | Cystic fibrosis |
| CI | Confidence interval |
| COPD | Chronic obstructive pulmonary disease |
| CT | Computed tomography |
| LC | Long-course (15–21 days) |
| LTOT | Long-term oxygen therapy |
| OR | Odds ratio |
| SC | Short-course (≤14 days) |
References
- Watt, A.P.; Brown, V.; Courtney, J.; Kelly, M.; Garske, L.; Elborn, J.S.; Ennis, M. Neutrophil apoptosis, proinflammatory mediators and cell counts in bronchiectasis. Thorax 2004, 59, 231–236. [Google Scholar] [CrossRef] [PubMed]
- Bell, S.C.; Elborn, J.S.; Byrnes, C.A. Bronchiectasis: Treatment decisions for pulmonary exacerbations and their prevention. Respirology 2018, 23, 1006–1022. [Google Scholar] [CrossRef] [PubMed]
- Chalmers, J.D.; Goeminne, P.; Aliberti, S.; McDonnell, M.J.; Lonni, S.; Davidson, J.; Poppelwell, L.; Salih, W.; Pesci, A.; Dupont, L.J.; et al. The bronchiectasis severity index. An international derivation and validation study. Am. J. Respir. Crit. Care Med. 2014, 189, 576–585. [Google Scholar] [CrossRef] [PubMed]
- Hill, A.T.; Haworth, C.S.; Aliberti, S.; Barker, A.; Blasi, F.; Boersma, W.; Chalmers, J.D.; De Soyza, A.; Dimakou, K.; Elborn, J.S.; et al. Pulmonary exacerbation in adults with bronchiectasis: A consensus definition for clinical research. Eur. Respir. J. 2017, 49, 1700051. [Google Scholar] [CrossRef] [PubMed]
- Martínez-García, M.Á.; Máiz, L.; Olveira, C.; Girón, R.M.; de la Rosa, D.; Blanco, M.; Cantón, R.; Vendrell, M.; Polverino, E.; de Gracia, J.; et al. Normativa sobre la valoración y eldiagnóstico de lasbronquiectasias en el adulto. Arch. Bronconeumol. 2018, 54, 79–87. [Google Scholar] [PubMed]
- Polverino, E.; Goeminne, P.C.; McDonnell, M.J.; Aliberti, S.; Marshall, S.E.; Loebinger, M.R.; Murris, M.; Cantón, R.; Torres, A.; Dimakou, K.; et al. European Respiratory Society guidelines for the management of adult bronchiectasis. Eur. Respir. J. 2017, 50, 1700629. [Google Scholar] [CrossRef] [PubMed]
- Bilton, D.; Henig, N.; Morrissey, B.; Gotfried, M. Addition of Inhaled Tobramycin to Ciprofloxacin for Acute Exacerbations of Pseudomonas aeruginosa Infection in Adult Bronchiectasis. Chest 2006, 130, 1503–1510. [Google Scholar] [CrossRef] [PubMed]
- Martínez-García, M.Á.; Máiz, L.; Olveira, C.; Girón, R.M.; de la Rosa, D.; Blanco, M.; Cantón, R.; Vendrell, M.; Polverino, E.; de Gracia, J.; et al. Spanish Guidelines on the Evaluation and Diagnosis/Treatment of Bronchiectasis in Adults. Arch. Bronconeumol. 2018, 54, 79–98. [Google Scholar] [CrossRef] [PubMed]
- Cantón, R.; Máiz, L.; Escribano, A.; Olveira, C.; Oliver, A.; Asensio, O.; Gartner, S.; Roma, E.; Quintana-Gallego, E.; Salcedo, A.; et al. Spanish Consensus on the Prevention and Treatment of Pseudomonas aeruginosa Bronchial Infections in Cystic Fibrosis Patients. Arch. Bronconeumol. 2015, 51, 140–150. [Google Scholar] [CrossRef] [PubMed]
- Wilson, R.; Anzueto, A.; Miravitlles, M.; Arvis, P.; Faragó, G.; Haverstock, D.; Trajanovic, M.; Sethi, S. A novel study design for antibiotic trials in acute exacerbations of COPD: MAESTRAL methodology. Int. J. Chron. Obstruct. Pulmon. Dis. 2011, 6, 373–383. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Martínez-García, M.Á.; de Gracia, J.; Vendrell Relat, M.; Girón, R.M.; Máiz Carro, L.; de la Rosa Carrillo, D.; Olveira, C. Multidimensional approach to non-cystic fibrosis bronchiectasis: The FACED score. Eur. Respir. J. 2014, 43, 1357–1367. [Google Scholar] [CrossRef] [PubMed]
- Hosmer, D.W.; Lemeshow, S. Applied Logistic Regression; John Wiley & Sons: New York, NY, USA, 2000. [Google Scholar]
- Sterne, J.A.; White, I.R.; Carlin, J.B.; Spratt, M.; Royston, P.; Kenward, M.G.; Wood, A.M.; Carpenter, J.R. Multiple imputation for missing data in epidemiological and clinical research: Potential and pitfalls. Br. Med. J. 2009, 338, b2393. [Google Scholar] [CrossRef] [PubMed]
- Efron, B.; Tibshirani, R. An Introduction to the Bootstrap; Chapman & Hall: London, UK, 1994. [Google Scholar]
- Chalmers, J.D.; Aliberti, S.; Filonenko, A.; Shteinberg, M.; Goeminne, P.C.; Hill, A.T.; Fardon, T.C.; Obradovic, D.; Gerlinger, C.; Sotgiu, G.; et al. Characterization of the “Frequent Exacerbator Phenotype” in Bronchiectasis. Am. J. Respir. Crit. Care Med. 2018, 197, 1410–1420. [Google Scholar] [CrossRef] [PubMed]
- Bhatt, J.M. Treatment of pulmonary exacerbations in cystic fibrosis. Eur. Respir. Rev. 2013, 22, 205–216. [Google Scholar] [CrossRef] [PubMed]
- Murray, M.P.; Turnbull, K.; MacQuarrie, S.; Hill, A.T. Assessing response to treatment of exacerbations of bronchiectasis in adults. Eur. Respir. J. 2008, 33, 312–318. [Google Scholar] [CrossRef] [PubMed]
- Chalmers, J.D.; Smith, M.P.; McHugh, B.J.; Doherty, C.; Govan, J.R.; Hill, A.T. Short- and long-term antibiotic treatment reduces airway and systemic inflammation in non-cystic fibrosis bronchiectasis. Am. J. Respir. Crit. Care Med. 2012, 186, 657–665. [Google Scholar] [CrossRef] [PubMed]
- Collaco, J.M.; Green, D.M.; Cutting, G.R.; Naughton, K.M.; Mogayzel, P.J. Location and Duration of Treatment of Cystic Fibrosis Respiratory Exacerbations Do Not Affect Outcomes. Am. J. Respir. Crit. Care Med. 2010, 182, 1137–1143. [Google Scholar] [CrossRef] [PubMed]
- Hill, A.T.; Sullivan, A.L.; Chalmers, J.D.; De Soyza, A.; Elborn, S.J.; Floto, A.R.; Grillo, L.; Gruffydd-Jones, K.; Harvey, A.; Haworth, C.S.; et al. British Thoracic Society Guideline for bronchiectasis in adults. Thorax 2019, 74 (Suppl. 1), 1–69. [Google Scholar] [CrossRef] [PubMed]
- Rosales-Mayor, E.; Polverino, E.; Raguer, L.; Alcaraz, V.; Gabarrus, A.; Ranzani, O.; Menendez, R.; Torres, A. Comparison of two prognostic scores (BSI and FACED) in a Spanish cohort of adult patients with bronchiectasis and improvement of the FACED predictive capacity for exacerbations. PLoS ONE 2017, 12, e0175171. [Google Scholar] [CrossRef] [PubMed]
- Crisafulli, E.; Ielpo, A.; Barbeta, E.; Ceccato, A.; Huerta, A.; Gabarrús, A.; Soler, N.; Chetta, A.; Torres, A. Clinical variables predicting the risk of a hospital stay for longer than 7 days in patients with severe acute exacerbations of chronic obstructive pulmonary disease: A prospective study. Respir. Res. 2018, 19, 261. [Google Scholar] [CrossRef] [PubMed]
- Sundh, J.; Ahmadi, Z.; Ekström, M. Daily duration of long-term oxygen therapy and risk of hospitalization in oxygen-dependent COPD patients. Int. J. Chron. Obstruct. Pulmon. Dis. 2018, 13, 2623–2628. [Google Scholar] [CrossRef] [PubMed]
- Martínez-García, M.A.; Soler-Cataluña, J.J.; Perpiñá-Tordera, M.; Román-Sánchez, P.; Soriano, J. Factors associated with lung function decline in adult patients with stable non-cystic fibrosis bronchiectasis. Chest 2007, 132, 1565–1572. [Google Scholar] [CrossRef] [PubMed]
- Wilson, C.B.; Jones, P.W.; O’Leary, C.J.; Hansell, D.M.; Cole, P.J.; Wilson, R. Effect of sputum bacteriology on the quality of life of patients with bronchiectasis. Eur. Respir. J. 1997, 10, 1754–1760. [Google Scholar] [CrossRef] [PubMed]

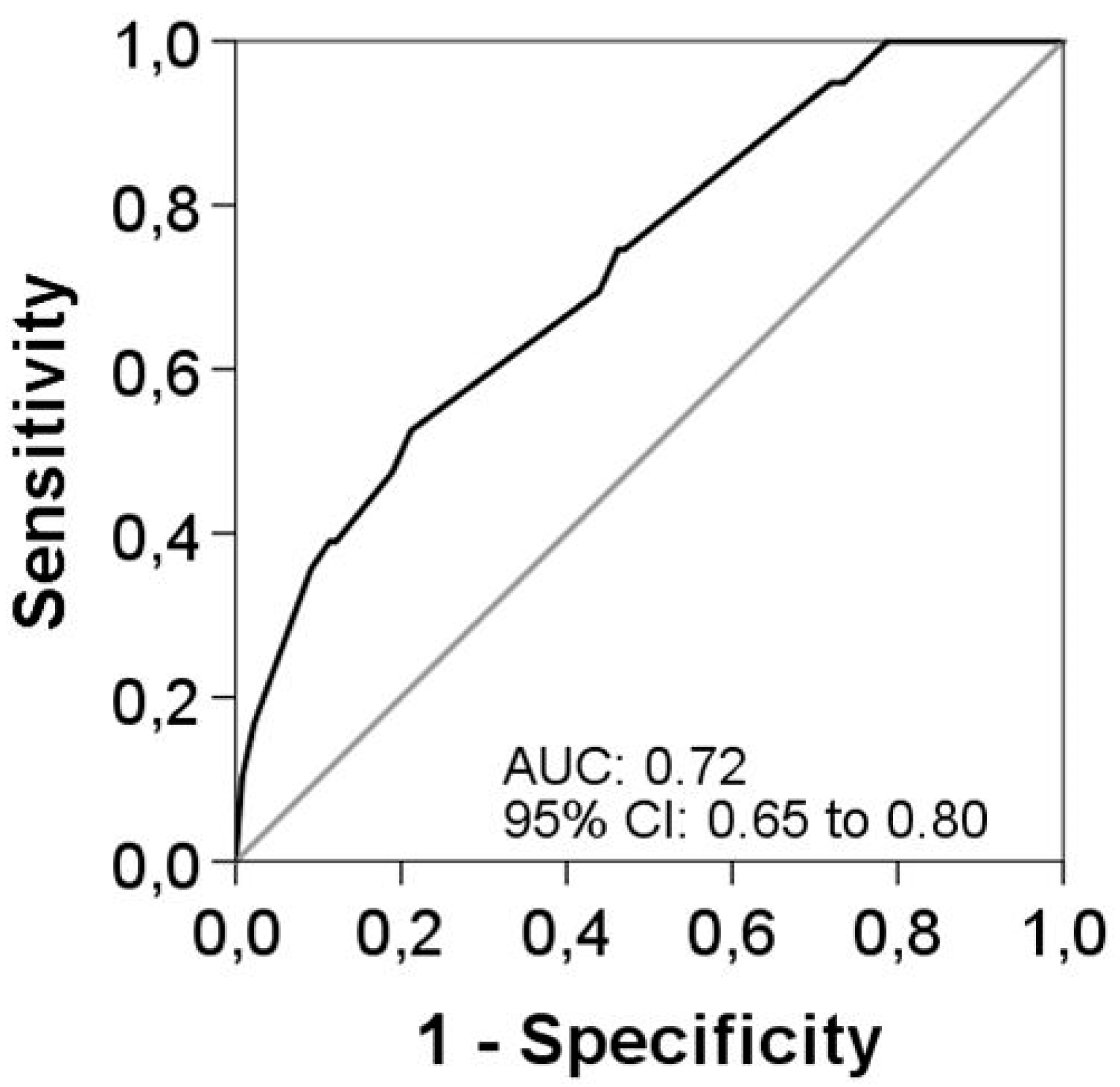
| All Patients | SC ≤ 14 Days | LC 15–21 Days | p-Value a | |
|---|---|---|---|---|
| Patients | n = 191 | n = 132 | n = 59 | |
| Clinical | ||||
| Female sex, n (%) | 108 (56.5) | 76 (57.6) | 32 (54.2) | 0.667 |
| Age, median (Q1; Q3), years | 72 (63; 79) | 72 (62.5; 79) | 73 (64; 78) | 0.881 |
| BMI, median (Q1; Q3), Kg/m2 | 25.3 (22.2; 28.9) | 25.5 (22.3; 29.4) | 25 (21.9; 28.9) | 0.576 |
| Smoking habit, n (%) | 0.738 | |||
| Actual | 7 (3.7) | 5 (3.8) | 2 (3.4) | |
| Former | 83 (43.5) | 55 (41.7) | 28 (47.5) | |
| Influenza vaccination, n (%) | 130 (68.4) | 94 (71.8) | 36 (61) | 0.141 |
| Pneumococcal vaccination, n (%) | 93 (57.4) | 67 (59.8) | 26 (52) | 0.352 |
| Etiology, n (%) | 0.535 | |||
| Post-infectious | 65 (34) | 49 (37.1) | 16 (27.1) | |
| Idiopathic | 61 (32) | 38 (28.8) | 23 (39) | |
| Asthma | 13 (6.8) | 8 (6.1) | 5 (8.5) | |
| COPD | 36 (18.8) | 25 (18.9) | 11 (18.6) | |
| Others | 16 (8.4) | 12 (0.08) | 4 (6.8) | |
| Comorbidities, n (%) b | ||||
| Arterial hypertension | 93 (48.7) | 59 (44.7) | 34 (57.6) | 0.099 |
| Arrhythmia | 20 (17.2) | 17 (22.7) | 3 (7.3) | 0.036 |
| Dyslipidemia | 43 (37.1) | 31 (41.3) | 12 (29.3) | 0.198 |
| Diabetes | 22 (11.5) | 14 (10.6) | 8 (13.6) | 0.555 |
| Therapy, n (%) c | ||||
| Mucolytics | 76 (39.8) | 47 (35.6) | 29 (49.2) | 0.077 |
| Inhaled Antibiotic | 43 (22.5) | 27 (20.5) | 16 (27.1) | 0.308 |
| Bronchodilators | 263 (68.8) | 179 (67.8) | 84 (71.18) | 0.268 |
| Inhaled steroid | 154 (80.6) | 108 (81.8) | 13 (22) | 0.534 |
| Oral corticosteroids (<28 days) | 16 (8.4) | 9 (6.8) | 7 (11.9) | 0.418 |
| LTOT | 26 (13.6) | 11 (8.3) | 15 (25.4) | 0.001 |
| Pulmonary function | ||||
| FVC pre-BD, median (Q1; Q3), % | 82.5 (67; 99.3) | 81.5 (69; 103) | 84 (66; 96.3) | 0.498 |
| FEV1 pre-BD, median (Q1; Q3), % | 61 (44.8; 76.6) | 63 (45; 79) | 58 (41; 68.6) | 0.181 |
| FEV1/FVC pre-BD, median (Q1; Q3), % | 57.3 (49.8; 65) | 58 (49; 65) | 56.5 (50; 62.50) | 0.546 |
| Severity | ||||
| Lobes affected (HRCT), n (%) | 0.312 | |||
| <3 | 77 (41) | 56 (43.4) | 21 (35.6) | |
| ≥3 | 111 (59) | 73 (56.6) | 38 (64.4) | |
| Type of BE (HRCT), n (%) | <0.001 | |||
| Cylindrical | 58 (30.7) | 33 (25.4) | 25 (42.4) | 0.019 |
| Varicose | 12 (6.3) | 7 (5.4) | 5 (8.5) | 0.521 |
| Cystic | 13 (6.9) | 5 (3.8) | 8 (13.6) | 0.026 |
| Chronic colonization, n (%) | ||||
| Pseudomonas aeruginosa | 89 (46.6) | 52 (39.4) | 37 (62.7) | 0.003 |
| Others | 46 (24.3) | 31 (23.8) | 15 (25.4) | 0.815 |
| History of exacerbations due to BE (1 year), n (%) | 143 (75.3) | 94 (71.8) | 49 (83) | 0.095 |
| History of pneumonia (1 year), n (%) | 32 (16.8) | 25 (19.1) | 7 (11.9) | 0.218 |
| History of hospitalizations due to BE (1 year), n (%) | 97 (50.8) | 60 (45.5) | 37 (62.7) | 0.028 |
| BSI stage, n (%) | 0.315 | |||
| Mild 0–4 | 19 (9.9) | 16 (12.1) | 3 (5.1) | |
| Moderate 5–8 | 45 (23.6) | 31 (23.5) | 14 (23.7) | |
| Severe ≥9 | 127 (66.5) | 85 (64.4) | 42 (71.2) | |
| FACED score, n (%) | 0.029 | |||
| Mild 0–2 | 94 (49.2) | 72 (54.5) | 22 (37.3) | 0.027 |
| Moderate 3–4 | 61 (31.9) | 41 (31.1) | 20 (33.9) | 0.697 |
| Severe 5–7 | 36 (18.8) | 19 (14.4) | 17 (28.8) | 0.019 |
| All Patients | SC ≤ 14 Days | LC 15–21 Days | p-Value a | |
|---|---|---|---|---|
| Patients | n = 191 | n = 132 | n = 59 | |
| Clinical variables, n (%) | ||||
| Increased sputum volume | 156 (83.4) | 106 (82.8) | 50 (84.7) | 0.741 |
| Increased sputum purulence | 135 (72.2) | 91 (71.1) | 44 (74.6) | 0.621 |
| Increased dyspnea | 145 (77.5) | 98 (76.6) | 47 (79.7) | 0.637 |
| Fever (>38 °C) | 109 (58.3) | 79 (61.7) | 30 (50.8) | 0.161 |
| Chest pain | 45 (24.1) | 39 (30.5) | 6 (10.2) | 0.003 |
| Hemoptysis | 15 (8) | 9 (7) | 6 (10.2) | 0.563 |
| Increased cough | 159 (85) | 108 (84.4) | 51 (86.4) | 0.713 |
| Blood analysis, median (Q1; Q3) | ||||
| C-Reactive Protein, mg/dL | 5.8 (1.9; 15.5) | 5.6 (2; 16.3) | 6.5 (1.5; 13.1) | 0.988 |
| Hemoglobin, g/L | 106 (13; 129) | 113 (13.7; 133) | 114.1 (12.3; 115) | 0.232 |
| Platelets, 109/L | 240 (202; 310) | 245 (204; 303) | 236 (200.5; 325.5) | 0.674 |
| Leukocytes, 109/L | 10.7 (8.1; 14.3) | 10.6 (8.4; 13.8) | 11.4 (7.5; 14.9) | 0.810 |
| PaO2 | 64.7 (57; 73.4) | 63 (56.5; 72.7) | 67.3 (58.4; 73.4) | 0.327 |
| Severity | ||||
| Severity of exacerbation | 0.005 | |||
| Mild | 48 (25.1) | 42 (31.8) | 6 (10.2) | 0.001 |
| Moderate to severe | 143(74.9) | 90 (61.2) | 53 (89.8) | 0.001 |
| Site of treatment | 0.013 | |||
| Outpatient treatment | 52 (27.2) | 43 (32.6) | 9 (15.3) | |
| Hospital ward/Intensive Care Unit/Intermediate Care Unit | 139 (72.8) | 89 (67.4) | 50 (84.7) | |
| Duration of exacerbation, median (Q1; Q3), days | 11 (8; 15) | 10 (7; 14) | 15 (11; 20) | <0.001 |
| Duration of hospitalization, median (Q1; Q3), days | 8 (6; 11) | 8 (6; 10) | 9.5 (7;17) | 0.002 |
| Diagnosis of CAP, n (%) | 49 (25.7) | 31 (23.5) | 18 (30.5) | 0.304 |
| Complications, n (%)b | ||||
| Intubation/MV | 4 (2.1) | 2 (1.5) | 2 (3.4) | 0.589 |
| NIV | 7 (3.7) | 3 (2.3) | 4 (6.8) | 0.205 |
| Sepsis | 7 (3.7) | 3 (2.3) | 4 (6.8) | 0.205 |
| Septic shock | 4 (2.1) | 1 (0.8) | 3 (5.1) | 0.088 |
| Acute myocardial infarction | 2 (1) | 0 | 2 (3.4) | 0.094 |
| Arrhythmia | 8 (4.2) | 6 (4.5) | 2 (3.4) | >0.999 |
| Microbiology, n (%) | ||||
| Patients with defined etiology | 141 (73.8) | 90 (68.2) | 51 (86.4) | 0.008 |
| Haemophilus influenzaec | 12 (8.5) | 11 (12.2) | 1 (2) | 0.056 |
| Escherichia colic | 5 (3.5) | 4 (4.4) | 1 (2) | 0.654 |
| Klebsiella pneumoniaec | 1 (0.7) | 1 (1.1) | 0 | >0.999 |
| Moraxella catarrhalisc | 3 (2.1) | 3 (3.3) | 0 | 0.553 |
| Pseudomonas aeruginosac | 28 (19.9) | 12 (13.39) | 16 (31.4) | 0.010 |
| Staphylococcus aureusc | 2 (1.4) | 0 | 2 (3.9) | 0.129 |
| Mycoplasma pneumoniaec | 2 (1.4) | 2 (2.2) | 0 | 0.535 |
| Streptococcus pneumoniaec | 11 (7.8) | 6 (6.7) | 5 (9.8) | 0.527 |
| Virus c | 6 (4.3) | 5 (5.69) | 1 (2) | 0.418 |
| Other c | 17 (12.1) | 13 (14.4) | 4 (7.8) | 0.247 |
| Polymicrobial c | 52 (36.9) | 32 (35.69) | 20 (39.2) | 0.665 |
| Antibiotic treatment | ||||
| Number of initial antibiotics, n (%) | <0.001 | |||
| 1 | 94 (49.2) | 75 (56.8) | 19 (32.2) | 0.002 |
| 2 | 74 (38.7) | 49 (37.1) | 25 (42.4) | 0.491 |
| ≥3 | 23 (12) | 8 (6.1) | 15 (25.4) | <0.001 |
| All Patients | SC ≤14 Days | LC 15–21 Days | p-Value a | |
|---|---|---|---|---|
| Patients | n = 191 | n = 132 | n = 59 | |
| Outcomes | ||||
| Poor clinical response, n (%) | 36 (22.2) | 18 (15.9) | 18 (36.7) | 0.003 |
| History of exacerbations for 1 year, n (%) | 114 (61.3) | 80 (62.5) | 34 (58.6) | 0.615 |
| History of hospitalization for 1 year, n (%) | 49 (50.5) | 29 (48.3) | 20 (54.1) | 0.584 |
| Mortality during the exacerbation, n (%) | 3 (1.6) | 1 (0.8) | 2 (3.4) | 0.176 |
| Mortality after 30 days, n (%) | 1 (0.5) | 1 (0.8) | 0 | 0.501 |
| Mortality after 1 year, n (%) | 16 (8.4) | 12 (9.2) | 4 (6.8) | 0.779 |
| Variable | Univariate | Multivariable a,b,c | ||||
|---|---|---|---|---|---|---|
| OR | 95% CI | p-Value | OR | 95% CI | p-Value | |
| LTOT | 3.75 | 1.60–8.78 | 0.002 | 2.51 | 1.02–6.22 | 0.046 |
| FACED score | 0.033 | – | – | – | ||
| Mild 0–2 | 1.00 | – | – | – | – | – |
| Moderate 3–4 | 1.60 | 0.78–3.27 | 0.20 | – | – | – |
| Severe 5–7 | 2.93 | 1.30–6.58 | 0.009 | – | – | – |
| Site of treatment: Hospital ward/Intensive Care Unit/Intermediate Care Unit | 2.68 | 1.21–5.96 | 0.015 | – | – | – |
| Moderate to severe exacerbation | 4.12 | 1.64–10.35 | 0.003 | 3.31 | 1.26–8.65 | 0.015 |
| Pseudomonas aeruginosa | 3.76 | 1.94–7.29 | <0.001 | 2.89 | 1.43–5.80 | 0.003 |
| MRSA | 6.02 | 1.13–31.98 | 0.035 | – | – | – |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Scioscia, G.; Amaro, R.; Alcaraz-Serrano, V.; Gabarrús, A.; Oscanoa, P.; Fernandez, L.; Menendez, R.; Mendez, R.; Foschino Barbaro, M.P.; Torres, A. Clinical Factors Associated with a Shorter or Longer Course of Antibiotic Treatment in Patients with Exacerbations of Bronchiectasis: A Prospective Cohort Study. J. Clin. Med. 2019, 8, 1950. https://doi.org/10.3390/jcm8111950
Scioscia G, Amaro R, Alcaraz-Serrano V, Gabarrús A, Oscanoa P, Fernandez L, Menendez R, Mendez R, Foschino Barbaro MP, Torres A. Clinical Factors Associated with a Shorter or Longer Course of Antibiotic Treatment in Patients with Exacerbations of Bronchiectasis: A Prospective Cohort Study. Journal of Clinical Medicine. 2019; 8(11):1950. https://doi.org/10.3390/jcm8111950
Chicago/Turabian StyleScioscia, Giulia, Rosanel Amaro, Victoria Alcaraz-Serrano, Albert Gabarrús, Patricia Oscanoa, Laia Fernandez, Rosario Menendez, Raul Mendez, Maria Pia Foschino Barbaro, and Antoni Torres. 2019. "Clinical Factors Associated with a Shorter or Longer Course of Antibiotic Treatment in Patients with Exacerbations of Bronchiectasis: A Prospective Cohort Study" Journal of Clinical Medicine 8, no. 11: 1950. https://doi.org/10.3390/jcm8111950
APA StyleScioscia, G., Amaro, R., Alcaraz-Serrano, V., Gabarrús, A., Oscanoa, P., Fernandez, L., Menendez, R., Mendez, R., Foschino Barbaro, M. P., & Torres, A. (2019). Clinical Factors Associated with a Shorter or Longer Course of Antibiotic Treatment in Patients with Exacerbations of Bronchiectasis: A Prospective Cohort Study. Journal of Clinical Medicine, 8(11), 1950. https://doi.org/10.3390/jcm8111950







