Pathophysiology of Hemophilic Arthropathy
Abstract
:1. Introduction
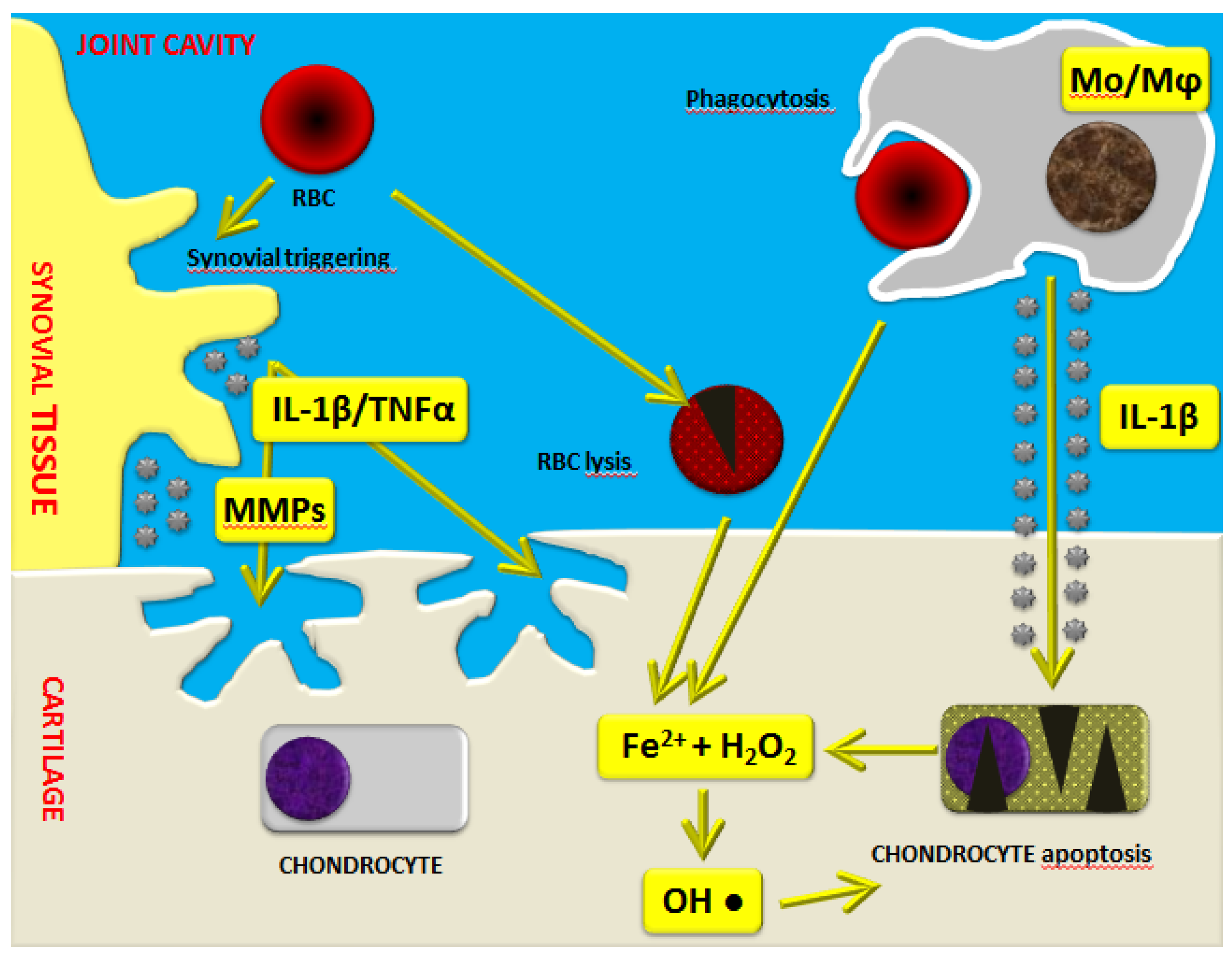
2. Pathophysiology of Hemophilic Arthropathy
2.1. From Synovitis to Articular Cartilage Damage
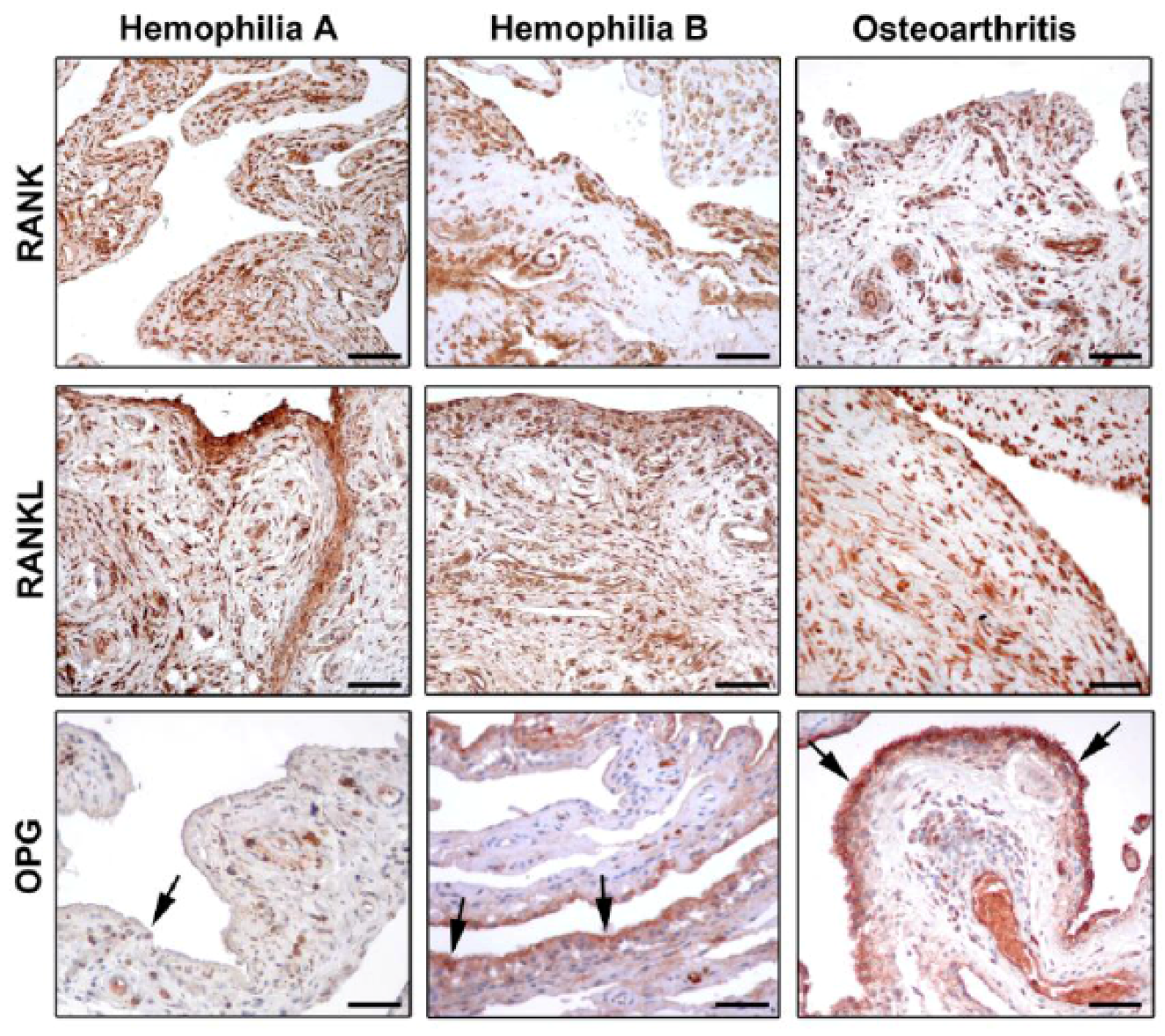
2.2. Bone Damage
3. Evidence That Arthropathy May be More Severe in Hemophilia A than in Hemophilia B
4. Conclusions
Author Contributions
Conflicts of Interest
References
- Bolton-Maggs, P.H.; Pasi, K.J. Haemophilias A and B. Lancet 2003, 361, 1801–1809. [Google Scholar] [CrossRef]
- Pollmann, H.; Richter, H.; Ringkamp, H.; Jurgens, H. When are children diagnosed as having severe haemophilia and when do they start to bleed? A 10-year single-centre PUP study. Eur. J. Pediatr. 1999, 158 (Suppl. 3), S166–S170. [Google Scholar] [CrossRef] [PubMed]
- Pulles, A.E.; Mastbergen, S.C.; Schtgens, R.E.; Lafeber, F.P.; van Vulpen, L.F. Pathophysiology of hemophilic arthropathy and potential targets for therapy. Pharmacol. Res. 2017, 115, 192–199. [Google Scholar] [CrossRef] [PubMed]
- Aledort, L.M.; Haschmeyer, R.H.; Pettersson, H. A longitudinal study of orthopaedic outcomes for severe factor-VIII-deficient haemophiliacs. The Orthopaedic Outcome Study Group. J. Intern. Med. 1994, 236, 391–399. [Google Scholar] [CrossRef] [PubMed]
- Fischer, K.; Bom, J.G.; Mauser-Bunschoten, E.P.; Roosendaal, G.; Berg, H.M. Effects of haemophilic arthropathy on health-related quality of life and socio-economic parameters. Haemophilia 2005, 11, 43–48. [Google Scholar] [CrossRef] [PubMed]
- Acharya, S.S.; Kaplan, R.N.; Macdonald, D.; Fabiyi, O.T.; DiMichele, D.; Lyden, D. Neoangiogenesis contributes to the development of hemophilic synovitis. Blood 2011, 117, 2484–2493. [Google Scholar] [CrossRef] [PubMed]
- Valentino, L.A.; Hakobyan, N.; Enockson, C. Blood-induced joint disease: The confluence of dysregulated oncogenes, inflammatory signals, and angiogenic cues. Semin. Hematol. 2008, 45, S50–S57. [Google Scholar] [CrossRef] [PubMed]
- Eubank, T.D.; Galloway, M.; Montague, C.M.; Waldman, W.J.; Marsh, C.B. M-CSF induces vascular endothelial growth factor production and angiogenic activity from human monocytes. J. Immunol. 2003, 171, 2637–2643. [Google Scholar] [CrossRef] [PubMed]
- Ruger, B.; Giurea, A.; Wanivenhaus, A.H.; Zehetgruber, H.; Hollemann, D.; Yanagida, G.; Groger, M.; Petzelbauer, P.; Smolen, J.S.; Hoecker, P.; et al. Endothelial precursor cells in the synovial tissue of patients with rheumatoid arthritis and osteoarthritis. Arthritis Rheum. 2004, 50, 2157–2166. [Google Scholar] [CrossRef] [PubMed]
- Maeno, N.; Takei, S.; Imanaka, H.; Takasaki, I.; Kitajima, I.; Maruyama, I.; Matsuo, K.; Miyata, K. Increased circulating vascular endothelial growth factor is correlated with disease activity in polyarticular juvenile rheumatoid arthritis. J. Rheumatol. 1999, 26, 2244–2248. [Google Scholar] [PubMed]
- Busso, N.; Morard, C.; Salvi, R.; Péclat, V.; So, A. Role of the tissue factor pathway in synovial inflammation. Arthritis Rheum. 2003, 48, 651–659. [Google Scholar] [CrossRef] [PubMed]
- Mannucci, P.M.; Franchini, M. Is haemophilia B less severe than haemophilia A? Haemophilia 2013, 19, 499–502. [Google Scholar] [CrossRef] [PubMed]
- Roosendaal, G.; Lafeber, F.P. Pathogenesis of haemophilic arthropathy. Haemophilia 2006, 12 (Suppl. 3), 117–121. [Google Scholar] [CrossRef] [PubMed]
- Roy, S.; Ghadially, F.N. Pathology of experimental haemarthrosis. Ann. Rheum. Dis. 1966, 25, 402–415. [Google Scholar] [CrossRef] [PubMed]
- Mainardi, C.L.; Levine, P.H.; Werb, Z.; Harris, E.D., Jr. Proliferative synovitis in hemophilia: Biochemical and morphologic observations. Arthritis Rheum. 1978, 21, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Morris, C.J.; Blake, D.R.; Wainwright, A.C.; Steven, M.M. Relationship between iron deposits and tissue damage in the synovium: An ultrastructural study. Ann. Rheum. Dis. 1986, 45, 21–26. [Google Scholar] [CrossRef] [PubMed]
- Melchiorre, D.; Linari, S.; Matassi, F.; Castaman, G. Pathogenesis of the haemophilic arthropathy. In Frontiers in Arthritis, 1st ed.; Carulli, C., Ed.; Bentham Science Publishers: Sharjah, United Arab Emirates, 2017; Volume 2, pp. 1–13. ISBN 978-1-68108-353-7. [Google Scholar]
- Mendonça, R.; Silveira, A.A.; Conran, N. Red cell DAMPs and inflammation. Inflamm. Res. 2016, 65, 665–678. [Google Scholar] [CrossRef] [PubMed]
- Ovlisen, K.; Kristensen, A.T.; Jensen, A.L.; Tranholm, M. IL-1 beta, IL-6, KC and MCP-1 are elevated in synovial fluid from haemophilic mice with experimentally induced haemarthrosis. Haemophilia 2009, 15, 802–810. [Google Scholar] [CrossRef] [PubMed]
- Hakobyan, N.; Kazarian, T.; Jabbar, A.A.; Jabbar, K.J.; Valentino, L.A. Pathobiology of hemophilic synovitis I: Overexpression of mdm2 oncogene. Blood 2004, 104, 2060–2064. [Google Scholar]
- Wen, F.Q.; Jabbar, A.A.; Chen, Y.X.; Kazarian, T.; Patel, D.A.; Valentino, L.A. C-myc proto-oncogene expression in hemophilic synovitis: In vitro studies of the effects of iron and ceramide. Blood 2002, 100, 912–916. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Merchan, E.C. Haemophilic synovitis: Basic concepts. Haemophilia 2007, 13 (Suppl. 3), 1–3. [Google Scholar] [CrossRef] [PubMed]
- Niibayashi, H.; Shimizu, K.; Suzuki, K.; Yamamoto, S.; Yasuda, T.; Yamamuro, T. Proteoglycan degradation in hemartrosis. Intraarticular, autologous blood injection in rat knees. Acta Orthop. Scand. 1995, 66, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Jansen, N.W.; Roosendaal, G.; Lafeber, F.P. Understanding haemophilic arthropathy: An exploration of current open issues. Br. J. Haematol. 2008, 143, 632–640. [Google Scholar] [CrossRef] [PubMed]
- Roosendaal, G.; Vianen, E.; van den Berg, H.M.; Lafeber, F.P.; Bijlsma, J.W. Cartilage damage as a result of haemarthrosis in a human in vitro model. J. Rheumatol. 1997, 24, 1350–1354. [Google Scholar] [PubMed]
- Hooiveld, M.; Roosendaal, G.; Wenting, M.; van den Berg, H.M.; Bijlsma, J.; Lafeber, F. Short-term exposure of cartilage to blood results in chondrocyte apoptosis. Am. J. Pathol. 2003, 162, 943–951. [Google Scholar] [CrossRef]
- Hooiveld, M.; Roosendaal, G.; van den Berg, H.M.; Bijlsma, J.; Lafeber, F. Hemoglobin-derived iron-dependent hydroxyl radical formation in blood-induced joint damage: An in vitro study. Rheumatology (Oxford) 2003, 42, 784–790. [Google Scholar] [CrossRef] [PubMed]
- Hooiveld, M.; Roosendaal, G.; Vianen, M.E.; van den Berg, H.M.; Bijlsma, J.; Lafeber, F. Immature articular cartilage is more susceptible to blood-induced damage than mature articular cartilage: An animal in vivo study. Arthritis Rheum. 2003, 48, 396–403. [Google Scholar] [CrossRef] [PubMed]
- Jansen, N.W.; Roosendaal, G.; Bijlsma, J.W.; DeGroot, J.; Lafeber, F.P. Exposure of human cartilage tissue to low concentrations of blood for a short period of time leads to prolonged cartilage damage. An in vitro study. Arthritis Rheum. 2007, 56, 199–207. [Google Scholar] [CrossRef] [PubMed]
- Elshabrawy, H.A.; Chen, Z.; Volin, M.V.; Ravella, S.; Virupannavar, S.; Shahrara, S. The pathogenic role of angiogenesis in rheumatoid arthritis. Angiogenesis 2015, 18, 433–448. [Google Scholar] [CrossRef] [PubMed]
- Acharya, S.S. Exploration of the pathogenesis of haemophilic joint arthropathy: Understanding implications for optimal clinical management. Br. J. Haematol. 2012, 156, 13–23. [Google Scholar] [CrossRef] [PubMed]
- Zetterberg, E.; Palmblad, J.; Wallensten, R.; Morfini, M.; Melchiorre, D.; Holmström, M. Angiogenesis is increased in advanced haemophilic joint disease and characterized by normal pericyte coverage. Eur. J. Haematol. 2014, 92, 256–262. [Google Scholar] [CrossRef] [PubMed]
- Linari, S.; Montorzi, G.; Bartolozzi, D.; Borderi, M.; Melchiorre, D.; Benelli, M.; Morfini, M. Hypovitaminosis D and osteopenia/osteoporosis in a haemophilia population: A study in HCV/HIV or HCV infected patients. Haemophilia 2013, 19, 126–133. [Google Scholar] [CrossRef] [PubMed]
- Roosendaal, G.; van Rinsum, A.C.; Vianen, M.E.; van den Berg, H.M.; Lafeber, F.P.; Bijlsma, J.W. Haemophilic arthropathy resembles degenerative rather than inflammatory joint disease. Histopathology 1999, 34, 144–153. [Google Scholar] [CrossRef] [PubMed]
- Valentino, L.A. Blood induced joint disease: The pathophysiology of haemophilic arthropathy. J. Thromb. Haemost. 2010, 8, 1895–1902. [Google Scholar] [CrossRef] [PubMed]
- Boyce, B.F.; Xing, L. Biology of RANK, RANKL, and osteoprotegerin. Arthritis Res. Ther. 2007, 9 (Suppl. 1), S1–S23. [Google Scholar] [CrossRef] [PubMed]
- Vandooren, B.; Cantaert, T.; Noordenbos, T.; Tak, P.P.; Baeten, D. The abundant synovial expression of the RANK/RANKL/Osteoprotegerin system in peripheral spondylarthritis is partially disconnected from inflammation. Arthritis Rheum. 2008, 58, 718–729. [Google Scholar] [CrossRef] [PubMed]
- Saidenberg-Kermanac’h, N.; Cohen-Solal, M.; Bessis, N.; De Vernejoul, M.C.; Boissier, M.C. Role for osteoprotegerin in rheumatoid inflammation. Joint Bone Spine 2004, 71, 9–13. [Google Scholar] [CrossRef]
- Lacey, D.L.; Timms, E.; Tan, H.L.; Kelley, M.J.; Dunstan, C.R.; Burgess, T.; Elliott, R.; Colombero, A.; Elliott, G.; Scully, S.; et al. Osteoprotegerin ligand is a cytokine that regulates osteoclast differentiation and activation. Cell 1998, 93, 165–176. [Google Scholar] [CrossRef]
- Jones, H.D.; Kong, Y.Y.; Penninger, J.M. Role of RANKL and RANK in bone loss and arthritis. Ann. Rheum. Dis. 2002, 61 (Suppl. 2), ii32–ii39. [Google Scholar] [CrossRef] [PubMed]
- Hofbauer, L.C.; Heufelder, A.E. Role of receptor activator of nuclear factor-kappaB ligand and osteoprotegerin in bone cell biology. J. Mol. Med. 2001, 79, 243–253. [Google Scholar] [CrossRef] [PubMed]
- Simonet, W.S.; Lacey, D.L.; Dunstan, C.R.; Kelley, M.; Chang, M.S.; Luthy, R.; Nguyen, H.Q.; Wooden, S.; Bennett, L.; Boone, T.; et al. Osteoprotegerin: A novel secreted protein involved in the regulation of bone density. Cell 1997, 89, 309–319. [Google Scholar] [CrossRef]
- Katsarou, O.; Terpos, E.; Chatzismalis, P.; Provelengios, S.; Adraktas, T.; Hadjidakis, D.; Kouramba, A.; Karafoulidou, A. Increased bone resorption is implicated in the pathogenesis of bone loss in hemophiliacs: Correlations with haemophilic arthropathy and HIV infection. Ann. Hematol. 2010, 89, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Christoforidis, A.; Economou, M.; Papadopoulou, E.; Kazantzidou, E.; Farmaki, E.; Tzimouli, V.; Tsatra, I.; Gompakis, N.; Athanassiou-Metaxa, M. Comparative study of dual energy X-ray absorptiometry and quantitative ultrasonography with the use of biochemical markers of bone turnover in boys with haemophilia. Haemophilia 2011, 17, e217–e222. [Google Scholar] [CrossRef] [PubMed]
- Melchiorre, D.; Milia, A.F.; Linari, S.; Romano, E.; Benelli, G.; Manetti, M.; Guiducci, S.; Ceccarelli, C.; Innocenti, M.; Carulli, C.; et al. RANK-RANKL-OPG in hemophilic arthropathy: From clinical and imaging diagnosis to histopathology. J. Rheumatol. 2012, 39, 1678–1686. [Google Scholar] [CrossRef] [PubMed]
- Melchiorre, D.; Linari, S.; Manetti, M.; Romano, E.; Sofi, F.; Matucci-Cerinic, M.; Carulli, C.; Innocenti, M.; Ibba-Manneschi, L.; Castaman, G. Clinical, instrumental, serological and histological findings suggest that hemophilia B may be less severe than hemophilia A. Haematologica 2016, 101, 219–225. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, M.S. Prophylaxis: Musculoskeletal evaluation. Semin. Hematol. 1993, 30, 3–6. [Google Scholar] [PubMed]
- Pettersson, H.; Ahlberg, A.; Nilsson, I.M. A radiologic classification of the hemophilic arthropathy. Clin. Orthop. Relat. Res. 1980, 149, 153–159. [Google Scholar] [CrossRef]
- Melchiorre, D.; Linari, S.; Innocenti, M.; Biscoglio, I.; Toigo, M.; Cerinic, M.M.; Morfini, M. Ultrasound detects joint damage and bleeding in haemophilic arthropathy: A proposal of a score. Haemophilia 2011, 17, 112–117. [Google Scholar] [CrossRef] [PubMed]
- Pavlova, A.; Oldenburg, J. Defining severity of hemophilia: More than factor levels. Semin. Thromb. Hemost. 2013, 39, 702–710. [Google Scholar] [CrossRef] [PubMed]
- Tagariello, G.; Iorio, A.; Santagostino, E.; Morfini, M.; Bisson, R.; Innocenti, M.; Mancuso, M.E.; Mazzucconi, M.G.; Pasta, G.L.; Radossi, P.; et al. Italian Association Hemophilia Centre (AICE). Comparison of the rates of joint arthroplasty in patients with severe factor VIII and IX deficiency: An index of different clinical severity of the 2 coagulation disorders. Blood 2009, 114, 779–784. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Melchiorre, D.; Manetti, M.; Matucci-Cerinic, M. Pathophysiology of Hemophilic Arthropathy. J. Clin. Med. 2017, 6, 63. https://doi.org/10.3390/jcm6070063
Melchiorre D, Manetti M, Matucci-Cerinic M. Pathophysiology of Hemophilic Arthropathy. Journal of Clinical Medicine. 2017; 6(7):63. https://doi.org/10.3390/jcm6070063
Chicago/Turabian StyleMelchiorre, Daniela, Mirko Manetti, and Marco Matucci-Cerinic. 2017. "Pathophysiology of Hemophilic Arthropathy" Journal of Clinical Medicine 6, no. 7: 63. https://doi.org/10.3390/jcm6070063
APA StyleMelchiorre, D., Manetti, M., & Matucci-Cerinic, M. (2017). Pathophysiology of Hemophilic Arthropathy. Journal of Clinical Medicine, 6(7), 63. https://doi.org/10.3390/jcm6070063






