Urinary Proteomics for the Early Diagnosis of Diabetic Nephropathy in Taiwanese Patients
Abstract
1. Introduction
2. Materials and Methods
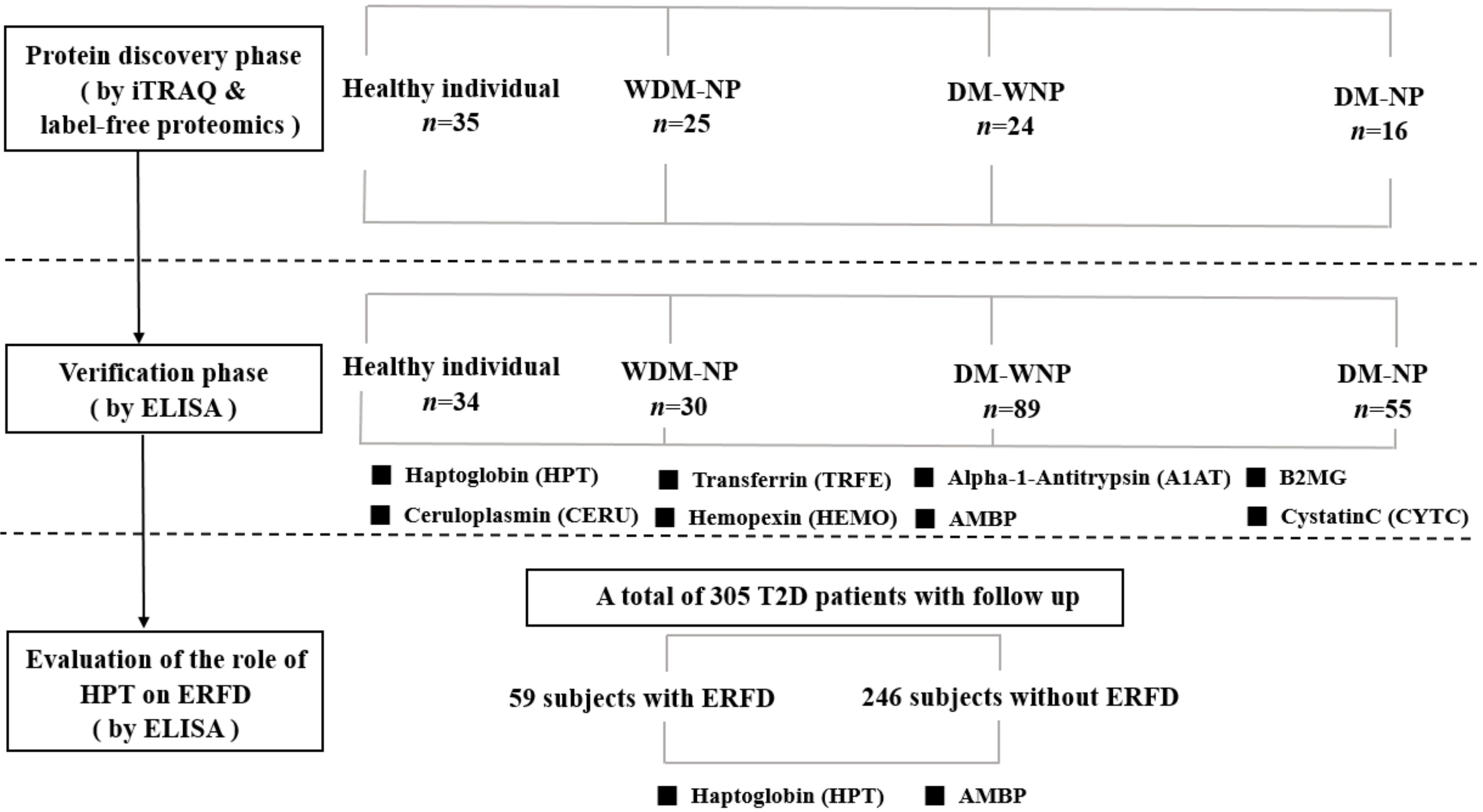
2.1. Study Population
2.1.1. Proteomic Discovery Phase
2.1.2. Verification Phase
2.1.3. Evaluation of the Role of HPT on ERFD
2.2. Method for the Proteomic Discovery Phase
2.2.1. Urine Sample Preparation for Proteomic Analysis
2.2.2. iTRAQ Labeling
2.2.3. Nano-LC-MS/MS
2.2.4. Label-Free Quantitative Proteomics
2.2.5. Protein Database Search
2.3. ELISA
2.4. Statistical Analysis
3. Results
3.1. Proteomic Discovery Phase
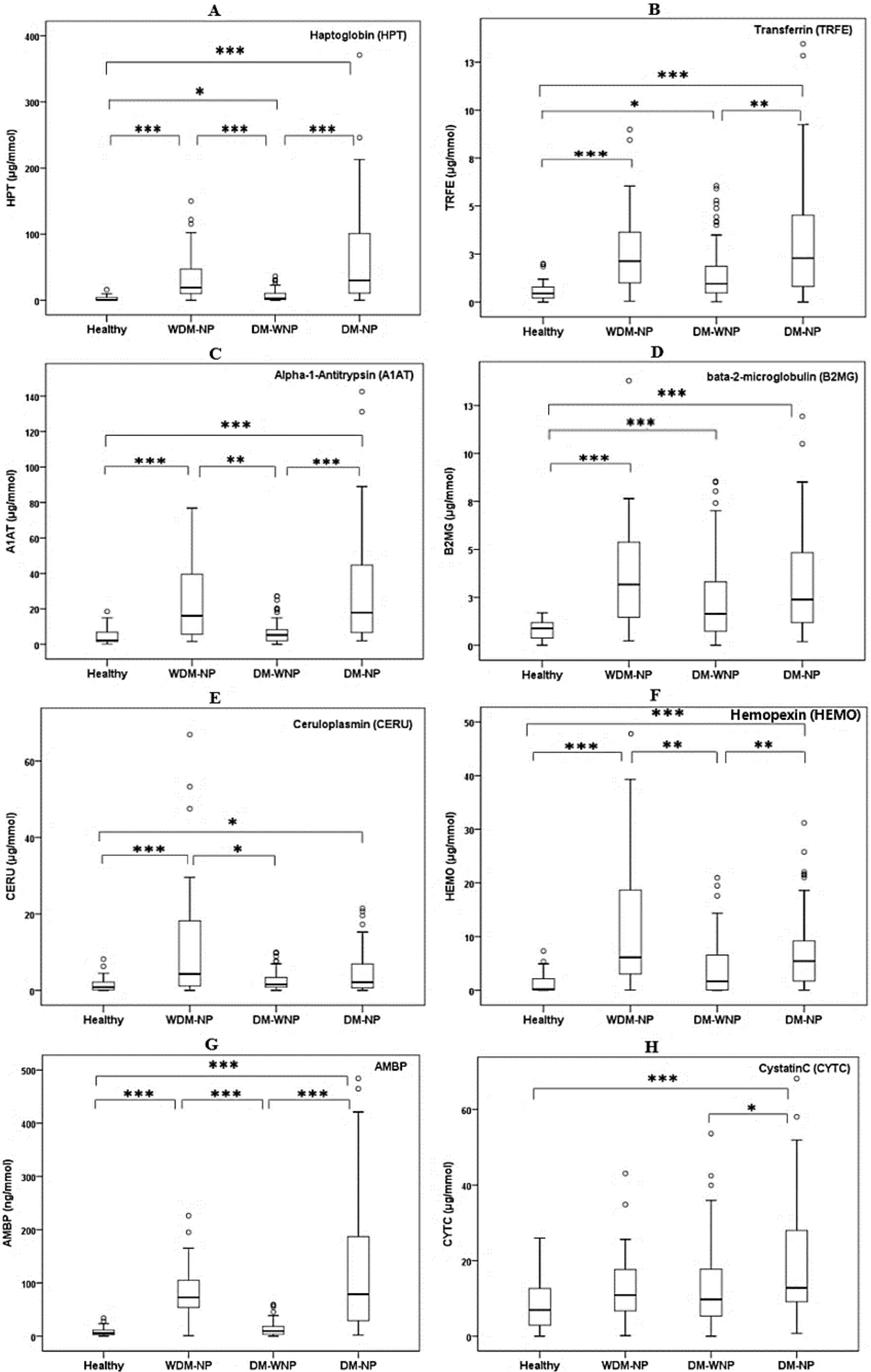
3.2. Verification Phase
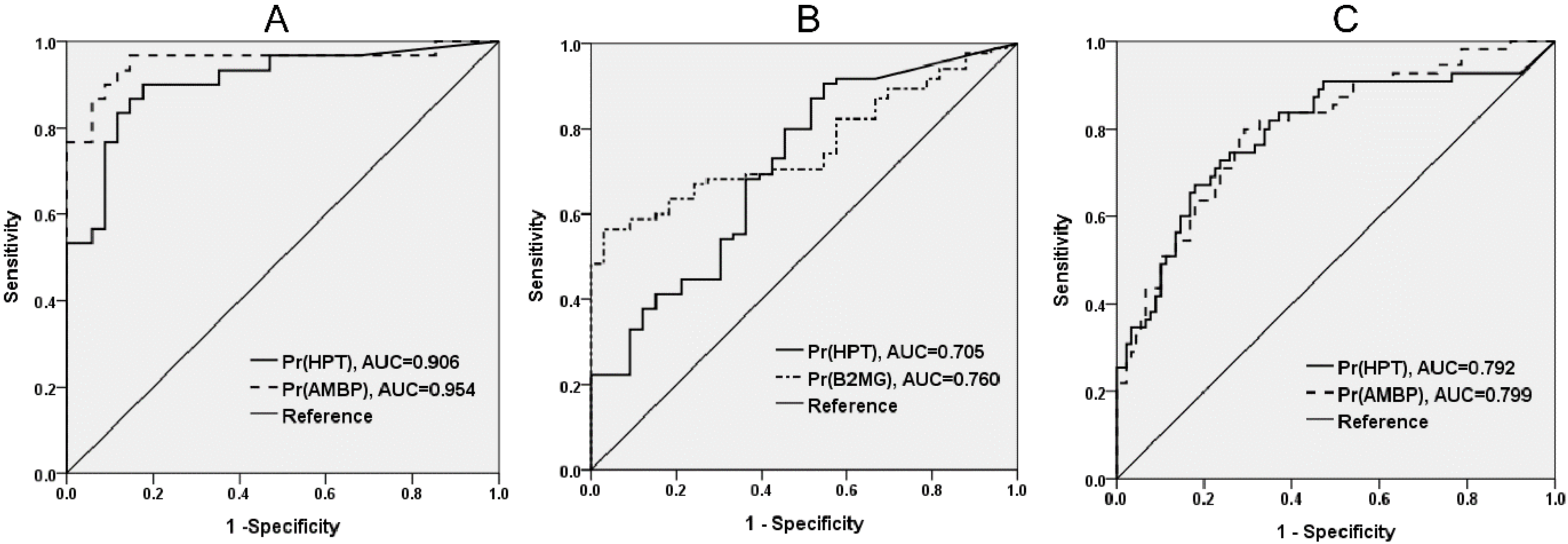
3.3. Evaluation of the Role of HPT on ERFD
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lewis, E.J.; Lewis, J.B. Treatment of diabetic nephropathy with angiotensin II receptor antagonist. Clin. Exp. Nephrol. 2003, 7, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Dihazi, H.; Muller, G.A.; Lindner, S.; Meyer, M.; Asif, A.R.; Oellerich, M.; Strutz, F. Characterization of diabetic nephropathy by urinary proteomic analysis: Identification of a processed ubiquitin form as a differentially excreted protein in diabetic nephropathy patients. Clin. Chem. 2007, 53, 1636–1645. [Google Scholar] [CrossRef] [PubMed]
- Rossing, K.; Christensen, P.K.; Hovind, P.; Tarnow, L.; Rossing, P.; Parving, H.H. Progression of nephropathy in type 2 diabetic patients. Kidney Int. 2004, 66, 1596–1605. [Google Scholar] [CrossRef] [PubMed]
- US Renal Data System. In Annual Data Report; National Institutes of Diabetes, Digestive and Kidney Diseases: Bethesda, MD, USA, 2002.
- Jain, S.; Rajput, A.; Kumar, Y.; Uppuluri, N.; Arvind, A.S.; Tatu, U. Proteomic analysis of urinary protein markers for accurate prediction of diabetic kidney disorder. J. Assoc. Physicians India 2005, 53, 513–520. [Google Scholar] [PubMed]
- Brosius, F.C.; Pennathur, S. How to find a prognostic biomarker for progressive diabetic nephropathy. Kidney Int. 2013, 83, 996–998. [Google Scholar] [CrossRef] [PubMed]
- Macisaac, R.J.; Jerums, G. Diabetic kidney disease with and without albuminuria. Curr. Opin. Nephrol. Hypertens. 2011, 20, 246–257. [Google Scholar] [CrossRef] [PubMed]
- Gerstein, H.C.; Mann, J.F.; Yi, Q.; Zinman, B.; Dinneen, S.F.; Hoogwerf, B.; Halle, J.P.; Young, J.; Rashkow, A.; Joyce, C.; et al. Albuminuria and risk of cardiovascular events, death, and heart failure in diabetic and nondiabetic individuals. JAMA 2001, 286, 421–426. [Google Scholar] [CrossRef] [PubMed]
- Al-Rubeaan, K.; Siddiqui, K.; Al-Ghonaim, M.A.; Youssef, A.M.; Al-Sharqawi, A.H.; AlNaqeb, D. Assessment of the diagnostic value of different biomarkers in relation to various stages of diabetic nephropathy in type 2 diabetic patients. Sci. Rep. 2017, 7, 2684. [Google Scholar] [CrossRef] [PubMed]
- Gluhovschi, C.; Gluhovschi, G.; Petrica, L.; Timar, R.; Velciov, S.; Ionita, I.; Kaycsa, A.; Timar, B. Urinary biomarkers in the assessment of early diabetic nephropathy. J. Diabetes Res. 2016, 2016, 4626125. [Google Scholar] [CrossRef] [PubMed]
- Stubendorff, B.; Finke, S.; Walter, M.; Kniemeyer, O.; von Eggeling, F.; Gruschwitz, T.; Steiner, T.; Ott, U.; Wolf, G.; Wunderlich, H.; et al. Urine protein profiling identified alpha-1-microglobulin and haptoglobin as biomarkers for early diagnosis of acute allograft rejection following kidney transplantation. World J. Urol. 2014, 32, 1619–1624. [Google Scholar] [CrossRef] [PubMed]
- Van der Leeuw, J.; Beulens, J.W.; van Dieren, S.; Schalkwijk, C.G.; Glatz, J.F.; Hofker, M.H.; Verschuren, W.M.; Boer, J.M.; van der Graaf, Y.; Visseren, F.L.; et al. Novel biomarkers to improve the prediction of cardiovascular event risk in type 2 diabetes mellitus. J. Am. Heart Assoc. 2016, 5. [Google Scholar] [CrossRef] [PubMed]
- Jin, J.; Ku, Y.H.; Kim, Y.; Kim, Y.; Kim, K.; Lee, J.Y.; Cho, Y.M.; Lee, H.K.; Park, K.S.; Kim, Y. Differential proteome profiling using iTRAQ in microalbuminuric and normoalbuminuric type 2 diabetic patients. Exp. Diabetes Res. 2012, 2012, 168602. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Cho, E.H.; Yoo, J.H.; Kim, P.K.; Shin, J.S.; Kim, M.R.; Kim, C.W. Proteome analysis of serum from type 2 diabetics with nephropathy. J. Proteome Res. 2007, 6, 735–743. [Google Scholar] [CrossRef] [PubMed]
- Nkuipou-Kenfack, E.; Duranton, F.; Gayrard, N.; Argiles, A.; Lundin, U.; Weinberger, K.M.; Dakna, M.; Delles, C.; Mullen, W.; Husi, H.; et al. Assessment of metabolomic and proteomic biomarkers in detection and prognosis of progression of renal function in chronic kidney disease. PloS ONE 2014, 9, e96955. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.H.; Huang, Y.H.; Chen, H.Y.; Khor, S.S.; Chang, Y.H.; Lin, Y.J.; Jen, C.L.; Lu, S.N.; Yang, H.I.; Nishida, N.; et al. Human leukocyte antigen variants and risk of hepatocellular carcinoma modified by hepatitis C virus genotypes: A genome-wide association study. Hepatology 2017. [Google Scholar] [CrossRef] [PubMed]
- Lindeman, R.D.; Tobin, J.; Shock, N.W. Longitudinal studies on the rate of decline in renal function with age. J. Am. Geriatr. Soc. 1985, 33, 278–285. [Google Scholar] [CrossRef] [PubMed]
- Levey, A.S.; Bosch, J.P.; Lewis, J.B.; Greene, T.; Rogers, N.; Roth, D. A more accurate method to estimate glomerular filtration rate from serum creatinine: A new prediction equation. Modification of diet in renal disease study group. Ann. Intern. Med. 1999, 130, 461–470. [Google Scholar] [CrossRef] [PubMed]
- DeLong, E.R.; DeLong, D.M.; Clarke-Pearson, D.L. Comparing the areas under two or more correlated receiver operating characteristic curves: A nonparametric approach. Biometrics 1988, 44, 837–845. [Google Scholar] [CrossRef] [PubMed]
- Levey, A.S.; de Jong, P.E.; Coresh, J.; El Nahas, M.; Astor, B.C.; Matsushita, K.; Gansevoort, R.T.; Kasiske, B.L.; Eckardt, K.U. The definition, classification, and prognosis of chronic kidney disease: A KDIGO controversies conference report. Kidney Int. 2011, 80, 17–28. [Google Scholar] [CrossRef] [PubMed]
- Shore, N.; Khurshid, R.; Saleem, M. Alpha-1 microglobulin: A marker for early detection of tubular disorders in diabetic nephropathy. JAMC 2010, 22, 53–55. [Google Scholar] [PubMed]
- Wu, J.; Chen, Y.D.; Yu, J.K.; Shi, X.L.; Gu, W. Analysis of urinary proteomic patterns for type 2 diabetic nephropathy by ProteinChip. Diabetes Res. Clin. Pract. 2011, 91, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.K.; Wang, Y.Y.; Liu, C.; Shi, T.T.; Lu, J.; Cao, X.; Yang, F.Y.; Feng, J.P.; Chen, C.; Ji, L.N.; et al. Urine proteome specific for eye damage can predict kidney damage in patients with type 2 diabetes: A case-control and a 5.3-year prospective cohort study. Diabetes Care 2017, 40, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Katnik, I.; Guszczynski, T.; Dobryszycka, W. Immunological comparison of glycopeptides obtained from haptoglobin types 1-1, 2-1 and 2-2. Arch. Immunol. Ther. Expe. 1984, 32, 111–120. [Google Scholar]
- Langlois, M.R.; Delanghe, J.R. Biological and clinical significance of haptoglobin polymorphism in humans. Clin. Chem. 1996, 42, 1589–1600. [Google Scholar] [PubMed]
- Lim, Y.K.; Jenner, A.; Ali, A.B.; Wang, Y.; Hsu, S.I.; Chong, S.M.; Baumman, H.; Halliwell, B.; Lim, S.K. Haptoglobin reduces renal oxidative DNA and tissue damage during phenylhydrazine-induced hemolysis. Kidney Int. 2000, 58, 1033–1044. [Google Scholar] [CrossRef] [PubMed]
- Bhensdadia, N.M.; Hunt, K.J.; Lopes-Virella, M.F.; Michael Tucker, J.; Mataria, M.R.; Alge, J.L.; Neely, B.A.; Janech, M.G.; Arthur, J.M. Urine haptoglobin levels predict early renal functional decline in patients with type 2 diabetes. Kidney Int. 2013, 83, 1136–1143. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.J.; Liu, S.; Wong, M.D.; Gurung, R.L.; Lim, S.C. Urinary haptoglobin predicts rapid renal function decline in asians with type 2 diabetes and early kidney disease. J. Clin. Endocrinol. Metab. 2016, 101, 3794–3802. [Google Scholar] [CrossRef] [PubMed]
- Zubiri, I.; Posada-Ayala, M.; Sanz-Maroto, A.; Calvo, E.; Martin-Lorenzo, M.; Gonzalez-Calero, L.; de la Cuesta, F.; Lopez, J.A.; Fernandez-Fernandez, B.; Ortiz, A.; et al. Diabetic nephropathy induces changes in the proteome of human urinary exosomes as revealed by label-free comparative analysis. J. Proteomics 2014, 96, 92–102. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Calero, L.; Martin-Lorenzo, M.; de la Cuesta, F.; Maroto, A.S.; Baldan-Martin, M.; Ruiz-Hurtado, G.; Pulido-Olmo, H.; Segura, J.; Barderas, M.G.; Ruilope, L.M.; et al. Urinary alpha-1 antitrypsin and CD59 glycoprotein predict albuminuria development in hypertensive patients under chronic renin-angiotensin system suppression. Cardiovasc. Diabetol. 2016, 15, 8. [Google Scholar] [CrossRef] [PubMed]
| Accession/ Protein | Proteomic Approach | WDM-NP: Healthy (Ratio) | Number of Quantitative Peptides | CV (%) | DM-WNP: Healthy (Ratio) | Number of Quantitative Peptides | CV (%) | DM-NP: DM-WNP (Ratio) | Number of Quantitative Peptides | CV (%) |
|---|---|---|---|---|---|---|---|---|---|---|
| TRFE_ HUMAN | ||||||||||
| Serotransferrin M.W.: 77.0 kDa | iTRAQ | 4.71 | 36 | 121.14 | 1.22 | 36 | 67.65 | 2.56 | 47 | 66.06 |
| label-free | 15.36 | 31 | 75.14 | 1.68 | 30 | 67.45 | 3.67 | 39 | 44.00 | |
| CERU_HUMAN | ||||||||||
| Ceruloplasmin M.W.: 122.1 kDa | iTRAQ | 1.87 | 4 | 28.26 | 1.27 | 4 | 25.30 | 1.72 | 5 | 29.68 |
| label-free | 5.18 | 15 | 46.62 | 2.08 | 11 | 36.90 | 1.98 | 14.00 | 18.50 | |
| HEMO_HUMAN | ||||||||||
| Hemopexin M.W.: 51.6 kDa | iTRAQ | 1.62 | 1 | N.D. | 1.08 | 1 | N.D. | N.D. | N.D. | N.D. |
| label-free | 2.08 | 12 | 28.91 | 0.99 | 12 | 63.44 | 3.08 | 11.00 | 21.52 | |
| A1AT_HUMAN | ||||||||||
| 𝛼-1-antitrypsin M.W.: 46.7 kDa | iTRAQ | 7.29 | 20 | 95.11 | 0.67 | 14 | 53.67 | 3.05 | 16 | 55.02 |
| label-free | 10.49 | 16 | 42.81 | 0.54 | 13 | 63.87 | 5.18 | 14.00 | 42.85 | |
| B2MG_HUMAN | ||||||||||
| β-2-microglobulin M.W.: 13.7 kDa | iTRAQ | 1.50 | 4 | 90.85 | 0.95 | 4 | 41.36 | 1.42 | 4 | 17.18 |
| label-free | 2.24 | 2 | 7.20 | 0.36 | 1 | N.D. | 1.62 | 2.00 | 19.02 | |
| HPT_HUMAN | ||||||||||
| Haptoglobin M.W.: 45.2 kDa | iTRAQ | 3.42 | 10 | 67.54 | 1.11 | 9 | 39.42 | 1.30 | 7 | 18.55 |
| label-free | 9.56 | 12 | 87.79 | 1.79 | 13 | 65.41 | 1.48 | 13.00 | 27.33 | |
| AMBP_HUMAN | ||||||||||
| Protein AMBP M.W.: 39.0 kDa | iTRAQ | 1.40 | 7 | 187.03 | 0.41 | 7 | 79.48 | 5.75 | 6 | 206.59 |
| label-free | 2.20 | 20 | 138.75 | 0.54 | 15 | 88.25 | 3.20 | 19.00 | 317.40 |
| Non-ERFD (n = 230) | ERFD (n = 59) | p-Value | |
|---|---|---|---|
| Age (years) | 55.06 (8.40) | 56.22 (7.94) | 0.340 |
| Diabetes duration (years) | 6.85 (6.92) | 8.11 (5.79) | 0.324 |
| Follow-up time (years) | 4.57 (1.52) | 3.37 (1.61) | <0.001 * |
| HbA1c (%) | 7.64 (1.52) | 7.91 (1.90) | 0.309 |
| Creatinine (mg/dL) | 0.77 (0.18) | 0.72 (0.19) | 0.053 |
| eGFR (mL/min per 1.73 m2) | 100.54 (22.39) | 113.36 (25.62) | <0.001 * |
| ACR (mg/g) | 25.68 (41.08) | 32.35 (51.24) | 0.293 |
| HCR (ng/mg) | 17.98 (64.80) | 42.97 (79.11) | 0.028 * |
| BMI (kg/m2) | 26.15 (3.71) | 26.20 (4.02) | 0.927 |
| SBP (mmHg) | 121.25 (17.20) | 127.63 (18.80) | 0.013 * |
| DBP (mmHg) | 72.70 (10.77) | 72.58 (10.59) | 0.939 |
| Model 1 ACR | Model 2 HCR | Model 3 HCR and ACR | |
|---|---|---|---|
| Follow-up time | 0.64 (0.53, 0.78) | 0.58 (0.47, 0.71) | 0.58 (0.47, 0.71) |
| SBP | 1.02 (1.00, 1.04) | 1.02 (1.00, 1.04) | 1.02 (1.00, 1.04) |
| eGFR | 1.02 (1.01, 1.04) | 1.02 (1.01, 1.04) | 1.02 (1.01, 1.04) |
| Lower conc. * | 1.00 | 1.00 | |
| Upper conc. * | 1.38 (0.65, 2.94) | 1.03 (0.47, 2.24) | |
| Lower conc. # | 1.00 | 1.00 | |
| Upper conc. # | 4.47 (2.20, 9.09) | 4.45 (2.17, 9.16) | |
| AUC value | 0.759 | 0.804 | 0.803 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liao, W.-L.; Chang, C.-T.; Chen, C.-C.; Lee, W.-J.; Lin, S.-Y.; Liao, H.-Y.; Wu, C.-M.; Chang, Y.-W.; Chen, C.-J.; Tsai, F.-J. Urinary Proteomics for the Early Diagnosis of Diabetic Nephropathy in Taiwanese Patients. J. Clin. Med. 2018, 7, 483. https://doi.org/10.3390/jcm7120483
Liao W-L, Chang C-T, Chen C-C, Lee W-J, Lin S-Y, Liao H-Y, Wu C-M, Chang Y-W, Chen C-J, Tsai F-J. Urinary Proteomics for the Early Diagnosis of Diabetic Nephropathy in Taiwanese Patients. Journal of Clinical Medicine. 2018; 7(12):483. https://doi.org/10.3390/jcm7120483
Chicago/Turabian StyleLiao, Wen-Ling, Chiz-Tzung Chang, Ching-Chu Chen, Wen-Jane Lee, Shih-Yi Lin, Hsin-Yi Liao, Chia-Ming Wu, Ya-Wen Chang, Chao-Jung Chen, and Fuu-Jen Tsai. 2018. "Urinary Proteomics for the Early Diagnosis of Diabetic Nephropathy in Taiwanese Patients" Journal of Clinical Medicine 7, no. 12: 483. https://doi.org/10.3390/jcm7120483
APA StyleLiao, W.-L., Chang, C.-T., Chen, C.-C., Lee, W.-J., Lin, S.-Y., Liao, H.-Y., Wu, C.-M., Chang, Y.-W., Chen, C.-J., & Tsai, F.-J. (2018). Urinary Proteomics for the Early Diagnosis of Diabetic Nephropathy in Taiwanese Patients. Journal of Clinical Medicine, 7(12), 483. https://doi.org/10.3390/jcm7120483




