Rehabilitation for Cardiorenal Multimorbidity: Epidemiology, Functional Phenotypes, and Effects on Physical Function, Renal Trajectory, and Prognosis
Abstract
1. Introduction
Scope and Methodological Boundaries (Focused Narrative Review)
2. Epidemiology and Clinical Relevance of Cardiorenal Overlap
3. Effects of Rehabilitation on Physical Function in Cardiorenal Populations
3.1. Evidence from CR
3.2. Evidence from Renal Rehabilitation
4. CR and Renal Outcomes
4.1. Center-Based CR and Renal Trajectory
4.2. Management of Habitual Physical Activity and Renal Trajectory in Cardiovascular Patients
5. Significance of Cystatin C Assessment When Evaluating Renal Endpoints
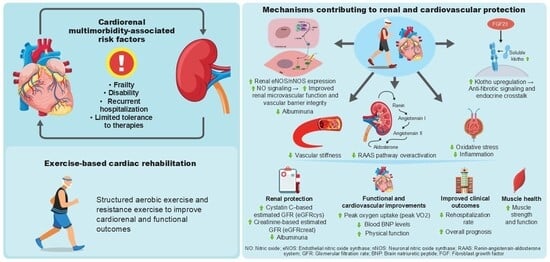
6. Mechanistic Pathways and Translational Evidence for CR-Mediated Renal Protection
7. Prognostic Implications
8. Evidence Gaps and Future Directions
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ACR | Albumin-to-creatinine ratio |
| AHA | American Heart Association |
| AMI | Acute myocardial infarction |
| AT | Anaerobic threshold |
| BNP | B-type natriuretic peptide |
| CKD | Chronic kidney disease |
| CKM | Cardiovascular–kidney–metabolic |
| CR | Cardiac rehabilitation |
| Cr | Creatinine |
| CRS | Cardiorenal syndrome |
| CVD | Cardiovascular disease |
| eGFR | Estimated glomerular filtration rate |
| eGFRcreat | Creatinine-based estimated glomerular filtration rate |
| eGFRcys | Cystatin C-based estimated glomerular filtration rate |
| eNOS | Endothelial nitric oxide synthase |
| FGF23 | Fibroblast growth factor 23 |
| Hct | Hematocrit |
| HDL-C | High-density lipoprotein cholesterol |
| JMDRD | Japanese Modification of Diet in Renal Disease equation |
| K | Potassium |
| KDIGO | Kidney Disease: Improving Global Outcomes |
| LVEF | Left ventricular ejection fraction |
| NO | Nitric oxide |
| nNOS | Neuronal nitric oxide synthase |
| Peak VO2 | Peak oxygen uptake |
| RAAS | Renin–angiotensin–aldosterone system |
| RCT | Randomized controlled trial |
| UN | Urea nitrogen |
References
- Rangaswami, J.; Bhalla, V.; Blair, J.E.A.; Chang, T.I.; Costa, S.; Lentine, K.L.; Lerma, E.V.; Mezue, K.; Molitch, M.; Mullens, W.; et al. Cardiorenal syndrome: Classification, pathophysiology, diagnosis, and treatment strategies: A scientific statement from the American Heart Association. Circulation 2019, 139, e840–e878. [Google Scholar] [CrossRef]
- Ndumele, C.E.; Neeland, I.J.; Tuttle, K.R.; Chow, S.L.; Mathew, R.O.; Khan, S.S.; Coresh, J.; Baker-Smith, C.M.; Carnethon, M.R.; Després, J.P.; et al. A synopsis of the evidence for the science and clinical management of cardiovascular-kidney-metabolic (CKM) syndrome: A scientific statement from the American Heart Association. Circulation 2023, 148, 1636–1664. [Google Scholar] [CrossRef]
- Zuo, X.; Li, X.; Tang, K.; Zhao, R.; Wu, M.; Wang, Y.; Li, T. Sarcopenia and cardiovascular diseases: A systematic review and meta-analysis. J. Cachexia Sarcopenia Muscle 2023, 14, 1183–1198. [Google Scholar] [CrossRef] [PubMed]
- Beddhu, S.; Wei, G.; Marcus, R.L.; Chonchol, M.; Greene, T. Light-intensity physical activities and mortality in the United States general population and CKD subpopulation. Clin. J. Am. Soc. Nephrol. 2015, 10, 1145–1153. [Google Scholar] [CrossRef]
- Pandey, A.; Kitzman, D.; Reeves, G. Frailty is intertwined with heart failure: Mechanisms, prevalence, prognosis, assessment, and management. JACC Heart Fail. 2019, 7, 1001–1011. [Google Scholar] [CrossRef]
- Kirkman, D.L.; Bohmke, N.; Carbone, S.; Garten, R.S.; Rodriguez-Miguelez, P.; Franco, R.L.; Kidd, J.M.; Abbate, A. Exercise intolerance in kidney diseases: Physiological contributors and therapeutic strategies. Am. J. Physiol. Ren. Physiol. 2021, 320, F161–F173. [Google Scholar] [CrossRef]
- Scrutinio, D.; Agostoni, P.; Gesualdo, L.; Corrà, U.; Mezzani, A.; Piepoli, M.; Di Lenarda, A.; Iorio, A.; Passino, C.; Magrì, D.; et al. Renal function and peak exercise oxygen consumption in chronic heart failure with reduced left ventricular ejection fraction. Circ. J. 2015, 79, 583–591. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Heidenreich, P.A.; Bozkurt, B.; Aguilar, D.; Allen, L.A.; Byun, J.J.; Colvin, M.M.; Deswal, A.; Drazner, M.H.; Dunlay, S.M.; Evers, L.R.; et al. 2022 AHA/ACC/HFSA guideline for the management of heart failure: A report of the American College of Cardiology/American Heart Association joint committee on clinical practice guidelines. Circulation 2022, 145, e895–e1032. [Google Scholar] [CrossRef] [PubMed]
- Tsutsui, H.; Ide, T.; Ito, H.; Kihara, Y.; Kinugawa, K.; Kinugawa, S.; Makaya, M.; Murohara, T.; Node, K.; Saito, Y.; et al. JCS/JHFS 2021 guideline focused update on diagnosis and treatment of acute and chronic heart failure. Circ. J. 2021, 85, 2252–2291. [Google Scholar] [CrossRef] [PubMed]
- Lamarche, F.; Brousseau-La Rosa, F.; Heitz, P.H.; Beruni, N.A.; Bondonno, N.P.; Wong, G.; Bernier-Jean, A. Exercise training for adults with chronic kidney disease not requiring dialysis. Cochrane Database Syst. Rev. 2026, 1, CD014654. [Google Scholar] [CrossRef]
- Baker, L.A.; March, D.S.; Wilkinson, T.J.; Billany, R.E.; Bishop, N.C.; Castle, E.M.; Chilcot, J.; Davies, M.D.; Graham-Brown, M.P.M.; Greenwood, S.A.; et al. Clinical practice guideline exercise and lifestyle in chronic kidney disease. BMC Nephrol. 2022, 23, 75. [Google Scholar] [CrossRef] [PubMed]
- Kouidi, E.; Hanssen, H.; Anding-Rost, K.; Cupisti, A.; Deligiannis, A.; Grupp, C.; Koufaki, P.; Leeson, P.; Segura-Orti, E.; Van Craenenbroeck, A.; et al. The role of exercise training on cardiovascular risk factors and heart disease in patients with chronic kidney disease G3-G5 and G5D: A Clinical Consensus Statement of the European Association of Preventive Cardiology of the ESC and the European Association of Rehabilitation in Chronic Kidney Diseasel. Eur. J. Prev. Cardiol. 2024, 31, 1493–1515. [Google Scholar] [CrossRef] [PubMed]
- Ronco, C.; Haapio, M.; House, A.A.; Anavekar, N.; Bellomo, R. Cardiorenal syndrome. J. Am. Coll. Cardiol. 2008, 52, 1527–1539. [Google Scholar] [CrossRef]
- Eijkelkamp, W.B.A.; de Graeff, P.A.; van Veldhuisen, D.J.; van Dokkum, R.P.E.; Gansevoort, R.T.; de Jong, P.E.; de Zeeuw, D.; Hillege, H.L.; Prevention of Renal and Vascular End-Stage Disease (PREVEND) Study Group. Effect of first myocardial ischemic event on renal function. Am. J. Cardiol. 2007, 100, 7–12. [Google Scholar] [CrossRef]
- Esmeijer, K.; Geleijnse, J.M.; de Fijter, J.W.; Giltay, E.J.; Kromhout, D.; Hoogeveen, E.K. Cardiovascular risk factors accelerate kidney function decline in post-myocardial infarction patients: The alpha omega cohort study. Kidney Int. Rep. 2018, 3, 879–888. [Google Scholar] [CrossRef]
- Klein, L.; Massie, B.M.; Leimberger, J.D.; O’Connor, C.M.; Piña, I.L.; Adams, K.F.; Califf, R.M.; Gheorghiade, M.; OPTIME-CHF Investigators. Admission or changes in renal function during hospitalization for worsening heart failure predict postdischarge survival: Results from the Outcomes of a Prospective Trial of intravenous milrinone for Exacerbations of Chronic Heart Failure (OPTIME-CHF). Circ. Heart Fail. 2008, 1, 25–33. [Google Scholar] [CrossRef]
- Heywood, J.T.; Fonarow, G.C.; Costanzo, M.R.; Mathur, V.S.; Wigneswaran, J.R.; Wynne, J.; ADHERE Scientific Advisory Committee and Investigators. High prevalence of renal dysfunction and its impact on outcome in 118,465 patients hospitalized with acute decompensated heart failure: A report from the ADHERE Database. J. Card. Fail. 2007, 13, 422–430. [Google Scholar] [CrossRef]
- Smith, G.L.; Lichtman, J.H.; Bracken, M.B.; Shlipak, M.G.; Phillips, C.O.; DiCapua, P.; Krumholz, H.M. Renal impairment and outcomes in heart failure: Systematic review and meta-analysis. J. Am. Coll. Cardiol. 2006, 47, 1987–1996. [Google Scholar] [CrossRef] [PubMed]
- Anavekar, N.S.; McMurray, J.J.V.; Velazquez, E.J.; Solomon, S.D.; Kober, L.; Rouleau, J.L.; White, H.D.; Nordlander, R.; Maggioni, A.; Dickstein, K.; et al. Relation between renal dysfunction and cardiovascular outcomes after myocardial infarction. N. Engl. J. Med. 2004, 351, 1285–1295. [Google Scholar] [CrossRef]
- Yamaguchi, J.; Kasanuki, H.; Ishii, Y.; Yagi, M.; Nagashima, M.; Fujii, S.; Koyanagi, R.; Ogawa, H.; Hagiwara, N.; Haze, K.; et al. Serum creatinine on admission predicts long-term mortality in acute myocardial infarction patients undergoing successful primary angioplasty: Data from the Heart Institute of Japan Acute Myocardial Infarction (HIJAMI) Registry. Circ. J. 2007, 71, 1354–1359. [Google Scholar] [CrossRef]
- Koganei, H.; Kasanuki, H.; Ogawa, H.; Tsurumi, Y. Association of glomerular filtration rate with unsuccessful primary percutaneous coronary intervention and subsequent mortality in patients with acute myocardial infarction: From the HIJAMI registry. Circ. J. 2008, 72, 179–185. [Google Scholar] [CrossRef][Green Version]
- Yamashita, Y.; Shiomi, H.; Morimoto, T.; Yaku, H.; Furukawa, Y.; Nakagawa, Y.; Ando, K.; Kadota, K.; Abe, M.; Nagao, K.; et al. Cardiac and noncardiac causes of long-term mortality in ST-segment-elevation acute myocardial infarction patients who underwent primary percutaneous coronary intervention. Circ. Cardiovasc. Qual. Outcomes 2017, 10, e002790. [Google Scholar] [CrossRef]
- Kooman, J.P.; Kotanko, P.; Schols, A.M.W.J.; Shiels, P.G.; Stenvinkel, P. Chronic kidney disease and premature ageing. Nat. Rev. Nephrol. 2014, 10, 732–742. [Google Scholar] [CrossRef]
- Saitoh, M.; Itoh, H.; Morotomi, N.; Ozawa, T.; Ishii, N.; Uewaki, R.; Hori, K.; Shiotani, Y.; Ando, M.; Nakashima, S.; et al. Impact of chronic kidney disease and anemia on physical function in patients with chronic heart failure. Cardioren. Med. 2014, 4, 73–81. [Google Scholar] [CrossRef]
- Kubo, I.; Izawa, K.P.; Kajisa, N.; Nakamura, H.; Kimura, K.; Ogura, A.; Kanai, M.; Makihara, A.; Nishio, R.; Matsumoto, D. Association between worsening renal function severity during hospitalization and low physical function at discharge: A retrospective cohort study of older patients with heart failure and chronic kidney disease from Japan. Eur. Geriatr. Med. 2023, 14, 869–878. [Google Scholar] [CrossRef]
- Odden, M.C.; Whooley, M.A.; Shlipak, M.G. Association of chronic kidney disease and anemia with physical capacity: The heart and soul study. J. Am. Soc. Nephrol. 2004, 15, 2908–2915. [Google Scholar] [CrossRef]
- Molloy, C.D.; Long, L.; Mordi, I.R.; Bridges, C.; Sagar, V.A.; Davies, E.J.; Coats, A.J.; Dalal, H.; Rees, K.; Singh, S.J.; et al. Exercise-based cardiac rehabilitation for adults with heart failure. Cochrane Database Syst. Rev. 2024, 3, CD003331. [Google Scholar] [CrossRef]
- Cersosimo, A.; Longo Elia, R.; Condello, F.; Colombo, F.; Pierucci, N.; Arabia, G.; Matteucci, A.; Metra, M.; Adamo, M.; Vizzardi, E.; et al. Cardiac rehabilitation in patients with atrial fibrillation. Minerva Cardiol. Angiol. 2025; Online ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Hamazaki, N.; Kamiya, K.; Yamamoto, S.; Nozaki, K.; Ichikawa, T.; Matsuzawa, R.; Yamashita, M.; Uchida, S.; Maekawa, E.; Meguro, K.; et al. Associations between kidney function and outcomes of comprehensive cardiac rehabilitation in patients with heart failure. Clin. Res. Cardiol. 2022, 111, 253–263. [Google Scholar] [CrossRef] [PubMed]
- Thompson, S.; Wiebe, N.; Arena, R.; Rouleau, C.; Aggarwal, S.; Wilton, S.B.; Graham, M.M.; Hemmelgarn, B.; James, M.T. Effectiveness and utilization of cardiac rehabilitation among people with CKD. Kidney Int. Rep. 2021, 6, 1537–1547. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.; Bai, Y.; Zhao, X.; Huang, L.; Wang, W.; Zhou, W.; Zhang, H. Therapeutic effects of exercise interventions for patients with chronic kidney disease: An umbrella review of systematic reviews and meta-analyses. BMJ Open 2022, 12, e054887. [Google Scholar] [CrossRef]
- Thompson, S.; Wiebe, N.; Padwal, R.S.; Gyenes, G.; Headley, S.A.E.; Radhakrishnan, J.; Graham, M. The effect of exercise on blood pressure in chronic kidney disease: A systematic review and meta-analysis of randomized controlled trials. PLoS ONE 2019, 14, e0211032. [Google Scholar] [CrossRef]
- Vanden Wyngaert, K.; Van Craenenbroeck, A.H.; Van Biesen, W.; Dhondt, A.; Tanghe, A.; Van Ginckel, A.; Celie, B.; Calders, P. The effects of aerobic exercise on eGFR, blood pressure and VO2peak in patients with chronic kidney disease stages 3–4: A systematic review and meta-analysis. PLoS ONE 2018, 13, e0203662. [Google Scholar] [CrossRef]
- Zoccali, C.; Manfredini, F.; Kanbay, M.; Mallamaci, F. Intradialysis exercise in haemodialysis patients: Effective but complex and costly. Nephrol. Dial. Transplant. 2023, 39, 7–9. [Google Scholar] [CrossRef]
- Greenwood, S.A.; Koufaki, P.; Macdonald, J.H.; Bhandari, S.; Burton, J.O.; Dasgupta, I.; Farrington, K.; Ford, I.; Kalra, P.A.; Kean, S.; et al. Randomized trial-PrEscription of intradialytic exercise to improve quAlity of Life in Patients Receiving hemodialysis. Kidney Int. Rep. 2021, 6, 2159–2170. [Google Scholar] [CrossRef]
- Tabibi, M.A.; Cheema, B.; Salimian, N.; Corrêa, H.L.; Ahmadi, S. The effect of intradialytic exercise on dialysis patient survival: A randomized controlled trial. BMC Nephrol. 2023, 24, 100. [Google Scholar] [CrossRef]
- Zhang, D.; Yu, L.; Xia, B.; Zhang, X.; Liang, P.; Hu, X. Systematic review and meta-analysis of the efficacy of exercise intervention in kidney transplant recipients. World J. Urol. 2023, 41, 3449–3469. [Google Scholar] [CrossRef] [PubMed]
- Knobbe, T.J.; Kremer, D.; Zelle, D.M.; Klaassen, G.; Dijkema, D.; van Vliet, I.M.Y.; Leurs, P.B.; Bemelman, F.J.; Christiaans, M.H.L.; Berger, S.P.; et al. Effect of an exercise intervention or combined exercise and diet intervention on health-related quality of life-physical functioning after kidney transplantation: The Active Care after Transplantation (ACT) multicentre randomised controlled trial. Lancet Healthy Longev. 2024, 5, 100622. [Google Scholar] [CrossRef] [PubMed]
- Billany, R.E.; Bishop, N.C.; Castle, E.M.; Graham-Brown, M.P.M.; Greenwood, S.A.; Lightfoot, C.J.; Wilkinson, T.J. Physical activity interventions in adult kidney transplant recipients: An updated systematic review and meta-analysis of randomized controlled trials. Ren. Fail. 2025, 47, 2480246. [Google Scholar] [CrossRef]
- Billany, R.E.; Macdonald, J.H.; Burns, S.; Chowdhury, R.; Ford, E.C.; Mubaarak, Z.; Sohansoha, G.K.; Vadaszy, N.; Young, H.M.L.; Bishop, N.C.; et al. A structured, home-based exercise programme in kidney transplant recipients (ECSERT): A randomised controlled feasibility study. PLoS ONE 2025, 20, e0316031. [Google Scholar] [CrossRef]
- De Smet, S.; Vandecruys, M.; De Beir, J.; Leunis, S.; Wyngaert, K.V.; Renier, M.; de Loor, H.; Goetschalckx, K.; Naesens, M.; De Smedt, D.; et al. Home-based exercise and physical activity intervention after kidney transplantation: Impact of exercise intensity (Phoenix-Kidney). Protocol for a multicentre randomized controlled trial. Clin. Kidney J. 2025, 18, sfaf114. [Google Scholar] [CrossRef] [PubMed]
- Toyama, K.; Sugiyama, S.; Oka, H.; Sumida, H.; Ogawa, H. Exercise therapy correlates with improving renal function through modifying lipid metabolism in patients with cardiovascular disease and chronic kidney disease. J. Cardiol. 2010, 56, 142–146. [Google Scholar] [CrossRef]
- Takaya, Y.; Kumasaka, R.; Arakawa, T.; Ohara, T.; Nakanishi, M.; Noguchi, T.; Yanase, M.; Takaki, H.; Kawano, Y.; Goto, Y. Impact of cardiac rehabilitation on renal function in patients with and without chronic kidney disease after acute myocardial infarction. Circ. J. 2014, 78, 377–384. [Google Scholar] [CrossRef] [PubMed]
- Kimura, S.; Ueda, Y.; Ise, T.; Yagi, S.; Iwase, T.; Nishikawa, K.; Yamaguchi, K.; Yamada, H.; Soeki, T.; Wakatsuki, T.; et al. Impact of supervised cardiac rehabilitation on urinary albumin excretion in patients with cardiovascular disease. Int. Heart J. 2015, 56, 105–109. [Google Scholar] [CrossRef][Green Version]
- Iso, Y.; Kitai, H.; Kowaita, H.; Kyuno, E.; Maezawa, H.; Hashimoto, T.; Takahashi, T.; Sanbe, T.; Suzuki, H. Association of aging with glomerular filtration changes in cardiac rehabilitation participants with chronic kidney disease. Int. J. Cardiol. 2015, 187, 283–285. [Google Scholar] [CrossRef]
- Fujimi, K.; Miura, S.I.; Matsuda, T.; Fujita, M.; Ura, Y.; Kaino, K.; Sakamoto, M.; Horita, T.; Arimura, T.; Shiga, Y.; et al. Influence of a cardiac rehabilitation program on renal function in patients with cardiovascular disease in a one-year follow-up. Cardiol. Res. 2015, 6, 311–315. [Google Scholar] [CrossRef][Green Version]
- Hama, T.; Oikawa, K.; Ushijima, A.; Morita, N.; Matsukage, T.; Ikari, Y.J.; Kobayashi, Y. Effect of cardiac rehabilitation on the renal function in chronic kidney disease—Analysis using serum cystatin-C based glomerular filtration rate. Int. J. Cardiol. Heart Vasc. 2018, 19, 27–33. [Google Scholar] [CrossRef]
- Kitajima, K.; Fujimi, K.; Matsuda, T.; Fujita, M.; Kaino, K.; Teshima, R.; Ujifuku, Y.; Horita, T.; Sakamoto, M.; Arimura, T.; et al. Possibility of cardio-renal protection by long-term cardiac rehabilitation in elderly patients with cardiovascular diseases. Intern. Med. 2019, 58, 2133–2138. [Google Scholar] [CrossRef] [PubMed]
- Sasamoto, Y.; Endo, N.; Kanazawa, K.; Utsumi, T.; Takahashi, T.; Endo, Y.; Nanba, M.; Takeda, H.; Takano, T.; Takahashi, K.; et al. Outpatient cardiac rehabilitation suppresses deterioration of renal function in patients ≥75 years of age with heart disease. Circ. J. 2021, 85, 612–622. [Google Scholar] [CrossRef]
- Hama, T.; Ushijima, A.; Goto, T.; Nagamatsu, H.; Morita, N.; Yoshimachi, F.; Ikari, Y.; Kobayashi, Y. Effect of cardiac rehabilitation on glomerular filtration rate using serum cystatin C concentration in patients with cardiovascular disease and renal dysfunction. J. Cardiopulm. Rehabil. Prev. 2022, 42, E15–E22. [Google Scholar] [CrossRef]
- Sato, T.; Morishita, S.; Ono, M.; Funami, Y.; Hayao, K.; Kusano, D.; Kohzuki, M. Peak exercise oxygen uptake and changes in renal function in patients after acute myocardial infarction. Heart Lung 2023, 57, 277–282. [Google Scholar] [CrossRef]
- Xu, J.; Nishitani-Yokoyama, M.; Kasuya, H.; Yamashita, M.; Sato, Y.; Nishimura, J.; Iida, M.; Fujiwara, K.; Kunimoto, M.; Sugita-Yamaguchi, Y.; et al. The relationship between health-related quality of life and frailty in older patients participating in early Phase II cardiac rehabilitation. Circ. Rep. 2025, 7, 411–418. [Google Scholar] [CrossRef] [PubMed]
- Sato, T.; Kohzuki, M.; Ono, M.; Muto, M.; Osugi, T.; Kawamura, K.; Naganuma, W.; Sato, M.; Shishito, N. Association between physical activity and change in renal function in patients after acute myocardial infarction. PLoS ONE 2019, 14, e0212100. [Google Scholar] [CrossRef]
- Sato, T.; Kohzuki, M.; Ono, M.; Muto, M.; Osugi, T.; Kawamura, K.; Naganuma, W.; Sato, M.; Tsuchikawa, M.; Shishito, N.; et al. Association between physical activity and changes in renal function in patients after acute myocardial infarction: A dual-center prospective study. J. Cardiol. 2021, 78, 120–128. [Google Scholar] [CrossRef]
- Sato, T.; Kohzuki, M. Author’s reply. J. Cardiol. 2021, 78, 598–599. [Google Scholar] [CrossRef] [PubMed]
- Sato, T.; Ono, M.; Kawamura, K.; Naganuma, W.; Shishito, N.; Morishita, S.; Sasamoto, Y.; Kohzuki, M. Impact of moderate to vigorous intensity physical activity on change in renal function in patients after acute myocardial infarction. Heart Vessel. 2024, 39, 393–403. [Google Scholar] [CrossRef] [PubMed]
- Kidney Disease: Improving Global Outcomes (KDIGO) CKD Work Group. KDIGO 2024 clinical practice guideline for the evaluation and management of chronic kidney disease. Kidney Int. 2024, 105, S117–S314. [Google Scholar] [CrossRef]
- Bishop, N.C.; Burton, J.O.; Graham-Brown, M.P.M.; Stensel, D.J.; Viana, J.L.; Watson, E.L. Exercise and chronic kidney disease: Potential mechanisms underlying the physiological benefits. Nat. Rev. Nephrol. 2023, 19, 244–256. [Google Scholar] [CrossRef]
- Zhao, J.; Guan, Y.; Jia, Y.; Chen, Y.; Cai, Y. Aerobic exercise up-regulates klotho to improve renal fibrosis associated with aging and its mechanism. PLoS ONE 2024, 19, e0311055. [Google Scholar] [CrossRef]
- Valenzuela, P.L.; Castillo-García, A.; Saco-Ledo, G.; Santos-Lozano, A.; Lucia, A. Physical exercise: A polypill against chronic kidney disease. Nephrol. Dial. Transplant. 2024, 39, 1384–1391. [Google Scholar] [CrossRef]
- Ito, D.; Ito, O.; Mori, N.; Cao, P.; Suda, C.; Muroya, Y.; Hao, K.; Shimokawa, H.; Kohzuki, M. Exercise training upregulates nitric oxide synthases in the kidney of rats with chronic heart failure. Clin. Exp. Pharmacol. Physiol. 2013, 40, 617–625. [Google Scholar] [CrossRef]
- Ito, D.; Ito, O.; Cao, P.; Mori, N.; Suda, C.; Muroya, Y.; Takashima, K.; Ito, S.; Kohzuki, M. Effects of exercise training on nitric oxide synthase in the kidney of spontaneously hypertensive rats. Clin. Exp. Pharmacol. Physiol. 2013, 40, 74–82. [Google Scholar] [CrossRef]
- Ogawa, Y.; Takahashi, J.; Sakuyama, A.; Xu, L.; Miura, T.; Muroya, Y.; Ito, D.; Kohzuki, M.; Ito, O. Exercise training delays renal disorders with decreasing oxidative stress and increasing production of 20-hydroxyeicosatetraenoic acid in Dahl salt-sensitive rats. J. Hypertens. 2020, 38, 1336–1346. [Google Scholar] [CrossRef] [PubMed]
- Ito, D.; Cao, P.; Kakihana, T.; Sato, E.; Suda, C.; Muroya, Y.; Ogawa, Y.; Hu, G.; Ishii, T.; Ito, O.; et al. Chronic running exercise alleviates early progression of nephropathy with upregulation of nitric oxide synthases and suppression of glycation in Zucker diabetic rats. PLoS ONE 2015, 10, e0138037. [Google Scholar] [CrossRef] [PubMed]
- Kohzuki, M.; Kamimoto, M.; Wu, X.M.; Xu, H.L.; Kawamura, T.; Mori, N.; Nagasaka, M.; Kurosawa, H.; Minami, N.; Kanazawa, M.; et al. Renal protective effects of chronic exercise and antihypertensive therapy in hypertensive rats with chronic renal failure. J. Hypertens. 2001, 19, 1877–1882. [Google Scholar] [CrossRef]
- Castillo, R.F.; Pérez, R.G.; González, A.L. Beneficial effects of physical exercise on the osteo-renal klotho–FGF-23 axis in chronic kidney disease: A systematic review with meta-analysis. Int. J. Med. Sci. 2024, 21, 332–340. [Google Scholar] [CrossRef]
- Davies, M.D.; Hughes, F.; Sandoo, A.; Alejmi, A.; Macdonald, J.H. The effect of exercise on vascular health in chronic kidney disease: A systematic review and meta-analysis of randomized controlled trials. Am. J. Physiol. Ren. Physiol. 2023, 325, F638–F655. [Google Scholar] [CrossRef]
- Kohzuki, M. Renal rehabilitation: Present and future perspectives. J. Clin. Med. 2024, 13, 552. [Google Scholar] [CrossRef]
- Hama, T.; Kakuta, T.; Amano, K.; Ushijima, A.; Yoshimachi, F.; Ikari, Y. Effect of cardiac rehabilitation on cardiovascular events in patients with advanced chronic kidney disease. Ther. Apher. Dial. 2025, 29, 856–865. [Google Scholar] [CrossRef]
- Kutner, N.G.; Zhang, R.; Huang, Y.; Herzog, C.A. Cardiac rehabilitation and survival of dialysis patients after coronary bypass. J. Am. Soc. Nephrol. 2006, 17, 1175–1180. [Google Scholar] [CrossRef]
- Huang, Y.; Zhang, R.; Culler, S.D.; Kutner, N.G. Costs and effectiveness of cardiac rehabilitation for dialysis patients following coronary bypass. Kidney Int. 2008, 74, 1079–1084. [Google Scholar] [CrossRef]
- Thomas, R.J.; Beatty, A.L.; Beckie, T.M.; Brewer, L.C.; Brown, T.M.; Forman, D.E.; Franklin, B.A.; Keteyian, S.J.; Kitzman, D.W.; Regensteiner, J.G.; et al. Home-based cardiac rehabilitation: A scientific statement from the American Association of Cardiovascular and Pulmonary Rehabilitation, the American Heart Association, and the American College of Cardiology. Circulation 2019, 140, e69–e89. [Google Scholar] [CrossRef] [PubMed]
- Hughes, J.W.; Berry, R.; Brown, T.M.; Carlin, B.; Drwal, K.; Keteyian, S.J.; Prince, D.Z.; Wu, W.C. Consensus statement on the virtual and remote delivery of cardiac and pulmonary rehabilitation and their components. J. Cardiopulm. Rehabil. Prev. 2025, 45, 387–396. [Google Scholar] [CrossRef] [PubMed]
- Scherrenberg, M.; Falter, M.; Abreu, A.; Aktaa, S.; Busnatu, S.; Casado-Arroyo, R.; Dendale, P.; Dilaveris, P.; Locati, E.T.; Marques-Sule, E.; et al. Standards for cardiac telerehabilitation. Eur. Heart J. 2025, 46, 3714–3737. [Google Scholar] [CrossRef] [PubMed]


| Domain | CRS | CKM Framework |
|---|---|---|
| Concept | Pathophysiologic syndrome describing bidirectional heart–kidney interactions | Population health framework integrating cardiovascular, kidney, and metabolic disease across a risk continuum |
| Primary focus | Heart–kidney cross-talk and acute or chronic organ dysfunction | Integrated cardiometabolic and kidney risk continuum with prevention-oriented staging |
| Disease scope | Primarily heart and kidney dysfunction | Cardiovascular disease, CKD, obesity, diabetes, and metabolic risk factors |
| Classification | Five subtypes based on primary organ involvement and time course (CRS Types 1–5) | Staged continuum from risk to clinical disease (Stages 0–4) |
| Pathophysiological emphasis | Hemodynamics, congestion, neurohormonal activation (RAAS/SNS), and inflammation | Excess or dysfunctional adiposity, insulin resistance, metabolic dysregulation, systemic inflammation, and multisystem interactions |
| Clinical context | Often applied in acute or advanced settings (e.g., acute HF with AKI; advanced CKD–CVD interplay) | Designed for prevention, early risk identification, and long-term management across the life course |
| Clinical goal | Guide understanding and management of cardiorenal dysfunction | Coordinate prevention and treatment strategies across cardiovascular, renal, and metabolic health |
| Relevance to rehabilitation | Mechanistic rationale for exercise effects on hemodynamics, endothelial, and neurohormonal pathways | Emphasizes lifestyle interventions (physical activity and rehabilitation) across stages, supporting earlier and broader implementation |
| Study (Year) | Population (Participants) | Design/Setting | Intervention/Exposure | Duration/Follow-Up | Renal Markers and Other Outcomes | Main Renal Finding(s) |
|---|---|---|---|---|---|---|
| Toyama et al. [42] | CVD + CKD (n = 19; exercise n = 10 vs. non-exercise n = 9) | Non-RCT; comparative (CR vs. non-CR) | CR vs. non-CR | 12 weeks | eGFR (creatinine-based); lipids; AT-O2 | CR: eGFR improved; ΔeGFR correlated positively with ΔAT-O2 and ΔHDL-C and negatively with Δtriglycerides. |
| Takaya et al. [43] | AMI (total n = 528; CKD subgroup n = 180) | Non-RCT; pre–post within CR participants; attendance stratification | 3-month CR; CKD attendance: non-active (≤1/wk) vs. active (≥1.1/wk) | 3 months | eGFR (creatinine-based); peak O2; BNP | CKD subgroup: eGFR improved 48 ± 12 → 53 ± 15 (p < 0.001); active CKD improved 50 ± 10 → 53 ± 13; non-active no change; non-CKD no change. |
| Kimura et al. [44] | Male CVD patients without macroalbuminuria (n = 98); CR n = 23 vs. non-CR n = 75 | Non-RCT; retrospective observational | CR (1–3/wk + home exercise) vs. non-CR | 6 months | Urinary ACR; eGFR | CR: ACR decreased 43 ± 71 → 17 ± 20 mg/g (p < 0.05); eGFR unchanged. Non-CR: eGFR decreased 72 ± 18 → 67 ± 17 (p < 0.001). |
| Iso et al. [45] | CR participants; n = 67 (non-CKD n = 34; mild CKD n = 14; mod–severe CKD n = 19) | Non-RCT; retrospective | CR: supervised aerobic 1–2 × /wk + daily home walking | 5 months (plus renal re-evaluation 3 months post program) | eGFR (JMDRD; creatinine-based) | Mod–severe CKD group: eGFR 40.8 ± 7.4 → 43.2 ± 12.6 (not significant); age inversely associated with eGFR change; <70 y showed significant eGFR increase. |
| Fujimi et al. [46] | CVD; n = 49 (CR group n = 23 vs. non-CR n = 26) | Non-RCT; comparative (CR vs. Non-CR) | 1-year CR program vs. non-CR | 1 year | eGFR (creatinine-based); UN/Cr/K/Hct | Overall: no significant changes in eGFR in either group; in the CR group, low baseline eGFR subgroup (<51) showed significant eGFR increase after 1 year. |
| Hama et al. [47] | CVD with CKD; n = 86 | Non-RCT; single-arm | 3-month CR program | 3 months | eGFRcys (primary); eGFRcreat | eGFRcys improved 45.2 ± 11 → 47.3 ± 13 (p = 0.023); eGFRcreat unchanged. |
| Kitajima et al. [48] | Elderly CVD outpatients >65 y; n = 88 | Non-RCT; longitudinal cohort | Long-term outpatient CR follow-up | Up to 5 years | eGFR (creatinine-based); AT; LVEF; BNP | eGFR, AT, LVEF, and BNP were maintained over 5 years; no significant change vs. baseline reported. |
| Sasamoto et al. [49] | CVD; n = 136 (≥75 y subgroup n = 55) | Non-RCT; prospective intervention (CR vs. non-CR) | CR and physical activity promotion | 3 months | eGFRcys | ΔeGFRcys: non-CR −2.27 vs. CR +0.48 mL/min/1.73 m2 (p = 0.022); ≥75 y: −3.83 vs. −1.08 (p = 0.039). |
| Hama et al. [50] | CVD with CKD (15 ≤ eGFRcys < 60); n = 203 (G3a n = 122; G3b n = 60; G4 n = 21) | Non-RCT; retrospective cohort | CR program (pre vs. post) with baseline-stage stratification | 3 months | eGFRcys | eGFRcys improvement: +1.3 (G3a), +3.1 (G3b), +4.8 (G4) mL/min/1.73 m2; lower baseline eGFRcys associated with greater %ΔeGFRcys. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Sato, T.; Kohzuki, M. Rehabilitation for Cardiorenal Multimorbidity: Epidemiology, Functional Phenotypes, and Effects on Physical Function, Renal Trajectory, and Prognosis. J. Clin. Med. 2026, 15, 2504. https://doi.org/10.3390/jcm15072504
Sato T, Kohzuki M. Rehabilitation for Cardiorenal Multimorbidity: Epidemiology, Functional Phenotypes, and Effects on Physical Function, Renal Trajectory, and Prognosis. Journal of Clinical Medicine. 2026; 15(7):2504. https://doi.org/10.3390/jcm15072504
Chicago/Turabian StyleSato, Toshimi, and Masahiro Kohzuki. 2026. "Rehabilitation for Cardiorenal Multimorbidity: Epidemiology, Functional Phenotypes, and Effects on Physical Function, Renal Trajectory, and Prognosis" Journal of Clinical Medicine 15, no. 7: 2504. https://doi.org/10.3390/jcm15072504
APA StyleSato, T., & Kohzuki, M. (2026). Rehabilitation for Cardiorenal Multimorbidity: Epidemiology, Functional Phenotypes, and Effects on Physical Function, Renal Trajectory, and Prognosis. Journal of Clinical Medicine, 15(7), 2504. https://doi.org/10.3390/jcm15072504