Association between Urinary Calcium Excretion and Estimated Glomerular Filtration Rate Decline in Patients with Type 2 Diabetes Mellitus: A Retrospective Single-center Observational Study
Abstract
:1. Introduction
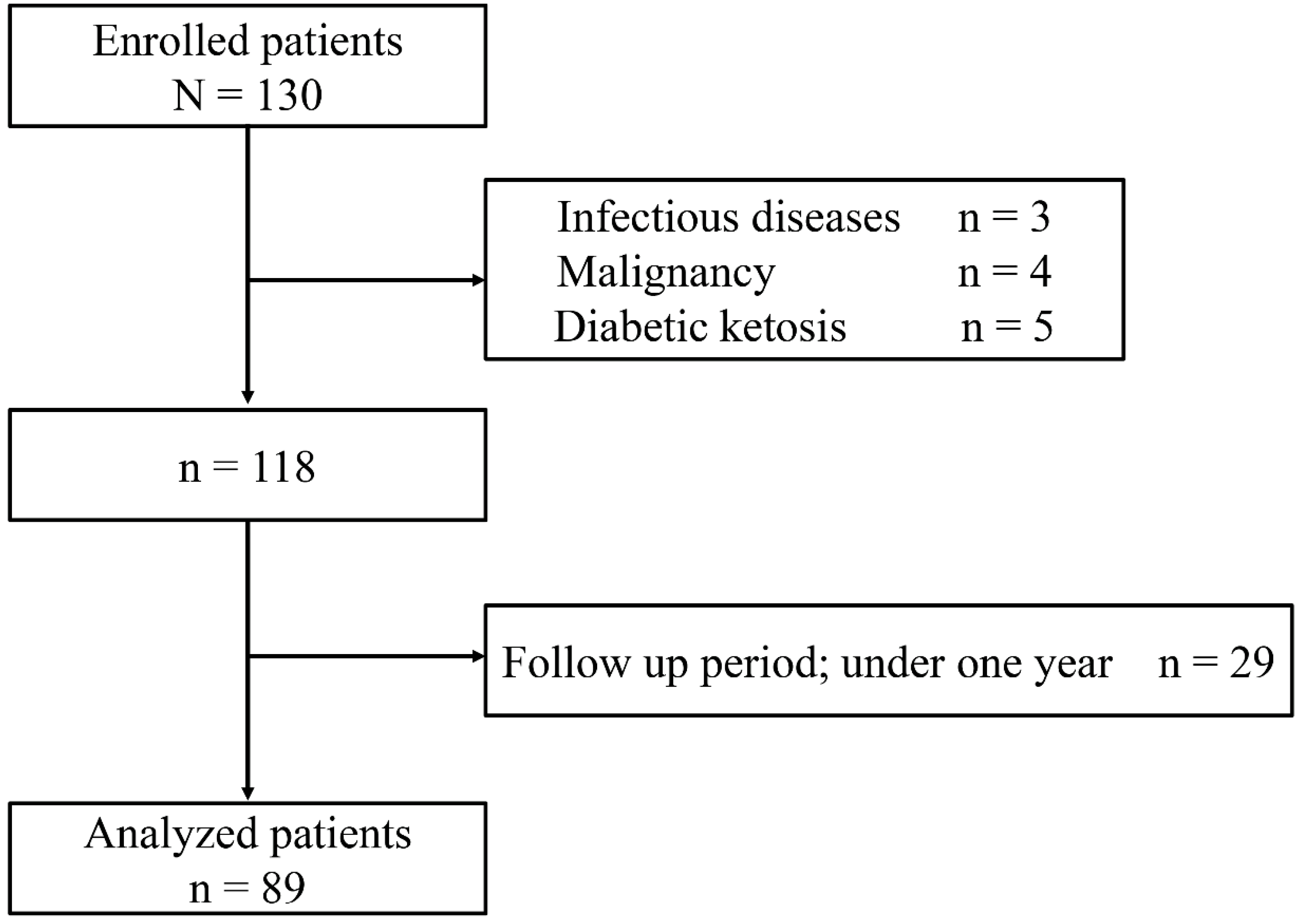
2. Materials and Methods
2.1. Study Participants
2.2. Data Collection
2.3. Ethical Approval
2.4. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Chamberlain, J.J.; Rhinehart, A.S.; Shaefer, C.F., Jr.; Neuman, A. Diagnosis and management of diabetes: Synopsis of the 2016 American diabetes association standards of medical care in diabetes. Ann. Clin. Med. 2016, 164, 542–552. [Google Scholar] [CrossRef] [PubMed]
- Gall, M.A.; Nielsen, F.S.; Smidt, U.M.; Parving, H.H. The course of kidney function in type 2 (non-insulin-dependent) diabetic patients with diabetic nephropathy. Diabetologia 1993, 36, 1071–1078. [Google Scholar] [CrossRef] [PubMed]
- Krolewski, A.S.; Skupien, J.; Rossing, P.; Warram, J.H. Fast renal decline to end-stage renal disease: An unrecognized feature of nephropathy in diabetes. Kidney Int. 2017, 91, 1300–1311. [Google Scholar] [CrossRef] [PubMed]
- Kuro-o, M. Klotho, phosphate and fgf-23 in ageing and disturbed mineral metabolism. Nat. Rev. Nephrol. 2013, 9, 650–660. [Google Scholar] [CrossRef] [PubMed]
- Araki, S.; Haneda, M.; Koya, D.; Kondo, K.; Tanaka, S.; Arima, H.; Kume, S.; Nakazawa, J.; Chin-Kanasaki, M.; Ugi, S.; et al. Urinary Potassium Excretion and Renal and Cardiovascular Complications in Patients with Type 2 Diabetes and Normal Renal Function. Clin. J. Am. Soc. Nephrol. 2015, 10, 2152–2158. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ueda, Y.; Ookawara, S.; Ito, K.; Miyazawa, H.; Kaku, Y.; Hoshino, T.; Tabei, K.; Morishita, Y. Changes in urinary potassium excretion in patients with chronic kidney disease. Kidney. Res. Clin. Pract. 2016, 35, 78–83. [Google Scholar] [CrossRef] [PubMed]
- Raskin, P.; Stevenson, M.R.; Barilla, D.E.; Pak, C.Y. The hypercalciuria of diabetes mellitus: Its amelioration with insulin. Clin. Endocrinol. 1978, 9, 329–335. [Google Scholar] [CrossRef]
- Wahl, P.; Xie, H.; Scialla, J.; Anderson, C.A.; Bellovich, K.; Brecklin, C.; Chen, J.; Feldman, H.; Gutierrez, O.M.; Lash, J.; et al. Earlier onset and greater severity of disordered mineral metabolism in diabetic patients with chronic kidney disease. Diabetes Care 2012, 35, 994–1001. [Google Scholar] [CrossRef] [PubMed]
- Saito, T.; Murata, M.; Otani, T.; Tamemoto, H.; Kawakami, M.; Ishikawa, S.E. Association of subcutaneous and visceral fat mass with serum concentrations of adipokines in subjects with type 2 diabetes mellitus. Endocr. J. 2012, 59, 39–45. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kanda, Y. Investigation of the freely available easy-to-use software ‘EZR’ for medical statistics. Bone Marrow Transplant. 2013, 48, 452–458. [Google Scholar] [CrossRef] [PubMed]
- Rathod, A.; Bonny, O.; Guessous, I.; Suter, P.M.; Conen, D.; Erne, P.; Binet, I.; Gabutti, L.; Gallino, A.; Muggli, F.; et al. Association of urinary calcium excretion with serum calcium and vitamin d levels. Clin. J. Am. Soc. Nephrol. 2015, 10, 452–462. [Google Scholar] [CrossRef] [PubMed]
- Jorgetti, V.; dos Reis, L.M.; Ott, S.M. Ethnic differences in bone and mineral metabolism in healthy people and patients with CKD. Kidney Int. 2014, 85, 1283–1289. [Google Scholar] [CrossRef] [PubMed]
- Moor, M.B.; Bonny, O. Ways of calcium reabsorption in the kidney. Am. J. Physiol. 2016, 310, F1337–F1350. [Google Scholar] [CrossRef] [PubMed]
- Vaidya, V.S.; Niewczas, M.A.; Ficociello, L.H.; Johnson, A.C.; Collings, F.B.; Warram, J.H.; Krolewski, A.S.; Bonventre, J.V. Regression of microalbuminuria in type 1 diabetes is associated with lower levels of urinary tubular injury biomarkers, kidney injury molecule-1, and n-acetyl-beta-d-glucosaminidase. Kidney Int. 2011, 79, 464–470. [Google Scholar] [CrossRef] [PubMed]
- Nowak, N.; Skupien, J.; Niewczas, M.A.; Yamanouchi, M.; Major, M.; Croall, S.; Smiles, A.; Warram, J.H.; Bonventre, J.V.; Krolewski, A.S. Increased plasma kidney injury molecule-1 suggests early progressive renal decline in non-proteinuric patients with type 1 diabetes. Kidney Int. 2016, 89, 459–467. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Carvalho, J.A.; Tatsch, E.; Hausen, B.S.; Bollick, Y.S.; Moretto, M.B.; Duarte, T.; Duarte, M.M.; Londero, S.W.; Premaor, M.O.; Comim, F.V.; et al. Urinary kidney injury molecule-1 and neutrophil gelatinase-associated lipocalin as indicators of tubular damage in normoalbuminuric patients with type 2 diabetes. Clin. Biochem. 2016, 49, 232–236. [Google Scholar] [CrossRef] [PubMed]
- Coca, S.G.; Nadkarni, G.N.; Huang, Y.; Moledina, D.G.; Rao, V.; Zhang, J.; Ferket, B.; Crowley, S.T.; Fried, L.F.; Parikh, C.R. Plasma biomarkers and kidney function decline in early and established diabetic kidney disease. J. Am. Soc. Nephrol. 2017, 28, 2786–2793. [Google Scholar] [CrossRef] [PubMed]
- Kendrick, J.; Cheung, A.K.; Kaufman, J.S.; Greene, T.; Roberts, W.L.; Smits, G.; Chonchol, M. Fgf-23 associates with death, cardiovascular events, and initiation of chronic dialysis. J. Am. Soc. Nephrol. 2011, 22, 1913–1922. [Google Scholar] [CrossRef] [PubMed]
- Fliser, D.; Kollerits, B.; Neyer, U.; Ankerst, D.P.; Lhotta, K.; Lingenhel, A.; Ritz, E.; Kronenberg, F.; Kuen, E.; Konig, P.; et al. Fibroblast growth factor 23 (FGF23) predicts progression of chronic kidney disease: The mild to moderate kidney disease (MMKD) study. J. Am. Soc. Nephrol. 2007, 18, 2600–2608. [Google Scholar] [CrossRef] [PubMed]
- Hamano, T.; Nakano, C.; Obi, Y.; Fujii, N.; Matsui, I.; Tomida, K.; Mikami, S.; Inoue, K.; Shimomura, A.; Kusunoki, Y.; et al. Fibroblast growth factor 23 and 25-hydroxyvitamin d levels are associated with estimated glomerular filtration rate decline. Kidney Int. Suppl. 2013, 3, 469–475. [Google Scholar] [CrossRef] [PubMed]
- Altemtam, N.; Russell, J.; El Nahas, M. A study of the natural history of diabetic kidney disease (DKD). Nephrol. Dial. Transplant. 2012, 27, 1847–1854. [Google Scholar] [CrossRef] [PubMed]
- Isakova, T.; Wahl, P.; Vargas, G.S.; Gutierrez, O.M.; Scialla, J.; Xie, H.; Appleby, D.; Nessel, L.; Bellovich, K.; Chen, J.; et al. Fibroblast growth factor 23 is elevated before parathyroid hormone and phosphate in chronic kidney disease. Kidney Int. 2011, 79, 1370–1378. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dhayat, N.A.; Ackermann, D.; Pruijm, M.; Ponte, B.; Ehret, G.; Guessous, I.; Leichtle, A.B.; Paccaud, F.; Mohaupt, M.; Fiedler, G.M.; et al. Fibroblast growth factor 23 and markers of mineral metabolism in individuals with preserved renal function. Kidney Int. 2016, 90, 648–657. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Andrukhova, O.; Smorodchenko, A.; Egerbacher, M.; Streicher, C.; Zeitz, U.; Goetz, R.; Shalhoub, V.; Mohammadi, M.; Pohl, E.E.; Lanske, B.; et al. FGF23 promotes renal calcium reabsorption through the trpv5 channel. EMBO J. 2014, 33, 229–246. [Google Scholar] [CrossRef] [PubMed]
- Drew, D.A.; Katz, R.; Kritchevsky, S.; Ix, J.; Shlipak, M.; Gutierrez, O.M.; Newman, A.; Hoofnagle, A.; Fried, L.; Semba, R.D.; et al. Association between soluble klotho and change in kidney function: The health aging and body composition study. J. Am. Soc. Nephrol. 2017, 28, 1859–1866. [Google Scholar] [CrossRef] [PubMed]
- John, G.B.; Cheng, C.Y.; Kuro-o, M. Role of klotho in aging, phosphate metabolism, and CKD. Am. J. Kidney Dis. 2011, 58, 127–134. [Google Scholar] [CrossRef] [PubMed]
- Van Abel, M.; Hoenderop, J.G.; van der Kemp, A.W.; Friedlaender, M.M.; van Leeuwen, J.P.; Bindels, R.J. Coordinated control of renal Ca2+ transport proteins by parathyroid hormone. Kidney Int. 2005, 68, 1708–1721. [Google Scholar] [CrossRef] [PubMed]
- Reyes-Garcia, R.; Garcia-Martin, A.; Garcia-Fontana, B.; Morales-Santana, S.; Rozas-Moreno, P.; Munoz-Torres, M. Fgf23 in type 2 diabetic patients: Relationship with bone metabolism and vascular disease. Diabetes Care 2014, 37, e89–e90. [Google Scholar] [CrossRef] [PubMed]
| Variables and Parameters | Over All (n = 89) | Non-Progressed (n = 44) | Progressed (n = 45) | p Value |
|---|---|---|---|---|
| Age (years) | 63 ± 11 | 59 ± 12 | 67 ± 8.3 | <0.001 |
| sex, male (%) | 45 (51) | 26 (59) | 19 (42) | 0.140 |
| BMI (kg/m2) | 25.1 ± 4.50 | 25.3 ± 5.1 | 24.9 ± 4.0 | 0.747 |
| Duration of Diabetes (years) | 12 ± 8.0 | 10 ± 7.1 | 14 ± 8.3 | 0.022 |
| Hypertension, n (%) | 49 (55) | 20 (45) | 29 (64) | 0.090 |
| Smoking status, n (%) | 27 (30) | 14 (32) | 13 (29) | 0.820 |
| HbA1c (mmol/L) | 78.0 ± 16.0 | 78.7 ± 17.6 | 77.2 ± 14.4 | 0.661 |
| Systolic BP (mmHg) | 132 ± 19 | 127 ± 18 | 136 ± 19 | 0.025 |
| Diastolic BP (mmHg) | 74 ± 13 | 75 ± 10 | 74 ± 16 | 0.795 |
| TC (mmol/L) | 5.05 ± 1.01 | 5.13 ± 1.02 | 4.98 ± 1.01 | 0.464 |
| TG (mmol/L) | 2.91 (2.09–4.14) | 2.90 (2.27–3.70) | 3.10 (2.07–4.60) | 0.733 |
| HDL-C (mmol/L) | 1.25 ± 0.42 | 1.30 ± 0.46 | 1.21 ± 0.37 | 0.275 |
| Baseline eGFR (mL/min/1.73 m2) | 74 (59–88) | 72 (58–86) | 80 (64–93) | 0.112 |
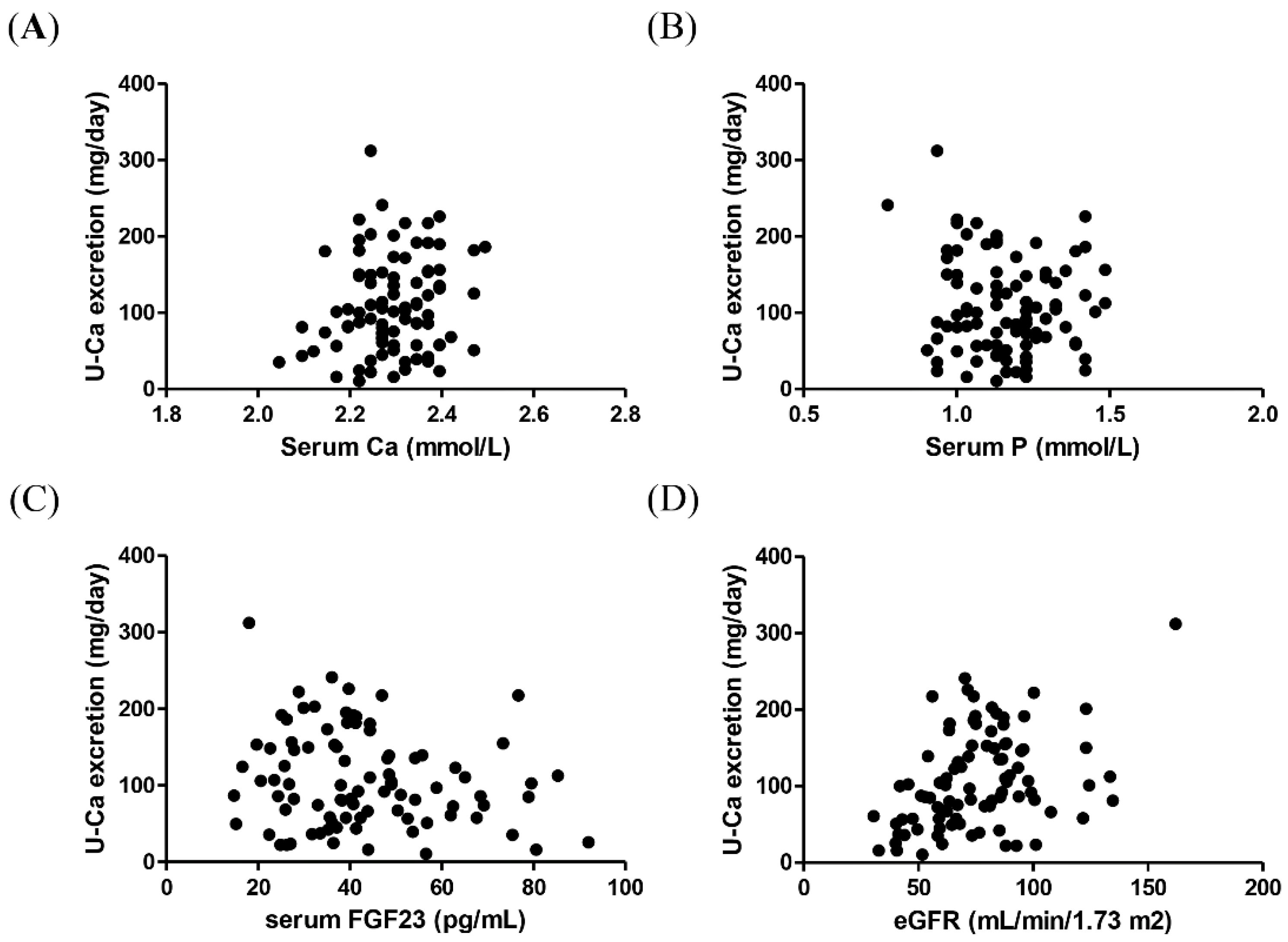
| Serum calcium (mmol/L) | 2.29 ± 0.09 | 2.29 ± 0.09 | 2.29 ± 0.09 | 0.945 |
| Serum phosphate (mmol/L) | 1.16 ± 0.154 | 1.17 ± 0.177 | 1.16 ± 0.130 | 0.791 |
| serum FGF23 (ng/L) | 40 (31.0–52.6) | 36 (27–45) | 44 (36–59) | 0.003 |
| Urinary albumin excretion (mg/day) | 12 (7.0–67) | 10 (4.8–19) | 16 (7.7–121) | 0.041 |
| Urinary calcium excretion (mg/day) | 97 (58–150) | 108 (72–154) | 82 (51–146) | 0.150 |
| Follow-up time (years) | 7.2 ± 1.0 | 7.3 ± 0.93 | 7.2 ± 1.1 | 0.578 |
| eGFR after follow up (mL/min/1.73 m2) | 62 (48–82) | 76 (57–88) | 54 (42–72) | <0.001 |
| eGFR decline speed (/year) | −1.1 (−2.6–0.19) | −0.17 (−0.44–0.94) | −2.6 (−5.1–2.0) | <0.001 |
| OR | 95% CI | p Value | OR | 95% CI | p Value | |
|---|---|---|---|---|---|---|
| Age (>62 years) | 1.9 | 0.65–5.6 | 0.244 | |||
| Male | 1.4 | 0.50–4.1 | 0.494 | |||
| BMI (>24.5 kg/m2) | 0.69 | 0.24–2.0 | 0.498 | |||
| Duration of diabetes (>10.5 years) | 1.4 | 0.49–4.2 | 0.521 | |||
| Hypertension | 1.5 | 0.53–4.2 | 0.454 | |||
| Smoking status | 1.1 | 0.38–3.2 | 0.862 | |||
| HbA1c (>76 mmol/L) | 0.96 | 0.37–2.5 | 0.925 | |||
| Baseline eGFR (>77 mL/min/1.73 m2) | 4.6 | 1.3–16.1 | 0.018 | 3.3 | 1.1–9.7 | 0.033 |
| Log serum FGF23 (>1.61) | 1.3 | 0.48–3.6 | 0.599 | |||
| Log Urinary albumin excretion (>1.14) | 1.2 | 0.45–3.4 | 0.692 | |||
| Log Urinary calcium excretion (≤1.99) | 4.3 | 1.5–12.2 | 0.005 | 6.1 | 2.1–18.0 | 0.001 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yamada, H.; Funazaki, S.; Suzuki, D.; Saikawa, R.; Yoshida, M.; Kakei, M.; Ishikawa, S.-e.; Morisita, Y.; Hara, K. Association between Urinary Calcium Excretion and Estimated Glomerular Filtration Rate Decline in Patients with Type 2 Diabetes Mellitus: A Retrospective Single-center Observational Study. J. Clin. Med. 2018, 7, 171. https://doi.org/10.3390/jcm7070171
Yamada H, Funazaki S, Suzuki D, Saikawa R, Yoshida M, Kakei M, Ishikawa S-e, Morisita Y, Hara K. Association between Urinary Calcium Excretion and Estimated Glomerular Filtration Rate Decline in Patients with Type 2 Diabetes Mellitus: A Retrospective Single-center Observational Study. Journal of Clinical Medicine. 2018; 7(7):171. https://doi.org/10.3390/jcm7070171
Chicago/Turabian StyleYamada, Hodaka, Shunsuke Funazaki, Daisuke Suzuki, Rika Saikawa, Masashi Yoshida, Masafumi Kakei, San-e Ishikawa, Yoshiyuki Morisita, and Kazuo Hara. 2018. "Association between Urinary Calcium Excretion and Estimated Glomerular Filtration Rate Decline in Patients with Type 2 Diabetes Mellitus: A Retrospective Single-center Observational Study" Journal of Clinical Medicine 7, no. 7: 171. https://doi.org/10.3390/jcm7070171





