Baseline Apparent Diffusion Coefficient as a Predictor of Response to Liver-Directed Therapies in Hepatocellular Carcinoma
Abstract
1. Introduction
2. Material and Methods
2.1. Study Population
2.2. MRI and DWI Protocol
2.3. Procedural Techniques
2.4. DEE Chemoembolization
2.5. Radioembolization
2.6. Image Analysis
2.7. Covariates
2.8. Statistical Analysis
3. Results
4. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Llovet, J.M.; Real, M.I.; Montañá, X.; Planas, R.; Coll, S.; Aponte, J.; Ayuso, C.; Sala, M.; Muchart, J.; Solà, R.; et al. Barcelona Liver Cancer Group Arterial embolisation or chemoembolisation versus symptomatic treatment in patients with unresectable hepatocellular carcinoma: A randomised controlled trial. Lancet 2002, 359, 1734–1739. [Google Scholar] [CrossRef]
- Lo, C.-M.; Ngan, H.; Tso, W.-K.; Liu, C.-L.; Lam, C.-M.; Poon, R.T.-P.; Fan, S.-T.; Wong, J. Randomized controlled trial of transarterial lipiodol chemoembolization for unresectable hepatocellular carcinoma. Hepatology 2002, 35, 1164–1171. [Google Scholar] [CrossRef] [PubMed]
- Burrel, M.; Reig, M.; Forner, A.; Barrufet, M.; de Lope, C.R.; Tremosini, S.; Ayuso, C.; Llovet, J.M.; Real, M.I.; Bruix, J. Survival of patients with hepatocellular carcinoma treated by transarterial chemoembolisation (TACE) using Drug Eluting Beads. Implications for clinical practice and trial design. J. Hepatol. 2012, 56, 1330–1335. [Google Scholar] [CrossRef] [PubMed]
- Bonekamp, S.; Jolepalem, P.; Lazo, M.; Gulsun, M.A.; Kiraly, A.P.; Kamel, I.R. Hepatocellular carcinoma: Response to TACE assessed with semiautomated volumetric and functional analysis of diffusion-weighted and contrast-enhanced MR imaging data. Radiology 2011, 260, 752–761. [Google Scholar] [CrossRef] [PubMed]
- Vandecaveye, V.; Michielsen, K.; De Keyzer, F.; Laleman, W.; Komuta, M.; Op de Beeck, K.; Roskams, T.; Nevens, F.; Verslype, C.; Maleux, G. Chemoembolization for hepatocellular carcinoma: 1-month response determined with apparent diffusion coefficient is an independent predictor of outcome. Radiology 2014, 270, 747–757. [Google Scholar] [CrossRef] [PubMed]
- Hompland, T.; Ellingsen, C.; Galappathi, K.; Rofstad, E.K. DW-MRI in assessment of the hypoxic fraction, interstitial fluid pressure, and metastatic propensity of melanoma xenografts. BMC Cancer 2014, 14, 92. [Google Scholar] [CrossRef] [PubMed]
- Sheth, R.A.; Hesketh, R.; Kong, D.S.; Wicky, S.; Oklu, R. Barriers to drug delivery in interventional oncology. J. Vasc. Interv. Radiol. 2013, 24, 1201–1207. [Google Scholar] [CrossRef] [PubMed]
- Minchinton, A.I.; Tannock, I.F. Drug penetration in solid tumours. Nat. Rev. Cancer 2006, 6, 583–592. [Google Scholar] [CrossRef] [PubMed]
- Ariffin, A.B.; Forde, P.F.; Jahangeer, S.; Soden, D.M.; Hinchion, J. Releasing pressure in tumors: What do we know so far and where do we go from here? A review. Cancer Res. 2014, 74, 2655–2662. [Google Scholar] [CrossRef] [PubMed]
- Salem, R.; Lewandowski, R.J.; Mulcahy, M.F.; Riaz, A.; Ryu, R.K.; Ibrahim, S.; Atassi, B.; Baker, T.; Gates, V.; Miller, F.H.; et al. Radioembolization for hepatocellular carcinoma using Yttrium-90 microspheres: A comprehensive report of long-term outcomes. Gastroenterology 2010, 138, 52–64. [Google Scholar] [CrossRef] [PubMed]
- Ho, S.; Lau, W.Y.; Leung, T.W.; Chan, M.; Ngar, Y.K.; Johnson, P.J.; Li, A.K. Partition model for estimating radiation doses from yttrium-90 microspheres in treating hepatic tumours. Eur. J. Nucl. Med. 1996, 23, 947–952. [Google Scholar] [CrossRef] [PubMed]
- Ludwig, J.M.; Camacho, J.C.; Kokabi, N.; Xing, M.; Kim, H.S. the role of diffusion-weighted imaging (dwi) in locoregional therapy outcome prediction and response assessment for Hepatocellular Carcinoma (HCC): The new era of functional imaging biomarkers. Diagnostics (Basel) 2015, 5, 546–563. [Google Scholar] [CrossRef] [PubMed]
- Kokabi, N.; Ludwig, J.M.; Camacho, J.C.; Xing, M.; Mittal, P.K.; Kim, H.S. Baseline and early MR apparent diffusion coefficient quantification as a predictor of response of unresectable hepatocellular carcinoma to doxorubicin drug-eluting bead chemoembolization. J. Vasc. Interv. Radiol. 2015, 26, 1777–1786. [Google Scholar] [CrossRef] [PubMed]
- Dong, S.; Ye, X.-D.; Yuan, Z.; Xu, L.-C.; Xiao, X.-S. Relationship of apparent diffusion coefficient to survival for patients with unresectable primary hepatocellular carcinoma after chemoembolization. Eur. J. Radiol. 2012, 81, 472–477. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Z.; Ye, X.-D.; Dong, S.; Xu, L.-C.; Xu, X.-Y.; Liu, S.-Y.; Xiao, X.-S. Role of magnetic resonance diffusion-weighted imaging in evaluating response after chemoembolization of hepatocellular carcinoma. Eur. J. Radiol. 2010, 75, e9–e14. [Google Scholar] [CrossRef] [PubMed]
- Mannelli, L.; Kim, S.; Hajdu, C.H.; Babb, J.S.; Taouli, B. Serial diffusion-weighted MRI in patients with hepatocellular carcinoma: Prediction and assessment of response to transarterial chemoembolization. Preliminary experience. Eur. J. Radiol. 2013, 82, 577–582. [Google Scholar] [CrossRef] [PubMed]
- Kokabi, N.; Camacho, J.C.; Xing, M.; Edalat, F.; Mittal, P.K.; Kim, H.S. Immediate post-doxorubicin drug-eluting beads chemoembolization MR Apparent diffusion coefficient quantification predicts response in unresectable hepatocellular carcinoma: A pilot study. J. Magn. Reson. Imaging 2015, 42, 981–989. [Google Scholar] [CrossRef] [PubMed]
- Sahin, H.; Harman, M.; Cinar, C.; Bozkaya, H.; Parildar, M.; Elmas, N. Evaluation of treatment response of chemoembolization in hepatocellular carcinoma with diffusion-weighted imaging on 3.0-T MR imaging. J. Vasc. Interv. Radiol. 2012, 23, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Kubota, K.; Yamanishi, T.; Itoh, S.; Murata, Y.; Miyatake, K.; Yasunami, H.; Morio, K.; Hamada, N.; Nishioka, A.; Ogawa, Y. Role of diffusion-weighted imaging in evaluating therapeutic efficacy after transcatheter arterial chemoembolization for hepatocellular carcinoma. Oncol. Rep. 2010, 24, 727–732. [Google Scholar] [CrossRef] [PubMed]
- Vouche, M.; Salem, R.; Lewandowski, R.J.; Miller, F.H. Can volumetric ADC measurement help predict response to Y90 radioembolization in HCC? Abdom. Imaging 2015, 40, 1471–1480. [Google Scholar] [CrossRef] [PubMed]
- Mori, Y.; Tamai, H.; Shingaki, N.; Moribata, K.; Deguchi, H.; Ueda, K.; Inoue, I.; Maekita, T.; Iguchi, M.; Kato, J.; et al. Signal intensity of small hepatocellular carcinoma on apparent diffusion coefficient mapping and outcome after radiofrequency ablation. Hepatol. Res. 2015, 45, 75–87. [Google Scholar] [CrossRef] [PubMed]
- Razek, A.A.K.A.; Gaballa, G.; Denewer, A.; Nada, N. Invasive ductal carcinoma: Correlation of apparent diffusion coefficient value with pathological prognostic factors. NMR Biomed. 2010, 23, 619–623. [Google Scholar] [CrossRef] [PubMed]
- Turkbey, B.; Shah, V.P.; Pang, Y.; Bernardo, M.; Xu, S.; Kruecker, J.; Locklin, J.; Baccala, A.A.; Rastinehad, A.R.; Merino, M.J.; et al. Is apparent diffusion coefficient associated with clinical risk scores for prostate cancers that are visible on 3-T MR images? Radiology 2011, 258, 488–495. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.-S.; Zhang, X.-P.; Tang, L.; Ji, J.-F.; Gu, J.; Cai, Y.; Zhang, X.-Y. Locally advanced rectal carcinoma treated with preoperative chemotherapy and radiation therapy: Preliminary analysis of diffusion-weighted MR imaging for early detection of tumor histopathologic downstaging. Radiology 2010, 254, 170–178. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, M.; Yamada, K.; Watanabe, Y.; Matsui, M.; Ida, M.; Fujiwara, S.; Shibata, E. Acute Stroke Imaging Standardization Group-Japan (ASIST-Japan) Investigators Variability in absolute apparent diffusion coefficient values across different platforms may be substantial: A multivendor, multi-institutional comparison study. Radiology 2008, 249, 624–630. [Google Scholar] [CrossRef] [PubMed]
- Lambregts, D.M.J.; Beets, G.L.; Maas, M.; Curvo-Semedo, L.; Kessels, A.G.H.; Thywissen, T.; Beets-Tan, R.G.H. Tumour ADC measurements in rectal cancer: Effect of ROI methods on ADC values and interobserver variability. Eur. Radiol. 2011, 21, 2567–2574. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zhou, M.; Liu, F.; Xiong, C.; Wang, W.; Cao, Q.; Wen, X.; Robertson, J.D.; Ji, X.; Wang, Y.A.; et al. Hepatocellular carcinoma: Intra-arterial delivery of doxorubicin-loaded hollow gold nanospheres for photothermal ablation-chemoembolization therapy in rats. Radiology 2016, 281, 427–435. [Google Scholar] [CrossRef] [PubMed]
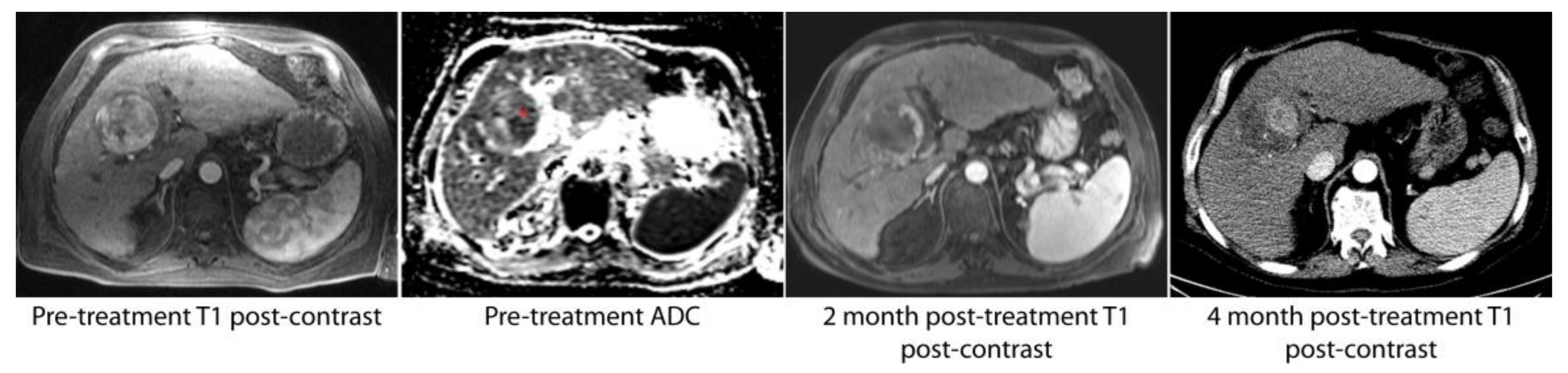
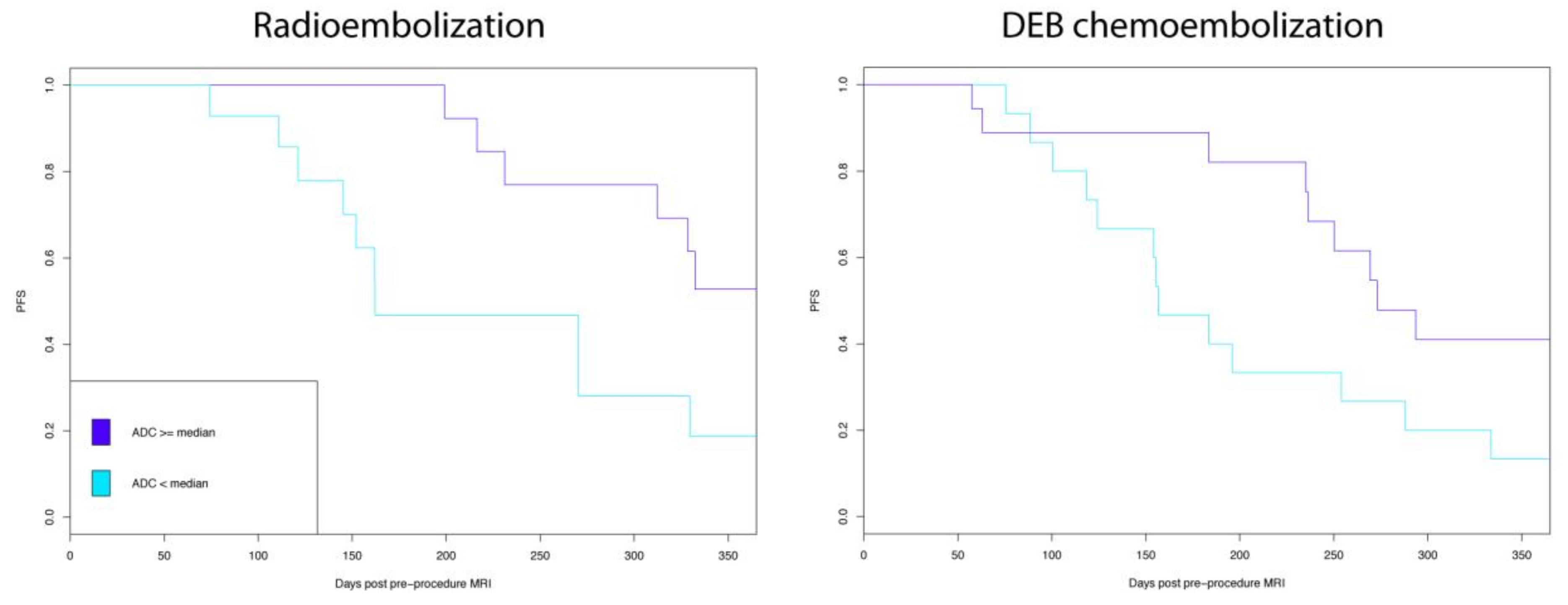
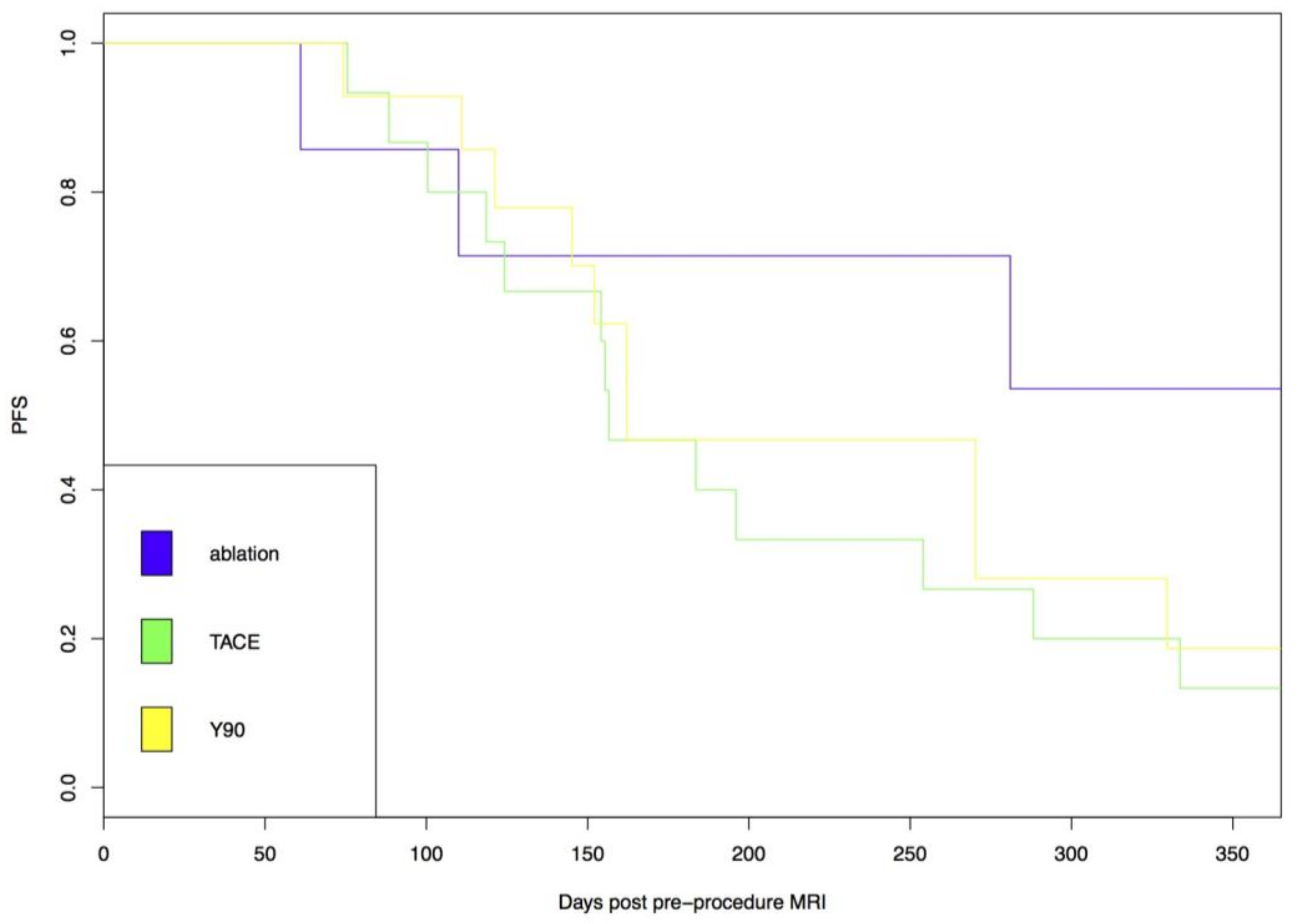
| DEE Chemoembolization | Radioembolization | P | |
|---|---|---|---|
| Patients (N/total, % total) | 32/58 (56%) | 26/58 (44%) | |
| Age (median, (range)in yrs) | 60 (21–81) | 69 (15–80) | 0.06 |
| Gender (M/F) | 27/5 | 19/7 | 0.34 |
| Index lesion size (median, (range) in cm) | 4 (1–17) | 6.7 (1.7–21.3) | <0.001 |
| Intrahepatic tumor multifocality | 0.12 | ||
| Single tumor | 10/32 | 4/26 | |
| 2–3 tumors | 10/32 | 5/26 | |
| >3 tumors | 12/32 | 17/26 | |
| Presence of extra-hepatic disease (N/total) | 1/32 | 8/26 | 0.007 |
| Presence of ascites (N/total) | 6/32 | 4/26 | 1.0 |
| Presence of vascular invasion (N/total) | 6/32 | 11/26 | 0.08 |
| Presence of lymph node metastasis (N, % total) | 0/32 | 6/26 | 0.005 |
| ADC value (median, (range) in 10−3 mm2/s) | 1.08 (0.528–2.71) | 0.99 (0.42–2.18) | 0.25 |
| ECOG status | 0.42 | ||
| ECOG 0 | 16/32 | 11/26 | |
| ECOG 1 | 14/32 | 15/26 | |
| ECOG 2 | 2/32 | 0/26 | |
| History of hepatic encephalopathy (N, % total) | 3/32 | 0/26 | 0.24 |
| Pre-procedure total bilirubin (median, (range) in mg/dL) | 1 (0.3–2.6) | 0.75 (0.3–2.5) | 0.06 |
| Pre-procedure serum albumin (median, (range) in g/dL) | 4.3 (3.0–5.1) | 4.2 (3.3–5.3) | 0.78 |
| Childs-Pugh score | 0.12 | ||
| Childs-Pugh A | 26/32 | 25/26 | |
| Childs-Pugh B | 6/32 | 1/26 | |
| Childs-Pugh C | 0/32 | 0/26 | |
| BCLC classification | 0.02 | ||
| BCLC A | 7/32 | 0/26 | |
| BCLC B | 7/32 | 5/26 | |
| BCLC C | 18/32 | 21/26 |
| Univariate P | Odds Ratio | Multivariate P | |
|---|---|---|---|
| Age | 0.59 | - | - |
| Gender | 0.08 | 2.7 (0.65–11.5) | 0.16 |
| Index lesion size | 0.86 | - | - |
| Childs-Pugh score | 0.61 | - | - |
| Tumor multifocality | 0.33 | - | - |
| Presence of vascular invasion | 0.53 | - | - |
| ADC ≥ 1.01 × 10−3 mm2/s | 0.03 | 4.4 (1.2–18.6) | 0.03 |
| ECOG status | 0.79 | - | - |
| Pre-procedure total bilirubin | 0.30 | - | - |
| BCLC classification | 0.20 | - | - |
| Treatment modality | 0.15 | ||
| Doxorubicin dose | 0.44 | - | - |
| Yttrium-90 absorbed dose | 0.11 | − | − |
| Univariate P | Hazard Ratio | Multivariate P | |
|---|---|---|---|
| Age | 0.55 | - | - |
| Gender | 0.89 | - | - |
| Index lesion size | 0.96 | - | - |
| Tumor multifocality | 0.14 | - | - |
| Presence of extra-hepatic disease | 0.05 | 1.0 (0.2–4.3) | 0.98 |
| Presence of ascites | 0.05 | 2.0 (0.5–4.2) | 0.08 |
| Presence of vascular invasion | 0.93 | - | - |
| Presence of lymph node metastasis | 0.006 | 2.9 (0.3–15.3) | 0.2 |
| ADC ≥ 1.01 × 10−3 mm2/s | 0.006 | 0.44 (0.23–0.84) | 0.01 |
| ECOG status | 0.81 | - | - |
| Childs-Pugh score | 1.0 | - | - |
| BCLC classification | 0.59 | - | - |
| Treatment modality | 0.47 | - | - |
| Doxorubicin dose | 0.46 | - | - |
| Yttrium-90 absorbed dose | 0.44 | − | − |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Niekamp, A.; Abdel-Wahab, R.; Kuban, J.; Odisio, B.C.; Mahvash, A.; Hassan, M.M.; Qayyum, A.; Kaseb, A.; Sheth, R.A. Baseline Apparent Diffusion Coefficient as a Predictor of Response to Liver-Directed Therapies in Hepatocellular Carcinoma. J. Clin. Med. 2018, 7, 83. https://doi.org/10.3390/jcm7040083
Niekamp A, Abdel-Wahab R, Kuban J, Odisio BC, Mahvash A, Hassan MM, Qayyum A, Kaseb A, Sheth RA. Baseline Apparent Diffusion Coefficient as a Predictor of Response to Liver-Directed Therapies in Hepatocellular Carcinoma. Journal of Clinical Medicine. 2018; 7(4):83. https://doi.org/10.3390/jcm7040083
Chicago/Turabian StyleNiekamp, Andrew, Reham Abdel-Wahab, Joshua Kuban, Bruno C. Odisio, Armeen Mahvash, Manal M. Hassan, Aliya Qayyum, Ahmed Kaseb, and Rahul A. Sheth. 2018. "Baseline Apparent Diffusion Coefficient as a Predictor of Response to Liver-Directed Therapies in Hepatocellular Carcinoma" Journal of Clinical Medicine 7, no. 4: 83. https://doi.org/10.3390/jcm7040083
APA StyleNiekamp, A., Abdel-Wahab, R., Kuban, J., Odisio, B. C., Mahvash, A., Hassan, M. M., Qayyum, A., Kaseb, A., & Sheth, R. A. (2018). Baseline Apparent Diffusion Coefficient as a Predictor of Response to Liver-Directed Therapies in Hepatocellular Carcinoma. Journal of Clinical Medicine, 7(4), 83. https://doi.org/10.3390/jcm7040083




