Resistant/Refractory Hypertension and Sleep Apnoea: Current Knowledge and Future Challenges
Abstract
1. Introduction
2. Resistant and Refractory Hypertension Phenotypes: Definition and Epidemiology
3. Clinical and Epidemiological Association with OSA
4. Pathophysiological Links with Obstructive Sleep Apnoea (OSA)
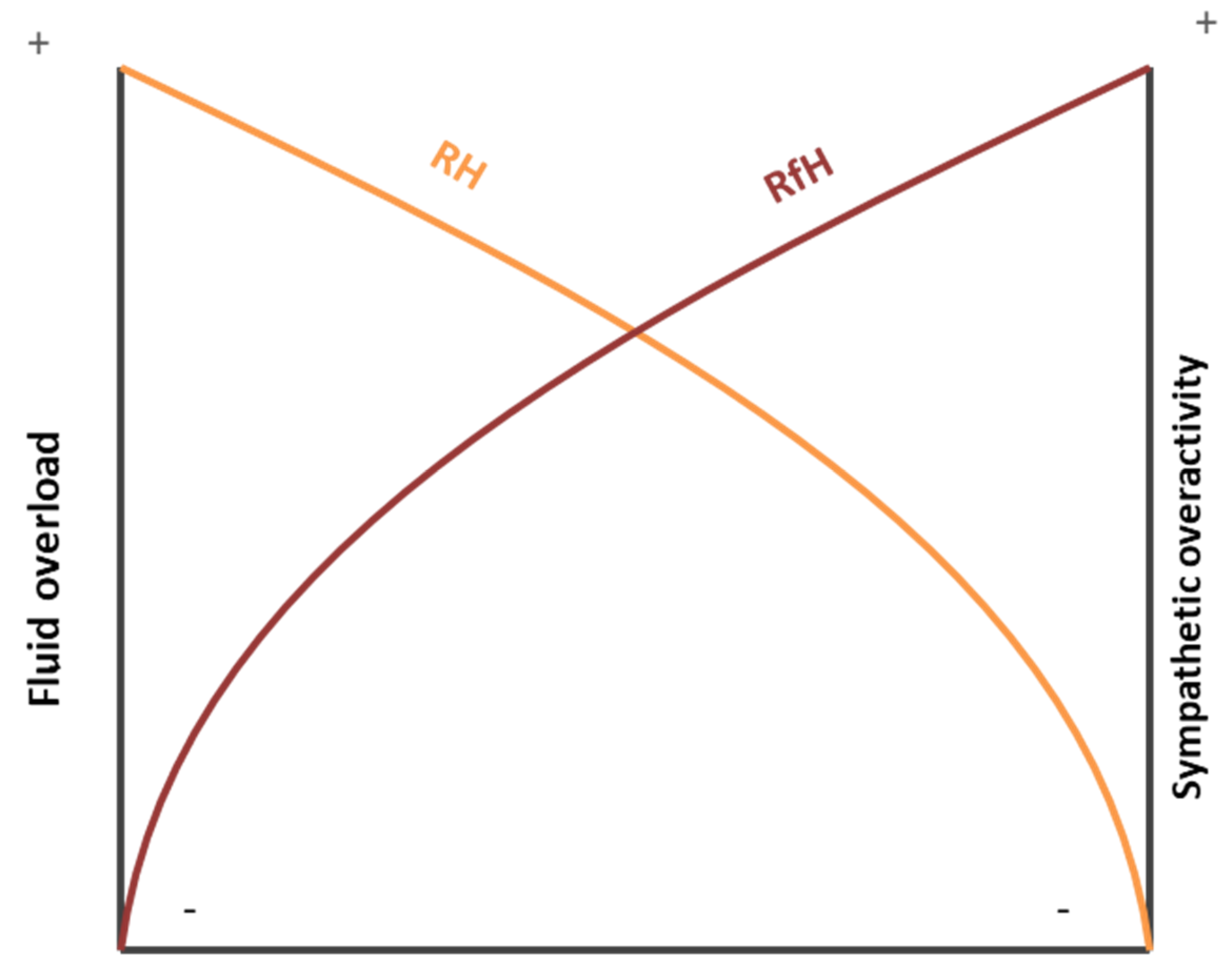
4.1. Resistant Hypertension
4.2. Refractory Hypertension
5. Multidimensional Treatment
5.1. Lifestyle Changes and Diet
5.2. Renal Denervation
5.3. Pharmacological Treatment
5.4. CPAP Treatment
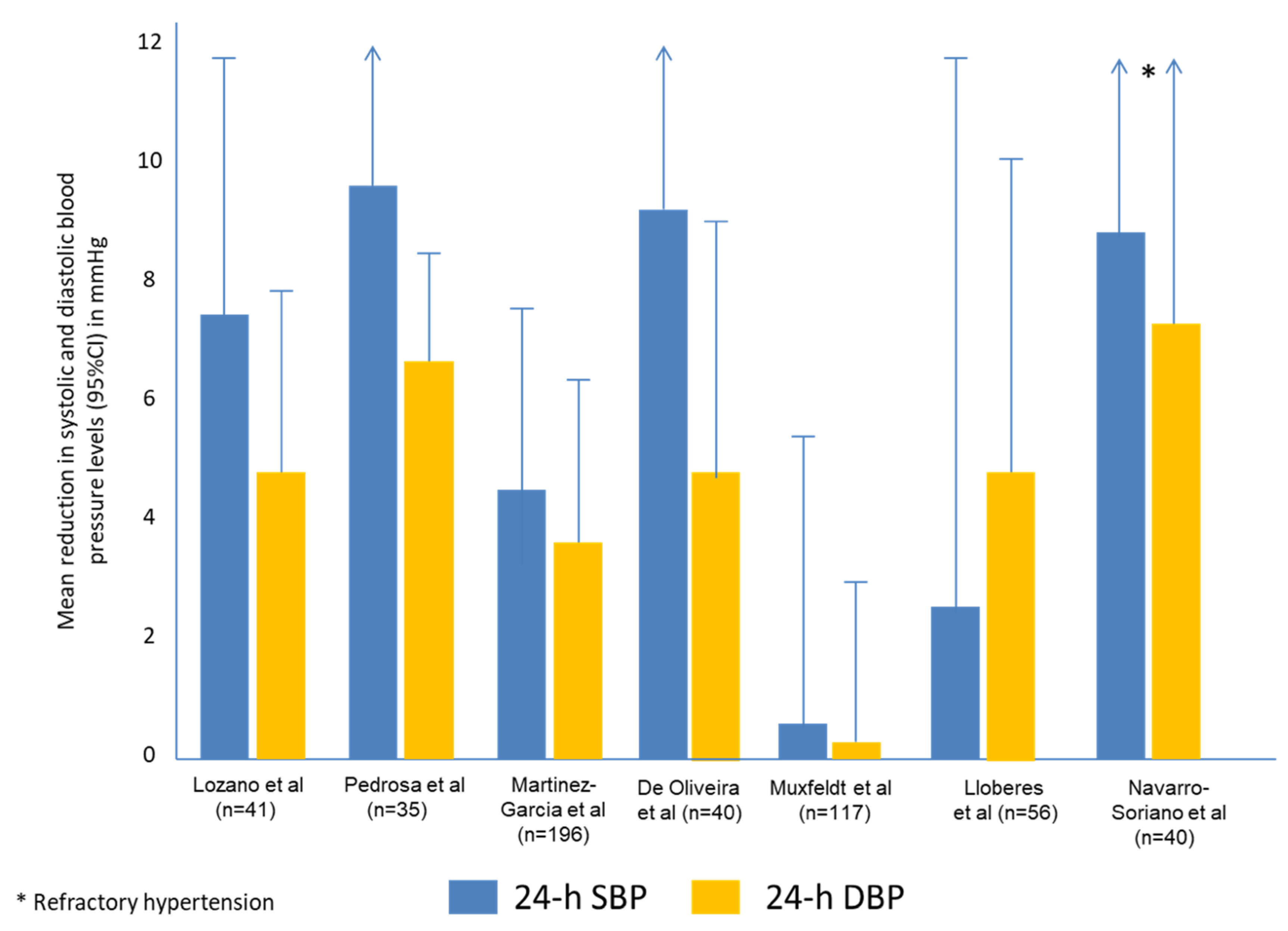
- The reduction in BP levels is greater in RH patients (especially in those with a lack of BP control) than in normotensive or well-controlled hypertensive patients (approximately 2 mmHg)
- The degree of reduction in BP levels is similar to those achieved by some antihypertensive drugs, especially in patients with RfH.
- There is a positive and linear correlation between the hours of use of CPAP and the reduction in BP levels.
- CPAP treatment produces an increase in the percentage of the normal nocturnal BP dipper pattern.
- CPAP decreases both the sympathetic activity and the level of aldosterone concentration in both RfH and RH patients, and these could constitute some of the pathophysiological action mechanisms involved in this treatment.
- The effect on RH of CPAP treatment combined with other pharmacological or non-pharmacological interventions such as renal denervation and antihypertensive drugs is still not known.
6. Future Challenges
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Williams, B.; Mancia, G.; Spiering, W.; Agabiti Rosei, E.; Azizi, M.; Burnier, M.; Clement, D.L.; Coca, A.; de Simone, G.; Dominiczak, A.; et al. 2018 ESC/ESH Guidelines for the management of arterial hypertension. Eur. Heart J. 2018, 39, 3021–3104. [Google Scholar] [CrossRef] [PubMed]
- Carey, R.M.; Sakhuja, S.; Calhoun, D.A.; Whelton, P.K.; Muntner, P. Prevalence of Apparent Treatment-Resistant Hypertension in the United States. Hypertension 2019, 73, 424–431. [Google Scholar] [CrossRef] [PubMed]
- Carey, R.M.; Calhoun, D.A.; Bakris, G.L.; Brook, R.D.; Daugherty, S.L.; Dennison-Himmelfarb, C.R.; Egan, B.M.; Flack, J.M.; Gidding, S.S.; Judd, E.; et al. American Heart Association Professional/Public Education and Publications Committee of the Council on Hypertension; Council on Cardiovascular and Stroke Nursing; Council on Clinical Cardiology; Council on Genomic and Precision Medicine; Council on Peripheral Vascular Disease; Council on Quality of Care and Outcomes Research; and Stroke Council. Resistant Hypertension: Detection, Evaluation, and Management: A Scientific Statement from the American Heart Association. Hypertension 2018, 72, e53–e90. [Google Scholar] [PubMed]
- Dudenbostel, T.; Siddiqui, M.; Oparil, S.; Calhoun, D.A. Refractory Hypertension: A Novel Phenotype of Antihypertensive Treatment Failure. Hypertension 2016, 67, 1085–1092. [Google Scholar] [CrossRef]
- Daugherty, S.L.; Powers, J.D.; Magid, D.J.; Tavel, H.M.; Masoudi, F.A.; Margolis, K.L.; O’Connor, P.J.; Selby, J.V.; Ho, P.M. Incidence and prognosis of resistant hypertension in hypertensive patients. Circulation 2012, 125, 1635–1642. [Google Scholar] [CrossRef]
- Javaheri, S.; Barbe, F.; Campos-Rodriguez, F.; Dempsey, J.A.; Khayat, R.; Javaheri, S.; Malhotra, A.; Martinez-Garcia, M.A.; Mehra, R.; Pack, A.I.; et al. Sleep Apnea: Types, Mechanisms, and Clinical Cardiovascular Consequences. J. Am. Coll. Cardiol. 2017, 69, 841–858. [Google Scholar] [CrossRef]
- Peppard, P.E.; Young, T.; Barnet, J.H.; Palta, M.; Hagen, E.W.; Hla, K.M. Increased prevalence of sleep-disordered breathing in adults. Am. J. Epidemiol. 2013, 177, 1006–1014. [Google Scholar] [CrossRef]
- Fietze, I.; Laharnar, N.; Obst, A.; Ewert, R.; Felix, S.B.; Garcia, C.; Gläser, S.; Glos, M.; Schmidt, C.O.; Stubbe, B.; et al. Prevalence and association analysis of obstructive sleep apnea with gender and age differences—Results of SHIP-Trend. J. Sleep Res. 2019, 28, e12770. [Google Scholar] [CrossRef]
- Heinzer, R.; Vat, S.; Marques-Vidal, P.; Marti-Soler, H.; Andries, D.; Tobback, N.; Mooser, V.; Preisig, M.; Malhotra, A.; Waeber, G.; et al. Prevalence of sleep-disordered breathing in the general population: The HypnoLaus study. Lancet Respir. Med. 2015, 3, 310–318. [Google Scholar] [CrossRef]
- Senaratna, C.V.; Perret, J.L.; Lodge, C.J.; Lowe, A.J.; Campbell, B.E.; Matheson, M.C.; Hamilton, G.S.; Dharmage, S.C. Prevalence of obstructive sleep apnea in the general population: A systematic review. Sleep Med Rev. 2017, 34, 70–81. [Google Scholar] [CrossRef]
- Benjafield, A.V.; Ayas, N.T.; Eastwood, P.R.; Heinzer, R.; Ip, M.S.M.; Morrell, M.J.; Nunez, C.M.; Patel, S.R.; Penzel, T.; Pépin, J.-L.; et al. Estimation of the global prevalence and burden of obstructive sleep apnoea: A literature-based analysis. Lancet Respir. Med. 2019, 7, 687–698. [Google Scholar] [CrossRef]
- Sjöström, C.; Lindberg, E.; Elmasry, A.; Hägg, A.; Svärdsudd, K.; Janson, C. Prevalence of sleep apnoea and snoring in hypertensive men: A population based study. Thorax 2002, 57, 602–607. [Google Scholar] [CrossRef] [PubMed]
- Logan, A.G.; Perlikowski, S.M.; Mente, A.; Tisler, A.; Tkacova, R.; Niroumand, M.; Leung, R.S.; Bradley, T.D. High prevalence of unrecognized sleep apnoea in drug-resistant hypertension. J. Hypertens. 2001, 19, 2271–2277. [Google Scholar] [CrossRef]
- Ruttanaumpawan, P.; Nopmaneejumruslers, C.; Logan, A.G.; Lazarescu, A.; Qian, I.; Bradley, T.D. Association between refractory hypertension and obstructive sleep apnea. J. Hypertens. 2009, 27, 1439–1445. [Google Scholar] [CrossRef] [PubMed]
- Martínez-García, M.-A.; Navarro-Soriano, C.; Torres, G.; Barbé, F.; Caballero-Eraso, C.; Lloberes, P.; Diaz-Cambriles, T.; Somoza, M.; Masa, J.F.; González, M.; et al. Beyond Resistant Hypertension. Hypertension 2018, 72, 618–624. [Google Scholar] [CrossRef] [PubMed]
- Pedrosa, R.P.; Drager, L.F.; Gonzaga, C.C.; Sousa, M.G.; de Paula, L.K.G.; Amaro, A.C.S.; Amodeo, C.; Bortolotto, L.A.; Krieger, E.M.; Bradley, T.D.; et al. Obstructive sleep apnea: The most common secondary cause of hypertension associated with resistant hypertension. Hypertension 2011, 58, 811–817. [Google Scholar] [CrossRef] [PubMed]
- Martínez-García, M.A.; Capote, F.; Campos-Rodríguez, F.; Lloberes, P.; Díaz de Atauri, M.J.; Somoza, M.; Masa, J.F.; González, M.; Sacristán, L.; Barbé, F.; et al. Effect of CPAP on blood pressure in patients with obstructive sleep apnea and resistant hypertension: The HIPARCO randomized clinical trial. JAMA 2013, 310, 2407–2415. [Google Scholar] [CrossRef] [PubMed]
- Calhoun, D.A.; Jones, D.; Textor, S.; Goff, D.C.; Murphy, T.P.; Toto, R.D.; White, A.; Cushman, W.C.; White, W.; Sica, D.; et al. Resistant Hypertension: Diagnosis, Evaluation and Treatment A Scientific Statement From the American Heart Association Professional Education Committee of the Council for High Blood Pressure Research. Circulation 2008, 117, e510–e526. [Google Scholar] [CrossRef]
- Kumbhani, D.J.; Steg, P.G.; Cannon, C.P.; Eagle, K.A.; Smith, S.C., Jr.; Crowley, K.; Goto, S.; Ohman, E.M.; Bakris, G.L.; Perlstein, T.S.; et al. Resistant hypertension: A frequent and ominous finding among hypertensive patients with atherothrombosis. Eur. Heart J. 2013, 34, 1204–1214. [Google Scholar] [CrossRef]
- Egan, B.M.; Zaho, Y.; Axon, R.N.; Brezinsky, W.A.; Ferdinand, K.C. Uncontrolled and apparent treatment resistant hypertension in the United States, 1988–2008. Circulation 2011, 124, 1046–1058. [Google Scholar] [CrossRef]
- de la Sierra, A.; Segura, J.; Banegas, J.R.; Gorostidi, M.; de la Cruz, J.J.; Armario, P.; Oliveras, A.; Ruilope, L.M. Clinical features of 8295 patients with resistant hypertension clasified on the basis of ambulatory blood pressure monitoring. Hypertension 2011, 57, 898–902. [Google Scholar] [CrossRef] [PubMed]
- Acelajado, M.C.; Pisoni, R.; Dudenbostel, T.; Dell’ Italia, L.J.; Cartmill, F.; Zhang, B.; Cofield, S.S.; Oparil, S.; Calhoun, D.A. Refractory hypertension: Definition, prevalence, and patient characteristics. J. Clin. Hypertens. 2012, 14, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Dudenbostel, T.; Acelajado, M.C.; Pisoni, R.; Li, P.; Oparil, S.; Calhoun, D.A. Refractory hypertension: Evidence of heightened sympathetic activity as a cause of antihypertensive treatment failure. Hypertension 2015, 66, 126–133. [Google Scholar] [CrossRef] [PubMed]
- Armario, P.; Calhoun, D.A.; Oliveras, A.; Blanch, P.; Vinyoles, E.; Banega, J.R.; Gorostidi, M.; Segura, J.; Ruilope, L.M.; Dudenbostel, T.; et al. Prevalence and clinical characteristics of refractory hypertension. J. Am. Heart Assoc. 2017, 6, e007365. [Google Scholar] [CrossRef] [PubMed]
- Calhoun, D.A.; Booth, J.N., III; Oparil, S.; Irvin, M.R.; Shimbo, D.; Lackland, D.T.; Howard, G.; Safford, M.M.; Muntner, P. Refractory hypertension: Determination of prevalence, risk factors, and comorbidities in a large, population-based cohort. Hypertension 2014, 63, 451–458. [Google Scholar] [CrossRef]
- Modolo, R.; de Faria, A.P.; Sabbatini, A.R.; Barbaro, N.R.; Ritter, A.M.; Moreno, H. Refractory and resistant hypertension. Characteristics and differences observed in a specialized clinic. J. Am. Soc. Hypertens. 2015, 9, 397–402. [Google Scholar] [CrossRef]
- Lavie, P. Obstructive sleep apnoea syndrome as a risk factor for hypertension: Population study. BMJ. 2000, 320, 479–482. [Google Scholar] [CrossRef]
- Nieto, F.J.; Young, T.B.; Lind, B.K.; Shahar, E.; Samet, J.M.; Redline, S.; D’Agostino, R.B.; Newman, A.B.; Lebowitz, M.D.; Pickering, T.G. Association of sleep-disordered breathing, sleep apnea, and hypertension in a large community-based study. Sleep Heart Health Study. JAMA 2000, 283, 1829–1836. [Google Scholar] [CrossRef]
- Martínez-García, M.A.; Gómez-Aldaraví, R.; Gil-Martínez, T.; Soler-Cataluña, J.J.; Bernácer, B.; Román-Sánchez, P. Sleep-disordered breathing in patients with difficult-to-control hypertension. Arch. Bronconeumol. 2006, 42, 14–20. [Google Scholar] [CrossRef]
- Calhoun, D.A.; Nishizaka, M.K.; Zaman, M.A.; Harding, S.M. Aldosterone excretion among subjects with resistant hypertension and symptoms of sleep apnea. Chest 2004, 125, 112–117. [Google Scholar] [CrossRef]
- Demede, M.; Pandey, A.; Zizi, F.; Bachmann, R.; Donat, M.; McFarlane, S.I.; Jean-Louis, G.; Ogedegbe, G. Resistant hypertension and obstructive sleep apnea in the primary-care setting. Int. J. Hypertens. 2011, 2011, 340929. [Google Scholar] [CrossRef] [PubMed]
- Florczak, E.; Prejbisz, A.; Szwench-Pietrasz, E.; Sliwiński, P.; Bieleń, P.; Klisiewicz, A.; Michałowska, I.; Warchoł, E.; Januszewicz, M.; Kała, M.; et al. Clinical characteristics of patients with resistant hypertension: The RESIST-POL study. J. Hum. Hypertens. 2013, 27, 678–685. [Google Scholar] [CrossRef] [PubMed]
- Lloberes, P.; Lozano, L.; Sampol, G.; Romero, O.; Jurado, M.J.; Ríos, J.; Untoria, M.D.; Tovar, J.L. Obstructive sleep apnoea and 24-h blood pressure in patients with resistant hypertension. J. Sleep Res. 2010, 19, 597–602. [Google Scholar] [CrossRef] [PubMed]
- Pratt-Ubunama, M.N.; Nishizaka, M.K.; Boedefeld, R.L.; Cofield, S.S.; Harding, S.M.; Calhoun, D.A. Plasma aldosterone is related to severity of obstructive sleep apnea in subjects with resistant hypertension. Chest 2007, 131, 453–459. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, S.C.; Martinez, D.; Gus, M.; de Abreu-Silva, E.O.; Bertoluci, C.; Dutra, I.; Branchi, T.; Moreira, L.B.; Fuchs, S.C.; de Oliveira, A.C.T.; et al. Obstructive sleep apnea and resistant hypertension: A case-control study. Chest 2007, 132, 1858–1862. [Google Scholar] [CrossRef]
- Bhandari, S.K.; Shi, J.; Molnar, M.Z.; Rasgon, S.A.; Derose, S.F.; Kovesdy, C.P.; Calhoun, D.A.; Kalantar-Zadeh, K.; Jacobsen, S.J.; Sim, J.J. Comparisons of sleep apnoea rate and outcomes among patients with resistant and non-resistant hypertension. Respirology 2016, 21, 1486–1492. [Google Scholar] [CrossRef]
- Johnson, D.A.; Thomas, S.J.; Abdalla, M.; Guo, N.; Yano, Y.; Rueschman, M.; Tanner, R.M.; Mittleman, M.A.; Calhoun, D.A.; Wilson, J.G.; et al. Association Between Sleep Apnea and Blood Pressure Control Among Blacks. Circulation 2019, 139, 1275–1284. [Google Scholar] [CrossRef]
- Abdel-Kader, K.; Dohar, S.; Shah, N.; Jhamb, M.; Reis, S.E.; Strollo, P.; Buysse, D.; Unruh, M.L. Resistant hypertension and obstructive sleep apnea in the setting of kidney disease. J. Hypertens. 2012, 30, 960–966. [Google Scholar] [CrossRef]
- Gus, M.; Gonçalves, S.C.; Martinez, D.; de Abreu Silva, E.O.; Moreira, L.B.; Fuchs, S.C.; Fuchs, F.D. Risk for Obstructive Sleep Apnea by Berlin Questionnaire, but not daytime sleepiness, is associated with resistant hypertension: A case-control study. Am. J. Hypertens. 2008, 21, 832–835. [Google Scholar] [CrossRef]
- Chobanian, A.V.; Bakris, G.L.; Black, H.R.; Cushman, W.C.; Green, L.A.; Izzo, J.L.; Jones, D.W.; Materson, B.J.; Oparil, S.; Wright, J.T.; et al. Seventh Report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure. Hypertension 2003, 42, 1206–1252. [Google Scholar] [CrossRef]
- Whelton, P.K.; Carey, R.M.; Aronow, W.S.; Casey, D.E.; Collins, K.J.; Dennison Himmelfarb, C.; DePalma, S.M.; Gidding, S.; Jamerson, K.A.; Jones, D.W.; et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA Guideline for the Prevention, Detection, Evaluation, and Management of High Blood Pressure in Adults: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Hypertension 2018, 71, e13–e115. [Google Scholar] [PubMed]
- Hwang, A.Y.; Dietrich, E.; Pepine, C.J.; Smith, S.M. Resistant Hypertension: Mechanisms and Treatment. Curr. Hypertens. Rep. 2017, 19, 56. [Google Scholar] [CrossRef] [PubMed]
- Tsioufis, C.; Kordalis, A.; Flessas, D. Pathophysiology of resistant hypertension: The role of sympathetic nervous system. Int. J. Hypertens. 2011, 2011, 642416. [Google Scholar] [CrossRef] [PubMed]
- Pimienta, E.; Gaddam, K.K.; Oparil, S.; Aban, I.; Husain, S.; Dell’ Italia, L.J.; Calhoun, D.A. Effects of dietary sodium reduction on blood pressure in subjects with resistant hypertension: Results from a randomized trial. Hypertension 2009, 54, 475–481. [Google Scholar] [CrossRef] [PubMed]
- Calhoun, D.A.; Nishizaka, M.K.; Zaman, M.A.; Thakkar, R.B.; Weisssmann, P. Hyperaldosteronism among black and white subjects with resistant hypertension. Hypertension 2002, 40, 892–896. [Google Scholar] [CrossRef] [PubMed]
- Václavík, J.; Sedlák, R.; Plachy, M.; Navrátil, K.; Plásek, J.; Jarkovsky, J.; Václavík, T.; Husár, R.; Kociánová, E.; Táborsky, M. Addition of spironolactone in patients with resistant arterial hypertension (ASPIRANT): A randomized, double-blind, placebo-controlled trial. Hypertension 2011, 57, 1069–1075. [Google Scholar] [CrossRef] [PubMed]
- Zhao, D.; Liu, H.; Dong, P.; Zhao, J. A meta-analysis of add-on use of spironolactone in patients with resistant hypertension. Int. J. Cardiol. 2017, 233, 113–117. [Google Scholar] [CrossRef] [PubMed]
- Williams, B.; MacDonald, T.M.; Morant, S.; Webb, D.J.; Sever, P.; McInnes, G.; Ford, I.; Cruickshank, J.K.; Caulfield, M.J.; Salsbury, J.; et al. Spironolactone versus placebo, bisoprolol, and doxazosin to determine the optimal treatment for drug-resistant hypertension (PATHWAY-2): A randomised, double-blind, crossover trial. Lancet 2015, 386, 2059–2068. [Google Scholar] [CrossRef]
- Friedman, O.; Bradley, T.D.; Chan, C.T.; Parkes, R.; Logan, A.G. Relationship between overnight rostral fluid shift and obstructive sleep apnea in drug-resistant hypertension. Hypertension 2010, 56, 1077–1082. [Google Scholar] [CrossRef]
- Dempsey, J.A.; Veasey, S.C.; Morgan, B.J.; O’Donnell, C.P. Pathophysiology of sleep apnea. Physiol. Rev. 2010, 90, 47–112. [Google Scholar] [CrossRef]
- Sánchez-de-la-Torre, M.; Campos-Rodriguez, F.; Barbé, F. Obstructive sleep apnoea and cardiovascular disease. Lancet Respir. Med. 2013, 1, 61–72. [Google Scholar] [CrossRef]
- Dudenbostel, T.; Siddiqui, M.; Gharpure, N.; Calhoun, D.A. Refractory versus resistant hypertension: Novel distinctive phenotypes. J Nat. Sci. 2017, 3, e430. [Google Scholar] [PubMed]
- Foster, G.D.; Borradaile, K.E.; Sanders, M.H.; Millman, R.; Zammit, G.; Newman, A.B.; Wadden, T.A.; Kelley, D.; Wing, R.; Pi-Sunyer, F.X.; et al. A randomized study on the effect of weight loss on obstructive sleep apnea among obese patients with Type 2 diabetes. Arch. Intern. Med. 2009, 169, 1619–1626. [Google Scholar] [CrossRef] [PubMed]
- Fiori, C.Z.; Martinez, D.; Montanari, C.C.; Lopez, P.; Camargo, R.; Sezerá, L.; Gonçalves, S.C.; Fuchs, F.D. Diuretic or sodium-restricted diet for obstructive sleep apnea-a randomized trial. Sleep 2018, 41. [Google Scholar] [CrossRef]
- McMahon, E.J.; Bauer, J.D.; Hawley, C.M.; Isbel, N.M.; Stowasser, M.; Johnson, D.W.; Campbell, K.L. A randomized trial of dietary sodium restriction in CKD. J. Am. Soc. Nephrol. 2013, 24, 2096–2103. [Google Scholar] [CrossRef]
- Mente, A.; O’Donnell, M.; Rangarajan, S.; Dagenais, G.; Lear, S.; McQueen, M.; Diaz, R.; Avezum, A.; Lopez-Jaramillo, P.; Lanas, F.; et al. Associations of urinary sodium excretion with cardiovascular events in individuals with and without hypertension: A pooled analysis of data from four studies. Lancet 2016, 388, 465–475. [Google Scholar] [CrossRef]
- Gay, H.C.; Rao, S.G.; Vaccarino, V.; Ali, M.K. Effects of Different Dietary Interventions on Blood Pressure: Systematic Review and Meta-Analysis of Randomized Controlled Trials. Hypertension 2016, 67, 733–739. [Google Scholar] [CrossRef]
- Saneei, P.; Salehi-Abargouei, A.; Esmaillzadeh, A.; Azadbakht, L. Influence of Dietary Approaches to Stop Hypertension (DASH) diet on blood pressure: A systematic review and meta-analysis on randomized controlled trials. Nutr. Metab. Cardiovasc. Dis. 2014, 24, 1253–1261. [Google Scholar] [CrossRef]
- De Pergola, G.; D’Alessandro, A. Influence of Mediterranean Diet on Blood Pressure. Nutrients 2018, 10, 1700. [Google Scholar] [CrossRef]
- Krum, H.; Schlaich, M.; Whitbourn, R.; Sobotka, P.A.; Sadowski, J.; Bartus, K.; Kapelak, B.; Walton, A.; Sievert, H.; Thambar, S.; et al. Catheter-based renal sympathetic denervation for resistant hypertension: A multicentre safety and proof-of-principle cohort study. Lancet 2009, 373, 1275–1281. [Google Scholar] [CrossRef]
- Symplicity HTN-2 Investigators; Esler, M.D.; Krum, H.; Sobotka, P.A.; Schlaich, M.P.; Schmieder, R.E.; Böhm, M. Renal sympathetic denervation in patients with treatment-resistant hypertension (The SymplicityHTN-2 Trial): A randomised controlled trial. Lancet 2010, 376, 1903–1909. [Google Scholar] [PubMed]
- Warchol-Celinska, E.; Prejbisz, A.; Kadziela, J.; Florczak, E.; Januszewicz, M.; Michalowska, I.; Dobrowolski, P.; Kabat, M.; Sliwinski, P.; Klisiewicz, A.; et al. Renal denervation in resistant hypertension and obstructive sleep apnea: Randomized proof-of-concept phase II trial. Hypertension 2018, 72, 381–390. [Google Scholar] [CrossRef] [PubMed]
- Linz, D.; Mancia, G.; Mahfoud, F.; Narkiewicz, K.; Ruilope, L.; Schlaich, M.; Kindermann, I.; Schmieder, R.E.; Ewen, S.; Williams, B.; et al. Renal artery denervation for treatment of patients with self-reported obstructive sleep apnea and resistant hypertension: Results from the Global SYMPLICITY Registry. J. Hypertens. 2017, 35, 148–153. [Google Scholar] [CrossRef] [PubMed]
- Bhatt, D.L.; Kandzari, D.E.; O’Neill, W.W.; D’Agostino, R.; Flack, J.M.; Katzen, B.T.; Leon, M.B.; Liu, M.; Mauri, L.; Negoita, M.; et al. A controlled trial of renal denervation for resistant hypertension. N. Engl. J. Med. 2014, 370, 1393–1401. [Google Scholar] [CrossRef]
- Gaddam, K.; Pimenta, E.; Thomas, S.J.; Cofield, S.S.; Oparil, S.; Harding, S.M.; Calhoun, D.A. Spironolactone reduces severity of obstructive sleep apnoea in patients with resistant hypertension: A preliminary report. J. Hum. Hypertens. 2010, 24, 532–537. [Google Scholar] [CrossRef]
- Yang, L.; Zhang, H.; Cai, M.; Zou, Y.; Jiang, X.; Song, L.; Liang, E.; Bian, J.; Wu, H.; Hui, R. Effect of spironolactone on patients with resistant hypertension and obstructive sleep apnea. Clin. Exp. Hypertens. 2016, 38, 464–468. [Google Scholar] [CrossRef]
- Bucca, C.B.; Brussino, L.; Battisti, A.; Mutani, R.; Rolla, G.; Mangiardi, L.; Cicolin, A. Diuretics in obstructive sleep apnea with diastolic heart failure. Chest 2007, 132, 440–446. [Google Scholar] [CrossRef]
- Gaisl, T.; Haile, S.R.; Thiel, S.; Osswald, M.; Kohler, M. Efficacy of pharmacotherapy for OSA in adults: A systematic review and network meta-analysis. Sleep Med. Rev. 2019, 46, 74–86. [Google Scholar] [CrossRef]
- Engbaek, M.; Hjerrild, M.; Hallas, J.; Jacobsen, I.A. The effect of low-dose spironolactone on resistant hypertension. J. Am. Soc. Hypertens. 2010, 4, 290–294. [Google Scholar] [CrossRef]
- Lazich, I.; Bakris, G.L. Prediction and management of hyperkalemia across the spectrum of chronic kidney disease. Semin. Nephrol. 2014, 34, 333–339. [Google Scholar] [CrossRef]
- Muxfeldt, E.S.; Margallo, V.; Costa, L.M.S.; Guimaraes, G.; Cavalcante, A.H.; Azevedo, J.C.; de Souza, F.; Cardoso, C.R.; Salles, G.F. Effects of continuous positive airway pressure treatment on clinic and ambulatory blood pressures in patients with obstructive sleep apnea and resistant hypertension. A randomized controlled trial. Hypertension 2015, 65, 736–742. [Google Scholar] [CrossRef] [PubMed]
- Iftikhar, I.H.; Valentine, C.W.; Bittencourt, L.R.A.; Cohen, D.L.; Fedson, A.C.; Gíslason, T.; Penzel, T.; Phillips, C.L.; Yu-sheng, L.; Pack, A.I.; et al. Effects of continuous positive airway pressure on blood pressure in patients with resistant hypertension and obstructive sleep apnea: A meta-analysis. J. Hypertens. 2014, 32, 2341–2350. [Google Scholar] [CrossRef] [PubMed]
- Fava, C.; Dorigoni, S.; Dalle Vedove, F.; Danese, E.; Montagnana, M.; Guidi, G.C.; Narkiewicz, K.; Minuz, P. Effect of CPAP on blood pressure in patients with OSA/hypopnea: A systematic review and meta-analysis. Chest 2014, 145, 762–771. [Google Scholar] [CrossRef] [PubMed]
- Montesi, S.B.; Edwards, B.A.; Malhotra, A.; Bakker, J.P. The effect of continuous positive airway pressure treatment on blood pressure: A systematic review and meta-analysis of randomized controlled trials. J. Clin. Sleep. Med. 2012, 8, 587–596. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-de-la-Torre, M.; Khalyfa, A.; Sánchez-de-la-Torre, A.; Martinez-Alonso, M.; Martinez-García, M.Á.; Barceló, A.; Lloberes, P.; Campos-Rodriguez, F.; Capote, F.; Diaz-de-Atauri, M.J.; et al. Precision Medicine in Patients with Resistant Hypertension and Obstructive Sleep Apnea: Blood Pressure Response to Continuous Positive Airway Pressure Treatment. J. Am. Coll. Cardiol. 2015, 66, 1023–1032. [Google Scholar] [CrossRef] [PubMed]
- Logan, A.G.; Tkacova, R.; Perlikowski, S.M.; Leung, R.S.; Tisler, A.; Floras, J.S.; Bradley, T.D. Refractory hypertension and sleep apnoea: Effect of CPAP on blood pressure and baroreflex. Eur. Respir. J. 2003, 21, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Lozano, L.; Tovar, J.L.; Sampol, G.; Romero, O.; Jurado, M.J.; Segarra, A.; Espinel, E.; Río, J.; Untoria, M.D.; Lloberes, P. Continuous positive airway pressure treatment in sleep apnea patients with resistant hypertension: A randomized, controlled trial. J. Hypertens. 2010, 28, 2161–2168. [Google Scholar] [CrossRef]
- de Oliveira, A.C.; Martinez, D.; Massierer, D.; Gus, M.; Gonçalves, S.C.; Ghizzoni, F.; Steinhorst, A.M.; Moreira, L.B.; Fuchs, S.C.; Fuchs, F.D. The antihypertensive effect of positive airway pressure on resistant hypertension of patients with obstructive sleep apnea: A randomized, double-blind, clinical trial. Am. J. Respir. Crit. Care. Med. 2014, 190, 345–347. [Google Scholar] [CrossRef]
- Lloberes, P.; Sampol, G.; Espinel, E.; Segarra, A.; Ramon, M.A.; Romero, O.; Ferrer, R.; Martínez-Garcia, M.A.; Tovar, J.L. A randomized controlled study of CPAP effect on plasma aldosterone concentration in patients with resistant hypertension and obstructive sleep apnea. J. Hypertens. 2014, 32, 1650–1657. [Google Scholar] [CrossRef]
- Navarro-Soriano, C.; Martínez-García, M.A.; Torres, G.; Barbé, F.; Caballero-Eraso, C.; Lloberes, P.; Diaz Cambriles, T.; Somoza, M.; Masa, J.F.; González, M.; et al. Effect of continuous positive airway pressure in patients with true refractory hypertension and sleep apnea: A post-hoc intention-to-treat analysis of the HIPARCO randomized clinical trial. J. Hypertens. 2019, 37, 1269–1275. [Google Scholar] [CrossRef]
- Joyeux-Faure, M.; Baguet, J.P.; Barone-Rochette, G.; Faure, P.; Sosner, P.; Mounier-Vehier, C.; Lévy, P.; Tamisier, R.; Pépin, J.L. Continuous Positive Airway Pressure Reduces Night-Time Blood Pressure and Heart Rate in Patients with Obstructive Sleep Apnea and Resistant Hypertension: The RHOOSAS Randomized Controlled Trial. Front. Neurol. 2018, 9, 318. [Google Scholar] [CrossRef] [PubMed]
- de Souza, F.; Muxfeldt, E.S.; Margallo, V.; Cortez, A.F.; Cavalcanti, A.H.; Salles, G.F. Effects of continuous positive airway pressure treatment on aldosterone excretion in patients with obstructive sleep apnoea and resistant hypertension: A randomized controlled trial. J. Hypertens. 2017, 35, 837–844. [Google Scholar] [CrossRef] [PubMed]
- Pedrosa, R.P.; Drager, L.F.; de Paula, L.K.G.; Amaro, A.C.S.; Bortolotto, L.A.; Lorenzi-Filho, G. Effects of OSA treatment on BP in patients with resistant hypertension. A randomized trial. Chest 2013, 14, 1487–1494. [Google Scholar] [CrossRef] [PubMed]
- Dernaika, T.A.; Kinasewitz, G.T.; Tawk, M.M. Effects of nocturnal continuous positive airway pressure therapy in patients with resistant hypertension and obstructive sleep apnea. J. Clin. Sleep Med. 2009, 5, 103–107. [Google Scholar]
- Martínez-García, M.A.; Gómez-Aldaraví, R.; Soler-Cataluña, J.J.; Martínez, T.G.; Bernácer-Alpera, B.; Román-Sánchez, P. Positive effect of CPAP treatment on the control of difficult-to-treat hypertension. Eur. Respir. J. 2007, 29, 951–957. [Google Scholar] [CrossRef]
- Walia, H.K.; Griffith, S.D.; Foldvary-Schaefer, N.; Thomas, G.; Bravo, E.L.; Moul, D.E.; Mehra, R. Longitudinal Effect of CPAP on BP in Resistant and Nonresistant Hypertension in a Large Clinic-Based Cohort. Chest 2016, 149, 747–755. [Google Scholar] [CrossRef]
- Frenţ, Ş.M.; Tudorache, V.M.; Ardelean, C.; Mihăicuţă, S. Long-term effects of nocturnal continuous positive airway pressure therapy in patients with resistant hypertension and obstructive sleep apnea. Pneumologia 2014, 204, 207–211. [Google Scholar]
- Lei, Q.; Lv, Y.; Li, K.; Ma, L.; Du, G.; Xiang, Y.; Li, X. Effects of continuous positive airway pressure on blood pressure in patients with resistant hypertension and obstructive sleep apnea: A systematic review and meta-analysis of six randomized controlled trials. J. Bras. Pneumol. 2017, 43, 373–379. [Google Scholar] [CrossRef]
- Feldstein, C.A. Blood pressure effects of CPAP in nonresistant and resistant hypertension associated with OSA: A systematic review of randomized clinical trials. Clin. Exp. Hypertens. 2016, 38, 337–346. [Google Scholar] [CrossRef]
- Varounis, C.; Katsi, V.; Kallikazaros, I.E.; Tousoulis, D.; Stefanadis, C.; Parissis, J.; Lekakis, J.; Siristatidis, C.; Manolis, A.J.; Makris, T. Effect of CPAP on blood pressure in patients with obstructive sleep apnea and resistant hypertension: A systematic review and meta-analysis. Int. J. Cardiol. 2014, 175, 195–198. [Google Scholar] [CrossRef]
- Liu, L.; Cao, Q.; Guo, Z.; Dai, Q. Continuous Positive Airway Pressure in Patients with Obstructive Sleep Apnea and Resistant Hypertension: A Meta-Analysis of Randomized Controlled Trials. J. Clin. Hypertens. 2016, 18, 153–158. [Google Scholar] [CrossRef] [PubMed]
- Sapiña-Beltrán, E.; Torres, G.; Martínez-Alonso, M.; Sánchez-de-la-Torre, M.; Franch, M.; Bravo, C.; Masa, J.F.; Felez, M.; Fortuna-Gutierrez, A.M.; Abad, J.; et al. Rationale and Methodology of the SARAH Trial: Long-Term Cardiovascular Outcomes in Patients with Resistant Hypertension and Obstructive Sleep Apnea. Arch. Bronconeumol. 2018, 54, 518–523. [Google Scholar] [CrossRef] [PubMed]
- Campos-Rodriguez, F.; Navarro-Soriano, C.; Reyes-Nuñez, N.; Torres, G.; Caballero-Eraso, C.; Lloberes, P.; Diaz-Cambriles, T.; Somoza, M.; Masa, J.F.; Gonzalez, M.; et al. Good long-term adherence to continuous positive airway pressure therapy in patients with resistant hypertension and sleep apnea. J. Sleep Res. 2019, 28, e12805. [Google Scholar] [CrossRef] [PubMed]
- Navarro-Soriano, C.; Martínez-García, M.A.; Torres, G.; Barbé, F.; Caballero-Eraso, C.; Lloberes, P.; Cambriles, T.D.; Somoza, M.; Masa, J.F.; González, M.; et al. Factors associated with the changes from a resistant to a refractory phenotype in hypertensive patients: A Pragmatic Longitudinal Study. Hypertens. Res. 2019, 42, 1708–1715. [Google Scholar] [CrossRef] [PubMed]
| Studies | Patients (n) | Age (Years) | Type of BP Measure SBP/DBP (mmHg) | Type of Sleep Study (AHI Threshold to Define OSA) | OSA Prevalence/AHI |
|---|---|---|---|---|---|
| Logan 2001 [13] | 41 patients with resistant HT (24 men, 17 women) | 57.2 (1.6) Men 54.6 (1.8) Women 58.3 (3.0) | 24 h ABPM SBP: 149.0 (2.6) in Men, 150.6 (3.7) in women DBP: 86.3 (2.0) in Men, 83.7 (1.9) in women | PSG (AHI ≥ 10) | 82.9% (96% in men, 65% in women) Mean AHI: 32.2 (4.5) in men, 14.0 (3.1) in women |
| Martinez-Garcia 2006 [29] | 49 pts with resistant HT (40.8% men) | 68.1 (9.1) | 24 h ABPM SBP: 152.5 (13) DBP: 89.2 (8.5) | RP (AHI ≥ 10) | AHI ≥ 10: 71.4% AHI ≥ 30: 40.8% Mean AHI: 26.2 (19.5) |
| Gonçalves 2007 [35] | 63 pts with resistant HT (21 men, 42 women) and 63 pts with controlled HT (23 men, 40 women) | 59 (7) in both the resistant and controlled HT groups | 24 h ABPM SBP: 141 (17) in the resistant HT group vs. 121 (10) in the controlled HT group DBP: 84 (12) in the resistant HT group vs. 74 (7) in the controlled HT group | RP (AHI ≥ 10) | 71% in the resistant HT group vs. 38% in the controlled HT group (p < 0.001) Men: 86% vs. 52% (p = 0.016) Women: 64% vs. 30% (p = 0.002) |
| Prat-Ubunama 2007 [34] | 71 pts with resistant HT | 56.0 (9.9) | Office BP measurement SBP: 155.8 (27) DBP: 88.3 (15) | PSG (AHI ≥ 5) | 85% (90% in men, 77% in women) Mean AHI: 24.1 (24.7) (Men 20.8, Women 10.8) |
| Lloberes 2010 [33] | 62 pts with resistant HT (67.3% men) | 59 (10) | 24 h ABPM SBP: 139.1 (1.6) DBP: 80.9 (1.2) | PSG (AHI ≥ 5) | AHI ≥ 5: 90.3% AHI ≥ 30: 70% Mean AHI: 47.8 (23.4) |
| Pedrosa 2011 [16] | 125 pts with resistant HT (43% men) | 52 (10) | 24 h ABPM SBP: 176 (31) DBP: 107 (19) | PSG (AHI ≥ 15) | AHI ≥ 15: 64% AHI ≥ 30: 32% Median AHI: 18 (interquartile range, 10–40) |
| Florczak 2013 [32] | 204 pts with resistant HT (123 men, 81 women) | 48.4 (10.6) | 24 h ABPM Daytime SBP: 145 (19), DBP: 90 (13) Nightime SBP: 132 (19), DBP: 79 (12) | PSG (AHI ≥ 5) | AHI ≥ 5: 72.1% AHI ≥ 30: 26.5% |
| Ruttanaumpawan 2009 [14] | 42 pts with resistant HT and 22 pts with controlled HT, matched for age, sex and BMI | 56.5 (1.6) in resistant HT group, 60.1 (1.8) in controlled HT group | 24 h ABPM in the resistant HT group SBP: 149 (2) DBP: 85 (1) | PSG (AHI ≥ 10) | 81% in the resistant HT group vs. 55% in the controlled HT group (p = 0.03) Mean AHI: 24.9 (3.2) in the resistant HT group vs. 16.5 (2.7) in the controlled HT group (p = 0.13) |
| Johnson 2019 [37] | 664 black participants with HT (205 men), of whom 96 (14.5%) had resistant HT | 64.9 (10.6) | Office BP measurement | RP (AHI ≥ 15) | 25.7% of all HT patients. Patients with resistant HT were 1.92 times more likely (95%CI 1.15–3.20) to have OSA, compared to those with controlled HT |
| Abdel-Kader 2012 [38] | 407 patients (229 men, 178 women), distributed in: 224 from general population without chronic kidney disease, 88 non-dialysis-dependent chronic kidney disease, and 95 with end-stage renal disease | 60.0 (7.2) for the non-chronic kidney disease, 52.2 (14) for the non-dialysis-dependent chronic kidney disease, and 53.8 (14.9) for the end-stage renal disease group | Office BP measurement Resistant HT was present in 4.9% of patients in the non-chronic kidney disease, 35.2% of the non-dialysis-dependent chronic kidney disease, and 22.1% of the end-stage renal disease group | PSG (AHI ≥ 30) | Resistant HT was associated with severe OSA in participants with end-stage renal disease (adjusted OR 7.1, 95%CI 2.2–23.2), but not in the non-chronic kidney disease (adjusted OR 3.5, 95%CI 0.8–15.4) or the non-dialysis-dependent chronic kidney disease groups (adjusted OR 1.2, 95%CI 0.4–3.7) |
| Bhandari 2016 [36] | Retrospective cohort study of 470,386 individuals from a health insurance database | 65 (11) | HT and Resistant HT were identified by ICD-9 specific diagnoses codes SBP: 139 (20) DBP: 75 (13) | Sleep apnoea was identified by ICD-9 specific diagnoses codes or by dispensation of positive pressure therapy | 9.6% in the resistant HT group vs. 6.8 in the non-resistant HT group (p < 0.01). Sleep apnoea was significantly more common in the resistant HT group compared to the non-resistant HT group (adjusted OR 1.16, 95%CI 1.12–1.19) |
| Martinez-Garcia 2018 * [15] | 229 pts with resistant HT (63% men). Of these, 42 (18.3%) had refractory HT | 58.3 (9.6) for the resistant HT group and 58.4 (8.5) for the refractory HT group | 24 h ABPM Resistant HT SBP: 141.6 (11.2) DBP: 82.2 (10) Refractory HT SBP: 152.4 (13.9) DBP: 85.6 (11.8) | RP (AHI ≥ 5) | AHI ≥ 5 Resistant HT: 89.3% Refractory HT: 100% (p = 0.027) AHI ≥ 30: Resistant HT: 48.6% Refractory HT: 64.3% (p = 0.044) |
| Studies | Randomisation (Complete Follow-Up) | Age | BMI | ESS | Anti-HT Drugs | SBP/DBP (mmHg) at Entry | BP Measure | AHI, Sleep Study | CPAP Use | Follow-Up |
|---|---|---|---|---|---|---|---|---|---|---|
| Lozano et al., 2010 [77] | 29 to CPAP 35 to control | 59.2 (9.9) | 30.8 (5) | 6.14 (3.3) | 3.48 (0.57) | 129.9 (13.7)/76 (10) | ABPM | 52.3 (21.5) Full PSG | 5.6 (1.52) | 3 months |
| Pedrosa et al., 2013 [83] | 19 to CPAP 16 to control | 56 (1) | 32 (28–39) | 10 (1) | 4 (4–5) | 162 (4)/97 (2) | ABPM | 29 (24–48) PSG | 6.01 (0.2) | 6 months |
| Martinez-Garcia et al., 2013 [17] | 98 to CPAP 96 to control | 56 (9.5) | 34.1 (5.4) | 9.1 (3.7) | 3.8 (0.9) | 144.2 (12.5)/83 (10.5) | ABPM | 40.4 (18.9) RP | 5 (1.9) | 3 months |
| De Oliveira et al., 2014 [78] | 24 to CPAP 23 to sham | 59.4 (7.7) | 29.8 (4.4) | 10 (6–15) | 4 (1) | 148 (17)/88 (13) | ABPM | 20 (18–1) RP | 5.3 (4.1–7.1) Median (IQR) | 8 weeks |
| Muxfeldt et al., 2015 [71] | 57 to CPAP 60 to control | 60.5 (8.2) | 33.4 (5.3) | 11 (6) | 5 (3–8) | 129 (16)/75 812) | ABPM | 41 (21) PSG | 4.8 (median) | 6 months |
| Navarro et al., 2019 * [80] | 23 to CPAP 19 to control | 61.1 (8.3) vs. 56.7 (9) | 34.9 (5.4) vs. 34.1 (6.8) | 9 (4) vs. 8.9 (3.8) | 5 (5–6) vs. 5 (5–6) (median, IQR) | 154.1 (12.2)/82.9 (14.2) 149(11.5)/84.1 (10.8) | ABPM | 42.7 (17.2) vs. 40.4 (20.3) RP | 5.2 (1.5) | 3 months |
| Treatment Strategy | Effect on BP | Effect on OSA |
|---|---|---|
| Lifestyle and diet measures | ||
| Sodium restriction | Decrease of BP (*) | Improvement of OSA severity |
| Weight reduction | Decrease of BP (*) | Improvement of OSA severity |
| Regular physical exercise | Decrease of BP | (*) |
| Specific diet (Mediterranean, DASH) | Decrease of BP (*) | (*) |
| Antihypertensive drugs | ||
| Diuretics | Decrease of BP | Improvement of OSA severity |
| Spironolactone | Decrease of BP (Fourth drug of choice) | Improvement of OSA severity |
| Other antihypertensive drugs | Decrease of BP | (*) |
| Invasive procedures | ||
| Renal ablation | Probable decrease of BP (*) | Improvement of OSA severity |
| OSA treatment (devices) | ||
| CPAP treatment | Decrease of BP especially in RfH | Control of the disease. |
| Other OSA treatments | Decrease of BP (*) | Control of the disease. |
| To understand the pathophysiological mechanisms that distinguish RH and RfH, and how OSA and CPAP treatment can influence them. |
| To assess the best combined therapeutic strategy in patients with RH/RfH and OSA. |
| To determine the added value of CPAP to the different antihypertensive treatments, including renal denervation. |
| To analyse the effect of long-term CPAP on blood pressure and cardiovascular events in patients with RH/RfH. |
| To determine the various biomarker predictors of a good BP response to CPAP. |
| To group homogeneous clinical phenotypes in terms of clinical presentation, prognosis and response to treatment. |
| To determine the role of confounders in the relationship between RH/RfH and OSA, particularly obesity. |
| The contrast in office BP measures and 24 h ABPM results makes it possible to define different phenotypes of RH, according to whether the monitoring results are congruent (controlled or sustained) or not (white coat or masked). At present, the predictive value of each phenotype with respect to the effect of CPAP is still unknown. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oscullo, G.; Torres, G.; Campos-Rodriguez, F.; Posadas, T.; Reina-González, A.; Sapiña-Beltrán, E.; Barbé, F.; Martinez-Garcia, M.A. Resistant/Refractory Hypertension and Sleep Apnoea: Current Knowledge and Future Challenges. J. Clin. Med. 2019, 8, 1872. https://doi.org/10.3390/jcm8111872
Oscullo G, Torres G, Campos-Rodriguez F, Posadas T, Reina-González A, Sapiña-Beltrán E, Barbé F, Martinez-Garcia MA. Resistant/Refractory Hypertension and Sleep Apnoea: Current Knowledge and Future Challenges. Journal of Clinical Medicine. 2019; 8(11):1872. https://doi.org/10.3390/jcm8111872
Chicago/Turabian StyleOscullo, Grace, Gerard Torres, Francisco Campos-Rodriguez, Tomás Posadas, Angela Reina-González, Esther Sapiña-Beltrán, Ferrán Barbé, and Miguel Angel Martinez-Garcia. 2019. "Resistant/Refractory Hypertension and Sleep Apnoea: Current Knowledge and Future Challenges" Journal of Clinical Medicine 8, no. 11: 1872. https://doi.org/10.3390/jcm8111872
APA StyleOscullo, G., Torres, G., Campos-Rodriguez, F., Posadas, T., Reina-González, A., Sapiña-Beltrán, E., Barbé, F., & Martinez-Garcia, M. A. (2019). Resistant/Refractory Hypertension and Sleep Apnoea: Current Knowledge and Future Challenges. Journal of Clinical Medicine, 8(11), 1872. https://doi.org/10.3390/jcm8111872





