Metformin Decreases Risk of Tuberculosis Infection in Type 2 Diabetes Patients
Abstract
1. Introduction
2. Materials and Methods
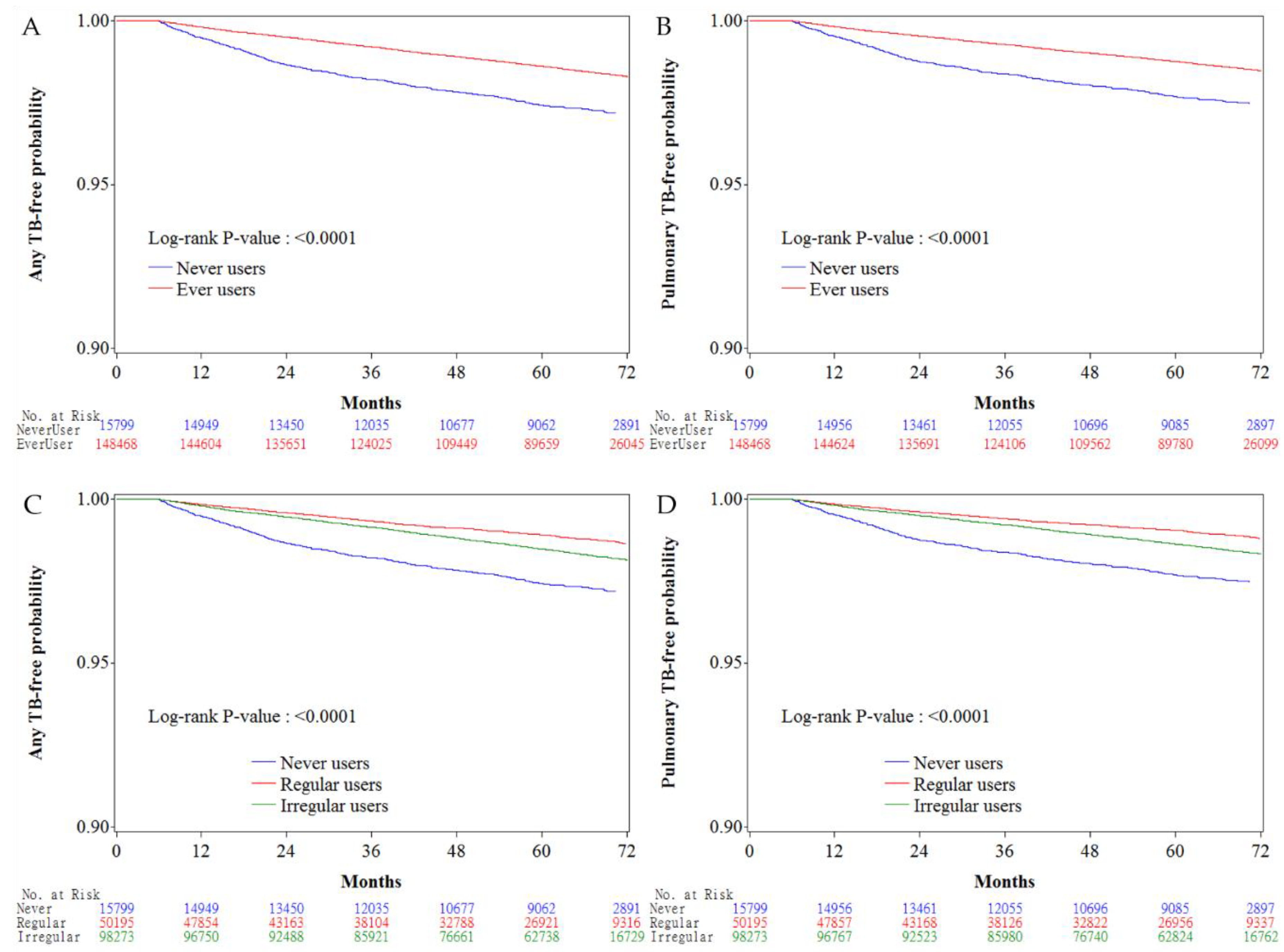
3. Results
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- GBD 2016 Causes of Death Collaborators. Global, regional, and national age-sex specific mortality for 264 causes of death, 1980–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet 2017, 390, 1151–1210. [Google Scholar] [CrossRef]
- World Health Organization. Global Tuberculosis Report 2017. Available online: http://www.who.int/tb/publications/global_report/en/ (accessed on 30 August 2018).
- Maniar, K.; Moideen, A.; Mittal, A.; Patil, A.; Chakrabarti, A.; Banerjee, D. A story of metformin-butyrate synergism to control various pathological conditions as a consequence of gut microbiome modification: Genesis of a wonder drug? Pharmacol. Res. 2017, 117, 103–128. [Google Scholar] [CrossRef] [PubMed]
- UK Prospective Diabetes Study (UKPDS) Group. Effect of intensive blood-glucose control with metformin on complications in overweight patients with type 2 diabetes (UKPDS 34). Lancet 1998, 352, 854–865. [Google Scholar] [CrossRef]
- Singhal, A.; Jie, L.; Kumar, P.; Hong, G.S.; Leow, M.K.; Paleja, B.; Tsenova, L.; Kurepina, N.; Chen, J.; Zolezzi, F.; et al. Metformin as adjunct antituberculosis therapy. Sci. Transl. Med. 2014, 6, 263ra159. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.Y.; Tu, H.P.; Lu, P.L.; Chen, T.C.; Wang, W.H.; Chong, I.W.; Chen, Y.H. Metformin is associated with a lower risk of active tuberculosis in patients with type 2 diabetes. Respirology 2018. [Google Scholar] [CrossRef] [PubMed]
- Tseng, C.H. Metformin is associated with a lower risk of colorectal cancer in Taiwanese patients with type 2 diabetes: A retrospective cohort analysis. Diabetes Metab. 2017, 43, 438–445. [Google Scholar] [CrossRef] [PubMed]
- Tseng, C.H. Metformin is associated with decreased skin cancer risk in Taiwanese patients with type 2 diabetes. J. Am. Acad. Dermatol. 2018, 78, 694–700. [Google Scholar] [CrossRef] [PubMed]
- Austin, P.C.; Stuart, E.A. Moving towards best practice when using inverse probability of treatment weighting (IPTW) using the propensity score to estimate causal treatment effects in observational studies. Stat. Med. 2015, 34, 3661–3679. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.; Dalton, J.E. A Unified Approach to Measuring the Effect Size between Two Groups Using SAS®. Available online: http://support.sas.com/resources/papers/proceedings12/335-2012.pdf (accessed on 30 August 2018).
- Austin, P.C. The performance of different propensity score methods for estimating marginal hazard ratios. Stat. Med. 2013, 32, 2837–2849. [Google Scholar] [CrossRef] [PubMed]
- Kumar Nathella, P.; Babu, S. Influence of diabetes mellitus on immunity to human tuberculosis. Immunology 2017, 152, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Lachmandas, E.; Van den Heuvel, C.N.; Damen, M.S.; Cleophas, M.C.; Netea, M.G.; Van Crevel, R. Diabetes mellitus and increased tuberculosis susceptibility: The role of short-chain fatty acids. J. Diabetes Res. 2016, 2016, 6014631. [Google Scholar] [CrossRef] [PubMed]
- Maniar, K.; Moideen, A.; Bhattacharyya, R.; Banerjee, D. Metformin exerts anti-obesity effect via gut microbiome modulation in prediabetics: A hypothesis. Med. Hypotheses 2017, 104, 117–120. [Google Scholar] [CrossRef] [PubMed]
- Sellamuthu, S.; Singh, M.; Kumar, A.; Singh, S.K. Type-II NADH Dehydrogenase (NDH-2): A promising therapeutic target for antitubercular and antibacterial drug discovery. Expert. Opin. Ther. Targets 2017, 21, 559–570. [Google Scholar] [CrossRef] [PubMed]
- Restrepo, B.I. Metformin: Candidate host-directed therapy for tuberculosis in diabetes and non-diabetes patients. Tuberculosis 2016, 101, S69–S72. [Google Scholar] [CrossRef] [PubMed]
- Dutta, N.K.; Pinn, M.L.; Karakousis, P.C. Metformin adjunctive therapy does not improve the sterilizing activity of the first-line antitubercular regimen in mice. Antimicrob. Agents Chemother. 2017, 61, e00652-17. [Google Scholar] [CrossRef] [PubMed]
- ENCePP Guide on Methodological Standards in Pharmacoepidemiology. Available online: http://www.encepp.eu/standards_and_guidances/methodologicalGuide4_2_3_1.shtml (accessed on 30 August 2018).
- Lévesque, L.E.; Hanley, J.A.; Kezouh, A.; Suissa, S. Problem of immortal time bias in cohort studies: Example using statins for preventing progression of diabetes. BMJ 2010, 340, b5087. [Google Scholar] [CrossRef] [PubMed]
- Niazi, A.K.; Kalra, S. Diabetes and tuberculosis: A review of the role of optimal glycemic control. J. Diabetes Metab. Disord. 2012, 11, 28. [Google Scholar] [CrossRef] [PubMed]
- Van Stralen, K.J.; Dekker, F.W.; Zoccali, C.; Jager, K.J. Confounding. Nephron. Clin. Pract. 2010, 116, c143–c147. [Google Scholar] [CrossRef] [PubMed]

| Variable | Never Users | Ever Users | SD | 95% CI of SD | |||
|---|---|---|---|---|---|---|---|
| (n = 15,799) | (n = 148,468) | ||||||
| n | % | n | % | Lower | Upper | ||
| Basic data | |||||||
| Age * (years) | 63.52 ± 10.43 | 61.77 ± 10.02 | −18.64 | −20.28 | −17.00 | ||
| Diabetes duration (years) * | 8.66 ± 2.31 | 9.67 ± 2.11 | 54.43 | 52.78 | 56.08 | ||
| Sex (men) | 9014 | 57.05 | 79,227 | 53.36 | −7.68 | −9.32 | −6.04 |
| Occupation | |||||||
| I | 6200 | 39.24 | 58,150 | 39.17 | |||
| II | 3179 | 20.12 | 34,292 | 23.10 | 7.90 | 6.26 | 9.54 |
| III | 3310 | 20.95 | 30,988 | 20.87 | 0.42 | −1.22 | 2.06 |
| IV | 3110 | 19.68 | 25,038 | 16.86 | −9.21 | −10.85 | −7.56 |
| Living region | |||||||
| Taipei | 5332 | 33.75 | 47,084 | 31.71 | |||
| Northern | 1614 | 10.22 | 16,930 | 11.40 | 4.16 | 2.52 | 5.80 |
| Central | 2742 | 17.36 | 27,167 | 18.30 | 2.36 | 0.72 | 4.00 |
| Southern | 2754 | 17.43 | 25,390 | 17.10 | 0.15 | −1.49 | 1.79 |
| Kao-Ping and Eastern | 3357 | 21.25 | 31,897 | 21.48 | 0.52 | −1.12 | 2.16 |
| Major comorbidities | |||||||
| Hypertension | 12,909 | 81.71 | 121,765 | 82.01 | −2.80 | −4.44 | −1.15 |
| Dyslipidemia | 11,450 | 72.47 | 123,483 | 83.17 | 32.38 | 30.73 | 34.02 |
| Obesity | 436 | 2.76 | 6797 | 4.58 | 10.71 | 9.07 | 12.35 |
| Diabetes-related complications | |||||||
| Nephropathy | 5431 | 34.38 | 40,602 | 27.35 | −27.84 | −29.48 | −26.19 |
| Peritoneal dialysis/hemodialysis | 1237 | 7.83 | 2231 | 1.50 | −50.29 | −51.94 | −48.64 |
| Diabetes with ophthalmic manifestations/diabetic retinopathy | 2840 | 17.98 | 47,581 | 32.05 | 32.87 | 31.23 | 34.51 |
| Glaucoma | 1822 | 11.53 | 17,868 | 12.03 | 1.23 | −0.41 | 2.87 |
| Cataract | 7196 | 45.55 | 69,901 | 47.08 | 1.06 | −0.58 | 2.70 |
| Blindness and low vision | 82 | 0.52 | 565 | 0.38 | −3.01 | −4.65 | −1.37 |
| Stroke | 5189 | 32.84 | 44,041 | 29.66 | −10.49 | −12.13 | −8.85 |
| Ischemic heart disease | 7502 | 47.48 | 68,162 | 45.91 | −6.95 | −8.59 | −5.31 |
| Peripheral arterial disease | 3636 | 23.01 | 38,379 | 25.85 | 3.65 | 2.01 | 5.29 |
| Antidiabetic drugs | |||||||
| Insulin | 1268 | 8.03 | 3374 | 2.27 | −40.01 | −41.66 | −38.37 |
| Sulfonylurea | 11,553 | 73.12 | 108,025 | 72.76 | 12.55 | 10.91 | 14.19 |
| Meglitinide | 1278 | 8.09 | 5810 | 3.91 | −24.69 | −26.33 | −23.05 |
| Acarbose | 1801 | 11.40 | 8103 | 5.46 | −22.00 | −23.64 | −20.36 |
| Rosiglitazone | 465 | 2.94 | 7360 | 4.96 | 10.98 | 9.34 | 12.62 |
| Pioglitazone | 392 | 2.48 | 3913 | 2.64 | 2.72 | 1.08 | 4.36 |
| Commonly encountered comorbidities | |||||||
| Chronic obstructive pulmonary disease | 7715 | 48.83 | 71,015 | 47.83 | −3.77 | −5.41 | −2.13 |
| Tobacco abuse | 429 | 2.72 | 5820 | 3.92 | 8.20 | 6.56 | 9.84 |
| Alcohol-related diagnoses | 1202 | 7.61 | 10,299 | 6.94 | −3.49 | −5.13 | −1.85 |
| Heart failure | 3328 | 21.06 | 25,419 | 17.12 | −18.27 | −19.91 | −16.63 |
| Gingival and periodontal diseases | 13,216 | 83.65 | 128,871 | 86.80 | 11.69 | 10.05 | 13.33 |
| Pneumonia | 2759 | 17.46 | 21,320 | 14.36 | −15.96 | −17.60 | −14.32 |
| Osteoporosis | 3571 | 22.60 | 30,299 | 20.41 | −6.26 | −7.90 | −4.62 |
| Rheumatologic diseases | 1176 | 7.44 | 10,456 | 7.04 | −3.30 | −4.94 | −1.66 |
| Liver cirrhosis | 1282 | 8.11 | 7961 | 5.36 | −15.49 | −17.13 | −13.85 |
| Other chronic non-alcoholic liver diseases | 1503 | 9.51 | 16,882 | 11.37 | 7.48 | 5.84 | 9.12 |
| Hepatitis B virus infection | 704 | 4.46 | 6028 | 4.06 | −4.36 | −6.00 | −2.72 |
| Hepatitis C virus infection | 1003 | 6.35 | 7772 | 5.23 | −7.54 | −9.18 | −5.90 |
| Human immunodeficiency virus infection | 18 | 0.11 | 94 | 0.06 | −3.51 | −5.15 | −1.87 |
| Organ transplantation | 137 | 0.87 | 380 | 0.26 | −12.54 | −14.18 | −10.90 |
| Commonly used medications in diabetes patients | |||||||
| Angiotensin converting enzyme inhibitor/angiotensin receptor blocker | 10,958 | 69.36 | 108,934 | 73.37 | 6.00 | 4.36 | 7.64 |
| Calcium channel blocker | 9879 | 62.53 | 89,335 | 60.17 | −9.32 | −10.96 | −7.68 |
| Statin | 8568 | 54.23 | 98,307 | 66.21 | 27.60 | 25.95 | 29.24 |
| Fibrate | 5416 | 34.28 | 64,562 | 43.49 | 20.53 | 18.89 | 22.17 |
| Aspirin | 9024 | 57.12 | 91,731 | 61.79 | 8.20 | 6.56 | 9.84 |
| Tuberculosis Infection/Metformin Exposure | n | N | Person–Years | Incidence Rate (Per 100,000 Person-Years) | HR | 95% CI | p Value |
|---|---|---|---|---|---|---|---|
| Any tuberculosis infection | |||||||
| Never users | 360 | 15,799 | 70,462.21 | 510.91 | 1.000 | ||
| Ever users | 1976 | 148,468 | 698,376.69 | 282.94 | 0.552 | (0.493–0.617) | <0.0001 |
| Tertiles of cumulative duration of metformin therapy (months) | |||||||
| Never users | 360 | 15,799 | 70,462.21 | 510.91 | 1.000 | ||
| <27.10 | 1001 | 48,988 | 171,052.60 | 585.20 | 1.116 | (0.989–1.261) | 0.0755 |
| 27.10–58.27 | 671 | 49,015 | 239,169.18 | 280.55 | 0.543 | (0.478–0.618) | <0.0001 |
| >58.27 | 304 | 50,465 | 288,154.92 | 105.50 | 0.200 | (0.171–0.233) | <0.0001 |
| Tertiles of cumulative dose of metformin therapy (mg) | |||||||
| Never users | 360 | 15,799 | 70,462.21 | 510.91 | 1.000 | ||
| <817,000 | 941 | 48,972 | 172,676.49 | 544.95 | 1.037 | (0.918–1.173) | 0.5558 |
| 817,000–2,047,180 | 663 | 49,016 | 240,955.45 | 275.15 | 0.533 | (0.469–0.606) | <0.0001 |
| >2,047,180 | 372 | 50,480 | 284,744.76 | 130.64 | 0.249 | (0.215–0.288) | <0.0001 |
| Pulmonary tuberculosis infection | |||||||
| Never users | 324 | 15,799 | 70,549.63 | 459.25 | 1.000 | ||
| Ever users | 1773 | 148,468 | 698,800.36 | 253.72 | 0.551 | (0.489–0.620) | <0.0001 |
| Tertiles of cumulative duration of metformin therapy (months) | |||||||
| Never users | 324 | 15,799 | 70,549.63 | 459.25 | 1.000 | ||
| <27.10 | 902 | 48,988 | 171,274.72 | 526.64 | 1.112 | (0.978–1.264) | 0.1042 |
| 27.10–58.27 | 605 | 49,015 | 239,298.85 | 252.82 | 0.545 | (0.477–0.624) | <0.0001 |
| >58.27 | 266 | 50,465 | 288,226.79 | 92.29 | 0.195 | (0.165–0.229) | <0.0001 |
| Tertiles of cumulative dose of metformin therapy (mg) | |||||||
| Never users | 324 | 15,799 | 70,549.63 | 459.25 | 1.000 | ||
| <817,000 | 847 | 48,972 | 172,887.94 | 489.91 | 1.032 | (0.907–1.175) | 0.6282 |
| 817,000–2,047,180 | 595 | 49,016 | 241,084.90 | 246.80 | 0.532 | (0.465–0.610) | <0.0001 |
| >2,047,180 | 331 | 50,480 | 284,827.52 | 116.21 | 0.247 | (0.211–0.288) | <0.0001 |
| Tuberculosis Infection/Metformin Exposure | n | N | HR | 95% CI | p Value |
|---|---|---|---|---|---|
| Any tuberculosis infection | |||||
| Never users | 360 | 15,799 | 1.000 | ||
| Ever users | 496 | 50,195 | 0.438 | (0.382–0.501) | <0.0001 |
| Tertiles of cumulative duration of metformin therapy (months) | |||||
| Never users | 360 | 15,799 | 1.000 | ||
| <27.10 | 217 | 16,349 | 0.889 | (0.748–1.055) | 0.1781 |
| 27.10–58.27 | 167 | 13,683 | 0.523 | (0.435–0.628) | <0.0001 |
| >58.27 | 112 | 20,163 | 0.190 | (0.153–0.235) | <0.0001 |
| Tertiles of cumulative dose of metformin therapy (mg) | |||||
| Never users | 360 | 15,799 | 1.000 | ||
| <817,000 | 218 | 16,919 | 0.847 | (0.713–1.005) | 0.0572 |
| 817,000–2,047,180 | 151 | 14,180 | 0.443 | (0.366–0.535) | <0.0001 |
| >2,047,180 | 127 | 19,096 | 0.230 | (0.188–0.282) | <0.0001 |
| Pulmonary tuberculosis infection | |||||
| Never users | 324 | 15,799 | 1.000 | ||
| Ever users | 439 | 50,195 | 0.431 | (0.373–0.497) | <0.0001 |
| Tertiles of cumulative duration of metformin therapy (months) | |||||
| Never users | 324 | 15,799 | 1.000 | ||
| <27.10 | 190 | 16,349 | 0.852 | (0.710–1.023) | 0.0853 |
| 27.10–58.27 | 149 | 13,683 | 0.517 | (0.426–0.629) | <0.0001 |
| >58.27 | 100 | 20,163 | 0.189 | (0.151–0.237) | <0.0001 |
| Tertiles of cumulative dose of metformin therapy (mg) | |||||
| Never users | 324 | 15,799 | 1.000 | ||
| <817,000 | 191 | 16,919 | 0.813 | (0.678–0.976) | 0.0262 |
| 817,000–2,047,180 | 135 | 14,180 | 0.440 | (0.360–0.537) | <0.0001 |
| >2,047,180 | 113 | 19,096 | 0.228 | (0.184–0.283) | <0.0001 |
| Tuberculosis Infection/Metformin Exposure | Irregular Metformin Users versus Never Users | Regular Metformin Users versus Never Users | ||||
|---|---|---|---|---|---|---|
| HR | 95% CI | p Value | HR | 95% CI | p Value | |
| Any tuberculosis infection | ||||||
| Never users | 1.000 | 1.000 | ||||
| Ever users | 0.603 | (0.538–0.677) | <0.0001 | 0.438 | (0.382–0.501) | <0.0001 |
| Tertiles of cumulative duration of metformin therapy (months) | ||||||
| Never users | 1.000 | 1.000 | ||||
| <27.1 | 1.179 | (1.040–1.336) | 0.0103 | 0.889 | (0.748–1.055) | 0.1781 |
| 27.1–58.3 | 0.547 | (0.478–0.627) | <0.0001 | 0.523 | (0.435–0.628) | <0.0001 |
| >58.3 | 0.214 | (0.180–0.255) | <0.0001 | 0.190 | (0.153–0.235) | <0.0001 |
| Tertiles of cumulative dose of metformin therapy (mg) | ||||||
| Never users | 1.000 | 1.000 | ||||
| <817,000 | 1.096 | (0.965–1.245) | 0.1570 | 0.847 | (0.713–1.005) | 0.0572 |
| 817,000–2,047,180 | 0.565 | (0.494–0.647) | <0.0001 | 0.443 | (0.366–0.535) | <0.0001 |
| >2,047,180 | 0.268 | (0.228–0.316) | <0.0001 | 0.230 | (0.188–0.282) | <0.0001 |
| Pulmonary tuberculosis infection | ||||||
| Never users | 1.000 | 1.000 | ||||
| Ever users | 0.605 | (0.536–0.683) | <0.0001 | 0.431 | (0.373–0.497) | <0.0001 |
| Tertiles of cumulative duration of metformin therapy (months) | ||||||
| Never users | 1.000 | 1.000 | ||||
| <27.1 | 1.185 | (1.038–1.352) | 0.0119 | 0.852 | (0.710–1.023) | 0.0853 |
| 27.1–58.3 | 0.551 | (0.478–0.636) | <0.0001 | 0.517 | (0.426–0.629) | <0.0001 |
| >58.3 | 0.207 | (0.171–0.249) | <0.0001 | 0.189 | (0.151–0.237) | <0.0001 |
| Tertiles of cumulative dose of metformin therapy (mg) | ||||||
| Never users | 1.000 | 1.000 | ||||
| <817,000 | 1.101 | (0.963–1.259) | 0.1579 | 0.813 | (0.678–0.976) | 0.0262 |
| 817,000–2,047,180 | 0.566 | (0.491–0.652) | <0.0001 | 0.440 | (0.360–0.537) | <0.0001 |
| >2,047,180 | 0.266 | (0.224–0.316) | <0.0001 | 0.228 | (0.184–0.283) | <0.0001 |
© 2018 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tseng, C.-H. Metformin Decreases Risk of Tuberculosis Infection in Type 2 Diabetes Patients. J. Clin. Med. 2018, 7, 264. https://doi.org/10.3390/jcm7090264
Tseng C-H. Metformin Decreases Risk of Tuberculosis Infection in Type 2 Diabetes Patients. Journal of Clinical Medicine. 2018; 7(9):264. https://doi.org/10.3390/jcm7090264
Chicago/Turabian StyleTseng, Chin-Hsiao. 2018. "Metformin Decreases Risk of Tuberculosis Infection in Type 2 Diabetes Patients" Journal of Clinical Medicine 7, no. 9: 264. https://doi.org/10.3390/jcm7090264
APA StyleTseng, C.-H. (2018). Metformin Decreases Risk of Tuberculosis Infection in Type 2 Diabetes Patients. Journal of Clinical Medicine, 7(9), 264. https://doi.org/10.3390/jcm7090264





