The Effect of Mitochondrial Supplements on Mitochondrial Activity in Children with Autism Spectrum Disorder
Abstract
:1. Introduction
2. Material and Methods
2.1. Participants
2.1.1. Autism Spectrum Disorder
2.1.2. Mitochondrial Disease
2.1.3. Historical Healthy Controls
2.2. Measures of Mitochondrial Function
2.3. Statistical Analysis
3. Results
3.1. Participants
3.2. Supplements
3.3. Supplement Effect on Mitochondrial Complexes and Citrate Synthase Activity
3.3.1. Normalized Complex I Activity
3.3.2. Normalized Citrate Synthase Activity
3.3.3. Normalized Complex IV Activity
3.4. Supplement Effect on Relationship between Mitochondrial Complexes and Citrate Synthase Activity
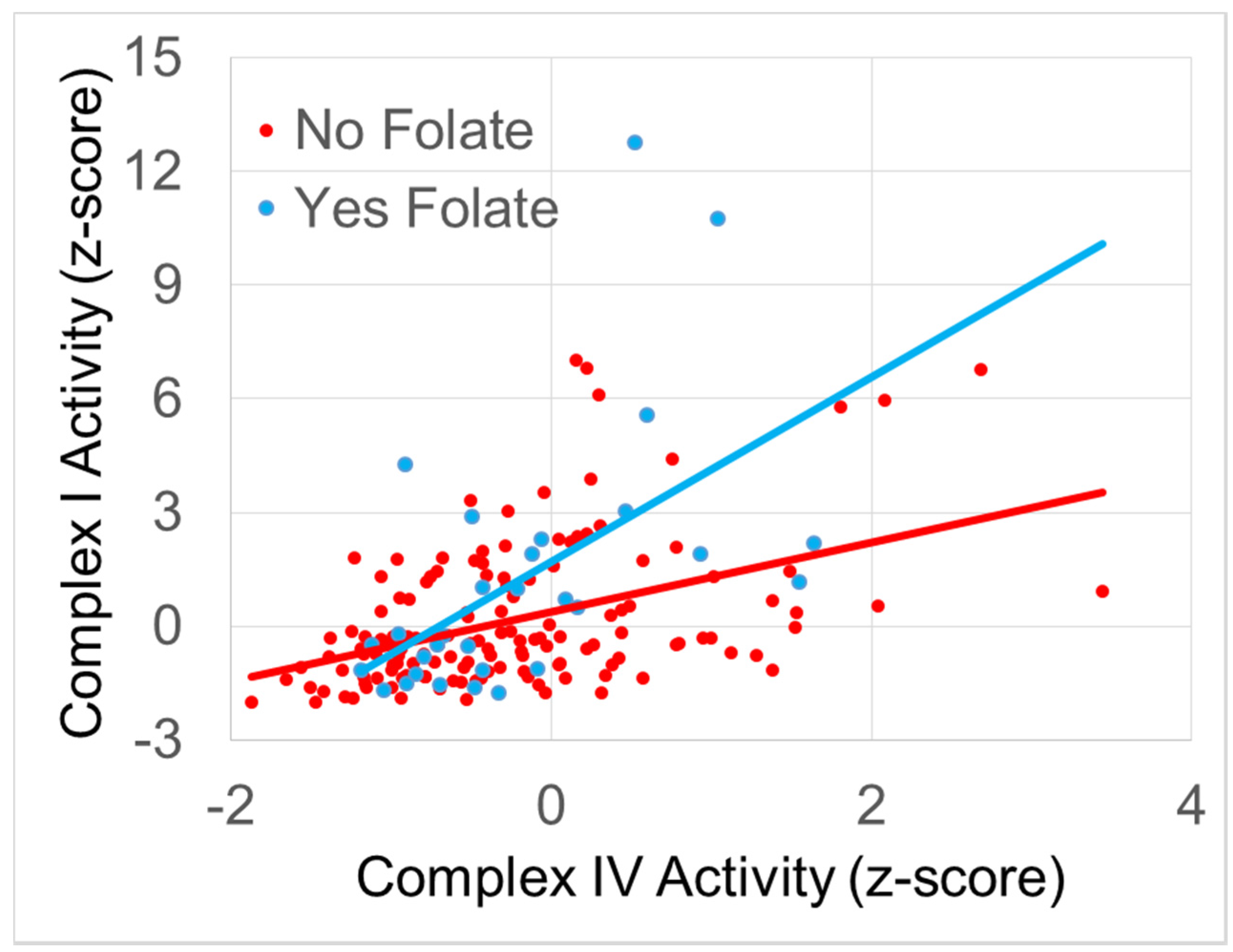
3.4.1. The Relationship between Normalized Complex I and Complex IV Activity
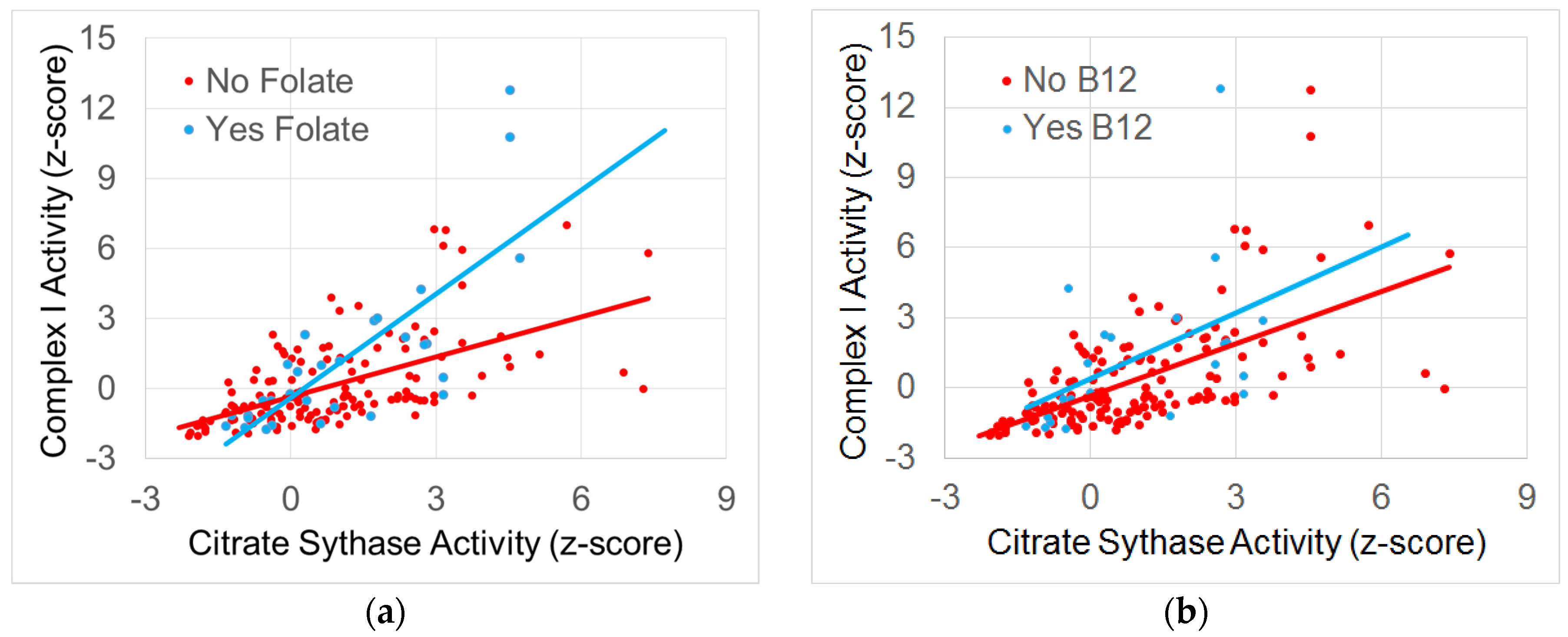
3.4.2. The Relationship between Normalized Complex I and Citrate Synthase Activity
3.4.3. The Relationship between Normalized Complex IV and Citrate Synthase Activity
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Niyazov, D.M.; Kahler, S.G.; Frye, R.E. Primary mitochondrial disease and secondary mitochondrial dysfunction: Importance of distinction for diagnosis and treatment. Mol. Syndromol. 2016, 7, 122–137. [Google Scholar] [CrossRef] [PubMed]
- Camp, K.M.; Krotoski, D.; Parisi, M.A.; Gwinn, K.A.; Cohen, B.H.; Cox, C.S.; Enns, G.M.; Falk, M.J.; Goldstein, A.C.; Gopal-Srivastava, R.; et al. Nutritional interventions in primary mitochondrial disorders: Developing an evidence base. Mol. Genet. Metab. 2016, 119, 187–206. [Google Scholar] [CrossRef] [PubMed]
- Parikh, S.; Goldstein, A.; Koenig, M.K.; Scaglia, F.; Enns, G.M.; Saneto, R.; Anselm, I.; Cohen, B.H.; Falk, M.J.; Greene, C.; et al. Diagnosis and management of mitochondrial disease: A consensus statement from the mitochondrial medicine society. Genet. Med. Off. J. Am. Coll. Med. Genet. 2015, 17, 689–701. [Google Scholar] [CrossRef] [PubMed]
- Viscomi, C. Toward a therapy for mitochondrial disease. Biochem. Soc. Trans. 2016, 44, 1483–1490. [Google Scholar] [CrossRef] [PubMed]
- Chinnery, P.; Majamaa, K.; Turnbull, D.; Thorburn, D. Treatment for mitochondrial disorders. Cochrane Database Syst. Rev. 2006. [Google Scholar] [CrossRef]
- Pfeffer, G.; Majamaa, K.; Turnbull, D.M.; Thorburn, D.; Chinnery, P.F. Treatment for mitochondrial disorders. Cochrane Database Syst. Rev. 2012. [Google Scholar] [CrossRef]
- Pfeffer, G.; Horvath, R.; Klopstock, T.; Mootha, V.K.; Suomalainen, A.; Koene, S.; Hirano, M.; Zeviani, M.; Bindoff, L.A.; Yu-Wai-Man, P.; et al. New treatments for mitochondrial disease-no time to drop our standards. Nat. Rev. Neurol. 2013, 9, 474–481. [Google Scholar] [CrossRef] [PubMed]
- Goldenthal, M.J.; Kuruvilla, T.; Damle, S.; Salganicoff, L.; Sheth, S.; Shah, N.; Marks, H.; Khurana, D.; Valencia, I.; Legido, A. Non-invasive evaluation of buccal respiratory chain enzyme dysfunction in mitochondrial disease: Comparison with studies in muscle biopsy. Mol. Genet. Metab. 2012, 105, 457–462. [Google Scholar] [CrossRef] [PubMed]
- Yorns, W.R., Jr.; Valencia, I.; Jayaraman, A.; Sheth, S.; Legido, A.; Goldenthal, M.J. Buccal swab analysis of mitochondrial enzyme deficiency and DNA defects in a child with suspected myoclonic epilepsy and ragged red fibers (merrf). J. Child. Neurol. 2012, 27, 398–401. [Google Scholar] [CrossRef] [PubMed]
- Ezugha, H.; Goldenthal, M.; Valencia, I.; Anderson, C.E.; Legido, A.; Marks, H. 5q14.3 deletion manifesting as mitochondrial disease and autism: Case report. J. Child. Neurol. 2010, 25, 1232–1235. [Google Scholar] [CrossRef] [PubMed]
- Frye, R.E.; Cox, D.; Slattery, J.; Tippett, M.; Kahler, S.; Granpeesheh, D.; Damle, S.; Legido, A.; Goldenthal, M.J. Mitochondrial dysfunction may explain symptom variation in phelan-mcdermid syndrome. Sci. Rep. 2016, 6, 19544. [Google Scholar] [CrossRef] [PubMed]
- Goldenthal, M.J.; Damle, S.; Sheth, S.; Shah, N.; Melvin, J.; Jethva, R.; Hardison, H.; Marks, H.; Legido, A. Mitochondrial enzyme dysfunction in autism spectrum disorders; a novel biomarker revealed from buccal swab analysis. Biomark. Med. 2015, 9, 957–965. [Google Scholar] [CrossRef] [PubMed]
- Legido, A.; Jethva, R.; Goldenthal, M.J. Mitochondrial dysfunction in autism. Semin. Pediatr. Neurol. 2013, 20, 163–175. [Google Scholar] [CrossRef] [PubMed]
- Zablotsky, B.; Black, L.I.; Maenner, M.J.; Schieve, L.A.; Blumberg, S.J. Estimated prevalence of autism and other developmental disabilities following questionnaire changes in the 2014 national health interview survey. Nat. Health Stat. Rep. 2015, 87, 1–20. [Google Scholar]
- Frye, R.E.; Rossignol, D.A. Mitochondrial dysfunction can connect the diverse medical symptoms associated with autism spectrum disorders. Pediatr. Res. 2011, 69, 41R–47R. [Google Scholar] [CrossRef] [PubMed]
- Rossignol, D.A.; Frye, R.E. Mitochondrial dysfunction in autism spectrum disorders: A systematic review and meta-analysis. Mol. Psychiatry 2012, 17, 290–314. [Google Scholar] [CrossRef] [PubMed]
- Frye, R.E. Biomarkers of abnormal energy metabolism in children with autism spectrum disorder. N. Am. J. Med. Sci. 2012, 5, 141–147. [Google Scholar] [CrossRef]
- Giulivi, C.; Zhang, Y.F.; Omanska-Klusek, A.; Ross-Inta, C.; Wong, S.; Hertz-Picciotto, I.; Tassone, F.; Pessah, I.N. Mitochondrial dysfunction in autism. JAMA 2010, 304, 2389–2396. [Google Scholar] [CrossRef] [PubMed]
- Napoli, E.; Wong, S.; Hertz-Picciotto, I.; Giulivi, C. Deficits in bioenergetics and impaired immune response in granulocytes from children with autism. Pediatrics 2014, 133, e1405–e1410. [Google Scholar] [CrossRef] [PubMed]
- Palmieri, L.; Papaleo, V.; Porcelli, V.; Scarcia, P.; Gaita, L.; Sacco, R.; Hager, J.; Rousseau, F.; Curatolo, P.; Manzi, B.; et al. Altered calcium homeostasis in autism-spectrum disorders: Evidence from biochemical and genetic studies of the mitochondrial aspartate/glutamate carrier agc1. Mol. Psychiatry 2010, 15, 38–52. [Google Scholar] [CrossRef] [PubMed]
- Frye, R.E. Novel cytochrome b gene mutations causing mitochondrial disease in autism. J. Pediatr. Neurol. 2012, 10, 35–40. [Google Scholar]
- Frye, R.E.; Naviaux, R.K. Autistic disorder with complex iv overactivity: A new mitochondrial syndrome. J. Pediatr. Neurol. 2011, 9, 427–434. [Google Scholar]
- Frye, R.E.; Melnyk, S.; Macfabe, D.F. Unique acyl-carnitine profiles are potential biomarkers for acquired mitochondrial disease in autism spectrum disorder. Transl. Psychiatry 2013, 3, e220. [Google Scholar] [CrossRef] [PubMed]
- Rose, S.; Frye, R.E.; Slattery, J.; Wynne, R.; Tippett, M.; Pavliv, O.; Melnyk, S.; James, S.J. Oxidative stress induces mitochondrial dysfunction in a subset of autism lymphoblastoid cell lines in a well-matched case control cohort. PLoS ONE 2014, 9, e85436. [Google Scholar] [CrossRef] [PubMed]
- Rose, S.; Frye, R.E.; Slattery, J.; Wynne, R.; Tippett, M.; Melnyk, S.; James, S.J. Oxidative stress induces mitochondrial dysfunction in a subset of autistic lymphoblastoid cell lines. Transl. Psychiatry 2014, 4, e377. [Google Scholar] [CrossRef] [PubMed]
- Rose, S.; Bennuri, S.C.; Wynne, R.; Melnyk, S.; James, S.J.; Frye, R.E. Mitochondrial and redox abnormalities in autism lymphoblastoid cells: A sibling control study. FASEB J. 2016. [Google Scholar] [CrossRef] [PubMed]
- Frye, R.E.; Rossignol, D.A. Treatments for biomedical abnormalities associated with autism spectrum disorder. Front. Pediatr. 2014, 2, 66. [Google Scholar] [CrossRef] [PubMed]
- Frye, R.E.; Delatorre, R.; Taylor, H.; Slattery, J.; Melnyk, S.; Chowdhury, N.; James, S.J. Redox metabolism abnormalities in autistic children associated with mitochondrial disease. Transl. Psychiatry 2013, 3, e273. [Google Scholar] [CrossRef] [PubMed]
- Frye, R.E.; Rossignol, D.; Casanova, M.F.; Brown, G.L.; Martin, V.; Edelson, S.; Coben, R.; Lewine, J.; Slattery, J.C.; Lau, C.; et al. A review of traditional and novel treatments for seizures in autism spectrum disorder: Findings from a systematic review and expert panel. Front. Public Health 2013, 1, 31. [Google Scholar] [CrossRef] [PubMed]
- Berressem, D.; Koch, K.; Franke, N.; Klein, J.; Eckert, G.P. Intravenous treatment with a long-chain omega-3 lipid emulsion provides neuroprotection in a murine model of ischemic stroke—A pilot study. PLoS ONE 2016, 11, e0167329. [Google Scholar] [CrossRef] [PubMed]
- Agnihotri, N.; Sharma, G.; Rani, I.; Renuka; Bhatnagar, A. Fish oil prevents colon cancer by modulation of structure and function of mitochondria. Biomed. Pharmacother. 2016, 82, 90–97. [Google Scholar] [CrossRef] [PubMed]
- Yoshino, J.; Smith, G.I.; Kelly, S.C.; Julliand, S.; Reeds, D.N.; Mittendorfer, B. Effect of dietary n-3 pufa supplementation on the muscle transcriptome in older adults. Physiol. Rep. 2016. [Google Scholar] [CrossRef] [PubMed]
- James, S.J.; Melnyk, S.; Fuchs, G.; Reid, T.; Jernigan, S.; Pavliv, O.; Hubanks, A.; Gaylor, D.W. Efficacy of methylcobalamin and folinic acid treatment on glutathione redox status in children with autism. Am. J. Clin. Nutr. 2009, 89, 425–430. [Google Scholar] [CrossRef] [PubMed]
- Hendren, R.L.; James, S.J.; Widjaja, F.; Lawton, B.; Rosenblatt, A.; Bent, S. Randomized, placebo-controlled trial of methyl b12 for children with autism. J. Child Adolesc. Psychopharmacol. 2016, 26, 774–783. [Google Scholar] [CrossRef] [PubMed]
- Desai, A.; Sequeira, J.M.; Quadros, E.V. The metabolic basis for developmental disorders due to defective folate transport. Biochimie 2016, 126, 31–42. [Google Scholar] [CrossRef] [PubMed]
| Supplement | % Taking Regularly | % Taking During Mitochondrial Testing | % Holding During Mitochondrial Testing |
|---|---|---|---|
| Amino Acids | 23% | 4% | 19% |
| B12 | 38% | 13% | 25% |
| B Vitamins | 36% | 10% | 26% |
| Carnitine | 42% | 13% | 29% |
| Coenzyme Q10 | 36% | 9% | 27% |
| Fatty Acids | 45% | 16% | 29% |
| Folate | 54% | 16% | 38% |
| Herbal | 17% | 6% | 11% |
| Multivitamin | 26% | 12% | 14% |
| Antioxidants | 46% | 14% | 32% |
| Other Vitamins | 49% | 14% | 35% |
| Supplement | Off Supplement | On Supplement |
|---|---|---|
| Carnitine | −0.1 (0.28) | 1.4 (0.46) |
| Antioxidants | 0.1 (0.26) | 1.5 (0.50) |
| Other Vitamins | 0.1 (0.27) | 1.4 (0.45) |
| Supplement | No Mitochondrial Disease | Mitochondrial Disease | ||
|---|---|---|---|---|
| Off Supplement | On Supplement | Off Supplement | On Supplement | |
| Fatty Acids | 0.1 (0.20) | 1.2 (0.48) | −0.3 (0.47) | 3.1 (0.84) |
| Folate | 0.2 (0.21) | 0.7 (0.5) | −0.3 (0.49) | 2.7 (0.79) |
| Supplement | No Mitochondrial Disease | Mitochondrial Disease | ||
|---|---|---|---|---|
| Off Supplement | On Supplement | Off Supplement | On Supplement | |
| Fatty Acids | 0.8 (0.17) | 1.2 (0.40) | 0.3 (0.40) | 2.6 (0.70) |
| Folate | 0.9 (0.18) | 0.8 (0.40) | 0.2 (0.42) | 2.4 (0.66) |
| Antioxidants | 0.9 (0.17) | 0.8 (0.42) | 0.2 (0.41) | 2.7 (0.71) |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Delhey, L.M.; Nur Kilinc, E.; Yin, L.; Slattery, J.C.; Tippett, M.L.; Rose, S.; Bennuri, S.C.; Kahler, S.G.; Damle, S.; Legido, A.; et al. The Effect of Mitochondrial Supplements on Mitochondrial Activity in Children with Autism Spectrum Disorder. J. Clin. Med. 2017, 6, 18. https://doi.org/10.3390/jcm6020018
Delhey LM, Nur Kilinc E, Yin L, Slattery JC, Tippett ML, Rose S, Bennuri SC, Kahler SG, Damle S, Legido A, et al. The Effect of Mitochondrial Supplements on Mitochondrial Activity in Children with Autism Spectrum Disorder. Journal of Clinical Medicine. 2017; 6(2):18. https://doi.org/10.3390/jcm6020018
Chicago/Turabian StyleDelhey, Leanna M., Ekim Nur Kilinc, Li Yin, John C. Slattery, Marie L. Tippett, Shannon Rose, Sirish C. Bennuri, Stephen G. Kahler, Shirish Damle, Agustin Legido, and et al. 2017. "The Effect of Mitochondrial Supplements on Mitochondrial Activity in Children with Autism Spectrum Disorder" Journal of Clinical Medicine 6, no. 2: 18. https://doi.org/10.3390/jcm6020018
APA StyleDelhey, L. M., Nur Kilinc, E., Yin, L., Slattery, J. C., Tippett, M. L., Rose, S., Bennuri, S. C., Kahler, S. G., Damle, S., Legido, A., Goldenthal, M. J., & Frye, R. E. (2017). The Effect of Mitochondrial Supplements on Mitochondrial Activity in Children with Autism Spectrum Disorder. Journal of Clinical Medicine, 6(2), 18. https://doi.org/10.3390/jcm6020018






