Modulating the Gut Micro-Environment in the Treatment of Intestinal Parasites
Abstract
:1. Introduction
2. Methodology
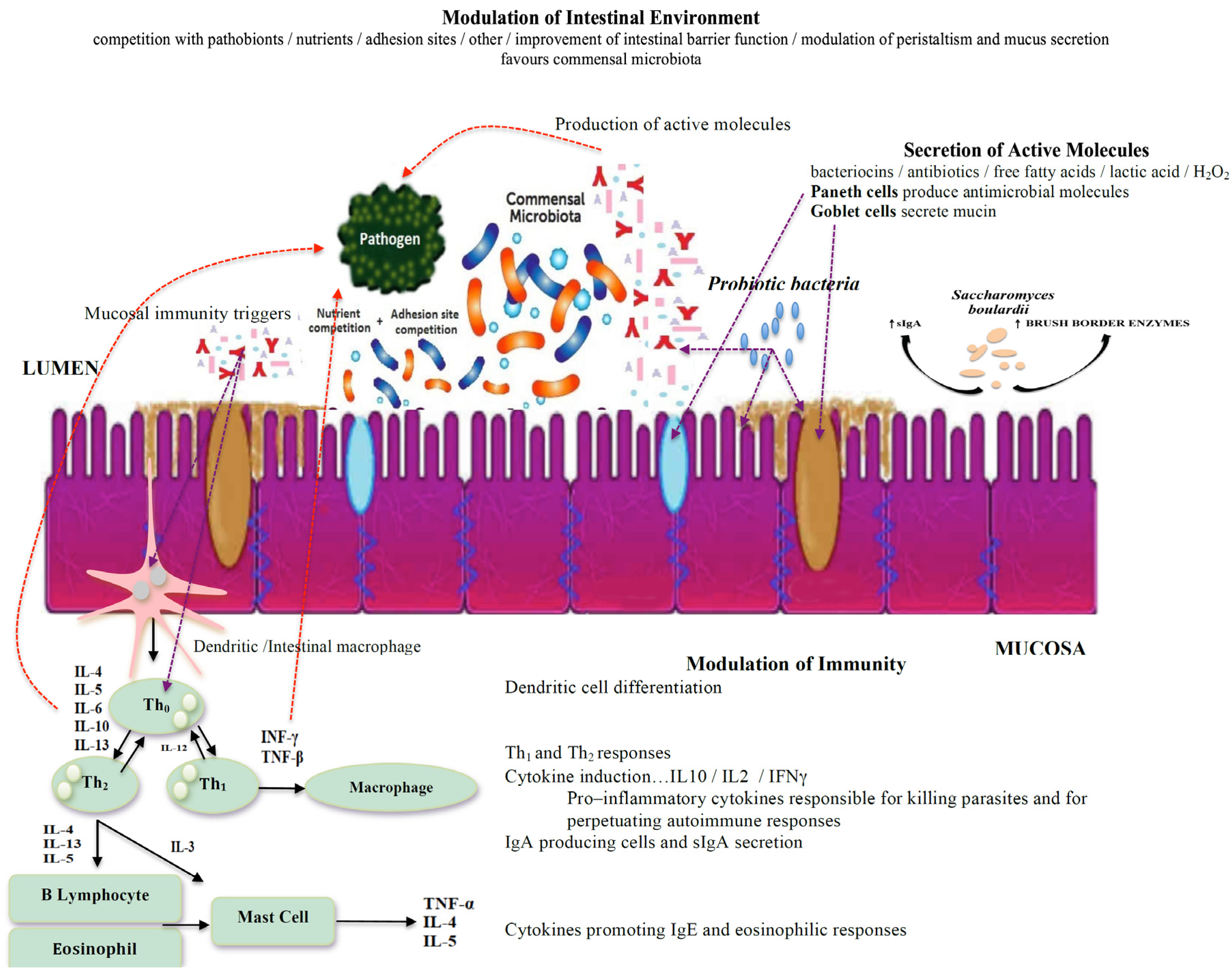
3. Intestinal Environment Interactions between the Microbiota and Protozoans
4. Probiotics for the Treatment of Protozoan Infections
5. Discussion
6. Conclusions
Conflicts of Interest
References
- Bjorksten, B. The gut microbiota: A complex ecosystem. Clin. Exp. Allergy 2006, 36, 1215–1217. [Google Scholar] [CrossRef] [PubMed]
- Christner, B.C.; Priscu, J.C.; Achberger, A.M.; Barbante, C.; Carter, S.P.; Christianson, K.; Michaud, A.B.; Mikucki, J.A.; Mitchell, A.C.; Skidmore, M.L.; et al. A microbial ecosystem beneath the West Antarctic ice sheet. Nature 2014, 512, 310–313. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Boyd, E.; Crane, S.; Lu-Irving, P.; Krabbenhoft, D.; King, S.; Dighton, J.; Geesey, G.; Barkay, T. Environmental conditions constrain the distribution and diversity of archaeal merA in Yellowstone National Park, Wyoming, USA. Microb. Ecol. 2011, 62, 739–752. [Google Scholar] [CrossRef] [PubMed]
- Whitman, W.B.; Coleman, D.C.; Wiebe, W.J. Prokaryotes: The unseen majority. Proc. Natl. Acad. Sci. USA 1998, 95, 6578–6583. [Google Scholar] [CrossRef] [PubMed]
- Turnbaugh, P.J.; Ley, R.E.; Hamady, M.; Fraser-Liggett, C.M.; Knight, R.; Gordon, J.I. The human microbiome project. Nature 2007, 449, 804–810. [Google Scholar] [CrossRef] [PubMed]
- Hooper, L.V.; Midtvedt, T.; Gordon, J.I. How host-microbial interactions shape the nutrient environment of the mammalian intestine. Annu. Rev. Nutr. 2002, 22, 283–307. [Google Scholar] [CrossRef] [PubMed]
- Vitetta, L.; Palacios, T.; Hall, S.; Coulson, S. Gastrointestinal Tract Commensal Bacteria and Probiotics: Influence on End-Organ Physiology. Prog. Drug Res. 2015, 70, 1–33. [Google Scholar] [PubMed]
- Berrilli, F.; Di Cave, D.; Cavallero, S.; D’Amelio, S. Interactions between parasites and microbial communities in the human gut. Front. Cell. Infect. Microbiol. 2012, 2, 141. [Google Scholar] [CrossRef] [PubMed]
- Scott, K.G.; Yu, L.C.; Buret, A.G. Role of CD8+ and CD4+ T lymphocytes in jejunal mucosal injury during murine giardiasis. Infect. Immun. 2004, 72, 3536–3542. [Google Scholar] [CrossRef] [PubMed]
- Chai, J.Y.; Guk, S.M.; Han, H.K.; Yun, C.K. Role of intraepithelial lymphocytes in mucosal immune responses of mice experimentally infected with Cryptosporidium parvum. J. Parasitol. 1999, 85, 234–239. [Google Scholar] [CrossRef] [PubMed]
- Zariffard, M.R.; Harwani, S.; Novak, R.M.; Graham, P.J.; Ji, X.; Spear, G.T. Trichomonas vaginalis infection activates cells through toll-like receptor 4. Clin. Immunol. 2004, 111, 103–107. [Google Scholar] [CrossRef] [PubMed]
- Tan, K.S.; Singh, M.; Yap, E.H. Recent advances in Blastocystis hominis research: Hot spots in terra incognita. Int. J. Parasitol. 2002, 32, 789–804. [Google Scholar] [CrossRef]
- Kaya, S.; Cetin, E.S.; Aridogan, B.C.; Arikan, S.; Demirci, M. Pathogenicity of Blastocystis hominis, a clinical reevaluation. Turkiye Parazitol. Derg. 2007, 31, 184–187. [Google Scholar] [PubMed]
- Correia, M.I.T.D.; Liboredo, J.C.; Consoli, M.L.D. The roof of probiotics in gastrointestinal surgery. Nutrition 2012, 28, 230–234. [Google Scholar] [CrossRef] [PubMed]
- Shukla, G.; Devi, P.; Sehgal, R. Effect of Lactobacillus casei as a probiotic on modulation of giardiasis. Dig. Dis. Sci. 2008, 53, 2671–2679. [Google Scholar] [CrossRef] [PubMed]
- Alak, J.I.; Wolf, B.W.; Mdurvwa, E.G.; Pimentel-Smith, G.E.; Adeyemo, O. Effect of Lactobacillus reuteri on intestinal resistance to Cryptosporidium parvum infection in a murine model of acquired immunodeficiency syndrome. J. Infect. Dis. 1997, 175, 218–221. [Google Scholar] [CrossRef] [PubMed]
- Alak, J.I.; Wolf, B.W.; Mdurvwa, E.G.; Pimentel-Smith, G.E.; Kolavala, S.; Abdelrahman, H.; Suppiramaniam, V. Supplementation with Lactobacillus reuteri or L. acidophilus reduced intestinal shedding of Cryptosporidium parvum oocysts in immunodeficient C57BL/6 mice. Cell. Mol. Biol. 1999, 45, 855–863. [Google Scholar] [PubMed]
- Besirbellioglu, B.A.; Ulcay, A.; Can, M.; Erdem, H.; Tanyuksel, M.; Avci, I.Y.; Araz, E.; Pahsa, A. Saccharomyces boulardii and infection due to Giardia lamblia. Scand. J. Infect. Dis. 2006, 38, 479–481. [Google Scholar] [CrossRef] [PubMed]
- Dinleyici, E.C.; Eren, M.; Dogan, N.; Reyhanioglu, S.; Yargic, Z.A.; Vandenplas, Y. Clinical efficacy of Saccharomyces boulardii or metronidazole in symptomatic children with Blastocystis hominis infection. Parasitol. Res. 2011, 108, 541–545. [Google Scholar] [CrossRef] [PubMed]
- Dinleyici, E.C.; Eren, M.; Yargic, Z.A.; Dogan, N.; Vandenplas, Y. Clinical efficacy of Saccharomyces boulardii and metronidazole compared to metronidazole alone in children with acute bloody diarrhea caused by amebiasis: A prospective, randomized, open label study. Am. J. Trop. Med. Hyg. 2009, 80, 953–955. [Google Scholar] [PubMed]
- Pickerd, N.; Tuthill, D. Resolution of cryptosporidiosis with probiotic treatment. Postgrad. Med. J. 2004, 80, 112–113. [Google Scholar] [CrossRef] [PubMed]
- Mansour-Ghanaei, F.; Dehbashi, N.; Yazdanparast, K.; Shafaghi, A. Efficacy of Saccharomyces boulardii with antibiotics in acute amoebiasis. World J. Gastroenterol. 2003, 9, 1832–1833. [Google Scholar] [CrossRef] [PubMed]
- Harp, J.A.; Chen, W.; Harmsen, A.G. Resistance of severe combined immunodeficient mice to infection with Cryptosporidium parvum: The importance of intestinal microflora. Infect. Immun. 1992, 60, 3509–3512. [Google Scholar] [PubMed]
- Phillips, B.P.; Zierdt, C.H. Blastocystis hominis: Pathogenic potential in human patients and in gnotobiotes. Exp. Parasitol. 1976, 39, 358–364. [Google Scholar] [CrossRef]
- Phillips, B.P.; Wolfe, P.A.; Rees, C.W.; Gordon, H.A.; Wright, W.H.; Reyniers, J.A. Studies on the ameba-bacteria relationship in amebiasis; comparative results of the intracecal inoculation of germfree, monocontaminated, and conventional guinea pigs with Entamoeba histolytica. Am. J. Trop. Med. Hyg. 1955, 4, 675–692. [Google Scholar] [PubMed]
- El-Shewy, K.A.; Eid, R.A. In vivo killing of Giardia trophozoites harbouring bacterial endosymbionts by intestinal Paneth cells: An ultrastructural study. Parasitology 2005, 130, 269–274. [Google Scholar] [CrossRef] [PubMed]
- Torres, M.F.; Uetanabaro, A.P.; Costa, A.F.; Alves, C.A.; Farias, L.M.; Bambirra, E.A.; Penna, F.J.; Vieira, E.C.; Nicoli, J.R. Influence of bacteria from the duodenal microbiota of patients with symptomatic giardiasis on the pathogenicity of Giardia duodenalis in gnotoxenic mice. J. Med. Microbiol. 2000, 49, 209–215. [Google Scholar] [CrossRef] [PubMed]
- Vitetta, L.; Hall, S.; Linnane, A.W. Live probiotic cultures and the gastrointestinal tract: Symbiotic preservation of tolerance whilst attenuating pathogenicity. Front. Cell. Infect. Microbiol. 2014, 4, 143. [Google Scholar] [CrossRef] [PubMed]
- Oelschlaeger, T.A. Mechanisms of probiotic actions—A review. Int. J. Med. Microbiol. 2010, 300, 57–62. [Google Scholar] [CrossRef] [PubMed]
- Elli M, Z.R.; Rytz, A.; Reniero, R.; Morelli, L. Iron requirement of Lactobacillus spp. in completely chemically defined growth media. J. Appl. Microbiol. 2000, 88, 695–703. [Google Scholar] [CrossRef] [PubMed]
- Wohlgemuth, S.; Loh, G.; Blaut, M. Recent developments and perspectives in the investigation of probiotic effects. Int. J. Med. Microbiol. 2010, 300, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Kuhbacher, T.; Ott, S.J.; Helwig, U.; Mimura, T.; Rizzello, F.; Kleessen, B.; Gionchetti, P.; Blaut, M.; Campieri, M.; Fölsch, U.R.; et al. Bacterial and fungal microbiota in relation to probiotic therapy (VSL#3) in pouchitis. Gut 2006, 55, 833–841. [Google Scholar] [PubMed]
- Drider, D.; Bendali, F.; Naghmouchi, K.; Chikindas, M.L. Bacteriocins: Not Only Antibacterial Agents. Probiotics Antimicrob. Proteins 2016, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Cleusix, V.; Lacroix, C.; Vollenweider, S.; Le Blay, G. Glycerol induces reuterin production and decreases Escherichia coli population in an in vitro model of colonic fermentation with immobilized human feces. FEMS Microbiol. Ecol. 2008, 63, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Perdigon, G.; Alvarez, S.; Rachid, M.; Aguero, G.; Gobbato, N. Immune system stimulation by probiotics. J. Dairy Sci. 1995, 78, 1597–1606. [Google Scholar] [CrossRef]
- Konstantinov, S.R.; Smidt, H.; de Vos, W.M.; Bruijns, S.C.; Singh, S.K.; Valence, F.; Molle, D.; Lortal, S.; Altermann, E.; Klaenhammer, T.R.; et al. S layer protein A of Lactobacillus acidophilus NCFM regulates immature dendritic cell and T cell functions. Proc. Natl. Acad. Sci. USA 2008, 105, 19474–19479. [Google Scholar] [CrossRef] [PubMed]
- Arvola, T.; Laiho, K.; Torkkeli, S.; Mykkanen, H.; Salminen, S.; Maunula, L.; Isolauri, E. Prophylactic Lactobacillus GG reduces antibiotic-associated diarrhea in children with respiratory infections: A randomized study. Pediatrics 1999, 104, e64. [Google Scholar] [CrossRef] [PubMed]
- Farthing, M.J. Treatment options for the eradication of intestinal protozoa. Nat. Clin. Pract. Gastroenterol. Hepatol. 2006, 3, 436–445. [Google Scholar] [CrossRef] [PubMed]
- Carding, S.; Verbeke, K.; Vipond, D.T.; Corfe, B.M.; Owen, L.J. Dysbiosis of the gut microbiota in disease. Microb. Ecol. Health Dis. 2015, 26, 26191. [Google Scholar] [CrossRef] [PubMed]
- Humen, M.A.; de Antoni, G.L.; Benyacoub, J.; Costas, M.E.; Cardozo, M.I.; Kozubsky, L.; Saudan, K.Y.; Boenzli-Bruand, A.; Blum, S.; Schiffrin, E.J.; et al. Lactobacillus johnsonii La1 antagonizes Giardia intestinalis in vivo. Infect. Immun. 2005, 73, 1265–1269. [Google Scholar] [CrossRef] [PubMed]
- Benyacoub, J.; Perez, P.F.; Rochat, F.; Saudan, K.Y.; Reuteler, G.; Antille, N.; Humen, M.; De Antoni, G.L.; Cavadini, C.; Blum, S.; et al. Enterococcus faecium SF68 enhances the immune response to Giardia intestinalis in mice. J. Nutr. 2005, 135, 1171–1176. [Google Scholar] [PubMed]
- Castaneda, C.; Guillot, E.G.B.; Santa Cruz, M.; Dominguez, M.F.; Garcia, E.; Gutierrez, P.M. Effects of Saccharomyces boulardii in children with chronic diarrhea, especially cases due to giardiasis. Revista Mexicana de Puericultura y Pediatria 1995, 2, 1–11. [Google Scholar]
- Vitetta, L.; Manuel, R.; Zhou, J.Y.; Linnane, A.W.; Hall, S.; Coulson, S. The overarching influence of the gut microbiome on end-organ function: The role of live probiotic cultures. Pharmaceuticals (Basel) 2014, 7, 954–989. [Google Scholar] [CrossRef] [PubMed]
- Bellafante, E.; Morgano, A.; Salvatore, L.; Murzilli, S.; Di Tullio, G.; D’Orazio, A.; Latorre, D.; Villani, G.; Moschetta, A. PGC-1β promotes enterocyte lifespan and tumorigenesis in the intestine. Proc. Natl. Acad. Sci. USA 2014, 111, E4523–E4531. [Google Scholar] [CrossRef] [PubMed]
- Boorom, K.F.; Smith, H.; Nimri, L.; Viscogliosi, E.; Spanakos, G.; Parkar, U.; Li, L.H.; Zhou, X.N.; Ok, U.Z.; Leelayoova, S.; et al. Oh my aching gut: Irritable bowel syndrome, Blastocystis, and asymptomatic infection. Parasit. Vectors 2008, 1, 40. [Google Scholar] [CrossRef] [PubMed]
- O’Flaherty, S.; Saulnier, D.M.; Pot, B.; Versalovic, J. How can probiotics and prebiotics impact mucosal immunity? Gut Microbes 2010, 1, 293–300. [Google Scholar] [CrossRef] [PubMed]
- Ohashi, Y.; Ushida, K. Health-beneficial effects of probiotics: Its mode of action. Anim. Sci. J. 2009, 80, 361–371. [Google Scholar] [CrossRef] [PubMed]
- Travers, M.A.; Florent, I.; Kohl, L.; Grellier, P. Probiotics for the control of parasites: An overview. J. Parasitol. Res. 2011, 2011, 610769. [Google Scholar] [CrossRef] [PubMed]
- McFarland, L.V. Systematic review and meta-analysis of Saccharomyces boulardii in adult patients. World J. Gastroenterol. 2010, 16, 2202–2222. [Google Scholar] [CrossRef] [PubMed]
- Maslowski, K.M.; Vieira, A.T.; Ng, A.; Kranich, J.; Sierro, F.; Yu, D.; Schilter, H.C.; Rolph, M.S.; Mackay, F.; Artis, D.; et al. Regulation of inflammatory responses by gut microbiota and chemoattractant receptor GPR43. Nature 2009, 461, 1282–1286. [Google Scholar] [CrossRef] [PubMed]
- Galdeano, C.M.; de Moreno de LeBlanc, A.; Vinderola, G.; Bonet, M.E.; Perdigon, G. Proposed model: Mechanisms of immunomodulation induced by probiotic bacteria. Clin. Vaccine Immunol. 2007, 14, 485–492. [Google Scholar] [CrossRef] [PubMed]
- Antoine, J.M. Probiotics: Beneficial factors of the defence system. Proc. Nutr. Soc. 2010, 69, 429–433. [Google Scholar] [CrossRef] [PubMed]
- Martin, R.; Miquel, S.; Ulmer, J.; Langella, P.; Bermudez-Humaran, L.G. Gut ecosystem: How microbes help us. Benef. Microbes 2014, 5, 219–233. [Google Scholar] [CrossRef] [PubMed]
| Reference | Year | Study | Methods | Results | Conclusion |
|---|---|---|---|---|---|
| [15] | 2008 | Effect of Lactobacillus casei as a probiotic on modulation of giardiasis | Group I = single dose of TYI-S-33 Group II = Giardia-infected mice—single dose of Giardia trophozoites qid. Group III = probiotic group
|
| Probiotics, particularly L. casei, modulate Giardia infection by minimising or preventing adherence of Giardia trophozoites to the mucosal surface, suggesting probiotics offer a safe and effective mode to prevent and treat Giardia infection. |
| [16] | 1997 | Effect of L. reuteri on intestinal resistance to C. parvum infection in acquired immunodeficient murine model |
|
| L. reuteri may help prevent C. parvum infection in immune-deficient subjects. |
| [17] | 1999 | Supplementation with Lactobacillus reuteri or L. acidophilus reduced intestinal shedding of cryptosporidium parvum oocysts in immunodeficient C57BL/6 mice. | C57BL/6 immunosuppressed female mice with LP-BM5 leukemia virus and randomly assigned to one of five groups
|
| L. reuteri or L. acidophilus can reduce C. parvum parasite burdens in epithelium during cryptosporidiosis. L. acidophilus was more efficacious in reducing fecal shedding than L. reuteri |
| Reference | Year | Study | Symptoms | Treatment | Results |
|---|---|---|---|---|---|
| [18] | 2006 | S. boulardii and infection due to Giardia lamblia |
|
|
|
| [19] | 2011 | Efficacy of S. boulardii or metronidazole in symptomatic children with Blastocystis hominis infection |
|
| On day 15 clinical cure observed at:
|
| [20] | 2009 | Efficacy of S. boulardii in children with acute bloody diarrhea caused by amebiasis |
|
|
|
| [21] | 2004 | Resolution of cryptosporidiosis with probiotic treatment—Case Study of 12 y.o. coeliac sufferer. |
|
|
|
| [22] | 2003 | Efficacy Saccharomyces boulardii with antibiotics in acute amoebiasis |
| Group 1:
|
|
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vitetta, L.; Saltzman, E.T.; Nikov, T.; Ibrahim, I.; Hall, S. Modulating the Gut Micro-Environment in the Treatment of Intestinal Parasites. J. Clin. Med. 2016, 5, 102. https://doi.org/10.3390/jcm5110102
Vitetta L, Saltzman ET, Nikov T, Ibrahim I, Hall S. Modulating the Gut Micro-Environment in the Treatment of Intestinal Parasites. Journal of Clinical Medicine. 2016; 5(11):102. https://doi.org/10.3390/jcm5110102
Chicago/Turabian StyleVitetta, Luis, Emma Tali Saltzman, Tessa Nikov, Isabelle Ibrahim, and Sean Hall. 2016. "Modulating the Gut Micro-Environment in the Treatment of Intestinal Parasites" Journal of Clinical Medicine 5, no. 11: 102. https://doi.org/10.3390/jcm5110102
APA StyleVitetta, L., Saltzman, E. T., Nikov, T., Ibrahim, I., & Hall, S. (2016). Modulating the Gut Micro-Environment in the Treatment of Intestinal Parasites. Journal of Clinical Medicine, 5(11), 102. https://doi.org/10.3390/jcm5110102






