Liver Grafts with Major Extended Donor Criteria May Expand the Organ Pool for Patients with Hepatocellular Carcinoma
Abstract
1. Introduction
2. Methods
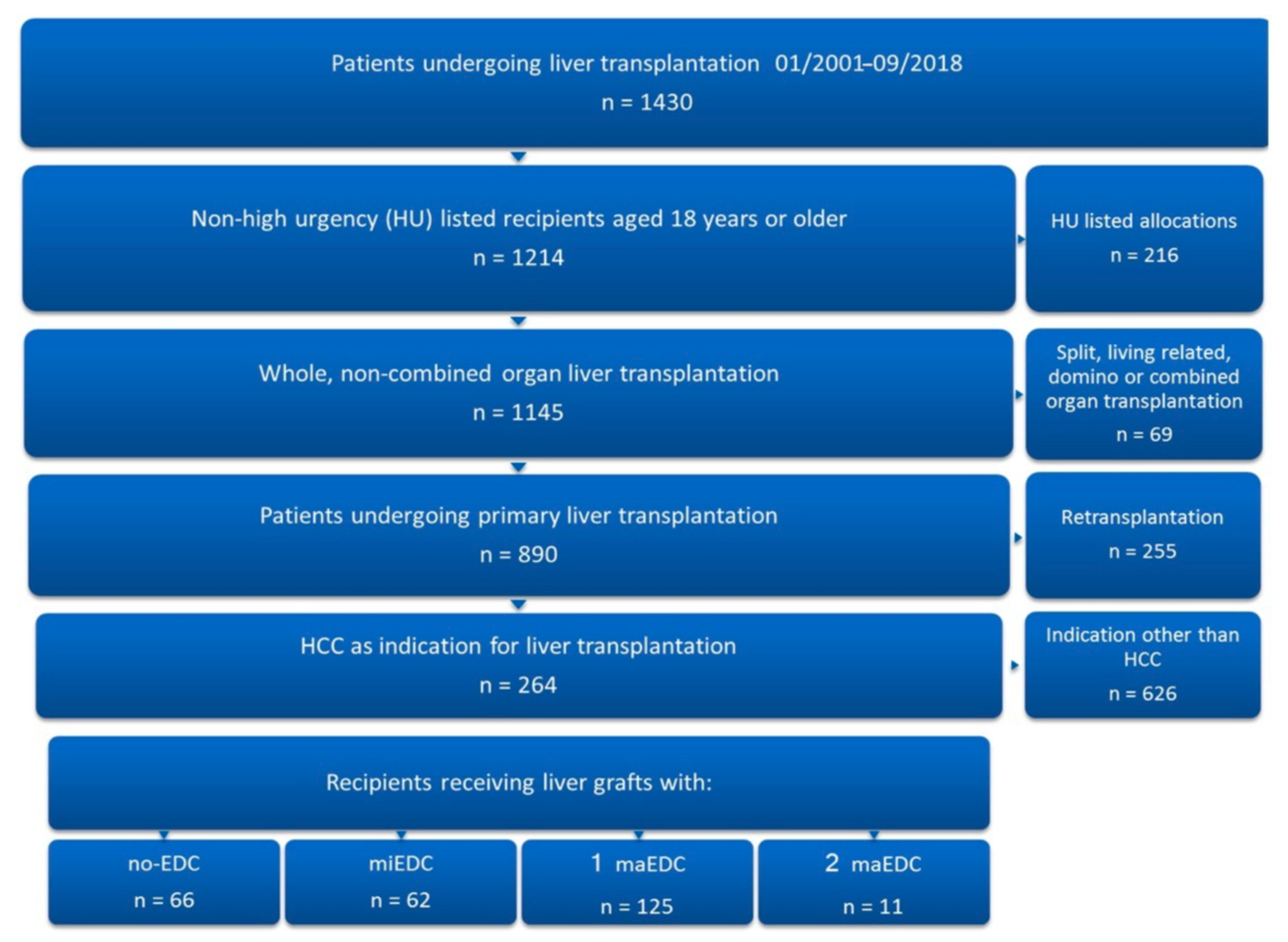
2.1. Patient Data Collection
2.2. Major EDC and Assessment of Clinical Outcome
2.3. Statistical Analysis
3. Results
3.1. Etiology of Liver Cirrhosis, Major EDC, and Comparison of No-maEDC and maEDC Groups
3.2. Outcome Following Liver Transplantation for HCC
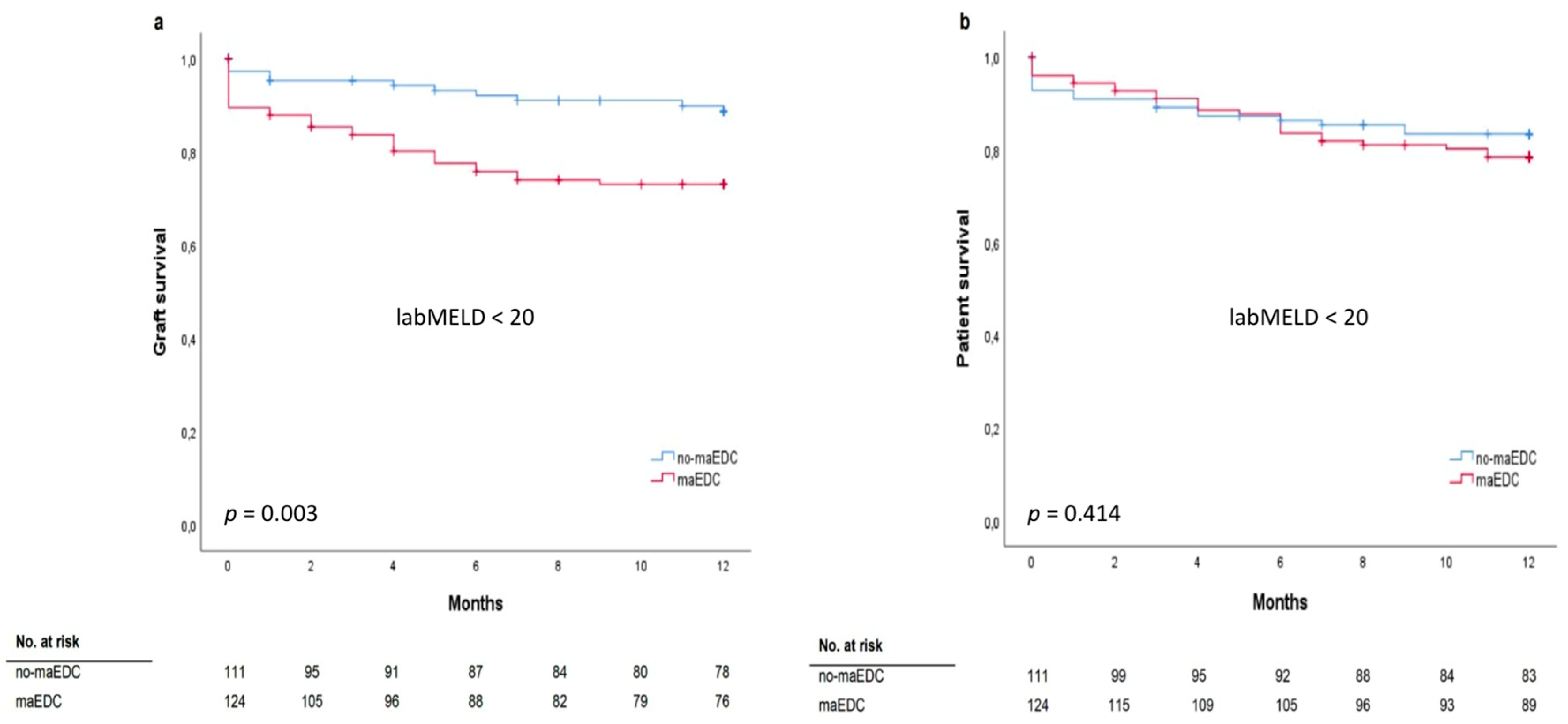
3.3. Graft Survival and Major EDC
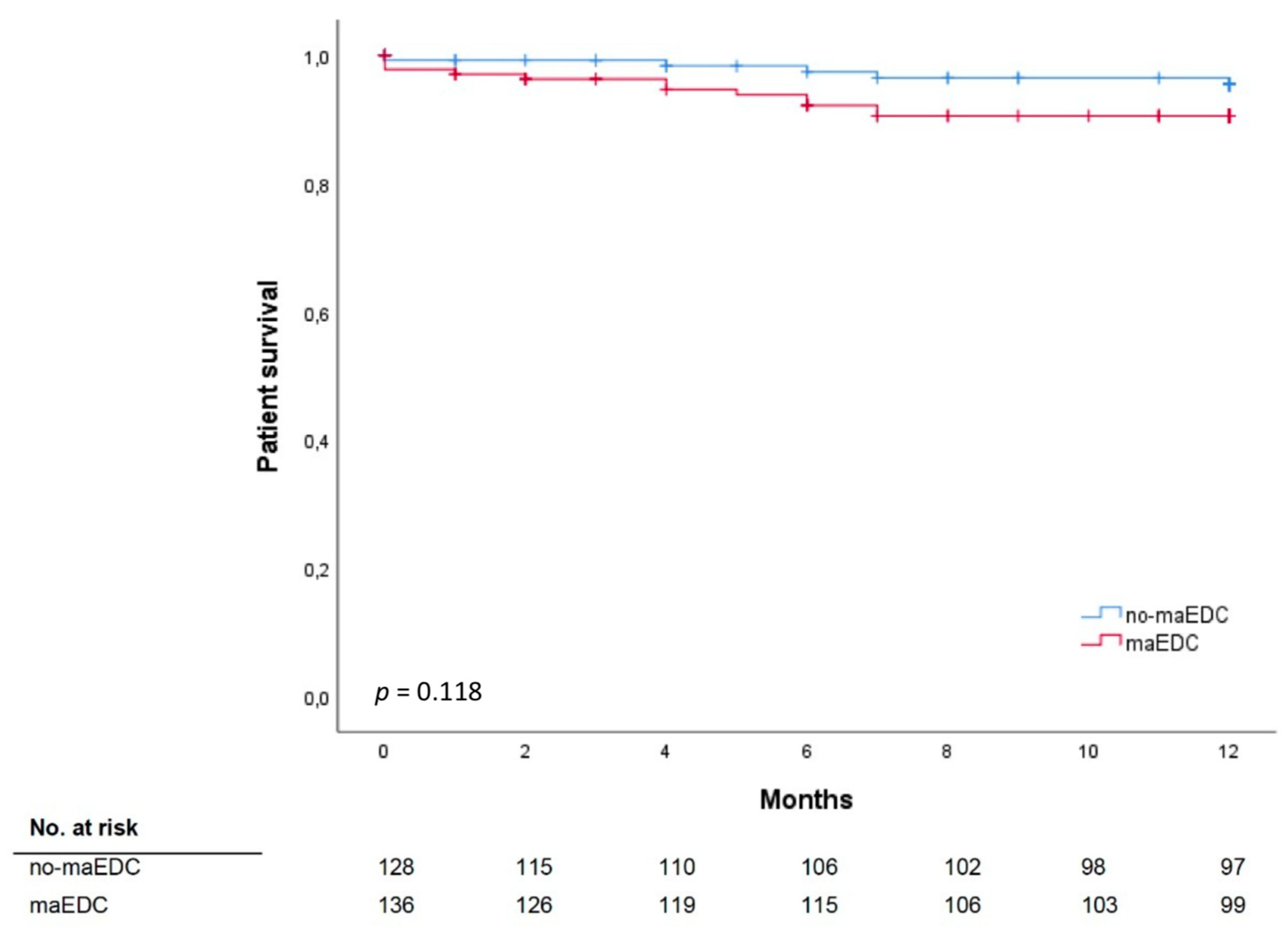
3.4. Patient Survival and Major EDC
3.5. Univariate and Multivariate Analyses
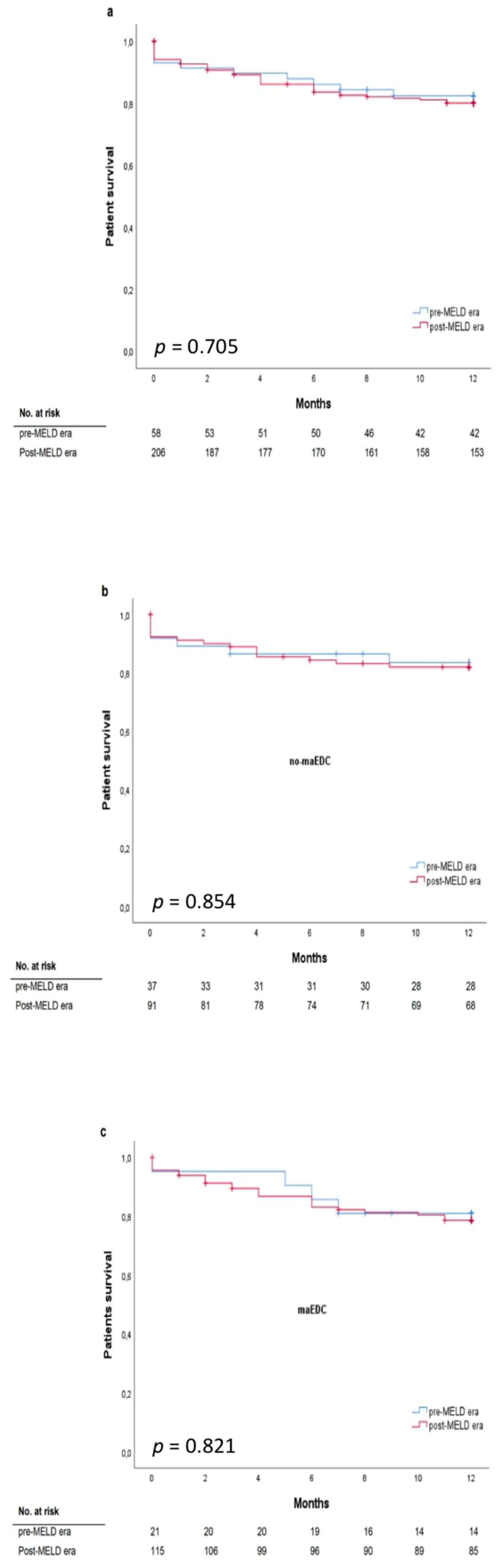
3.6. MELD Score and Major EDC
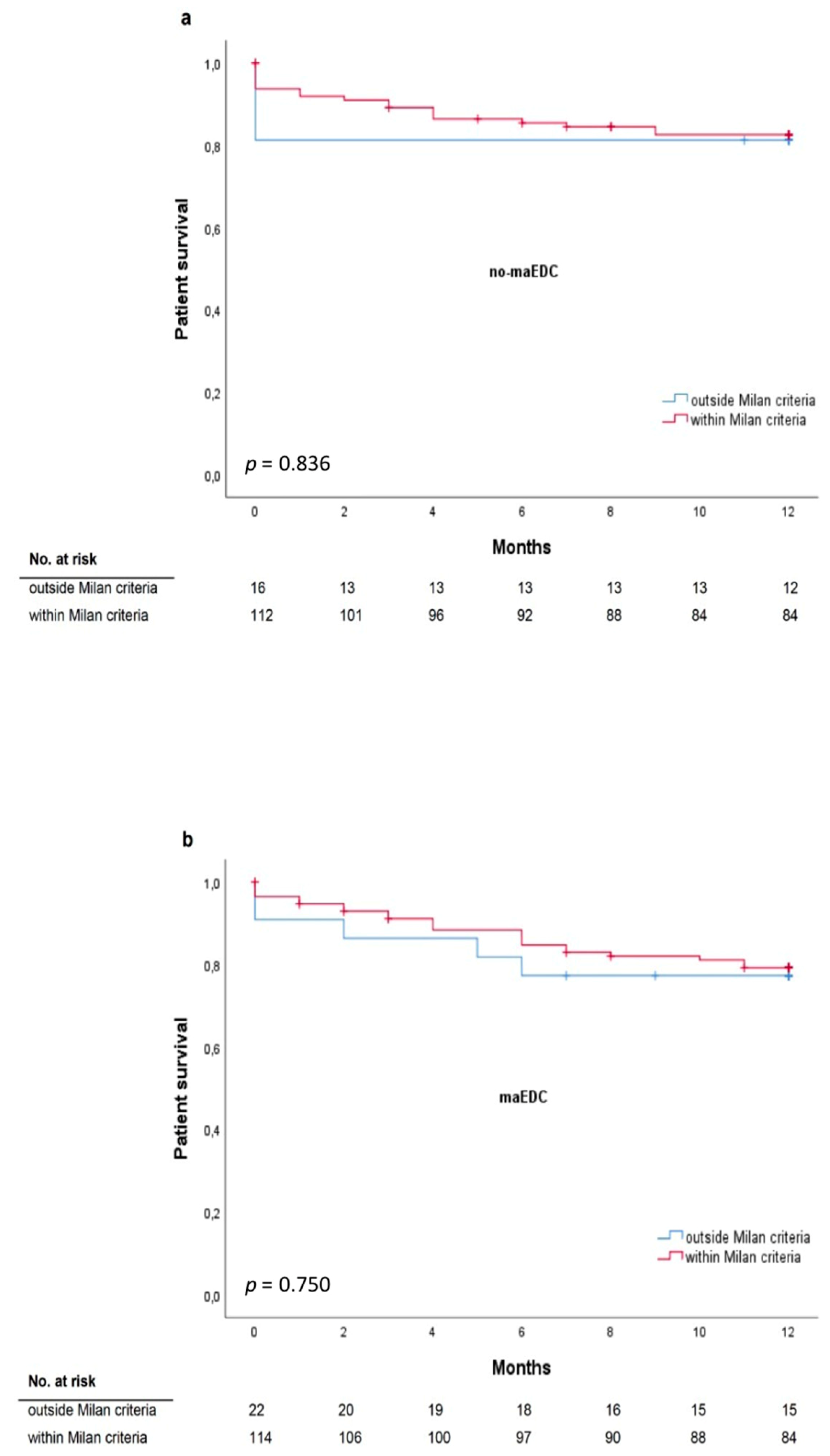
3.7. Milan Criteria and Major EDC
4. Discussion
4.1. Major EDC and Clinical Outcome After Liver Transplantation
4.2. Major EDC and the MELD Score
4.3. Milan and UCSF Criteria and Risk Factors for Increased Mortality after Liver Transplantation
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Lafaro, K.J.; Demirjian, A.N.; Pawlik, T.M. Epidemiology of Hepatocellular Carcinoma. Surg. Oncol. Clin. North Am. 2015, 24, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Pinna, A.D.; Yang, T.; Mazzaferro, V.; De Carlis, L.; Zhou, J.; Roayaie, S.; Shen, F.; Sposito, C.; Cescon, M.; Di Sandro, S.; et al. Liver Transplantation and Hepatic Resection can Achieve Cure for Hepatocellular Carcinoma. Ann. Surg. 2018, 268, 868–875. [Google Scholar] [CrossRef] [PubMed]
- Lozanovski, V.J.; Khajeh, E.; Fonouni, H.; Pfeiffenberger, J.; Von Haken, R.; Brenner, T.; Mieth, M.; Schirmacher, P.; Michalski, C.W.; Weiss, K.H.; et al. The impact of major extended donor criteria on graft failure and patient mortality after liver transplantation. Langenbeck’s Arch. Surg. 2018, 403, 719–731. [Google Scholar] [CrossRef] [PubMed]
- Sotiropoulos, G.C.; Paul, A.; Molmenti, E.; Lang, H.; Frilling, A.; Napieralski, B.P.; Nadalin, S.; Treckmann, J.; Brokalaki, E.I.; Gerling, T.; et al. Liver Transplantation for Hepatocellular Carcinoma in Cirrhosis within the Eurotransplant Area: An Additional Option with “Livers that Nobody Wants”. Transplant 2005, 80, 897–902. [Google Scholar] [CrossRef]
- Jochmans, I.; Pirenne, J.; Samuel, U.; Van Rosmalen, M. Adult Liver Allocation in Eurotransplant. Transplantation 2017, 101, 1542–1550. [Google Scholar] [CrossRef]
- Mehrabi, A.; Mood, Z.A.; Fonouni, H.; Kashfi, A.; Hillebrand, N.; Müller, S.A.; Encke, J.; Büchler, M.W.; Schmidt, J. A single-center experience of 500 liver transplants using the modified piggyback technique by Belghiti. Liver Transplant. 2009, 15, 466–474. [Google Scholar] [CrossRef]
- Braat, A.E.; Blok, J.J.; Putter, H.; Adam, R.; Burroughs, A.K.; Rahmel, A.O.; Porte, R.J.; Rogiers, X.; Ringers, J.; European Liver and Intestine Transplant Association (ELITA); et al. The Eurotransplant Donor Risk Index in Liver Transplantation: ET-DRI. Arab. Archaeol. Epigr. 2012, 12, 2789–2796. [Google Scholar]
- Der Bundesärztekammer, B. Richtlinien zur Organtransplantation gem. § 16 TPG. Dtsch Arztebl 2008, 105, 1461–1464. Available online: https://www.bundesaerztekammer.de/fileadmin/user_upload/downloads/pdf-Ordner/RL/RiliOrgaWlOvLeberTx20190924.pdf (accessed on 21 January 2019).
- Schemmer, P.; Nickkholgh, A.; Hinz, U.; Gerling, T.; Mehrabi, A.; Sauer, P.; Encke, J.; Friess, H.; Weitz, J.; Büchler, M. Extended donor criteria have no negative impact on early outcome after liver transplantation: A single-center multivariate analysis. Transpl. Proc. 2007, 39, 529–534. [Google Scholar] [CrossRef]
- Klempnauer, J.; Schrem, H.; Reichert, B.; Frühauf, N.; Becker, T.; Lehner, F.; Kleine, M.; Bektas, H.; Zachau, L. The Donor-Risk-Index, ECD-Score and D-MELD-Score all fail to predict short-term outcome after liver transplantation with acceptable sensitivity and specificity. Ann. Transplant. 2012, 17, 5–13. [Google Scholar] [CrossRef]
- Bruns, H.; Lozanovski, V.J.; Schultze, D.; Hillebrand, N.; Hinz, U.; Büchler, M.W.; Schemmer, P. Prediction of Postoperative Mortality in Liver Transplantation in the Era of MELD-Based Liver Allocation: A Multivariate Analysis. PLoS ONE 2014, 9, e98782. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.D.; Croome, K.P.; Shalev, J.A.; Musto, K.R.; Sharma, M.; Keaveny, A.P.; Taner, C.B. Early allograft dysfunction after liver transplantation: An intermediate outcome measure for targeted improvements. Ann. Hepatol. 2016, 15, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Goldberg, D.; French, B.; Newcomb, C.; Liu, Q.; Sahota, G.; Wallace, A.E.; Forde, K.A.; Lewis, J.D.; Halpern, S.D. Patients With Hepatocellular Carcinoma Have Highest Rates of Wait-listing for Liver Transplantation Among Patients With End-Stage Liver Disease. Clin. Gastroenterol. Hepatol. 2016, 14, 1638–1646.e2. [Google Scholar] [CrossRef] [PubMed]
- Rahbari, N.N.; Mehrabi, A.; Mollberg, N.M.; Müller, S.A.; Koch, M.; Büchler, M.W.; Weitz, J. Hepatocellular carcinoma: Current management and perspectives for the future. Ann. Surg. 2011, 253, 453–469. [Google Scholar] [CrossRef]
- Yao, F.; Bass, N.M.; Nikolai, B.; Merriman, R.; Davern, T.J.; Kerlan, R.; Ascher, N.L.; Roberts, J.P. A follow-up analysis of the pattern and predictors of dropout from the waiting list for liver transplantation in patients with hepatocellular carcinoma: Implications for the current organ allocation policy. Liver Transplant. 2003, 9, 684–692. [Google Scholar] [CrossRef]
- Adam, R.; Karam, V.; Cailliez, V.; O Grady, J.G.; Mirza, D.; Cherqui, D.; Klempnauer, J.; Salizzoni, M.; Pratschke, J.; Jamieson, N.; et al. 2018 Annual Report of the European Liver Transplant Registry (ELTR)—50-year evolution of liver transplantation. Transpl. Int. 2018, 31, 1293–1317. [Google Scholar] [CrossRef]
- Kim, W.; Lake, J.; Smith, J.; Schladt, D.; Skeans, M.; Noreen, S.; Robinson, A.; Miller, E.; Snyder, J.; Israni, A. OPTN/SRTR 2017 Annual Data Report: Liver. Am. J. Transplant. 2019, 19, 184–283. [Google Scholar] [CrossRef]
- Mittal, S.; El-Serag, H.B. Epidemiology of HCC: Consider the population. J. Clin. Gastroenterol. 2013, 47, S2–S6. [Google Scholar] [CrossRef]
- Statistics Report Library. Available online: http://statistics.eurotransplant.org/index.php?search_type=&search_organ=liver&search_region=by+country&search_period=2018&search_characteristic=&search_text (accessed on 15 January 2019).
- Adler, M.; De Pauw, F.; Vereerstraeten, P.; Fancello, A.; Lerut, J.; Starkel, P.; Van Vlierberghe, H.; Troisi, R.; Donckier, V.; Detry, O.; et al. Outcome of patients with hepatocellular carcinoma listed for liver transplantation within the Eurotransplant allocation system. Liver Transpl. 2008, 14, 526–533. [Google Scholar] [CrossRef]
- De Boer, J.D.; Blok, J.J.; Putter, H.; Koopman, J.J.; van Hoek, B.; Samuel, U.; van Rosmalen, M.; Metselaar, H.J.; Alwayn, I.P.; Guba, M.; et al. Optimizing the use of geriatric livers for transplantation in the Eurotransplant region. Liver Transpl. 2019, 25, 260–274. [Google Scholar] [CrossRef]
- Goldaracena, N.; Gorgen, A.; Doyle, A.; Hansen, B.E.; Tomiyama, K.; Zhang, W.; Ghanekar, A.; Lilly, L.; Cattral, M.; Galvin, Z.; et al. Live donor liver transplantation for patients with hepatocellular carcinoma offers increased survival vs. deceased donation. J. Hepatol. 2019, 70, 666–673. [Google Scholar] [CrossRef] [PubMed]
- Shan, Y.; Huang, L.; Xia, Q. Salvage Liver Transplantation Leads to Poorer Outcome in Hepatocellular Carcinoma Compared with Primary Liver Transplantation. Sci. Rep. 2017, 7, 44652. [Google Scholar] [CrossRef] [PubMed]
- Merion, R.; Wolfe, R.A.; Dykstra, D.M.; Leichtman, A.B.; Gillespie, B.; Held, P.J. Longitudinal assessment of mortality risk among candidates for liver transplantation. Liver Transpl. 2003, 9, 12–18. [Google Scholar] [CrossRef] [PubMed]
- Nocito, A.; El-Badry, A.M.; Clavien, P.-A. When is steatosis too much for transplantation? J. Hepatol. 2006, 45, 494–499. [Google Scholar] [CrossRef]
- Linares, I.; Hamar, M.; Selzner, N.; Selzner, M. Steatosis in Liver Transplantation: Current Limitations and Future Strategies. Transplantation 2019, 103, 78–90. [Google Scholar] [CrossRef]
- Dutkowski, P.; Schlegel, A.; Slankamenac, K.; Oberkofler, C.E.; Adam, R.; Burroughs, A.K.; Schadde, E.; Müllhaupt, B.; Clavien, P.-A. The use of fatty liver grafts in modern allocation systems: Risk assessment by the balance of risk (BAR) score. Ann. Surg. 2012, 256, 861–868. [Google Scholar] [CrossRef]
- Pratschke, S.; Bender, A.; Boesch, F.; Andrassy, J.; Van Rosmalen, M.; Samuel, U.; Rogiers, X.; Meiser, B.; Küchenhoff, H.; Driesslein, D.; et al. Association between donor age and risk of graft failure after liver transplantation: An analysis of the Eurotransplant database. Transpl. Int. 2018, 32, 270–279. [Google Scholar] [CrossRef]
- Durand, F.; Levitsky, J.; Cauchy, F.; Gilgenkrantz, H.; Soubrane, O.; Francoz, C. Age and liver transplantation. J. Hepatol. 2019, 70, 745–758. [Google Scholar] [CrossRef]
- Feng, S.; Lai, J.C. Expanded criteria donors. Clin. Liver Dis. 2014, 18, 633–649. [Google Scholar] [CrossRef]
- Haugen, C.E.; Bowring, M.G.; Holscher, C.M.; Jackson, K.R.; Garonzik-Wang, J.; Cameron, A.M.; Philosophe, B.; McAdams-DeMarco, M.; Segev, D.L. Survival Benefit of Accepting Livers from Deceased Donors over 70 Years Old. Am. J. Transpl. 2019, 19, 2020–2028. [Google Scholar] [CrossRef]
- Halazun, K.; Rana, A.; Fortune, B.; Quillin III, R.; Verna, E.; Samstein, B.; Guarrera, J.; Kato, T.; Griesemer, A.; Fox, A. No country for old livers? Examining and optimizing the utilization of elderly liver grafts. Am. J. Transpl. 2018, 18, 669–678. [Google Scholar] [CrossRef] [PubMed]
- Paterno, F.; Wima, K.; Hoehn, R.S.; Cuffy, M.C.; Diwan, T.S.; Woodle, S.E.; Abbott, D.E.; Shah, S.A. Use of Elderly Allografts in Liver Transplantation. Transplantation 2016, 100, 153–158. [Google Scholar] [CrossRef] [PubMed]
- Lake, J.R.; Shorr, J.S.; Steffen, B.J.; Chu, A.H.; Gordon, R.D.; Wiesner, R.H. Differential Effects of Donor Age in Liver Transplant Recipients Infected With Hepatitis B, Hepatitis C and Without Viral Hepatitis. Arab. Archaeol. Epigr. 2005, 5, 549–557. [Google Scholar] [CrossRef] [PubMed]
- Rupp, C.; Hippchen, T.; Neuberger, M.; Sauer, P.; Pfeiffenberger, J.; Stremmel, W.; Gotthardt, D.N.; Mehrabi, A.; Weiss, K.-H. Successful combination of direct antiviral agents in liver-transplanted patients with recurrent hepatitis C virus. World J. Gastroenterol. 2018, 24, 1353–1360. [Google Scholar] [CrossRef] [PubMed]
- Gao, Q.; Mulvihill, M.S.; Scheuermann, U.; Davis, R.P.; Yerxa, J.; Yerokun, B.A.; Hartwig, M.G.; Sudan, D.L.; Knechtle, S.J.; Barbas, A.S. Improvement in Liver Transplant Outcomes From Older Donors: A US National Analysis. Ann. Surg. 2019, 270, 333–339. [Google Scholar] [CrossRef] [PubMed]
- Nagai, S.; Yoshida, A.; Facciuto, M.; Moonka, D.; Abouljoud, M.S.; Schwartz, M.E.; Florman, S.S. Ischemia time impacts recurrence of hepatocellular carcinoma after liver transplantation. Hepatology 2015, 61, 895–904. [Google Scholar] [CrossRef] [PubMed]
- Oweira, H.; Lahdou, I.; Daniel, V.; Opelz, G.; Schmidt, J.; Zidan, A.; Mehrabi, A.; Sadeghi, M. Early post-operative acute phase response in patients with early graft dysfunction is predictive of 6-month and 12-month mortality in liver transplant recipients. Hum. Immunol. 2016, 77, 952–960. [Google Scholar] [CrossRef]
- Kern, B.; Feurstein, B.; Fritz, J.; Fabritius, C.; Sucher, R.; Graziadei, I.; Bale, R.; Tilg, H.; Zoller, H.; Newsome, P.; et al. High incidence of hepatocellular carcinoma and postoperative complications in patients with nonalcoholic steatohepatitis as a primary indication for deceased liver transplantation. Eur. J. Gastroenterol. Hepatol. 2019, 31, 205–210. [Google Scholar] [CrossRef]
- Selck, F.W.; Grossman, E.B.; Ratner, L.E.; Renz, J.F. Utilization, outcomes, and retransplantation of liver allografts from donation after cardiac death: Implications for further expansion of the deceased-donor pool. Ann. Surg. 2008, 248, 599–607. [Google Scholar] [CrossRef]
- Moosburner, S.; Ritschl, P.; Wiering, L.; Gassner, J.; Öllinger, R.; Pratschke, J.; Sauer, I.; Raschzok, N. High donor age for liver transplantation: Tackling organ scarcity in Germany. Chirurg 2019, 90, 744–751. [Google Scholar] [CrossRef]
- Mehta, N.; Heimbach, J.; Lee, D.; Dodge, J.L.; Harnois, D.; Burns, J.; Sanchez, W.; Roberts, J.P.; Yao, F.Y. Wait time of less than 6 and greater than 18 months predicts hepatocellular carcinoma recurrence after liver transplantation: Proposing a wait time “Sweet Spot”. Transplantation 2017, 101, 2071–2078. [Google Scholar] [CrossRef] [PubMed]
- De Carlis, L.; Di Sandro, S.; Centonze, L.; Lauterio, A.; Buscemi, V.; De Carlis, R.; Ferla, F.; Sguinzi, R.; Okolicsanyi, S.; Belli, L.; et al. Liver-allocation policies for patients affected by HCC in Europe. Curr. Transplant. Rep. 2016, 3, 313–318. [Google Scholar] [CrossRef] [PubMed]
- Regalia, E.; Doci, R.; Andreola, S.; Montalto, F.; Ammatuna, M.; Mazzaferro, V.M.; Pulvirenti, A.; Bozzetti, F.; Morabito, A.; Gennari, L. Liver Transplantation for the Treatment of Small Hepatocellular Carcinomas in Patients with Cirrhosis. New Engl. J. Med. 1996, 334, 693–700. [Google Scholar]
- Mazzaferro, V.M.; Llovet, J.M.; Miceli, R.; Bhoori, S.; Schiavo, M.; Mariani, L.; Camerini, T.; Roayaie, S.; Schwartz, M.E.; Grazi, G.L.; et al. Predicting survival after liver transplantation in patients with hepatocellular carcinoma beyond the Milan criteria: A retrospective, exploratory analysis. Lancet Oncol. 2009, 10, 35–43. [Google Scholar] [CrossRef]
- Jin, B.; Liu, C.Z.; Hu, S.Y.; Wang, T.T.; Wang, L.; Jiang, X.S.; Frezza, E.E. Influence of estrogen and androgen on the outcome of liver transplantation. Hepatogastroenterology 2008, 55, 207–211. [Google Scholar]
- Brooks, B.K.; Levy, M.F.; Jennings, L.W.; Abbasoglu, O.; Vodapally, M.; Goldstein, R.M.; Husberg, B.S.; Gonwa, T.A.; Klintmalm, G.B. Influence of Donor and Recipient Gender on the Outcome of Liver Transplantation12. Transplantation 1996, 62, 1784–1787. [Google Scholar] [CrossRef]
- Reese, P.P.; Sonawane, S.B.; Thomasson, A.; Yeh, H.; Markmann, J.F. Donor Age and Cold Ischemia Interact to Produce Inferior 90-Day Liver Allograft Survival. Transplantation 2008, 85, 1737–1744. [Google Scholar] [CrossRef]
| Total n = 264 | maEDC = 0 n = 128 | maEDC ≥ 1 n = 136 | p | |
|---|---|---|---|---|
| Donor, n = 264 | ||||
| BPS > 40% | 14 (5.3%) | 0% 50 ± 13 | 10.3% 71 ± 12 | <0.001 <0.001 |
| Age (years, mean ± SD) | 61 ± 16 | |||
| >65 years | 59 (39.6%) | |||
| CIT (hours, mean ± SD) | 10.2 ± 2.53 | 9.7 ± 2.2 | 10.8 ± 2.8 | <0.001 0.303 |
| >14 h | 15 (10.1%) | |||
| Gender | ||||
| Male | 58% | 61% | 55% | |
| Female | 42% | 39% | 45% | |
| BMI (kg/m2, mean ± SD) | 26.4 ± 3.7 | 26.1 ± 3.6 | 26.7 ± 3.8 | 0.196 |
| ICU/MV (days, mean ± SD) | 4.2 ± 4.1 | 4.4 ± 4 | 4.0 ± 4.1 | 0.436 |
| Recipient, n = 264 | ||||
| Age (years, mean ± SD) | 57 ± 7 | 57 ± 7 | 56 ± 7 | 0.318 |
| Gender | 0.84 | |||
| Male | 86% | 85% | 86% | |
| Female | 14% | 15% | 14% | |
| BMI (kg/m2, mean ± SD) | 27.4 ± 4.3 | 27.7 ± 4.1 | 27.1 ± 4.4 | 0.201 |
| labMELD score (mean ± SD) | 12.45 ± 6.2 | 12.75 ± 6.8 | 12.2 ± 5.5 | 0.444 0.247 |
| <20 | 89% | 87% | 91% | |
| ≥20 | 11% | 13% | 9% | |
| eMELD | 27.24 ± 4.2 | 28.4 ± 4.8 | 26.4 ± 3.6 | 0.004 |
| MatchMELD | 23.2 ± 8.4 | 22.4 ± 9.7 | 23.9 ± 6.9 | 0.137 |
| Recipients meeting Milan criteria * | 85.6% | 87.5% | 83.8% | 0.395 |
| Time on the waiting list (days ± SD) | 375 ± 528 | 314 ± 303 | 435 ± 678 | 0.062 |
| Major morbidity (Clavien–Dindo cl. ≥ IIIb) | 35.2% | 40.4% | 45.6% | 0.068 |
| 1-year graft survival | 82.1% | 89.5% | 75.3% | 0.003 |
| 1-year patient survival (liver failure ass.) | 92.9% | 95.5% | 90.5% | 0.118 |
| 1-year patient mortality | 18.9% | 17.2% | 20.6% | 0.531 |
| Etiology of liver cirrhosis | ||||
| Hepatitis | 139 (52.6%) | 70 (54.7%) | 69 (50.7%) | 0.539 |
| HBV-induced cirrhosis | 37 (14%) | 18 (14.1%) | 19 (14%) | |
| HCV-induced cirrhosis | 102 (38.6%) | 52 (40.6%) | 50 (36.8%) | |
| Alcohol-induced cirrhosis | 90 (34.1%) | 45 (35.2%) | 45 (33.1%) | 0.795 |
| Cryptogenic liver cirrhosis | 22 (8.3%) | 10 (7.8%) | 12 (8.8%) | 0.826 |
| Autoimmune hepatitis | 5 (1.9%) | 1 (0.8%) | 4 (2.9%) | 0.371 |
| Nonalcoholic steatohepatitis | 3 (1.1%) | 1 (0.8%) | 2 (1.5%) | 0.999 |
| Hemochromatosis | 2 (0.8%) | 1 (0.8%) | 1 (0.7%) | 0.999 |
| Biliary atresia | 1 (0.4%) | 0 (0%) | 1 (0.7%) | 0.999 |
| Inflammatory adenoma HCC | 1 (0.4%) | 0 (0%) | 1 (0.7%) | 0.999 |
| Primary biliary cholangitis | 1 (0.4%) | 0 (0%) | 1 (0.7%) | 0.999 |
| no-EDC (n = 66) | miEDC (n = 62) | maEDC = 1 (n = 125) | maEDC = 2 (n = 11) | p | |
|---|---|---|---|---|---|
| EAD, n (%) | 29 (43.9) | 37 (59.7) | 61 (48.8) | 7 (63.6) | 0.249 |
| PNF, n (%) | 1 (1.5) | 1 (1.6) | 9 (7.2) | 1 (9.1) | 0.159 |
| 30-day graft loss, n (%) | 1 (1.5) | 3 (4.8) | 11 (8.8) | 2 (18.2) | 0.086 |
| 90-day graft loss, n (%) | 2 (3.0) | 4 (6.5) | 15 (12.0) | 3 (27.3) | 0.028 |
| no-EDC (n = 66) | miEDC (n = 62) | maEDC = 1 (n = 125) | maEDC = 2 (n = 11) | p | |
|---|---|---|---|---|---|
| 30-day mortality, n (%) | 6 (9.1) | 4 (6.5) | 5 (4.0) | 1 (9.1) | 0.534 |
| 90-day mortality, n (%) | 8 (12.1) | 5 (8.1) | 10 (8.0) | 1 (9.1) | 0.803 |
| 1-year mortality, n (%) | 13 (19.7) | 9 (14.5) | 25 (20.0) | 3 (27.3) | 0.705 |
| no-EDC (n = 66) | miEDC (n = 62) | maEDC = 1 (n = 125) | maEDC = 2 (n = 11) | p | |
|---|---|---|---|---|---|
| 30-day mortality, n (%) | 0 (0) | 1 (1.6) | 2 (1.6) | 1 (9.1) | 0.154 |
| 90-day mortality, n (%) | 0 (0) | 1 (1.6) | 4 (3.2) | 1 (9.1) | 0.217 |
| 1-year mortality, n (%) | 4 (6.1) | 1 (1.6) | 11 (8.8) | 1 (9.1) | 0.296 |
| Univariate Analysis | Multivariate Analysis | |||||
|---|---|---|---|---|---|---|
| HR | 95% CI | p | HR | 95% CI | p | |
| Recipient age | 1.001 | 0.959–1.044 | 0.971 | |||
| Recipient female gender | 1.587 | 0.763–3.301 | 0.217 | |||
| maEDC | 2.605 | 1.342–5.059 | 0.005 | 2.603 | 1.340–5.055 | 0.005 |
| HCC grade G3 | 1.075 | 0.424–2.727 | 0.879 | |||
| Recipient BMI > 30 kg/m2 | 0.780 | 0.373–1.630 | 0.509 | |||
| labMELD | 0.919 | 0.851–0.991 | 0.029 | 0.915 | 0.846–0.991 | 0.029 |
| Underlying disease (cause of LC) | ||||||
| Other * | baseline | baseline | baseline | |||
| Hepatitis | 0.704 | 0.237–2.091 | 0.527 | |||
| Alcohol | 0.875 | 0.468–1.639 | 0.677 | |||
| Recipients meeting Milan criteria | 1.580 | 0.759–3.287 | 0.221 | |||
| LC Child-Pugh score (B/C vs. A) | 0.783 | 0.429–1.428 | 0.425 | |||
| Univariate Analysis | Multivariate Analysis | |||||
|---|---|---|---|---|---|---|
| HR | 95% CI | p | HR | 95% CI | p | |
| Recipient age | 0.992 | 0.926–1.064 | 0.827 | |||
| Recipient female gender | 3.267 | 1.208–8.836 | 0.02 | 3.671 | 1.4–13.231 | 0.011 |
| maEDC | 2.266 | 0.798–6.434 | 0.124 | 2.556 | 0.872–8.281 | 0.081 |
| HCC grade G3 | 1.031 | 0.236–4.507 | 0.968 | |||
| Recipient BMI > 30 kg/m2 | 0.876 | 0.286–2.686 | 0.817 | |||
| labMELD | 0.92 | 0.814–1.038 | 0.176 | 0.916 | 0.813–1.031 | 0.145 |
| Underlying disease (cause of LC) | ||||||
| Other * | baseline | baseline | baseline | |||
| Hepatitis | 1.563 | 0.192–12.706 | 0.654 | |||
| Alcohol | 3.027 | 0.383–23.893 | 0.293 | |||
| Recipients meeting Milan criteria | 1.922 | 0.627–5.895 | 0.253 | |||
| LC Child-Pugh score (B/C vs. A) | 0.618 | 0.228–1.671 | 0.343 | |||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lozanovski, V.J.; Kerr, L.T.B.; Khajeh, E.; Ghamarnejad, O.; Pfeiffenberger, J.; Hoffmann, K.; Chang, D.-H.; Mieth, M.; Longerich, T.; Strobel, O.; et al. Liver Grafts with Major Extended Donor Criteria May Expand the Organ Pool for Patients with Hepatocellular Carcinoma. J. Clin. Med. 2019, 8, 1692. https://doi.org/10.3390/jcm8101692
Lozanovski VJ, Kerr LTB, Khajeh E, Ghamarnejad O, Pfeiffenberger J, Hoffmann K, Chang D-H, Mieth M, Longerich T, Strobel O, et al. Liver Grafts with Major Extended Donor Criteria May Expand the Organ Pool for Patients with Hepatocellular Carcinoma. Journal of Clinical Medicine. 2019; 8(10):1692. https://doi.org/10.3390/jcm8101692
Chicago/Turabian StyleLozanovski, Vladimir J., Larissa T.B. Kerr, Elias Khajeh, Omid Ghamarnejad, Jan Pfeiffenberger, Katrin Hoffmann, De-Hua Chang, Markus Mieth, Thomas Longerich, Oliver Strobel, and et al. 2019. "Liver Grafts with Major Extended Donor Criteria May Expand the Organ Pool for Patients with Hepatocellular Carcinoma" Journal of Clinical Medicine 8, no. 10: 1692. https://doi.org/10.3390/jcm8101692
APA StyleLozanovski, V. J., Kerr, L. T. B., Khajeh, E., Ghamarnejad, O., Pfeiffenberger, J., Hoffmann, K., Chang, D.-H., Mieth, M., Longerich, T., Strobel, O., Weiss, K. H., Büchler, M. W., & Mehrabi, A. (2019). Liver Grafts with Major Extended Donor Criteria May Expand the Organ Pool for Patients with Hepatocellular Carcinoma. Journal of Clinical Medicine, 8(10), 1692. https://doi.org/10.3390/jcm8101692





