Cardiovascular Comorbidities in Chronic Obstructive Pulmonary Disease (COPD)—Current Considerations for Clinical Practice
Abstract
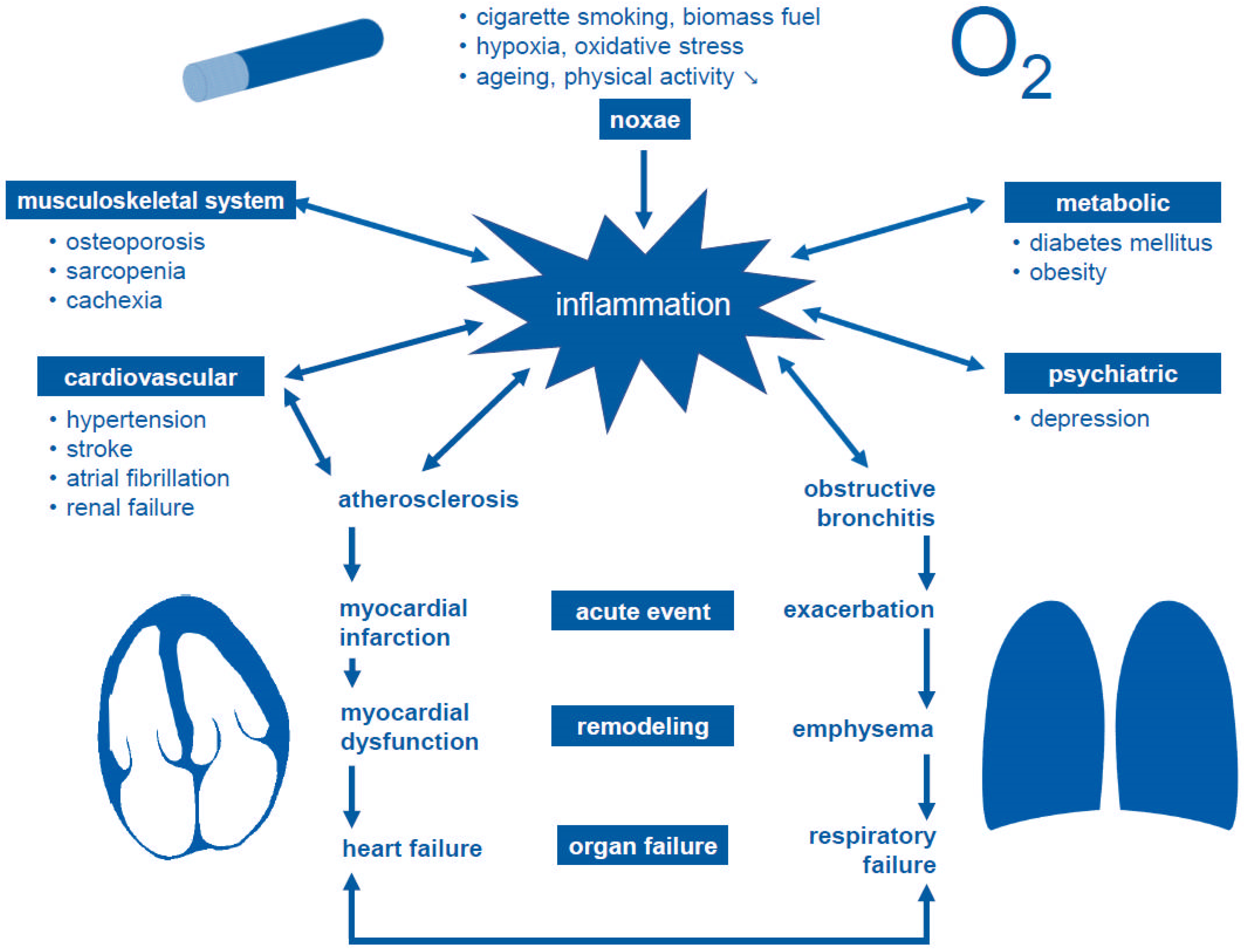
1. Introduction
2. Cardiovascular Risk
3. Coronary Heart Disease
4. Heart Failure
5. Arterial Hypertension
6. Pulmonary Hypertension
7. Cardiac Arrhythmias
8. Summary
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ukena, C.; Mahfoud, F.; Kindermann, M.; Kindermann, I.; Bals, R.; Voors, A.A.; van Veldhuisen, D.J.; Bohm, M. The cardiopulmonary continuum systemic inflammation as ‘common soil’ of heart and lung disease. Int. J. Cardiol. 2010, 145, 172–176. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.P.; Martinez, C.H. Chronic obstructive pulmonary disease comorbidities. Curr. Opin. Pulm. Med. 2016, 22, 113–118. [Google Scholar] [CrossRef] [PubMed]
- Camiciottoli, G.; Bigazzi, F.; Magni, C.; Bonti, V.; Diciotti, S.; Bartolucci, M.; Mascalchi, M.; Pistolesi, M. Prevalence of comorbidities according to predominant phenotype and severity of chronic obstructive pulmonary disease. Int. J. Chronic Obstruct. Pulm. Dis. 2016, 11, 2229–2236. [Google Scholar] [CrossRef] [PubMed]
- Boschetto, P.; Beghe, B.; Fabbri, L.M.; Ceconi, C. Link between chronic obstructive pulmonary disease and coronary artery disease: Implication for clinical practice. Respirology 2012, 17, 422–431. [Google Scholar] [CrossRef] [PubMed]
- Huiart, L.; Ernst, P.; Suissa, S. Cardiovascular morbidity and mortality in COPD. Chest 2005, 128, 2640–2646. [Google Scholar] [CrossRef] [PubMed]
- Curkendall, S.M.; Lanes, S.; de Luise, C.; Stang, M.R.; Jones, J.K.; She, D.; Goehring, E., Jr. Chronic obstructive pulmonary disease severity and cardiovascular outcomes. Eur. J. Epidemiol. 2006, 21, 803–813. [Google Scholar] [CrossRef] [PubMed]
- Gan, W.Q.; Man, S.F.; Senthilselvan, A.; Sin, D.D. Association between chronic obstructive pulmonary disease and systemic inflammation: A systematic review and a meta-analysis. Thorax 2004, 59, 574–580. [Google Scholar] [CrossRef]
- Global Strategy for the Diagnosis, Management and Prevention of COPD, Global Initiative for Chronic Obstructive Lung Disease (GOLD). 2019. Available online: https://goldcopd.org (accessed on 15 December 2018).
- Price, D.B.; Buhl, R.; Chan, A.; Freeman, D.; Gardener, E.; Godley, C.; Gruffydd-Jones, K.; McGarvey, L.; Ohta, K.; Ryan, D.; et al. Fractional exhaled nitric oxide as a predictor of response to inhaled corticosteroids in patients with non-specific respiratory symptoms and insignificant bronchodilator reversibility: A randomised controlled trial. Lancet Respir. Med. 2018, 6, 29–39. [Google Scholar] [CrossRef]
- Vestbo, J.; Anderson, J.A.; Brook, R.D.; Calverley, P.M.; Celli, B.R.; Crim, C.; Martinez, F.; Yates, J.; Newby, D.E.; Investigators, S. Fluticasone furoate and vilanterol and survival in chronic obstructive pulmonary disease with heightened cardiovascular risk (SUMMIT): A double-blind randomised controlled trial. Lancet 2016, 387, 1817–1826. [Google Scholar] [CrossRef]
- Adamson, P.D.; Anderson, J.A.; Brook, R.D.; Calverley, P.M.A.; Celli, B.R.; Cowans, N.J.; Crim, C.; Dixon, I.J.; Martinez, F.J.; Newby, D.E.; et al. Cardiac Troponin I and Cardiovascular Risk in Patients with Chronic Obstructive Pulmonary Disease. J. Am. Coll. Cardiol. 2018, 72, 1126–1137. [Google Scholar] [CrossRef]
- White, W.B.; Cooke, G.E.; Kowey, P.R.; Calverley, P.M.; Bredenbroker, D.; Goehring, U.M.; Zhu, H.; Lakkis, H.; Mosberg, H.; Rowe, P.; et al. Cardiovascular safety in patients receiving roflumilast for the treatment of COPD. Chest 2013, 144, 758–765. [Google Scholar] [CrossRef] [PubMed]
- Wedzicha, J.A.; Banerji, D.; Chapman, K.R.; Vestbo, J.; Roche, N.; Ayers, R.T.; Thach, C.; Fogel, R.; Patalano, F.; Vogelmeier, C.F.; et al. Indacaterol-Glycopyrronium versus Salmeterol-Fluticasone for COPD. N. Engl. J. Med. 2016, 374, 2222–2234. [Google Scholar] [CrossRef] [PubMed]
- Liao, J.K.; Laufs, U. Pleiotropic effects of statins. Annu. Rev. Pharmacol. Toxicol. 2005, 45, 89–118. [Google Scholar] [CrossRef]
- Oesterle, A.; Laufs, U.; Liao, J.K. Pleiotropic Effects of Statins on the Cardiovascular System. Circ. Res. 2017, 120, 229–243. [Google Scholar] [CrossRef] [PubMed]
- Cao, C.; Wu, Y.; Xu, Z.; Lv, D.; Zhang, C.; Lai, T.; Li, W.; Shen, H. The effect of statins on chronic obstructive pulmonary disease exacerbation and mortality: A systematic review and meta-analysis of observational research. Sci. Rep. 2015, 5, 16461. [Google Scholar] [CrossRef]
- Anthonisen, N.R.; Connett, J.E.; Enright, P.L.; Manfreda, J. Lung Health Study Research Group. Hospitalizations and mortality in the Lung Health Study. Am. J. Respir. Crit. Care Med. 2002, 166, 333–339. [Google Scholar] [CrossRef] [PubMed]
- Neukamm, A.; Hoiseth, A.D.; Einvik, G.; Lehmann, S.; Hagve, T.A.; Soyseth, V.; Omland, T. Rosuvastatin treatment in stable chronic obstructive pulmonary disease (RODEO): A randomized controlled trial. J. Intern. Med. 2015, 278, 59–67. [Google Scholar] [CrossRef] [PubMed]
- Carlson, A.A.; Smith, E.A.; Reid, D.J. The stats are in: An update on statin use in COPD. Int. J. Chronic Obstruct. Pulm. Dis. 2015, 10, 2277–2284. [Google Scholar] [CrossRef]
- Lee, T.M.; Lin, M.S.; Chang, N.C. Usefulness of C-reactive protein and interleukin-6 as predictors of outcomes in patients with chronic obstructive pulmonary disease receiving pravastatin. Am. J. Cardiol. 2008, 101, 530–535. [Google Scholar] [CrossRef]
- Lahousse, L.; Loth, D.W.; Joos, G.F.; Hofman, A.; Leufkens, H.G.; Brusselle, G.G.; Stricker, B.H. Statins, systemic inflammation and risk of death in COPD: The Rotterdam study. Pulm. Pharmacol. Ther. 2013, 26, 212–217. [Google Scholar] [CrossRef]
- John, M.E.; Hussain, S.; Haddad, M.A.; Bolton, C.E. M138 Do Standard Cardiovascular Risk Scores Identify Risk in Patients with Copd? Thorax 2014, 69, A212. [Google Scholar] [CrossRef]
- Sheng, X.; Murphy, M.J.; MacDonald, T.M.; Schembri, S.; Simpson, W.; Winter, J.; Winter, J.H.; Wei, L. Effect of statins on total cholesterol concentrations, cardiovascular morbidity, and all-cause mortality in chronic obstructive pulmonary disease: A population-based cohort study. Clin. Ther. 2012, 34, 374–384. [Google Scholar] [CrossRef] [PubMed]
- Catapano, A.L.; Graham, I.; De Backer, G.; Wiklund, O.; Chapman, M.J.; Drexel, H.; Hoes, A.W.; Jennings, C.S.; Landmesser, U.; Pedersen, T.R.; et al. 2016 ESC/EAS Guidelines for the Management of Dyslipidaemias. Eur. Heart J. 2016, 37, 2999–3058. [Google Scholar] [CrossRef] [PubMed]
- Jellinger, P.S.; Handelsman, Y.; Rosenblit, P.D.; Bloomgarden, Z.T.; Fonseca, V.A.; Garber, A.J.; Grunberger, G.; Guerin, C.K.; Bell, D.S.H.; Mechanick, J.I.; et al. American Association of Clinical Endocrinologists and American College of Endocrinology Guidelines for management of dyslipidemia and prevention of cardiovascular disease—Executive summary. Complete Appendix to Guidelines. Endocr. Pract. 2017, 23, 479–497. [Google Scholar] [CrossRef] [PubMed]
- Stone, N.J.; Robinson, J.G.; Lichtenstein, A.H.; Bairey Merz, C.N.; Blum, C.B.; Eckel, R.H.; Goldberg, A.C.; Gordon, D.; Levy, D.; Lloyd-Jones, D.M.; et al. 2013 ACC/AHA guideline on the treatment of blood cholesterol to reduce atherosclerotic cardiovascular risk in adults: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation 2014, 129, S1–S45. [Google Scholar] [CrossRef] [PubMed]
- De Lucas-Ramos, P.; Izquierdo-Alonso, J.L.; Rodriguez-Gonzalez Moro, J.M.; Frances, J.F.; Lozano, P.V.; Bellon-Cano, J.M. Chronic obstructive pulmonary disease as a cardiovascular risk factor. Results of a case-control study (CONSISTE study). Int. J. Chronic Obstruct. Pulm. Dis. 2012, 7, 679–686. [Google Scholar] [CrossRef]
- Durheim, M.T.; Cyr, D.D.; Lopes, R.D.; Thomas, L.E.; Tsuang, W.M.; Gersh, B.J.; Held, C.; Wallentin, L.; Granger, C.B.; Palmer, S.M.; et al. Chronic obstructive pulmonary disease in patients with atrial fibrillation: Insights from the ARISTOTLE trial. Int. J. Cardiol. 2016, 202, 589–594. [Google Scholar] [CrossRef] [PubMed]
- Andrade, J.; Ignaszewski, A. Cardiovascular risk assessment: Identification of individuals at increased risk. BCMJ 2008, 50, 246–251. [Google Scholar]
- Karch, A.; Vogelmeier, C.; Welte, T.; Bals, R.; Kauczor, H.U.; Biederer, J.; Heinrich, J.; Schulz, H.; Glaser, S.; Holle, R.; et al. The German COPD cohort COSYCONET: Aims, methods and descriptive analysis of the study population at baseline. Respir. Med. 2016, 114, 27–37. [Google Scholar] [CrossRef]
- Putcha, N.; Han, M.K.; Martinez, C.H.; Foreman, M.G.; Anzueto, A.R.; Casaburi, R.; Cho, M.H.; Hanania, N.A.; Hersh, C.P.; Kinney, G.L.; et al. Comorbidities of COPD have a major impact on clinical outcomes, particularly in African Americans. Chronic Obstr. Pulm. Dis. 2014, 1, 105–114. [Google Scholar] [CrossRef]
- McAllister, D.A.; Maclay, J.D.; Mills, N.L.; Mair, G.; Miller, J.; Anderson, D.; Newby, D.E.; Murchison, J.T.; Macnee, W. Arterial stiffness is independently associated with emphysema severity in patients with chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 2007, 176, 1208–1214. [Google Scholar] [CrossRef] [PubMed]
- Pavasini, R.; d’Ascenzo, F.; Campo, G.; Biscaglia, S.; Ferri, A.; Contoli, M.; Papi, A.; Ceconi, C.; Ferrari, R. Cardiac troponin elevation predicts all-cause mortality in patients with acute exacerbation of chronic obstructive pulmonary disease: Systematic review and meta-analysis. Int. J. Cardiol. 2015, 191, 187–193. [Google Scholar] [CrossRef] [PubMed]
- Williams, M.C.; Murchison, J.T.; Edwards, L.D.; Agusti, A.; Bakke, P.; Calverley, P.M.; Celli, B.; Coxson, H.O.; Crim, C.; Lomas, D.A.; et al. Coronary artery calcification is increased in patients with COPD and associated with increased morbidity and mortality. Thorax 2014, 69, 718–723. [Google Scholar] [CrossRef] [PubMed]
- Montalescot, G.; Sechtem, U.; Achenbach, S.; Andreotti, F.; Arden, C.; Budaj, A.; Bugiardini, R.; Crea, F.; Cuisset, T.; Di Mario, C.; et al. 2013 ESC guidelines on the management of stable coronary artery disease: The Task Force on the management of stable coronary artery disease of the European Society of Cardiology. Eur. Heart J. 2013, 34, 2949–3003. [Google Scholar] [CrossRef] [PubMed]
- Almagro, P.; Lapuente, A.; Pareja, J.; Yun, S.; Garcia, M.E.; Padilla, F.; Heredia, J.L.; De la Sierra, A.; Soriano, J.B. Underdiagnosis and prognosis of chronic obstructive pulmonary disease after percutaneous coronary intervention: A prospective study. Int. J. Chronic Obstr. Pulm. Dis. 2015, 10, 1353–1361. [Google Scholar] [CrossRef] [PubMed]
- Brekke, P.H.; Omland, T.; Smith, P.; Soyseth, V. Underdiagnosis of myocardial infarction in COPD—Cardiac Infarction Injury Score (CIIS) in patients hospitalised for COPD exacerbation. Respir. Med. 2008, 102, 1243–1247. [Google Scholar] [CrossRef] [PubMed]
- Andell, P.; Erlinge, D.; Smith, J.G.; Sundstrom, J.; Lindahl, B.; James, S.; Koul, S. Beta-blocker use and mortality in COPD patients after myocardial infarction: A Swedish nationwide observational study. J. Am. Heart Assoc. 2015, 4. [Google Scholar] [CrossRef] [PubMed]
- Goldberger, J.J.; Bonow, R.O.; Cuffe, M.; Dyer, A.; Rosenberg, Y.; O’Rourke, R.; Shah, P.K.; Smith, S.C., Jr.; Investigators, P.-M. β-Blocker use following myocardial infarction: Low prevalence of evidence-based dosing. Am. Heart J. 2010, 160, 435–442. [Google Scholar] [CrossRef] [PubMed]
- Salpeter, S.R.; Ormiston, T.M.; Salpeter, E.E. Cardioselective β-blockers in patients with reactive airway disease: A meta-analysis. Ann. Intern. Med. 2002, 137, 715–725. [Google Scholar] [CrossRef]
- Rutten, F.H.; Zuithoff, N.P.; Hak, E.; Grobbee, D.E.; Hoes, A.W. Beta-blockers may reduce mortality and risk of exacerbations in patients with chronic obstructive pulmonary disease. Arch. Intern. Med. 2010, 170, 880–887. [Google Scholar] [CrossRef]
- Ferrari, R.; Camici, P.G.; Crea, F.; Danchin, N.; Fox, K.; Maggioni, A.P.; Manolis, A.J.; Marzilli, M.; Rosano, G.M.C.; Lopez-Sendon, J.L. Expert consensus document: A ‘diamond’ approach to personalized treatment of angina. Nat. Rev. Cardiol. 2018, 15, 120–132. [Google Scholar] [CrossRef] [PubMed]
- Mascitelli, L.; Pezzetta, F.; Goldstein, M.R. Inhibition of the renin-angiotensin system in severe COPD. Eur. Respir. J. 2008, 32, 1130. [Google Scholar] [CrossRef] [PubMed]
- Shrikrishna, D.; Astin, R.; Kemp, P.R.; Hopkinson, N.S. Renin-angiotensin system blockade: A novel therapeutic approach in chronic obstructive pulmonary disease. Clin. Sci. 2012, 123, 487–498. [Google Scholar] [CrossRef] [PubMed]
- Packard, K.A.; Wurdeman, R.L.; Arouni, A.J. ACE inhibitor-induced bronchial reactivity in patients with respiratory dysfunction. Ann. Pharmacother. 2002, 36, 1058–1067. [Google Scholar] [CrossRef]
- Tanaka, H.; Teramoto, S.; Oashi, K.; Saikai, T.; Tanaka, S.; Suzuki, K.; Hashimoto, M.; Abe, S. Effects of candesartan on cough and bronchial hyperresponsiveness in mildly to moderately hypertensive patients with symptomatic asthma. Circulation 2001, 104, 281–285. [Google Scholar] [CrossRef] [PubMed]
- Caldeira, D.; Pinto, F.J.; Ferreira, J.J. Dyspnea and reversibility profile of P2Y(1)(2) antagonists: Systematic review of new antiplatelet drugs. Am. J. Cardiovasc. Drugs 2014, 14, 303–311. [Google Scholar] [CrossRef] [PubMed]
- Gershon, A.; Croxford, R.; Calzavara, A.; To, T.; Stanbrook, M.B.; Upshur, R.; Stukel, T.A. Cardiovascular safety of inhaled long-acting bronchodilators in individuals with chronic obstructive pulmonary disease. JAMA Intern. Med. 2013, 173, 1175–1185. [Google Scholar] [CrossRef]
- Salpeter, S.R.; Ormiston, T.M.; Salpeter, E.E. Cardiovascular effects of beta-agonists in patients with asthma and COPD: A meta-analysis. Chest 2004, 125, 2309–2321. [Google Scholar] [CrossRef]
- Rottenkolber, M.; Rottenkolber, D.; Fischer, R.; Ibanez, L.; Fortuny, J.; Ballarin, E.; Sabate, M.; Ferrer, P.; Thurmann, P.; Hasford, J.; et al. Inhaled beta-2-agonists/muscarinic antagonists and acute myocardial infarction in COPD patients. Respir. Med. 2014, 108, 1075–1090. [Google Scholar] [CrossRef]
- Lee, C.H.; Choi, S.; Jang, E.J.; Yang, H.M.; Il Yoon, H.; Kim, Y.J.; Kim, J.; Yim, J.J.; Kim, D.K. Inhaled bronchodilators and acute myocardial infarction: A nested case-control study. Sci. Rep. 2017, 7, 17915. [Google Scholar] [CrossRef]
- Rogliani, P.; Matera, M.G.; Ora, J.; Cazzola, M.; Calzetta, L. The impact of dual bronchodilation on cardiovascular serious adverse events and mortality in COPD: A quantitative synthesis. Int. J. Chronic Obstr. Pulm. Dis. 2017, 12, 3469–3485. [Google Scholar] [CrossRef] [PubMed]
- Leavitt, B.J.; Ross, C.S.; Spence, B.; Surgenor, S.D.; Olmstead, E.M.; Clough, R.A.; Charlesworth, D.C.; Kramer, R.S.; O’Connor, G.T. Northern New England Cardiovascular Disease Study Group. Long-term survival of patients with chronic obstructive pulmonary disease undergoing coronary artery bypass surgery. Circulation 2006, 114, I430–I434. [Google Scholar] [CrossRef] [PubMed]
- Selvaraj, C.L.; Gurm, H.S.; Gupta, R.; Ellis, S.G.; Bhatt, D.L. Chronic obstructive pulmonary disease as a predictor of mortality in patients undergoing percutaneous coronary intervention. Am. J. Cardiol. 2005, 96, 756–759. [Google Scholar] [CrossRef]
- Enriquez, J.R.; Parikh, S.V.; Selzer, F.; Jacobs, A.K.; Marroquin, O.; Mulukutla, S.; Srinivas, V.; Holper, E.M. Increased adverse events after percutaneous coronary intervention in patients with COPD: Insights from the National Heart, Lung, and Blood Institute dynamic registry. Chest 2011, 140, 604–610. [Google Scholar] [CrossRef] [PubMed]
- Windecker, S.; Kolh, P.; Alfonso, F.; Collet, J.P.; Cremer, J.; Falk, V.; Filippatos, G.; Hamm, C.; Head, S.J.; Juni, P.; et al. 2014 ESC/EACTS Guidelines on myocardial revascularization: The Task Force on Myocardial Revascularization of the European Society of Cardiology (ESC) and the European Association for Cardio-Thoracic Surgery (EACTS)Developed with the special contribution of the European Association of Percutaneous Cardiovascular Interventions (EAPCI). Eur. Heart J. 2014, 35, 2541–2619. [Google Scholar] [CrossRef] [PubMed]
- Huerta, C.; Lanes, S.F.; Garcia Rodriguez, L.A. Respiratory medications and the risk of cardiac arrhythmias. Epidemiology 2005, 16, 360–366. [Google Scholar] [CrossRef] [PubMed]
- Van der Hooft, C.S.; Heeringa, J.; Brusselle, G.G.; Hofman, A.; Witteman, J.C.; Kingma, J.H.; Sturkenboom, M.C.; Stricker, B.H. Corticosteroids and the risk of atrial fibrillation. Arch. Intern. Med. 2006, 166, 1016–1020. [Google Scholar] [CrossRef]
- Brook, R.D.; Anderson, J.A.; Calverley, P.M.; Celli, B.R.; Crim, C.; Denvir, M.A.; Magder, S.; Martinez, F.J.; Rajagopalan, S.; Vestbo, J.; et al. Cardiovascular outcomes with an inhaled beta2-agonist/corticosteroid in patients with COPD at high cardiovascular risk. Heart 2017, 103, 1536–1542. [Google Scholar] [CrossRef]
- Salpeter, S.R. Do inhaled anticholinergics increase or decrease the risk of major cardiovascular events? A synthesis of the available evidence. Drugs 2009, 69, 2025–2033. [Google Scholar] [CrossRef]
- Sessler, C.N.; Cohen, M.D. Cardiac arrhythmias during theophylline toxicity. A prospective continuous electrocardiographic study. Chest 1990, 98, 672–678. [Google Scholar] [CrossRef]
- Liang, M.; Puri, A.; Devlin, G. Heart rate and cardiovascular disease: An alternative to Beta blockers. Cardiol. Res. Pract. 2009, 2009, 179350. [Google Scholar] [CrossRef] [PubMed]
- Camus, P.; Martin, W.J., 2nd; Rosenow, E.C., 3rd. Amiodarone pulmonary toxicity. Clin. Chest Med. 2004, 25, 65–75. [Google Scholar] [CrossRef]
- Rutten, F.H.; Cramer, M.J.; Grobbee, D.E.; Sachs, A.P.; Kirkels, J.H.; Lammers, J.W.; Hoes, A.W. Unrecognized heart failure in elderly patients with stable chronic obstructive pulmonary disease. Eur. Heart J. 2005, 26, 1887–1894. [Google Scholar] [CrossRef] [PubMed]
- Macchia, A.; Rodriguez Moncalvo, J.J.; Kleinert, M.; Comignani, P.D.; Gimeno, G.; Arakaki, D.; Laffaye, N.; Fuselli, J.J.; Massolin, H.P.; Gambarte, J.; et al. Unrecognised ventricular dysfunction in COPD. Eur. Respir. J. 2012, 39, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Rusinaru, D.; Saaidi, I.; Godard, S.; Mahjoub, H.; Battle, C.; Tribouilloy, C. Impact of chronic obstructive pulmonary disease on long-term outcome of patients hospitalized for heart failure. Am. J. Cardiol. 2008, 101, 353–358. [Google Scholar] [CrossRef] [PubMed]
- Brenner, S.; Guder, G.; Berliner, D.; Deubner, N.; Frohlich, K.; Ertl, G.; Jany, B.; Angermann, C.E.; Stork, S. Airway obstruction in systolic heart failure—COPD or congestion? Int. J. Cardiol. 2013, 168, 1910–1916. [Google Scholar] [CrossRef] [PubMed]
- Le Jemtel, T.H.; Padeletti, M.; Jelic, S. Diagnostic and therapeutic challenges in patients with coexistent chronic obstructive pulmonary disease and chronic heart failure. J. Am. Coll. Cardiol. 2007, 49, 171–180. [Google Scholar] [CrossRef] [PubMed]
- Flessas, N.; Alexanian, I.; Parissis, J.; Kremastinos, D.; Lekakis, J.; Filippatos, G. Plasma activity of B-type natriuretic peptide in patients with biventricular heart failure versus those with right heart failure due to chronic obstructive pulmonary disease. J. Cardiovasc. Med. 2014, 15, 476–480. [Google Scholar] [CrossRef]
- Hawkins, N.M.; Virani, S.; Ceconi, C. Heart failure and chronic obstructive pulmonary disease: The challenges facing physicians and health services. Eur. Heart J. 2013, 34, 2795–2803. [Google Scholar] [CrossRef]
- Caruana, L.; Petrie, M.C.; Davie, A.P.; McMurray, J.J. Do patients with suspected heart failure and preserved left ventricular systolic function suffer from “diastolic heart failure” or from misdiagnosis? A prospective descriptive study. BMJ 2000, 321, 215–218. [Google Scholar] [CrossRef]
- Mühle, A.; Obst, A.; Winkler, J.; Ewert, R. Cardiopulmonary Exercise Testing in Chronic Obstructive Pulmonary Disease (COPD)—Breath-functional Characterization and Disease Severity Assessment. Pneumologie 2015, 69, 534–544. [Google Scholar] [CrossRef] [PubMed]
- Rutten, F.H.; Cramer, M.J.; Zuithoff, N.P.; Lammers, J.W.; Verweij, W.; Grobbee, D.E.; Hoes, A.W. Comparison of B-type natriuretic peptide assays for identifying heart failure in stable elderly patients with a clinical diagnosis of chronic obstructive pulmonary disease. Eur. J. Heart Fail. 2007, 9, 651–659. [Google Scholar] [CrossRef] [PubMed]
- Lipworth, B.; Skinner, D.; Devereux, G.; Thomas, V.; Jie, J.L.; Martin, J.; Carter, V.; Price, D.B. Underuse of beta-blockers in heart failure and chronic obstructive pulmonary disease. Heart 2016, 102, 1909–1914. [Google Scholar] [CrossRef] [PubMed]
- Ponikowski, P.; Voors, A.A.; Anker, S.D.; Bueno, H.; Cleland, J.G.; Coats, A.J.; Falk, V.; Gonzalez-Juanatey, J.R.; Harjola, V.P.; Jankowska, E.A.; et al. 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: The Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC) Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur. Heart J. 2016, 37, 2129–2200. [Google Scholar] [CrossRef] [PubMed]
- Lainscak, M.; Podbregar, M.; Kovacic, D.; Rozman, J.; von Haehling, S. Differences between bisoprolol and carvedilol in patients with chronic heart failure and chronic obstructive pulmonary disease: A randomized trial. Respir. Med. 2011, 105 (Suppl. 1), S44–S49. [Google Scholar] [CrossRef]
- Hohlfeld, J.M.; Vogel-Claussen, J.; Biller, H.; Berliner, D.; Berschneider, K.; Tillmann, H.C.; Hiltl, S.; Bauersachs, J.; Welte, T. Effect of lung deflation with indacaterol plus glycopyrronium on ventricular filling in patients with hyperinflation and COPD (CLAIM): A double-blind, randomised, crossover, placebo-controlled, single-centre trial. Lancet Respir. Med. 2018, 6, 368–378. [Google Scholar] [CrossRef]
- Wheaton, A.G.; Ford, E.S.; Cunningham, T.J.; Croft, J.B. Chronic obstructive pulmonary disease, hospital visits, and comorbidities: National Survey of Residential Care Facilities, 2010. J. Aging Health 2015, 27, 480–499. [Google Scholar] [CrossRef] [PubMed]
- Mills, N.L.; Miller, J.J.; Anand, A.; Robinson, S.D.; Frazer, G.A.; Anderson, D.; Breen, L.; Wilkinson, I.B.; McEniery, C.M.; Donaldson, K.; et al. Increased arterial stiffness in patients with chronic obstructive pulmonary disease: A mechanism for increased cardiovascular risk. Thorax 2008, 63, 306–311. [Google Scholar] [CrossRef]
- De Luca, N.; Asmar, R.G.; London, G.M.; O’Rourke, M.F.; Safar, M.E. Selective reduction of cardiac mass and central blood pressure on low-dose combination perindopril/indapamide in hypertensive subjects. J. Hypertens. 2004, 22, 1623–1630. [Google Scholar] [CrossRef]
- Roman, M.J.; Devereux, R.B.; Kizer, J.R.; Lee, E.T.; Galloway, J.M.; Ali, T.; Umans, J.G.; Howard, B.V. Central pressure more strongly relates to vascular disease and outcome than does brachial pressure: The Strong Heart Study. Hypertension 2007, 50, 197–203. [Google Scholar] [CrossRef]
- Wang, K.L.; Cheng, H.M.; Chuang, S.Y.; Spurgeon, H.A.; Ting, C.T.; Lakatta, E.G.; Yin, F.C.; Chou, P.; Chen, C.H. Central or peripheral systolic or pulse pressure: Which best relates to target organs and future mortality? J. Hypertens. 2009, 27, 461–467. [Google Scholar] [CrossRef] [PubMed]
- Pini, R.; Cavallini, M.C.; Palmieri, V.; Marchionni, N.; Di Bari, M.; Devereux, R.B.; Masotti, G.; Roman, M.J. Central but not brachial blood pressure predicts cardiovascular events in an unselected geriatric population: The ICARe Dicomano Study. J. Am. Coll. Cardiol. 2008, 51, 2432–2439. [Google Scholar] [CrossRef] [PubMed]
- Protogerou, A.D.; Stergiou, G.S.; Vlachopoulos, C.; Blacher, J.; Achimastos, A. The effect of antihypertensive drugs on central blood pressure beyond peripheral blood pressure. Part II: Evidence for specific class-effects of antihypertensive drugs on pressure amplification. Curr. Pharm. Des. 2009, 15, 272–289. [Google Scholar] [CrossRef] [PubMed]
- Manisty, C.H.; Hughes, A.D. Meta-analysis of the comparative effects of different classes of antihypertensive agents on brachial and central systolic blood pressure, and augmentation index. Br. J. Clin. Pharmacol. 2013, 75, 79–92. [Google Scholar] [CrossRef]
- Boutouyrie, P.; Achouba, A.; Trunet, P.; Laurent, S. Amlodipine-valsartan combination decreases central systolic blood pressure more effectively than the amlodipine-atenolol combination: The EXPLOR study. Hypertension 2010, 55, 1314–1322. [Google Scholar] [CrossRef]
- Williams, B.; Lacy, P.S.; Thom, S.M.; Cruickshank, K.; Stanton, A.; Collier, D.; Hughes, A.D.; Thurston, H.; O’Rourke, M. Differential impact of blood pressure-lowering drugs on central aortic pressure and clinical outcomes: Principal results of the Conduit Artery Function Evaluation (CAFE) study. Circulation 2006, 113, 1213–1225. [Google Scholar] [CrossRef]
- Chandy, D.; Aronow, W.S.; Banach, M. Current perspectives on treatment of hypertensive patients with chronic obstructive pulmonary disease. Integr. Blood Press. Control 2013, 6, 101–109. [Google Scholar] [CrossRef]
- Cheng, H.M.; Lang, D.; Tufanaru, C.; Pearson, A. Measurement accuracy of non-invasively obtained central blood pressure by applanation tonometry: A systematic review and meta-analysis. Int. J. Cardiol. 2013, 167, 1867–1876. [Google Scholar] [CrossRef]
- Papaioannou, T.G.; Karageorgopoulou, T.D.; Sergentanis, T.N.; Protogerou, A.D.; Psaltopoulou, T.; Sharman, J.E.; Weber, T.; Blacher, J.; Daskalopoulou, S.S.; Wassertheurer, S.; et al. Accuracy of commercial devices and methods for noninvasive estimation of aortic systolic blood pressure a systematic review and meta-analysis of invasive validation studies. J. Hypertens. 2016, 34, 1237–1248. [Google Scholar] [CrossRef]
- Schumacher, G.; Kaden, J.J.; Trinkmann, F. Multiple coupled resonances in the human vascular tree—Refining the Westerhof model of the arterial system. J. Appl. Physiol. 2017. [Google Scholar] [CrossRef]
- Seeger, W.; Adir, Y.; Barbera, J.A.; Champion, H.; Coghlan, J.G.; Cottin, V.; De Marco, T.; Galie, N.; Ghio, S.; Gibbs, S.; et al. Pulmonary hypertension in chronic lung diseases. J. Am. Coll. Cardiol. 2013, 62, D109–D116. [Google Scholar] [CrossRef] [PubMed]
- Guazzi, M.; Borlaug, B.A. Pulmonary hypertension due to left heart disease. Circulation 2012, 126, 975–990. [Google Scholar] [CrossRef]
- Terzano, C.; Romani, S.; Conti, V.; Paone, G.; Oriolo, F.; Vitarelli, A. Atrial fibrillation in the acute, hypercapnic exacerbations of COPD. Eur. Rev. Med. Pharmacol. Sci. 2014, 18, 2908–2917. [Google Scholar] [PubMed]
- Buch, P.; Friberg, J.; Scharling, H.; Lange, P.; Prescott, E. Reduced lung function and risk of atrial fibrillation in the Copenhagen City Heart Study. Eur. Respir. J. 2003, 21, 1012–1016. [Google Scholar] [CrossRef]
- Kirchhof, P.; Benussi, S.; Kotecha, D.; Ahlsson, A.; Atar, D.; Casadei, B.; Castella, M.; Diener, H.C.; Heidbuchel, H.; Hendriks, J.; et al. 2016 ESC Guidelines for the management of atrial fibrillation developed in collaboration with EACTS. Eur. Heart J. 2016, 37, 2893–2962. [Google Scholar] [CrossRef]
- Gu, J.; Liu, X.; Tan, H.; Zhou, L.; Jiang, W.; Wang, Y.; Liu, Y.; Gu, J. Impact of chronic obstructive pulmonary disease on procedural outcomes and quality of life in patients with atrial fibrillation undergoing catheter ablation. J. Cardiovasc. Electrophysiol. 2013, 24, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Pisters, R.; Nieuwlaat, R.; Prins, M.H.; Le Heuzey, J.Y.; Maggioni, A.P.; Camm, A.J.; Crijns, H.J. Clinical correlates of immediate success and outcome at 1-year follow-up of real-world cardioversion of atrial fibrillation: The Euro Heart Survey. Europace 2012, 14, 666–674. [Google Scholar] [CrossRef]
- Roh, S.Y.; Choi, J.I.; Lee, J.Y.; Kwak, J.J.; Park, J.S.; Kim, J.B.; Lim, H.E.; Kim, Y.H. Catheter ablation of atrial fibrillation in patients with chronic lung disease. Circulation 2011, 4, 815–822. [Google Scholar] [CrossRef]
- McCord, J.; Borzak, S. Multifocal atrial tachycardia. Chest 1998, 113, 203–209. [Google Scholar] [CrossRef]
- Lahousse, L.; Niemeijer, M.N.; van den Berg, M.E.; Rijnbeek, P.R.; Joos, G.F.; Hofman, A.; Franco, O.H.; Deckers, J.W.; Eijgelsheim, M.; Stricker, B.H.; et al. Chronic obstructive pulmonary disease and sudden cardiac death: The Rotterdam study. Eur. Heart J. 2015, 36, 1754–1761. [Google Scholar] [CrossRef]
- Priori, S.G.; Blomstrom-Lundqvist, C.; Mazzanti, A.; Blom, N.; Borggrefe, M.; Camm, J.; Elliott, P.M.; Fitzsimons, D.; Hatala, R.; Hindricks, G.; et al. 2015 ESC Guidelines for the management of patients with ventricular arrhythmias and the prevention of sudden cardiac death: The Task Force for the Management of Patients with Ventricular Arrhythmias and the Prevention of Sudden Cardiac Death of the European Society of Cardiology (ESC) Endorsed by: Association for European Paediatric and Congenital Cardiology (AEPC). Europace 2015, 17, 1601–1687. [Google Scholar] [CrossRef] [PubMed]
| Medication | Indication | Comment | References | ||
|---|---|---|---|---|---|
| COPD | steroids | inhaled | long-term therapy # | good safety profile | [57] |
| systemic | exacerbation | pro-arrhythmic potential | [58] | ||
| beta agonists | short acting | exacerbation long-term therapy | pro-arrhythmic potential (high doses) | [50,51,52,59] | |
| long acting | long-term therapy | acceptable safety profile | |||
| muscarinic antagonists | short acting | exacerbation long-term therapy | pro-arrhythmic potential (high doses) | [50,52,60] | |
| long acting | long-term therapy | acceptable safety profile | |||
| PDE inhibitors | Roflumilast | long-term therapy | reduction of cardiovascular events | [12] | |
| Theophylline | (long-term therapy) | narrow therapeutic range and considerable pro-arrhythmic potential | [57,61] | ||
| Cardiovascular Disease | beta blockers | selective | heart failure, CHD, ACS, AHT, SVT, VT | often withheld or under dosed, prefer selective substances, overall good safety profile | [40,41] |
| non-selective | |||||
| antiplatelet therapy | CHD, ACS | dyspnea caused by reversible P2Y₁₂-antagonists (Ticagrelor, Cangrelor) | [47] | ||
| Ivabradine | CHD, heart failure | alternative to beta blockers for anti-anginal and frequency control (sinus rhythm only) | [42,62] | ||
| statins | CHD, dyslipidemia | secondary prevention, pleiotropic effects of immune system and inflammation | [14,15] | ||
| ACE inhibitors | AHT, heart failure | no bronchoconstriction | [45,46] | ||
| angiotensin receptor blockers | alternative (ACE inhibitor induced cough) | ||||
| calcium channel blockers | AHT, CHD, SVT | alternative to beta blockers for anti-anginal and frequency control, smooth muscle relaxation (small clinical effect) | [42] | ||
| nitrates | CHD | alternative to beta blockers for anti-anginal control | [42] | ||
| amiodarone | SVT, VT | pulmonary toxicity | [63] | ||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Trinkmann, F.; Saur, J.; Borggrefe, M.; Akin, I. Cardiovascular Comorbidities in Chronic Obstructive Pulmonary Disease (COPD)—Current Considerations for Clinical Practice. J. Clin. Med. 2019, 8, 69. https://doi.org/10.3390/jcm8010069
Trinkmann F, Saur J, Borggrefe M, Akin I. Cardiovascular Comorbidities in Chronic Obstructive Pulmonary Disease (COPD)—Current Considerations for Clinical Practice. Journal of Clinical Medicine. 2019; 8(1):69. https://doi.org/10.3390/jcm8010069
Chicago/Turabian StyleTrinkmann, Frederik, Joachim Saur, Martin Borggrefe, and Ibrahim Akin. 2019. "Cardiovascular Comorbidities in Chronic Obstructive Pulmonary Disease (COPD)—Current Considerations for Clinical Practice" Journal of Clinical Medicine 8, no. 1: 69. https://doi.org/10.3390/jcm8010069
APA StyleTrinkmann, F., Saur, J., Borggrefe, M., & Akin, I. (2019). Cardiovascular Comorbidities in Chronic Obstructive Pulmonary Disease (COPD)—Current Considerations for Clinical Practice. Journal of Clinical Medicine, 8(1), 69. https://doi.org/10.3390/jcm8010069





