Potential Role of Induced Pluripotent Stem Cells (IPSCs) for Cell-Based Therapy of the Ocular Surface
Abstract
:1. Introduction
1.1. Limbal Stem Cells
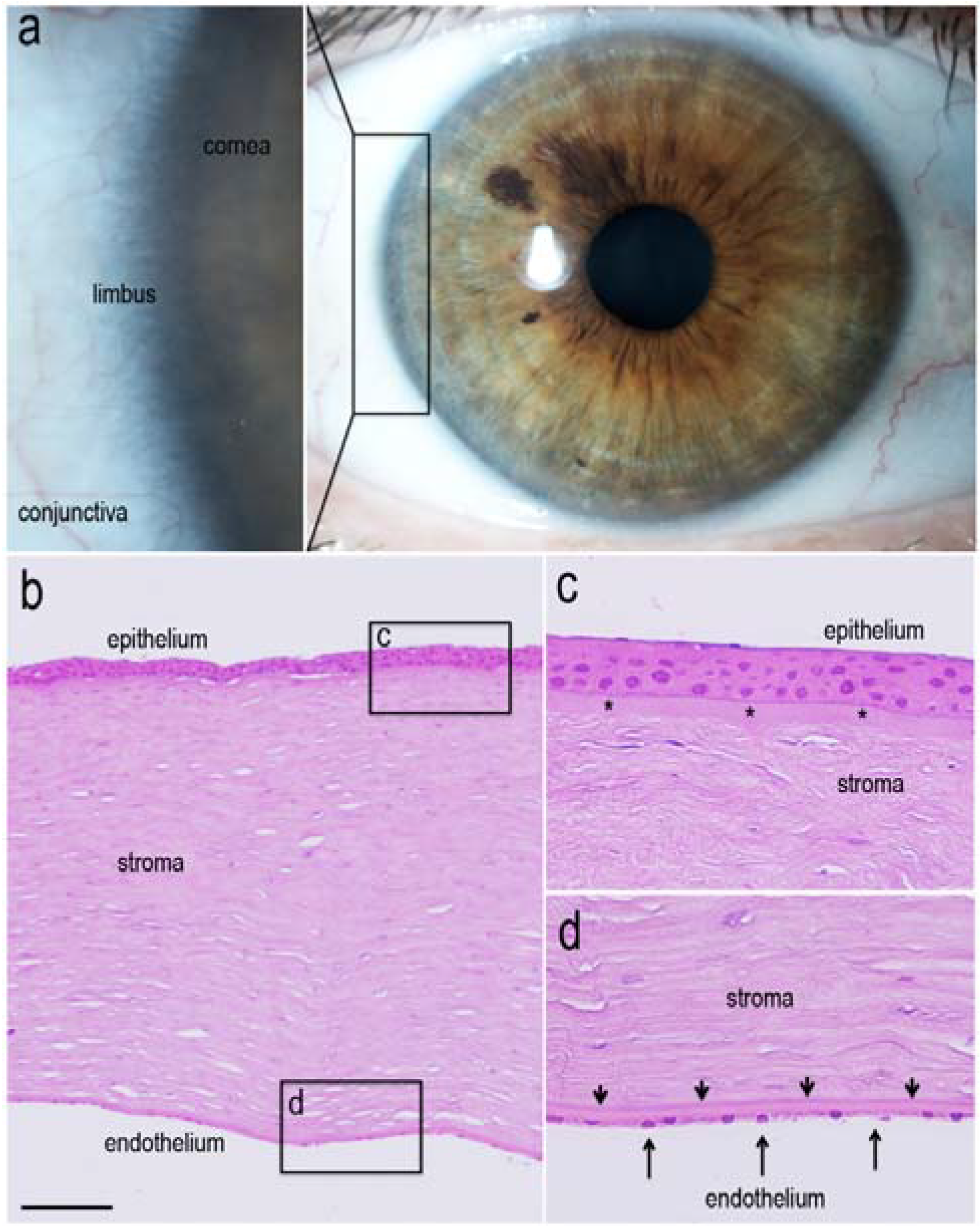
1.2. Renewal of Corneal Epithelium
1.3. Limbal Stem Cell Multipotency
2. Limbal Stem Cell Deficiency (LSCD)
| Etiology | Ocular Pathology |
|---|---|
| Idiopathic | - |
| Hereditary | Aniridia |
| Autosomal dominant keratitis | |
| Gelatinous drop-like corneal dystrophy | |
| Iris coloboma | |
| Xeroderma pigmentosa | |
| Epidermolysis bullosa | |
| Dyskeratosis congenita | |
| Ectodermic dysplasia | |
| Multiple endocrine neoplasia | |
| Polyglandular autoimmune syndromes | |
| Neoplasic | Intraepithelial neoplasia |
| Conjuntival tumors (melanoma) | |
| Limbal dermoid | |
| Degenerative | Recurrent pterygium |
| Salzmann nodular corneal dystrophy | |
| Infections | Severe infeccious keratitis |
| Chlamydia conjunctivitis | |
| Mechanical | Alkali, acid, thermal burns |
| Bullous keratopathy | |
| Tumor excision | |
| Cryotherapy, radioterapy | |
| Systemic and local chemotherapy (MMC, 5FU) | |
| UV radition | |
| Phototherapeutic keratectomy | |
| Anoxic | Contact lenses misuse or prolonged use |
| Trophic | Neurotrophic keratopathy |
| Inflammation | Superior limbic keratoconjunctivitis |
| Collagen diseases related ulcers | |
| Mooren ulcer | |
| Atopic keratoconjunctivitis | |
| Ocular pemphigoid | |
| Ocular rosacea | |
| Stevens-Johnson syndrome | |
| Graft-versus-host disease | |
| Vitamin A deficiency |
2.1. Cell-Based Treatments for LSCD
2.2. Alternative Cell Sources for LSCD Treatment
| Cell Sources | Application | References |
|---|---|---|
| Cultured Limbal Epithelial Cells (CLET) | Clinical application | [17,18,19,20,21] |
| Cultured Oral Mucosal Epithelial Cells (COMET) | Clinical application | [30,31,33,34,35,36,37] |
| Cultured Conjunctival Epithelial Cells | Clinical application | [38,39,40,41] |
| Cultured Embryonic Stem Cells | Mice model | [42,43,44,45] |
| Cultured Adult Epidermal Stem Cells | Goat model | [46,47,48] |
| Cultured Bone-Marrow Derived Mesenchymal Stem Cells | Rat and rabbit models | [49,50,51,52,53] |
| Cultured Adipose Derived Mesenchymal Stem Cells | In vitro model | [54,55] |
| Cultured Orbital Fat Mesenchymal Progenitor Cells | Mice model; in vitro model | [56,57,58] |
| Cultured Immature Dental Pulp Stem Cells | Rabbit model | [59,60] |
| Cultured Hair Follicle-Derived Stem Cells | Mice model | [61,62] |
| Cultured Umbilical Cord Stem Cells | Rabbit model | [63,64] |
| Clinical Trial | Identifier | Phase | Study Characteristics | Cell Source | Situation |
|---|---|---|---|---|---|
| Corneal Epithelium Repair and Therapy Using Autologous Limbal Stem Cell Transplantation. | NCT02148016 | Phase 1, Phase 2 | Open label, Interventional Non-randomized, SGA | Autologous LSCs | Currently recruiting |
| Multicenter Study of CAOMECS Transplantation to Patients With Total Limbal Stem Cell Deficiency. | NCT01489501 | Phase 3 | Open label, Interventional Non-randomized, SGA | Autologous OMC | Not yet open |
| The Improvement of Limbal Epithelial Culture Technique by Using Collagenase to Isolate Limbal Stem Cells. | NCT02202642 | Phase 1 | Open label, Interventional Non-randomized, SGA | Autologous LSCs | Currently recruiting |
| Autologous Transplantation of Cultivated Limbal Stem Cells on Amniotic Membrane in Limbal Stem Cell Deficiency (LSD) Patients. | NCT00736307 | Phase 1, Phase 2 | Open label, Interventional Non-randomized, SGA | Autologous LSCs | Completed |
| Clinical Trial on the Effect of Autologous Oral Mucosal Epithelial Sheet Transplantation. | NCT02149732 | Phase 1, Phase 2 | Open label, Interventional Non-randomized, SGA | Autologous OMEC | Currently recruiting |
| Cultivated Stem Cell Transplantation for the Treatment of Limbal Stem Cell Deficiency (LECT). | NCT00845117 | Phase 1, Phase 2 | Open label, Interventional Non-randomized, SGA | Autologous LSCs | Ongoing, but not recruiting |
| Limbal Epithelial Stem Cell Transplantation: a Phase II Multicenter Trial (MLEC) | NCT02318485 | Phase 2 | Open label, Interventional Non-randomized, SGA | Allogenic or autologous LSCs | Not yet open |
| Cell Therapy in Failure Syndromes in Limbal Stem Cells (TC181). | NCT01619189 | Phase 2 | Single blind, Interventional Non-randomized, SGA | Allogenic or autologous LSCs | Currently recruiting |
| Autologous Cultured Corneal Epithelium (CECA) for the Treatment of Limbal Stem Cell Deficiency. | NCT01756365 | Phase 1, Phase 2 | Open label, Interventional Non-randomized, SGA | Autologous cultured corneal epithelium | Enrolling by invitation |
| Ocular Surface Reconstruction With Cultivated Autologus Mucosal Epithelial Transplantation. | NCT01942421 | Phase 2, Phase 3 | Open label, Interventional Non-randomized, SGA | Autologous OMEC | Ongoing, but not recruiting |
| Efficacy of Cultivated Corneal Epithelial Stem Cell for Ocular Surface Reconstruction. | NCT01237600 | Phase 2, Phase 3 | Open label, Interventional Non-randomized, SGA | Allogenic or autologous LSCs | Completed |
| Safety Study of Stem Cell Transplant to Treat Limbus Insufficiency Syndrome. | NCT01562002 | Phase 1, Phase 2 | Double blind, Interventional Randomized, Parallel assignment | Allogenic LSCs vs. BM-MSCs | Ongoing, but not recruiting |
| The Application of Oral Mucosal Epithelial Cell Sheets Cultivated on Amino Membrane in Patients Suffering From Corneal Stem Cell Insufficiency or Symblepharon. | NCT00491959 | Phase 1 | Open label, Interventional Non-randomized, SGA | Autologous OMEC | Completed |
| Transplantation of Cultivated Corneal Epithelial Sheet in Patients With Ocular Surface Disease (CLET). | NCT01123044 | Phase 3 | Open label, Interventional Randomized, Parallel assignment | Autologous LSCs | Unknown |
| Application of Cell Therapy for Ocular Surface Repair Using Progenitor Cells of Sclerocorneal Limbus Amplified ex vivo (MeRSO09). | NCT01470573 | Phase 2 | Open label, Interventional Non-randomized, SGA | Autologous LSCs | Completed |
| The Application of Cultured Cornea Stem Cells in Patients Suffering From Corneal Stem Cell Insufficiency. | NCT01377311 | Phase 1 | Open label, Interventional Non-randomized, SGA | Autologous LSCs | Completed |
3. IPSCs and Corneal Epithelial Differentiation
3.1. Application of IPSCs for Ocular Pathology
3.2. Molecular Mechanisms of Corneal Epithelial Reprogramming
3.3. Restoration of Corneal Stromal Transparency
3.4. Future Trends for IPSC Technology
4. Conclusions
Acknowledgements
Author Contributions
Conflicts of Interest
References
- Notara, M.; Alatza, A.; Gilfillan, J.; Harris, A.R.; Levis, H.J.; Schrader, S.; Vernon, A.; Daniels, J.T. In sickness and in health: Corneal epithelial stem cell biology, pathology and therapy. Exp. Eye Res. 2010, 90, 188–195. [Google Scholar] [CrossRef] [PubMed]
- Osei-Bempong, C.; Figueiredo, F.C.; Lako, M. The limbal epithelium of the eye—A review of limbal stem cell biology, disease and treatment. Bioessays 2013, 35, 211–219. [Google Scholar] [CrossRef] [PubMed]
- Casaroli-Marano, R.P.; Nieto-Nicolau, N.; Martínez-Conesa, E.M. Progenitor cells for ocular surface regenerative therapy. Ophthalmic Res. 2013, 49, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, S.; Kolli, S.; Lako, M.; Figueiredo, F.C.; Daniels, J.T. Stem cell therapies for ocular surface disease. Drug Discov. Today 2010, 15, 306–313. [Google Scholar] [CrossRef] [PubMed]
- Utheim, T.P. Limbal epithelial cell therapy: Past, present, and future. Methods Mol. Biol. 2013, 1014, 3–43. [Google Scholar] [PubMed]
- Li, W.; Hayashida, Y.; Chen, Y.T.; Tseng, S.C. Niche regulation of corneal epithelial stem cells at the limbus. Cell Res. 2007, 17, 26–36. [Google Scholar] [CrossRef] [PubMed]
- Dua, H.S.; Shanmuganathan, V.A.; Powell-Richards, A.O.; Tighe, P.J.; Joseph, A. Limbal epithelial crypts: A novel anatomical structure and a putative limbal stem cell niche. Br. J. Ophthalmol. 2005, 89, 529–532. [Google Scholar] [CrossRef] [PubMed]
- Shortt, A.J.; Secker, G.A.; Munro, P.M.; Khaw, P.T.; Tuft, S.J.; Daniels, J.T. Characterization of the limbal epithelial stem cell niche: Novel imaging techniques permit in vivo observation and targeted biopsy of limbal epithelial stem cells. Stem Cells 2007, 25, 1402–1409. [Google Scholar] [CrossRef] [PubMed]
- Thoft, R.A.; Friend, J. The X, Y, Z hypothesis of corneal epithelial maintenance. Invest. Ophthalmol. Vis. Sci. 1987, 24, 1442–1443. [Google Scholar]
- Majo, F.; Rochat, A.; Nicolas, M.; Jaoude, G.A.; Barrandon, Y. Oligopotent stem cells are distributed throughout the mammalian ocular surface. Nature 2008, 456, 250–254. [Google Scholar] [CrossRef] [PubMed]
- Dua, H.S.; Miri, A.; Alomar, T.; Yeung, A.M.; Said, D.G. The role of limbal stem cells in corneal epithelial maintenance: Testing the dogma. Ophthalmology 2009, 116, 856–863. [Google Scholar] [CrossRef] [PubMed]
- Pellegrini, G.; Golisano, O.; Paterna, P.; Lambiase, A.; Bonini, S.; Rama, P.; De Luca, M. Location and clonal analysis of stem cells and their differentiated progeny in the human ocular surface. J. Cell Biol. 1999, 145, 769–782. [Google Scholar] [CrossRef] [PubMed]
- Lindberg, K.; Brown, M.E.; Chaves, H.V.; Kenyon, K.R.; Rheinwald, J.G. In vitro propagation of human ocular surface epithelial cells for transplantation. Invest. Ophthalmol. Vis. Sci. 1993, 34, 2672–2679. [Google Scholar] [PubMed]
- Kruse, F.E. Stem cells and corneal epithelial regeneration. Eye 1994, 8, 170–183. [Google Scholar] [CrossRef] [PubMed]
- Kiritoshi, A.; Sundar-Raj, N.; Thoft, R.A. Differentiation in cultured limbal epithelium as defined by keratin expression. Invest. Ophthalmol. Vis. Sci. 1991, 32, 3073–3077. [Google Scholar] [PubMed]
- Cotsarelis, G.; Cheng, S.Z.; Dong, G.; Sun, T.T.; Lavker, R.M. Existence of slow-cycling limbal epithelial basal cells that can be preferentially stimulated to proliferate: Implications on epithelial stem cells. Cell 1989, 57, 201–209. [Google Scholar] [CrossRef] [PubMed]
- Pellegrini, G.; Traverso, C.E.; Franzi, A.T.; Zingirian, M.; Cancedda, R.; de Luca, M. Long-term restoration of damaged corneal surfaces with autologous cultivated corneal epithelium. Lancet 1997, 349, 990–993. [Google Scholar] [CrossRef] [PubMed]
- Shortt, A.J.; Secker, G.A.; Notara, M.D.; Limb, G.A.; Khaw, P.T.; Tuft, S.J.; Daniels, J.T. Transplatation of ex vivo cultured limbal epitehlial stem cells: A review of techniques and clinical results. Surv. Ophthalmol. 2007, 52, 483–502. [Google Scholar] [CrossRef] [PubMed]
- Rama, P.; Matuska, S.; Paganoni, G.; Spinelli, A.; de Luca, M.; Pellegrini, G. Limbal stem-cell therapy and long-term corneal regeneration. N. Engl. J. Med. 2010, 363, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Sangwan, V.S.; Basu, S.; Vemuganti, G.K.; Sejpal, K.; Subramaniam, S.V.; Bandyopadhyay, S.; Krishnaiah, S.; Gaddipati, S.; Tiwari, S.; Balasubramanian, D. Clinical outcomes of xeno-free autologous cultivated limbal epithelial transplantation: A 10-year study. Br. J. Ophthalmol. 2011, 95, 1525–1529. [Google Scholar] [CrossRef] [PubMed]
- Baylis, O.; Figueiredo, F.; Henein, C.; Lako, M.; Ahmad, S. 13 years of cultured limbal epithelial cell therapy: A review of the outcomes. J. Cell Biochem. 2011, 112, 993–1002. [Google Scholar] [CrossRef] [PubMed]
- Pellegrini, G.; Rama, P.; di Rocco, A.; Panaras, A.; de Luca, M. Concise review: Hurdles in a successful example of limbal stem cell-based regenerative medicine. Stem Cells 2014, 32, 26–34. [Google Scholar] [CrossRef] [PubMed]
- Menzel-Severing, J.; Kruse, F.E.; Schlötzer-Schrehardt, U. Stem cell-based therapy for corneal epithelial reconstruction: Present and future. Can. J. Ophthalmol. 2013, 48, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, S. Concise review: Limbal stem cell deficiency, dysfunction, and distress. Stem Cells Transl. Med. 2012, 1, 110–115. [Google Scholar] [CrossRef] [PubMed]
- Mönks, T.; Busin, M. Modified technique of autologous conjunctival transplantation after corneal chemical burn. Klin. Monbl. Augenheilkd. 1994, 204, 121–125. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, S.; Osei-Bempong, C.; Dana, R.; Jurkunas, U. The culture and transplantation of human limbal stem cells. J. Cell Physiol. 2010, 225, 15–19. [Google Scholar] [CrossRef] [PubMed]
- Tseng, S.C.; Chen, S.Y.; Shen, Y.C.; Chen, W.L.; Hu, F.R. Critical appraisal of ex vivo expansion of human limbal epitelial stem cells. Curr. Mol. Med. 2010, 10, 841–850. [Google Scholar] [CrossRef] [PubMed]
- Di Iorio, E.; Ferrari, S.; Fasolo, A.; Böhm, E.; Ponzin, D.; Barbaro, V. Techniques for culture and assessment of limbal stem cell grafts. Ocul. Surf. 2010, 8, 146–153. [Google Scholar] [CrossRef] [PubMed]
- O’Callaghan, A.R.; Daniels, J.T. Limbal epithelial stem cell therapy: Controversies and challenges. Stem Cells 2011, 29, 1923–1932. [Google Scholar] [CrossRef] [PubMed]
- Nishida, K.; Yamato, M.; Hayashida, Y.; Watanabe, K.; Yamamoto, K.; Adachi, E.; Nagai, S.; Kikuchi, A.; Maeda, N.; Watanabe, H.; et al. Corneal reconstruction with tissue engineered cell sheets composed of autologous oral mucosal epithelium. N. Engl. J. Med. 2004, 351, 1187–1196. [Google Scholar] [CrossRef] [PubMed]
- Kolli, S.; Ahmad, S.; Mudhar, H.S.; Meeny, A.; Lako, M.; Figueiredo, F.C. Successful application of ex vivo expanded human autologous oral mucosal epithelium for the treatment of total bilateral limbal stem cell deficiency. Stem Cells 2014, 32, 2135–2146. [Google Scholar] [CrossRef] [PubMed]
- Vemuganti, G.K.; Fatima, A.; Madhira, S.L.; Basti, S.; Sangwan, V.S. Limbal stem cells: Application in ocular biomedicine. Int. Rev. Cell Mol. Biol. 2009, 275, 133–181. [Google Scholar] [PubMed]
- Priya, C.G.; Arpitha, P.; Vaishali, S.; Prajna, N.V.; Usha, K.; Sheetal, K.; Muthukkaruppan, V.; et al. Adult human buccal epithelial stem cells: Identification, ex-vivo expansion, and transplantation for corneal surface reconstruction. Eye 2011, 25, 1641–1649. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, T.; Takeda, K.; Inatomi, T.; Sotozono, C.; Kinoshita, S. Long-term results of autologous cultivated oral mucosal epithelial transplantation in the scar phase of severe ocular surface disorders. Br. J. Ophthalmol. 2011, 95, 942–946. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Sheha, H.; Fu, Y.; Giegengack, M.; Tseng, S.C. Oral mucosal graft with amniotic membrane transplantation for total limbal stem cell deficiency. Am. J. Ophthalmol. 2011, 152, 739–747. [Google Scholar] [CrossRef] [PubMed]
- Inatomi, T.; Nakamura, T.; Kojyo, M.; Koizumi, N.; Sotozono, C.; Kinoshita, S. Ocular surface reconstruction with combination of cultivated autologous oral mucosal epithelial transplantation and penetrating keratoplasty. Am. J. Ophthalmol. 2006, 142, 757–764. [Google Scholar] [CrossRef] [PubMed]
- Takeda, K.; Nakamura, T.; Inatomi, T.; Sotozono, C.; Watanabe, A.; Kinoshita, S. Ocular surface reconstruction using the combination of autologous cultivated oral mucosal epithelial transplantation and eyelid surgery for severe ocular surface disease. Am. J. Ophthalmol. 2011, 152, 195–201. [Google Scholar] [CrossRef] [PubMed]
- Sangwan, V.S.; Vemuganti, G.K.; Iftekhar, G.; Bansal, A.K.; Rao, G.N. Use of autologous cultured limbal and conjunctival epithelium in a patient with severe bilateral ocular surface disease induced by acid injury: A case report of unique application. Cornea 2003, 22, 478–481. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.T.; Ang, L.P.; Beuerman, R.W. Reconstruction of the ocular surface by transplantation of a serum-free derived cultivated conjunctival epithelial equivalent. Transplantation 2004, 77, 1729–1734. [Google Scholar] [CrossRef] [PubMed]
- Ang, L.P.; Tanioka, H.; Kawasaki, S.; Ang, L.P.; Yamasaki, K.; Do, T.P.; Thein, Z.M.; Koizumi, N.; Nakamura, T.; Yokoi, N.; et al. Cultivated human conjunctival epithelial transplantation for total limbal stem cell deficiency. Invest. Ophthalmol. Vis. Sci. 2010, 51, 758–764. [Google Scholar] [CrossRef] [PubMed]
- Subramaniam, S.V.; Sejpal, K.; Fatima, A.; Gaddipati, S.; Vemuganti, G.K.; Sangwan, V.S. Coculture of autologous limbal and conjunctival epithelial cells to treat severe ocular surface disorders: Long-term survival analysis. Indian J. Ophthalmol. 2013, 61, 202–207. [Google Scholar] [CrossRef] [PubMed]
- Homma, R.; Yoshikawa, H.; Takeno, M.; Kurokawa, M.S.; Masuda, C.; Takada, E.; Tsubota, K.; Ueno, S.; Suzuki, N. Induction of epithelial progenitors in vitro from mouse embryonic stem cells and application for reconstruction of damaged cornea in mice. Invest. Ophthalmol. Vis. Sci. 2004, 45, 4320–4326. [Google Scholar] [CrossRef] [PubMed]
- Ueno, H.; Kurokawa, M.S.; Kayama, M.; Homma, R.; Kumagai, Y.; Masuda, C.; Takada, E.; Tsubota, K.; Ueno, S.; Suzuki, N. Experimental transplantation of corneal epithelium-like cells induced by Pax6 gene transfection of mouse embryonic stem cells. Cornea 2007, 26, 1220–1227. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, S.; Stewart, S.R.; Yung, S.; Kolli, S.; Armstrong, L.; Stojkovic, M.; Figueiredo, F.; Lako, M. Differentiation of human embryonic stem cells into corneal epithelial-like cells by in vitro replication of the corneal epithelial stem cell niche. Stem Cells 2007, 25, 1145–1155. [Google Scholar] [CrossRef] [PubMed]
- Hanson, C.; Hardarson, T.; Ellerstrom, C.; Nordberg, M.; Caisander, G.; Rao, M.; Hyllner, J.; Stenevi, U. Transplantation of human embryonic stem cells onto a partially wounded human cornea in vitro. Acta Ophthalmol. 2013, 91, 127–130. [Google Scholar] [CrossRef] [PubMed]
- Gao, N.; Wang, Z.; Huang, B.; Ge, J.; Lu, R.; Zhang, R.; Fan, Z.; Lu, L.; Peng, Z.; Cui, G. Putative epidermal stem cell convert into corneal epithelium-like cell under corneal tissue in vitro. Sci. China C Life Sci. 2007, 50, 101–110. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Qu, L.; Wang, X.; Zhao, M.; Li, W.; Hua, J.; Shi, M.; Moldovan, N.; Wang, H.; Dou, Z. Plasticity of epidermal adult stem cells derived from adult goat ear skin. Mol. Reprod. Dev. 2007, 74, 386–396. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Moldovan, N.I.; Zhao, Q.; Mi, S.; Zhou, Z.; Chen, D.; Gao, Z.; Tong, D.; Dou, Z. Reconstruction of damaged cornea by autologous transplantation of epidermal adult stem cells. Mol. Vis. 2008, 14, 1064–1070. [Google Scholar] [PubMed]
- Ma, Y.; Xu, Y.; Xiao, Z.; Yang, W.; Zhang, C.; Song, E.; Du, Y.; Li, L. Reconstruction of chemically burned rat corneal surface by bone marrow-derived human mesenchymal stem cells. Stem Cells 2006, 24, 315–321. [Google Scholar] [CrossRef] [PubMed]
- Jiang, T.S.; Cai, L.; Ji, W.Y.; Hui, Y.N.; Wang, Y.S.; Hu, D.; Zhu, J. Reconstruction of the corneal epithelium with induced marrow mesenchymal stem cells in rats. Mol. Vis. 2010, 16, 1304–1316. [Google Scholar] [PubMed]
- Gu, S.; Xing, C.; Han, J.; Tso, M.O.; Hong, J. Differentiation of rabbit bone marrow mesenchymal stem cells into corneal epithelial cells in vivo and ex vivo. Mol. Vis. 2009, 15, 99–107. [Google Scholar] [PubMed]
- Omoto, M.; Miyashita, H.; Shimmura, S.; Higa, K.; Kawakita, T.; Yoshida, S.; McGrogan, M.; Shimazaki, J.; Tsubota, K. The use of human mesenchymal stem cell-derived feeder cells for the cultivation of transplantable epithelial sheets. Invest. Ophthalmol. Vis. Sci. 2009, 50, 2109–2115. [Google Scholar] [CrossRef] [PubMed]
- Hu, N.; Zhang, Y.Y.; Gu, H.W.; Guan, H.J. Effects of bone marrow mesenchymal stem cells on cell proliferation and growth factor expression of limbal epithelial cells in vitro. Ophthalmic Res. 2012, 48, 82–88. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Conesa, E.M.; Espel, E.; Reina, M.; Casaroli-Marano, R.P. Characterization of ocular surface epithelial and progenitor cell markers in human adipose stromal cells derived from lipoaspirates. Invest. Ophthalmol. Vis. Sci. 2012, 53, 513–520. [Google Scholar] [CrossRef] [PubMed]
- Nieto-Miguel, T.; Galindo, S.; Reinoso, R.; Corell, A.; Martino, M.; Pérez-Simón, J.A.; Calonge, M. In vitro simulation of corneal epithelium microenvironment induces a corneal epithelial-like cell phenotype from human adipose tissue mesenchymal stem cells. Curr. Eye Res. 2013, 38, 933–944. [Google Scholar] [CrossRef] [PubMed]
- Ho, J.H.; Ma, W.H.; Tseng, T.C.; Chen, Y.F.; Chen, M.H.; Lee, O.K. Isolation and characterization of multi-potent stem cells from human orbital fat tissues. Tissue Eng. Part A 2011, 17, 255–266. [Google Scholar] [CrossRef] [PubMed]
- Lin, K.J.; Loi, M.X.; Lien, G.S.; Cheng, C.F.; Pao, H.Y.; Chang, Y.C.; Ji, A.T.; Ho, J.H. Topical administration of orbital fat-derived stem cells promotes corneal tissue regeneration. Stem Cell Res. Ther. 2013, 4. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.Y.; Mahabole, M.; Horesh, E.; Wester, S.; Goldberg, J.L.; Tseng, S.C. Isolation and Characterization of Mesenchymal Progenitor Cells from Human Orbital Adipose Tissue. Invest Ophthalmol. Vis. Sci. 2014, 55, 4842–4852. [Google Scholar] [CrossRef] [PubMed]
- Monteiro, B.G.; Serafim, R.C.; Melo, G.B.; Silva, M.C.; Lizier, N.F.; Maranduba, C.M.; Smith, R.L.; Kerkis, A.; Cerruti, H.; Gomes, J.A.; et al. Human immature dental pulp stem cells share key characteristic features with limbal stem cells. Cell Prolif. 2009, 42, 587–594. [Google Scholar] [CrossRef] [PubMed]
- Gomes, J.A.; Geraldes Monteiro, B.; Melo, G.B.; Smith, R.L.; Cavenaghi Pereira da Silva, M.; Lizier, N.F.; Kerkis, A.; Cerruti, H.; Kerkis, I. Corneal reconstruction with tissue-engineered cell sheets composed of human immature dental pulp stem cells. Invest. Ophthalmol. Vis. Sci. 2010, 51, 1408–1414. [Google Scholar] [CrossRef] [PubMed]
- Blazejewska, E.A.; Schlotzer-Schrehardt, U.; Zenkel, M.; Bachmann, B.; Chankiewitz, E.; Jacobi, C.; Kruse, F.E. Corneal limbal microenvironment can induce transdifferentiation of hair follicle stem cells into corneal epithelial-like cells. Stem Cells 2009, 27, 642–652. [Google Scholar] [CrossRef] [PubMed]
- Meyer-Blazejewska, E.A.; Call, M.K.; Yamanaka, O.; Liu, H.; Schlotzer-Schrehardt, U.; Kruse, F.E.; Kao, W.W. From hair to cornea: Toward the therapeutic use of hair follicle-derived stem cells in the treatment of limbal stem cell deficiency. Stem Cells 2011, 29, 57–66. [Google Scholar] [CrossRef] [PubMed]
- Reza, H.M.; Ng, B.Y.; Phan, T.T.; Tan, D.T.; Beuerman, R.W.; Ang, L.P. Characterization of a novel umbilical cord lining cell with CD227 positivity and unique pattern of P63 expression and function. Stem Cell Rev. 2011, 7, 624–638. [Google Scholar] [CrossRef] [PubMed]
- Reza, H.M.; Ng, B.Y.; Gimeno, F.L.; Phan, T.T.; Ang, L.P. Umbilical cord lining stem cells as a novel and promising source for ocular surface regeneration. Stem Cell Rev. 2011, 7, 935–947. [Google Scholar] [CrossRef] [PubMed]
- Kanayama, S.; Nishida, K.; Yamato, M.; Hayashi, R.; Sugiyama, H.; Soma, T.; Maeda, N.; Okano, T.; Tano, Y. Analysis of angiogenesis induced by cultured corneal and oral mucosal epithelial cell sheets in vitro. Exp. Eye Res. 2007, 85, 772–781. [Google Scholar] [CrossRef] [PubMed]
- Kanayama, S.; Nishida, K.; Yamato, M.; Hayashi, R.; Maeda, N.; Okano, T.; Tano, Y. Analysis of soluble vascular endothelial growth factor receptor-1 secreted from cultured corneal and oral mucosal epithelial cell sheets in vitro. Br. J. Ophthalmol. 2009, 93, 263–267. [Google Scholar] [CrossRef] [PubMed]
- ClinicalTrials.gov, a service of US National Institute of Health. Available online: https://clinicaltrials.gov/ (accessed 30 December 2014).
- Takahashi, K.; Yamanaka, S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 2006, 126, 663–676. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yu, J.; Vodyanik, M.A.; Smuga-Otto, K.; Antosiewicz-Bourget, J.; Frane, J.L.; Tian, S.; Nie, J.; Jonsdottir, G.A.; Ruotti, V.; Stewart, R.; et al. Induced pluripotent stem cell lines derived from human somatic cells. Science 2007, 318, 1917–1920. [Google Scholar] [CrossRef] [PubMed]
- Yamanaka, S. Induced pluripotent stem cells: Past, present, and future. Cell Stem Cell 2012, 10, 678–684. [Google Scholar] [CrossRef] [PubMed]
- Kelaini, S.; Cochrane, A.; Margariti, A. Direct reprogramming of adult cells: Avoiding the pluripotent state. Stem Cells Cloning 2014, 15, 19–29. [Google Scholar]
- Pietronave, S.; Prat, M. Advances and applications of induced pluripotent stem cells. Can. J. Physiol. Pharmacol. 2012, 90, 317–325. [Google Scholar] [CrossRef] [PubMed]
- Yang, K.; Jiang, Z.; Wang, D.; Lian, X.; Yang, T. Corneal epithelial-like transdifferentiation of hair follicle stem cells is mediated by pax6 and beta-catenin/Lef-1. Cell Biol. Int. 2009, 33, 861–866. [Google Scholar] [CrossRef] [PubMed]
- Saichanma, S.; Bunyaratvej, A.; Sila-Asna, M. In vitro transdifferentiation of corneal epithelial-like cells from human skin-derived precursor cells. Int. J. Ophthalmol. 2012, 5, 158–163. [Google Scholar] [PubMed]
- Casaroli-Marano, R.P.; Zarbin, M.A. Cell-based therapy for retinal degenerative disease. In Developments in Ophthalmology; Karger: Basel, Switzerland, 2014; Volume 53. [Google Scholar]
- Zhong, X.; Gutierrez, C.; Xue, T.; Hampton, C.; Vergara, M.N.; Cao, L.H.; Peters, A.; Park, T.S.; Zambidis, E.T.; Meyer, J.S.; et al. Generation of three-dimensional retinal tissue with functional photoreceptors from human iPSCs. Nat. Commun. 2014, 10. [Google Scholar] [CrossRef] [Green Version]
- Mekala, S.R.; Vauhini, V.; Nagarajan, U.; Maddileti, S.; Gaddipati, S.; Mariappan, I. Derivation, characterization and retinal differentiation of induced pluripotent stem cells. J. Biosci. 2013, 38, 123–134. [Google Scholar] [CrossRef] [PubMed]
- Notara, M.; Hernandez, D.; Mason, C.; Daniels, J.T. Characterization of the phenotype and functionality of corneal epithelial cells derived from mouse embryonic stem cells. Regen. Med. 2012, 7, 167–178. [Google Scholar] [CrossRef] [PubMed]
- Yu, D.; Chen, M.; Sun, X.; Ge, J. Differentiation of mouse induced pluripotent stem cells into corneal epithelial-like cells. Cell Biol. Int. 2013, 37, 87–94. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, R.; Ishikawa, Y.; Ito, M.; Kageyama, T.; Takashiba, K.; Fujioka, T.; Tsujikawa, M.; Miyoshi, H.; Yamato, M.; Nakamura, Y.; et al. Generation of corneal epithelial cells from induced pluripotent stem cells derived from human dermal fibroblast and corneal limbal epithelium. PLoS One 2012, 7, e45435. [Google Scholar] [CrossRef] [PubMed]
- Sareen, D.; Saghizadeh, M.; Ornelas, L.; Winkler, M.A.; Narwani, K.; Sahabian, A.; Funari, V.A.; Tang, J.; Spurka, L.; Punj, V.; et al. Differentiation of Human Limbal-Derived Induced Pluripotent Stem Cells Into Limbal-Like Epithelium. Stem Cells Transl. Med. 2014, 3, 1002–1012. [Google Scholar] [CrossRef] [PubMed]
- Mikhailova, A.; Ilmarinen, T.; Uusitalo, H.; Skottman, H. Small-molecule induction promotes corneal epithelial cell differentiation from human induced pluripotent stem cells. Stem Cell Rep. 2014, 6, 219–231. [Google Scholar] [CrossRef]
- Zhu, J.; Zhang, K.; Sun, Y.; Gao, X.; Li, Y.; Chen, Z.; Wu, X. Reconstruction of functional ocular surface by acellular porcine cornea matrix scaffold and limbal stem cells derived from human embryonic stem cells. Tissue Eng. Part A 2013, 19, 2412–2425. [Google Scholar] [CrossRef] [PubMed]
- Taylor, C.J.; Peacock, S.; Chaudhry, A.N.; Bradley, J.A.; Bolton, E.M. Generating an iPSC bank for HLA-matched tissue transplantation based on known donor and recipient HLA types. Cell Stem Cell 2012, 11, 147–152. [Google Scholar] [CrossRef] [PubMed]
- Alexandrova, E.M.; Petrenko, O.; Nemajerova, A.; Romano, R.A.; Sinha, S.; Moll, U.M. ΔNp63 regulates select routes of reprogramming via multiple mechanisms. Cell Death Differ. 2013, 20, 1698–1708. [Google Scholar] [CrossRef] [PubMed]
- Ng, W.L.; Chen, G.; Wang, M.; Wang, H.; Story, M.; Shay, J.W.; Zhang, X.; Wang, J.; Amin, A.R.; Hu, B.; et al. OCT4 as a target of miR-34a stimulates p63 but inhibits p53 to promote human cell transformation. Cell Death Dis. 2014, 5. [Google Scholar] [CrossRef]
- Shalom-Feuerstein, R.; Serror, L.; Aberdam, E.; Müller, F.J.; van Bokhoven, H.; Wiman, K.G.; Zhou, H.; Aberdam, D.; Petit, I. Impaired epithelial differentiation of induced pluripotent stem cells from ectodermal dysplasia-related patients is rescued by the small compound APR-246/PRIMA-1MET. Proc. Natl. Acad. Sci. USA 2013, 110, 2152–2156. [Google Scholar] [CrossRef] [PubMed]
- Shalom-Feuerstein, R.; Serror, L.; de La Forest Divonne, S.; Petit, I.; Aberdam, E.; Camargo, L.; Damour, O.; Vigouroux, C.; Solomon, A.; Gaggioli, C.; et al. Pluripotent stem cell model reveals essential roles for miR-450b-5p and miR-184 in embryonic corneal lineage specification. Stem Cell 2012, 30, 898–909. [Google Scholar] [CrossRef]
- Li, X.; Pei, D.; Zheng, H. Transitions between epithelial and mesenchymal states during cell fate conversions. Protein Cell 2014, 5, 580–591. [Google Scholar] [CrossRef] [PubMed]
- Grocott, T.; Johnson, S.; Bailey, A.P.; Streit, A. Neural crest cells organize the eye via TGF-β and canonical Wnt signalling. Nat. Commun. 2011, 2. [Google Scholar] [CrossRef]
- Chan, A.A.; Hertsenberg, A.J.; Funderburgh, M.L.; Mann, M.M.; Du, Y.; Davoli, K.A.; Mich-Basso, J.D.; Yang, L.; Funderburgh, J.L. Differentiation of human embryonic stem cells into cells with corneal keratocyte phenotype. PLoS One 2013, 8, e56831. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chien, Y.; Liao, Y.W.; Liu, D.M.; Lin, H.L.; Chen, S.J.; Chen, H.L.; Peng, C.H.; Liang, C.M.; Mou, C.Y.; Chiou, S.H. Corneal repair by human corneal keratocyte-reprogrammed iPSCs and amphiphatic carboxymethyl-hexanoyl chitosan hydrogel. Biomaterials 2012, 33, 8003–8016. [Google Scholar] [CrossRef] [PubMed]
- Fukuta, M.; Nakai, Y.; Kirino, K.; Nakagawa, M.; Sekiguchi, K.; Nagata, S.; Matsumoto, Y.; Yamamoto, T.; Umeda, K.; Heike, T.; et al. Derivation of Mesenchymal Stromal Cells from Pluripotent Stem Cells through a Neural Crest Lineage using Small Molecule Compounds with Defined Media. PLoS One 2014, 9, e112291. [Google Scholar] [CrossRef] [PubMed]
- Gore, A.; Li, Z.; Fung, H.; Young, J.E.; Agarwal, S.; Antosiewicz-Bourget, J.; Canto, I.; Giorgetti, A.; Israel, M.A.; Kiskinis, E.; et al. Somatic coding mutations in human induced pluripotent stem cells. Nature 2011, 471, 63–67. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Casaroli-Marano, R.P.; Nieto-Nicolau, N.; Martínez-Conesa, E.M.; Edel, M.; B.Álvarez-Palomo, A. Potential Role of Induced Pluripotent Stem Cells (IPSCs) for Cell-Based Therapy of the Ocular Surface. J. Clin. Med. 2015, 4, 318-342. https://doi.org/10.3390/jcm4020318
Casaroli-Marano RP, Nieto-Nicolau N, Martínez-Conesa EM, Edel M, B.Álvarez-Palomo A. Potential Role of Induced Pluripotent Stem Cells (IPSCs) for Cell-Based Therapy of the Ocular Surface. Journal of Clinical Medicine. 2015; 4(2):318-342. https://doi.org/10.3390/jcm4020318
Chicago/Turabian StyleCasaroli-Marano, Ricardo P., Núria Nieto-Nicolau, Eva M. Martínez-Conesa, Michael Edel, and Ana B.Álvarez-Palomo. 2015. "Potential Role of Induced Pluripotent Stem Cells (IPSCs) for Cell-Based Therapy of the Ocular Surface" Journal of Clinical Medicine 4, no. 2: 318-342. https://doi.org/10.3390/jcm4020318
APA StyleCasaroli-Marano, R. P., Nieto-Nicolau, N., Martínez-Conesa, E. M., Edel, M., & B.Álvarez-Palomo, A. (2015). Potential Role of Induced Pluripotent Stem Cells (IPSCs) for Cell-Based Therapy of the Ocular Surface. Journal of Clinical Medicine, 4(2), 318-342. https://doi.org/10.3390/jcm4020318







