Plasma Aldosterone Concentration as a Determinant for Statin Use among Middle-Aged Hypertensive Patients for Atherosclerotic Cardiovascular Disease
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Participants
2.2. Outcome Measures
2.3. Ethics Committee Approval
2.4. Statistical Analysis
3. Results
3.1. Patient Characteristics
3.2. The Predictive Power of the ACC/AHA Guidelines
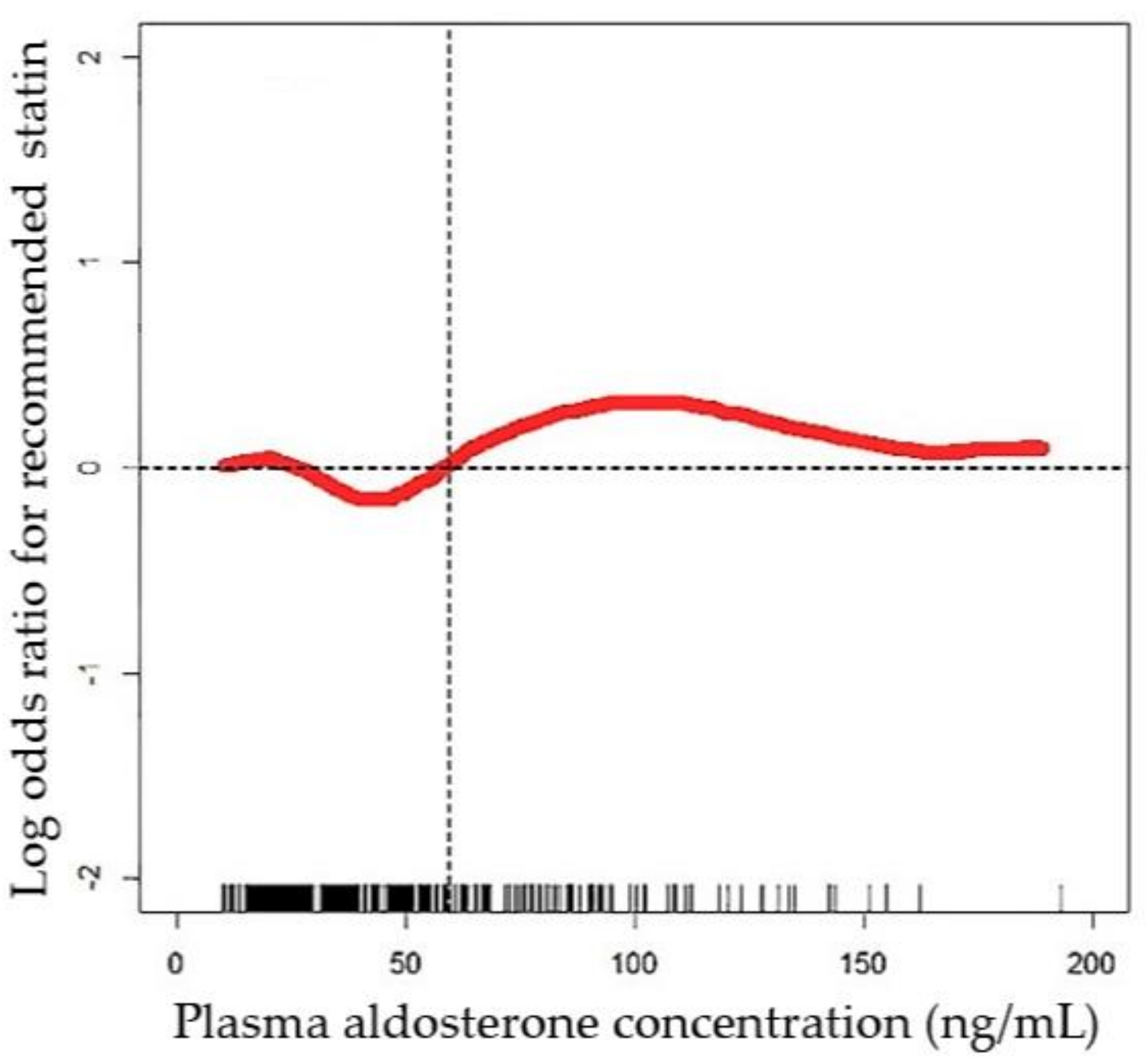
3.3. Effect of Plasma Aldosterone Concentration on Recommended Statin Use
3.4. The Comparison of Middle-Aged High-Risk Hypertensive Patients by the Plasma Aldosterone Concentration
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wu, K.D.; Liao, T.S.; Chen, Y.M.; Lai, M.K.; Chen, S.J.; Su, C.T.; Chu, T.S.; Chang, C.C.; Hsieh, B.S. Preoperative diagnosis and localization of aldosterone-producing adenoma by adrenal venous sampling after administration of metoclopramide. J. Formos. Med. Assoc. 2001, 100, 598–603. [Google Scholar] [PubMed]
- D’Agostino, R.B., Sr.; Vasan, R.S.; Pencina, M.J.; Wolf, P.A.; Cobain, M.; Massaro, J.M.; Kannel, W.B. General cardiovascular risk profile for use in primary care: The Framingham Heart Study. Circulation 2008, 117, 743–753. [Google Scholar] [CrossRef] [PubMed]
- Young, W.F., Jr. Minireview: Primary aldosteronism—Changing concepts in diagnosis and treatment. Endocrinology 2003, 144, 2008–2213. [Google Scholar] [CrossRef] [PubMed]
- Milliez, P.; Girerd, X.; Plouin, P.F.; Blacher, J.; Safar, M.E.; Mourad, J.J. Evidence for an increased rate of cardiovascular events in patients with primary aldosteronism. J. Am. Coll. Cardiol. 2005, 45, 1243–1248. [Google Scholar] [CrossRef] [PubMed]
- Wu, V.C.; Wang, S.M.; Chang, C.H.; Hu, Y.H.; Lin, L.Y.; Lin, Y.H.; Chueh, S.C.; Chen, L.; Wu, K.D. Long term outcome of Aldosteronism after target treatments. Sci. Rep. 2016, 6, 32103. [Google Scholar] [CrossRef] [PubMed]
- Wu, V.C.; Chueh, S.J.; Chen, L.; Chang, C.H.; Hu, Y.H.; Lin, Y.H.; Wu, K.D.; Yang, W.S.; Group, T.S. Risk of new-onset diabetes mellitus in primary aldosteronism: A population study over 5 years. J. Hypertens. 2017, 35, 1698–1708. [Google Scholar] [CrossRef] [PubMed]
- Stone, N.J.; Robinson, J.G.; Lichtenstein, A.H.; Bairey Merz, C.N.; Blum, C.B.; Eckel, R.H.; Goldberg, A.C.; Gordon, D.; Levy, D.; Lloyd-Jones, D.M.; et al. 2013 ACC/AHA Guideline on the Treatment of Blood Cholesterol to Reduce Atherosclerotic Cardiovascular Risk in Adults: A Report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J. Am. Coll. Cardiol. 2014, 63, 3024–3025. [Google Scholar] [CrossRef] [PubMed]
- Pencina, M.J.; Navar-Boggan, A.M.; D’Agostino, R.B., Sr.; Williams, K.; Neely, B.; Sniderman, A.D.; Peterson, E.D. Application of new cholesterol guidelines to a population-based sample. N. Engl. J. Med. 2014, 370, 1422–1431. [Google Scholar] [CrossRef] [PubMed]
- Johnson, K.M.; Dowe, D.A. Accuracy of Statin Assignment Using the 2013 AHA/ACC Cholesterol Guideline Versus the 2001 NCEP ATP III Guideline: Correlation With Atherosclerotic Plaque Imaging. J. Am. Coll. Cardiol. 2014, 64, 910–919. [Google Scholar] [CrossRef] [PubMed]
- Goff, D.C., Jr.; Lloyd-Jones, D.M.; Bennett, G.; Coady, S.; D’Agostino, R.B.; Gibbons, R.; Greenland, P.; Lackland, D.T.; Levy, D.; O’Donnell, C.J.; et al. 2013 American College of Cardiology/American Heart Association guideline on the assessment of cardiovascular risk: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation 2014, 129, S49–S73. [Google Scholar] [CrossRef] [PubMed]
- Wu, V.C.; Chang, H.W.; Liu, K.L.; Lin, Y.H.; Chueh, S.C.; Lin, W.C.; Ho, Y.L.; Huang, J.W.; Chiang, C.K.; Yang, S.Y.; et al. Primary Aldosteronism: Diagnostic Accuracy of the Losartan and Captopril Tests. Am. J. Hypertens. 2009, 22, 821–827. [Google Scholar] [CrossRef] [PubMed]
- Kuo, C.C.; Wu, V.C.; Huang, K.H.; Wang, S.M.; Chang, C.C.; Lu, C.C.; Yang, W.S.; Tsai, C.W.; Lai, C.F.; Lee, T.Y.; et al. Verification and evaluation of aldosteronism demographics in the Taiwan Primary Aldosteronism Investigation Group (TAIPAI Group). J. Renin-Angiotensin-Aldosterone Syst. 2011, 12, 348–357. [Google Scholar] [CrossRef] [PubMed]
- Peng, K.Y.; Chang, H.M.; Lin, Y.F.; Chan, C.K.; Chang, C.H.; Chueh, S.C.; Yang, S.Y.; Huang, K.H.; Lin, Y.H.; Wu, V.C.; et al. miRNA-203 Modulates Aldosterone Levels and Cell Proliferation by Targeting Wnt5a in Aldosterone-Producing Adenomas. J. Clin. Endocrinol. Metab. 2018, 103, 3737–3747. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.H.; Yang, Y.W.; Hu, Y.H.; Tsai, Y.C.; Kuo, K.L.; Lin, Y.H.; Hung, S.C.; Wu, V.C.; Wu, K.D. Comparison of 24-h urinary aldosterone level and random urinary aldosterone-to-creatinine ratio in the diagnosis of primary aldosteronism. PLoS ONE 2013, 8, e67417. [Google Scholar] [CrossRef] [PubMed]
- Wu, V.C.; Hu, Y.H.; Er, L.K.; Yen, R.F.; Chang, C.H.; Chang, Y.L.; Lu, C.C.; Chang, C.C.; Lin, J.H.; Lin, Y.H.; et al. Case detection and diagnosis of primary aldosteronism—The consensus of Taiwan Society of Aldosteronism. J. Formos. Med. Assoc. 2017, 116, 993–1005. [Google Scholar] [CrossRef] [PubMed]
- Wu, V.C.; Chueh, S.C.; Chang, H.W.; Lin, L.Y.; Liu, K.L.; Lin, Y.H.; Ho, Y.L.; Lin, W.C.; Wang, S.M.; Huang, K.H.; et al. Association of kidney function with residual hypertension after treatment of aldosterone-producing adenoma. Am. J. Kidney Dis. 2009, 54, 665–673. [Google Scholar] [CrossRef] [PubMed]
- Wu, V.C.; Kuo, C.C.; Wang, S.M.; Liu, K.L.; Huang, K.H.; Lin, Y.H.; Chu, T.S.; Chang, H.W.; Lin, C.Y.; Tsai, C.T.; et al. Primary aldosteronism: Changes in cystatin C-based kidney filtration, proteinuria, and renal duplex indices with treatment. J. Hypertens. 2011, 29, 1778–1786. [Google Scholar] [CrossRef] [PubMed]
- Wu, V.C.; Lo, S.C.; Chen, Y.L.; Huang, P.H.; Tsai, C.T.; Liang, C.J.; Kuo, C.C.; Kuo, Y.S.; Lee, B.C.; Wu, E.L.; et al. Endothelial progenitor cells in primary aldosteronism: A biomarker of severity for aldosterone vasculopathy and prognosis. J. Clin. Endocrinol. Metab. 2011, 96, 3175–3183. [Google Scholar] [CrossRef] [PubMed]
- Wu, V.C.; Huang, K.H.; Peng, K.Y.; Tsai, Y.C.; Wu, C.H.; Wang, S.M.; Yang, S.Y.; Lin, L.Y.; Chang, C.C.; Lin, Y.H.; et al. Prevalence and clinical correlates of somatic mutation in aldosterone producing adenoma-Taiwanese population. Sci. Rep. 2015, 5, 11396. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.H.; Wu, X.M.; Lee, H.H.; Lee, J.K.; Liu, Y.C.; Chang, H.W.; Lin, C.Y.; Wu, V.C.; Chueh, S.C.; Lin, L.C.; et al. Adrenalectomy reverses myocardial fibrosis in patients with primary aldosteronism. J. Hypertens. 2012, 30, 1606–1613. [Google Scholar] [CrossRef] [PubMed]
- Foppa, M.; Duncan, B.; Rohde, L. Echocardiography-based left ventricular mass estimation. How should we define hypertrophy? Cardiovasc. Ultrasound 2005, 3, 17. [Google Scholar] [CrossRef] [PubMed]
- Health Promotion Administration; Ministry of Health and Welfare. Metabolic Syndrome. Available online: http://www.hpa.gov.tw/BHPNet/Web/HealthTopic/Topic.aspx?id=201109140001 (accessed on 8 April 2018).
- Shu, K.H.; Wang, C.H.; Wu, C.H.; Huang, T.M.; Wu, P.C.; Lai, C.H.; Tseng, L.J.; Tsai, P.R.; Connolly, R.; Wu, V.C. Urinary π-glutathione S-transferase Predicts Advanced Acute Kidney Injury Following Cardiovascular Surgery. Sci. Rep. 2016, 2016, 626335. [Google Scholar] [CrossRef] [PubMed]
- Tomaschitz, A.; Ritz, E.; Pieske, B.; Rus-Machan, J.; Kienreich, K.; Verheyen, N.; Gaksch, M.; Grübler, M.; Fahrleitner-Pammer, A.; Mrak, P.; et al. Aldosterone and parathyroid hormone interactions as mediators of metabolic and cardiovascular disease. Metabolism 2014, 63, 20–31. [Google Scholar] [CrossRef] [PubMed]
- Catena, C.; Colussi, G.; Nadalini, E.; Chiuch, A.; Baroselli, S.; Lapenna, R.; Sechi, L.A. Relationships of plasma renin levels with renal function in patients with primary aldosteronism. Clin. J. Am. Soc. Nephrol. 2007, 2, 722–731. [Google Scholar] [CrossRef] [PubMed]
- Iacobellis, G.; Petramala, L.; Cotesta, D.; Pergolini, M.; Zinnamosca, L.; Cianci, R.; De Toma, G.; Sciomer, S.; Letizia, C. Adipokines and cardiometabolic profile in primary hyperaldosteronism. J. Clin. Endocrinol. Metab. 2010, 95, 2391–2398. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.F.; Chen, L.; Lin, S.L.; Yeh, Y.C.; Huang, T.M.; Chou, Y.H.; Chang, F.C.; Chen, Y.T.; Yang, S.Y.; Lai, T.S.; et al. Potential target-organ protection of mineralocorticoid receptor antagonist in acute kidney disease. J. Hypertens. 2018. [Google Scholar] [CrossRef] [PubMed]
- Syed, S.B.; Qureshi, M.A. Association of aldosterone and cortisol with cardiovascular risk factors in prehypertension stage. Int. J. Hypertens. 2012, 2012, 906327. [Google Scholar] [CrossRef] [PubMed]
- Baudrand, R.; Pojoga, L.H.; Vaidya, A.; Garza, A.E.; Vöhringer, P.A.; Jeunemaitre, X.; Hopkins, P.N.; Yao, T.M.; Williams, J.; Adler, G.K.; et al. Statin Use and Adrenal Aldosterone Production in Hypertensive and Diabetic Subjects. Circulation 2015, 132, 1825–1833. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.F.; Lin, S.L.; Huang, T.M.; Yang, S.Y.; Lai, T.S.; Chen, L.; Wu, V.C.; Chu, T.S.; Wu, K.D. National Taiwan University Hospital Study Group on Acute Renal Failure (NSARF). New-Onset Diabetes after Acute Kidney Injury Requiring Dialysis. Diabetes Care 2018, 41, 2105–2110. [Google Scholar] [CrossRef] [PubMed]
- Fallo, F.; Pilon, C.; Urbanet, R. Primary aldosteronism and metabolic syndrome. Horm. Metab. Res. 2012, 44, 208–214. [Google Scholar] [CrossRef] [PubMed]
- Rossi, G.P.; Bernini, G.; Caliumi, C.; Desideri, G.; Fabris, B.; Ferri, C.; Ganzaroli, C.; Giacchetti, G.; Letizia, C.; Maccario, M.; et al. A prospective study of the prevalence of primary aldosteronism in 1125 hypertensive patients. J. Am. Coll. Cardiol. 2006, 48, 2293–2300. [Google Scholar] [CrossRef] [PubMed]

| EH (n = 533) | PA (n = 461) | p | |
|---|---|---|---|
| Age (years) | 57.0 ± 9.5 | 57.4 ± 8.4 | 0.484 |
| Male gender (%) | 266 (49.9) | 207 (44.9) | 0.115 |
| Body mass index (Kg/m2) | 25.4 ± 4.0 | 25.4 ± 3.6 | 0.911 |
| Obesity (%) | 65 (12.2) | 48 (10.4) | 0.377 |
| Waist circumference (cm) | 83.8 ± 11.0 | 82.8 ± 10.9 | 0.742 |
| Systolic blood pressure (mmHg) | 144.5 ± 21.5 | 151.5 ± 22.4 | <0.001 * |
| Diastolic blood pressure (mmHg) | 86.1 ± 13.1 | 91.0 ± 14.1 | <0.001 * |
| Smoking status (%) | 64 (12.0) | 68 (14.8) | 0.204 |
| Diabetes mellitus (n, %) | 60 (11.3) | 69 (15.0) | 0.083 |
| Left ventricular hypertrophy (n, %) | 53 (9.9) | 90 (19.5) | <0.001 * |
| Laboratory data | |||
| PAC (ng/dL) (nmol/L) | 36.9 ± 83.81.0 ± 2.3 | 48.2 ± 32.7 1.3 ± 0.9 | 0.008 * |
| PRA (ng/mL/h) (pmol/L/h) | 4.5 ± 1.2 106.7 ± 28.4 | 1.2 ± 4.3 28.4 ± 101.9 | <0.001 * |
| Log ARR | 2.9 ± 2.0 | 4.9 ± 2.0 | <0.001 * |
| Potassium (mmol/L) (mEq/L) | 4.3 ± 2.0 4.3 ± 2.0 | 3.7 ± 0.7 3.7 ± 0.7 | <0.001 * |
| Fasting blood glucose (mg/dL) (mmol/L) | 99.2 ± 20.0 5.5 ± 1.1 | 101.9 ± 25.6 5.7 ± 1.4 | 0.108 |
| Total cholesterol (mg/dL) (mmol/L) | 197.0 ± 35.8 5.1 ± 0.9 | 194.7 ± 38.1 5.0 ± 1.0 | 0.328 |
| Low density lipoprotein (mg/dL) (mmol/L) | 114.6 ± 32.6 3.0 ± 0.8 | 119.0 ± 31.9 3.1 ± 0.8 | 0.033 * |
| Low density lipoprotein ≥ 190 mg/dL (n, %) | 12 (2.3) | 9 (2.0) | 0.744 |
| High density lipoprotein (mg/dL) (mmol/L) | 48.0 ± 12.4 1.2 ± 0.3 | 46.1 ± 13.6 1.2 ± 0.4 | 0.018 * |
| Triglyceride (mg/dL) (mmol/L) | 148.1 ± 110.6 1.7 ± 1.2 | 140.5 ± 97.4 1.6 ± 1.1 | 0.250 |
| Estimated glomerular filtration rate (mL/s per 1.73 m2) | 78.4 ± 20.4 | 78.8 ± 22.4 | 0.777 |
| Medication | |||
| Aspirin (n, %) | 62 (11.6) | 60 (13.0) | 0.587 |
| Antihypertensive drugs (n, %) | 449 (84.2) | 415 (90.0) | 0.009 * |
| Metabolic syndrome (n, %) | 200 (37.5) | 198 (43.0) | 0.093 |
| Outcome | |||
| 10-year ASCVD (%) a | 13.8 | 20.6 | <0.001 * |
| EH (n = 533) | PA (n = 461) | p | |
|---|---|---|---|
| Recommend statin use by ACC/AHA guidelines | 332 (62.3) | 305 (66.2) | 0.229 |
| Predicted 10-year ASCVD risk a (%) | |||
| ACC/AHA estimated 10-year ASCVD risk | 10.9 ± 7.0 | 12.8 ± 8.3 | <0.001 * |
| ACC/AHA estimated 10-year ASCVD risk > 7.5% | 14.6 ± 6.2 | 16.5 ± 7.4 | <0.001 * |
| ASCVD Risk prediction (AUROC, 95% CI) | |||
| ACC/AHA estimated 10-year ASCVD risk | 0.64 (0.58–0.71) | 0.76 (0.70–0.82) | |
| ACC/AHA estimated 10-year ASCVD risk > 7.5% | 0.61 (0.54–0.67) | 0.65 (0.59–0.71) | |
| Recommend statin use by ACC/AHA guidelines | 0.93 (0.91–0.95) | 0.94 (0.91–0.96) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lin, J.-H.; Lin, Y.-F.; Wang, W.-J.; Lin, Y.-F.; Chueh, S.-C.J.; Wu, V.-C.; Chu, T.-S.; Wu, K.-D.; On behalf of the Taiwan Primary Aldosteronism Investigation (TAIPAI) Study Group. Plasma Aldosterone Concentration as a Determinant for Statin Use among Middle-Aged Hypertensive Patients for Atherosclerotic Cardiovascular Disease. J. Clin. Med. 2018, 7, 382. https://doi.org/10.3390/jcm7110382
Lin J-H, Lin Y-F, Wang W-J, Lin Y-F, Chueh S-CJ, Wu V-C, Chu T-S, Wu K-D, On behalf of the Taiwan Primary Aldosteronism Investigation (TAIPAI) Study Group. Plasma Aldosterone Concentration as a Determinant for Statin Use among Middle-Aged Hypertensive Patients for Atherosclerotic Cardiovascular Disease. Journal of Clinical Medicine. 2018; 7(11):382. https://doi.org/10.3390/jcm7110382
Chicago/Turabian StyleLin, Jui-Hsiang, Yu-Feng Lin, Wei-Jie Wang, Yuh-Feng Lin, Shih-Chieh Jeff Chueh, Vin-Cent Wu, Tzong-Shinn Chu, Kwan-Dun Wu, and On behalf of the Taiwan Primary Aldosteronism Investigation (TAIPAI) Study Group. 2018. "Plasma Aldosterone Concentration as a Determinant for Statin Use among Middle-Aged Hypertensive Patients for Atherosclerotic Cardiovascular Disease" Journal of Clinical Medicine 7, no. 11: 382. https://doi.org/10.3390/jcm7110382
APA StyleLin, J.-H., Lin, Y.-F., Wang, W.-J., Lin, Y.-F., Chueh, S.-C. J., Wu, V.-C., Chu, T.-S., Wu, K.-D., & On behalf of the Taiwan Primary Aldosteronism Investigation (TAIPAI) Study Group. (2018). Plasma Aldosterone Concentration as a Determinant for Statin Use among Middle-Aged Hypertensive Patients for Atherosclerotic Cardiovascular Disease. Journal of Clinical Medicine, 7(11), 382. https://doi.org/10.3390/jcm7110382





