Inborn Errors of Amino Acid Metabolism Revisited: Clinical Implications and Insights into Current Therapies
Abstract
1. Introduction
2. Key Disorders of Amino Acid Metabolism: Clinical Features and Therapeutic Approaches
2.1. Phenylketonuria
| Disorder | Treatment | Rationale/ Mechanism | Dose | Biochemical Monitoring |
|---|---|---|---|---|
| Phenylketonuria (PKU) | Phenylalanine-free amino acid supplements Dietary restriction of phenylalanine | To limit intake of offending amino acid | Small, frequent doses (3–4) spaced evenly across day [21]. | Blood phenylalanine levels |
| Sapropterin dihydrochloride (Kuvan®, San Rafael, CA, USA) | Synthetic form of cofactor tetrahydrobiopterin (BH4) | Recommended starting dose in patients is 10 mg/kg body weight/day. Dose is adjusted, usually between 5 and 20 mg/kg/day, to achieve and maintain blood Phe control [9,22,23]. | Blood phenylalanine levels | |
| Pegvaliase (Palynziq®) | Recombinant phenylalanine ammonia lyase (PAL) enzyme (patients ≥ 16 years) | Recommended starting dose is 2.5 mg once per week for 4 weeks. Dose escalated gradually based on tolerability to daily maintenance dose needed to achieve blood Phe control. Maintenance dose is individualised to achieve blood Phe control [10,24,25]. | Blood phenylalanine levels | |
| Sepiapterin (Sephience™) | Synthetic BH4 precursor | <6 months: 7.5 mg/kg; 6–12 months: 15 mg/kg; 1–2 years: 30 mg/kg; >2 years: 60 mg/kg [26]. | Blood phenylalanine levels | |
| Biopterin defects causing hyperphenylalaninaemia [27] | Dietary restriction of phenylalanine (GTPCH and DHPR deficiency patients); phenylalanine-free amino acid supplements | To limit intake of offending amino acid | Small, frequent doses (3–4) spaced evenly across day [21]. | Blood phenylalanine levels |
| Sapropterin dihydrochloride (Kuvan®) | Synthetic form of cofactor tetrahydrobiopterin (BH4) | Recommended starting dose in adult patients is 10 mg/kg body weight/day. Dose is adjusted, usually between 5 and 20 mg/kg/day, to achieve and maintain blood phenylalanine control. | Blood phenylalanine levels | |
| l-3,4- dihydroxyphenylalanine/carbidopa (L-DOPA) and 5-OH-Tryptophan | For neurotransmitter related movement disorders | L-DOPA in four divided doses with similar dosing for 5-OH-Tryptophan [28]; age-dependent. | LP for CSF neurotransmitters measurement (HVA, 5-HIAA); prolactin levels | |
| Folinic acid | For movement disorders, to prevent cerebral folate deficiency | Dose of 10–15 mg/day [28]. | Monitoring of CSF folate and folinic acid status | |
| Hyperphenylalaninaemia due to DNAJC12 | Dietary restriction of phenylalanine; phenylalanine-free amino acid supplements | To limit intake of offending amino acid | Small, frequent doses (3–4) spaced evenly across day [21]. | Blood phenylalanine levels |
| Sapropterin dihydrochloride (Kuvan®) | Synthetic form of cofactor tetrahydrobiopterin (BH4) | Recommended starting dose in adult patients is 10 mg/kg body weight/day. Dose is adjusted, usually between 5 and 20 mg/kg/day, to achieve and maintain blood Phe control. | Blood phenylalanine levels | |
| L-DOPA and tryptophan | For neurotransmitter related movement disorder | Starting dose of 2.5 mg/kg/day (can be increased to 6 mg/kg/day) [29]. | LP for CSF neurotransmitters measurement (HVA, 5-HIAA) | |
| Alkaptonuria (AKU) | Dietary restriction of phenylalanine; tyrosine/phenylalanine-free amino acid supplements | To limit intake of offending amino acid | Moderate restriction of natural protein. | Plasma amino acids (phenylalanine, tyrosine) |
| Nitisinone (currently nitisinone is approved for alkaptonuria treatment in adults only) | Inhibits 4- hydroxyphenylpyruvate dioxygenase | The recommended dose in the adult AKU population is 10 mg once daily [30,31]. | Plasma amino acids (phenylalanine, tyrosine) | |
| Bisphosphonate [32] | Inhibit bone resorption by preventing hydroxyapatite breakdown | As clinically indicated. | Bone turnover markers (BTMs) | |
| Teriparatide [32] | Promotes bone anabolism through protein kinase A and protein kinase C pathways [33] | Dose of 20 mcg/day SC (approved in adults). | BTMs, plasma calcium levels | |
| Tyrosinaemia type I | Dietary restriction of phenylalanine and tyrosine; tyrosine/phenylalanine-free amino acid supplements | To limit intake of offending amino acids | Plasma amino acids (phenylalanine, tyrosine, methionine), liver function, blood/urine succinylacetone | |
| Nitisinone (nitisinone is approved for tyrosinaemia type I treatment in children) | Inhibits 4- hydroxyphenylpyruvate dioxygenase | Recommended starting dose in adult patients is 1 mg/kg body weight/day. Dose should be adjusted individually. Maximum of dose of 2 mg/kg body weight/day [34,35]. | Blood tyrosine levels, blood/urine succinylacetone, NTBC drug levels, liver function, alpha-fetoprotein | |
| Liver transplant | If end-stage liver disease, liver failure, or hepatocellular carcinoma develops | |||
| Tyrosinaemia type II | Dietary restriction of phenylalanine and tyrosine; tyrosine/phenylalanine-free amino acid supplements | To limit intake of offending amino acids | Blood tyrosine and phenylalanine levels | |
| Tyrosinaemia type III | A restrictive tyrosine and phenylalanine diet has been suggested during childhood [20], while other authors argue that such restriction is not recommended |
2.2. Alkaptonuria
2.3. Tyrosinaemia Type I, Type II, and Type III
2.4. Homocystinuria
| Disorder | Treatment | Rationale/Mechanism | Dose | Monitoring |
|---|---|---|---|---|
| Homocystinuria (HCU) due to cystathionine beta-synthase (CBS) deficiency | Methionine-free amino acid supplements; dietary restriction of methionine/proteind supplementation of cysteine, B12, folate | To limit intake of offending amino acid | Individualised to patient | Methionine and cystine levels, B12, folate |
| Pyridoxine (vitamin B6) (in pyridoxine-responsive patients) | Cofactor of cystathionine β-synthase | Recommended dose of up to 10 mg/kg/day; recommended to avoid doses >500 mg/day (risk of peripheral neuropathy) [59]. | Plasma tHcy | |
| Betaine | Betaine donates a methyl group via betaine homocysteine methyl transferase (BHMT) | Recommended starting dose of 3 g BD; can increase up to 200 mg/kg/day; rarely benefits from higher dose [59] | Plasma tHcy | |
| Homocystinuria due to methylene tetrahydrofolate reductase deficiency | Betaine | Betaine donates a methyl group via betaine homocysteine methyl transferase (BHMT) | Recommended starting dose of 3 g BD; can increase up to 200 mg/kg/day; rarely benefits from higher dose [59] | Plasma tHcy |
| Aspirin | Antiplatelet therapy post-stroke | 40.5 mg per second day [68] | Routine monitoring not recommended | |
| Supplementation of creatine, B6, B12, folate, 5MTHF | To achieve target plasma tHcy levels | Creatine (75–100 mg/kg/day), B6 (25 mg/day), B12 (25 mg/day), folate (4 mg/day), 5MTHF (2.4–3.2 mg/day) [68] | Creatinine, B6, B12, folate, 5MTHF levels | |
| Methionine S-adenosyltransferase deficiency | S-adenosyl-L-methionine disulfate tosylate (SAM) supplementation | For neurological manifestations | 400–800 mg BD [69] | SAM concentration in plasma and CSF |
| Methionine-free amino acid supplements; dietary restriction of methionine/protein | To limit intake of offending amino acid (although may decrease S-adenosyl-L-methionine (SAM) synthesis [70]) | Individualised to patient | Methionine levels | |
| S-adenosylhomocysteine hydrolase deficiency | Methionine-free amino acid supplements; dietary restriction of methionine/protein | To limit intake of offending amino acids; to reduce toxic SAH levels | Individualised to patient | Methionine levels |
| Phosphatidylcholine and creatine supplementation | Low levels of creatine and choline in SAH hydrolase deficiency | Creatine—e.g., 375 mg/kg/d Phosphatidylcholine—e.g., 150 mg/kg/d [71] | Creatinine, choline levels; blood/urine creatine | |
| Cystinosis | Cysteamine | Depletes lysosomal cystine levels | 1.30 g/m2/day; maximum of 1.95 g/m2/day [72] | WBC cystine assay |
| Symptomatic treatment | Management of symptoms | E.g., ACE inhibitors for proteinuria; kidney transplant in ESRD; HRT for endocrinopathies | Depends on symptoms |
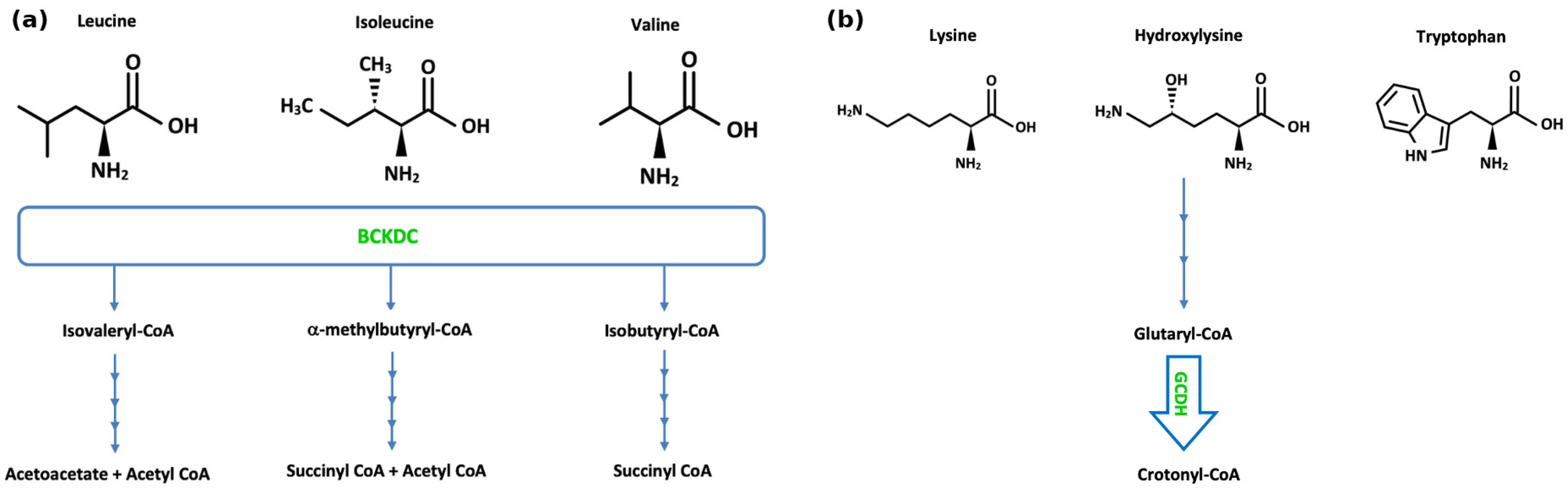
2.5. Methylmalonic Acidaemia
2.6. Maple Syrup Urine Disease
| Disorder | Treatment | Rationale/Mechanism | Dose | Monitoring |
|---|---|---|---|---|
| Maple syrup urine disease (MSUD) | Synthetic formula with all amino acids except leucine, isoleucine, valine; valine and isoleucine supplementation; protein-free foods | To limit intake of offending amino acids | Valine: 15–30 mg/kg Isoleucine: 10–30 mg/kg [92], individualised to patient | Plasma levels of BCAAs |
| Thiamine (vitamin B1) (in thiamine-responsive patients) | Increases stability of branched-chain alpha-ketoacid dehydrogenase complex (BCKDC) | Additional thiamine challenge of 150–300 mg/day for one month; continue thiamine supplementation in responsive patients [85] | Plasma levels of BCAAs | |
| Liver transplantation | Hepatic enzyme replacement | |||
| Management of acute crises: BCAA-free formula (PO or NG if not tolerating formula); provide all amino acids except leucine; supplement isoleucine and valine [37]. Reverse catabolism: increase calorie intake—IV calories (typically dextrose at high concentration); may start insulin drip if hyperglycaemic; use of normal or hypertonic saline; avoid hypotonic solutions; mannitol; diuretics; haemodialysis/haemofiltration. | ||||
| Methylmalonic acidaemia | Protein-restricted diet using synthetic propiogenic-devoid formulas | Reduce MMA production | Urine MMA, plasma amino acid concentrations | |
| Hydroxocobalamin | Enhance activity of methylmalonyl-CoA mutase | 1 mg intramuscularly, regular continuation depends on metabolic response [76] | Urine MMA, plasma amino acid concentrations | |
| Carnitine | To correct secondary carnitine deficiency | 50–100 mg/kg/day and up to ~300 mg/kg/day divided into 3–4 doses [76] | Plasma free carnitine level, acylcarnitine profile in dried blood spots | |
| Metronidazole | Reduce propionate production by gut flora | 10–15 mg/kg/day typically administered in 7–10 day courses every 1–3 months [76] | Urine MMA, propionylcarnitine | |
2.7. Nonketotic Hyperglycinaemia
| Disorder | Treatment | Rationale/Mechanism | Dose | Monitoring |
|---|---|---|---|---|
| Nonketotic hyperglycinemia (NKH) | Sodium benzoate Ketogenic diet (high in fat and low in carbohydrates) in some cases | Glycine reduction Alternate energy source for brain, epilepsy treatment with clobazam/ multidrug regimen | Glycine Blood glucose and ketones | |
| Sodium benzoate | Forms conjugated metabolite (hippurate), which is excreted by kidneys | Attenuated NKH—200–550 mg/kg/day Severe NKH—550 (–750) mg/kg/day (maximum dose 16.5 g/m2/day) [102] | Glycine in plasma and CSF | |
| Dextromethorphan (gene–drug interactions: CYP2D6, CYP3A4, CYPUGT) | Weak, non-competitive inhibitor of NMDA receptors | 3–15 mg/kg/day (high individual variability) [27] | Glycine in plasma and CSF | |
| Pyridoxal phosphate (active form of vitamin B6) | Cofactor of glycine decarboxylase (GLDC) | Glycine in plasma and CSF | ||
| PDE-ALDH7A1 | Pyridoxine (vitamin B6) | Pyridoxal 5′-phosphate (PLP) is a cofactor of enzymatic reactions involved in neurotransmitter synthesis | Adults: 200–500 mg/day (maximum dose 500 mg/day) [103] | Serum/plasma pipecolic acid levels, alpha-aminoadipic semialdehyde [AASA] in serum/plasma, urine, or CSF |
| Lysine reduction therapies (LRTs)—lysine restriction, arginine supplementation | Arginine is a competitive inhibitor of lysine transport | Start at 4 g/m2/day (Maximum dose 5.5 g/m2/day) [103] | Plasma lysine, arginine | |
| 3-Phosphoglycerate dehydrogenase deficiency | L-Serine and glycine | Seizure control, correction of behavioural abnormalities | Infantile 3-PGDH deficiency: 500–700 mg L-serine/kg/d and 200– 300 mg glycine/kg/d Juvenile 3-PGDH deficiency: 100–150 mg L-serine/kg/d [104] | CSF serine and glycine; plasma serine and glycine |
| Phosphoserine aminotransferase deficiency | L-Serine and glycine | Prevention of neurological abnormalities in presymptomatic patients | L-serine: 500 mg/kg/day Glycine: 200 mg/kg/day [105] | CSF serine and glycine; plasma serine and glycine |
| 3-Phosphoserine phosphatase deficiency | L-Serine | May prevent onset of neurological symptoms | 200–300 mg/kg/day [105] | CSF and plasma serine |
2.8. Pyridoxine-Dependent Epilepsy
2.9. Serine Deficiency
2.10. Cystinuria
| Disorder | Treatment | Rationale/Mechanism | Dose | Monitoring |
|---|---|---|---|---|
| Cystinuria [123] | Potassium citrate | Urine alkalisation | Children: 60–80 mEq/1.73 m2/d Adults: 60–80 mEq/d TDS/QDS | Urine pH |
| Penicillamine | Increases cystine solubility | Children: 20–30 mg/kg/d (max 4000 mg/d) Adults: 1–4 g/d TDS/QDS | Urine cystine excretion | |
| Tiopronin | Increases cystine solubility | Children: 15–40 mg/kg/d (max 1500 mg/d) Adults: 800–1500 mg/kg/d TDS | Urine cystine excretion | |
| Alpha-lipoic acid | Increases cystine solubility | Children: 30 mg/kg/d (max 1200 mg/d) Adults: 1200 mg/d BD | Urine cystine excretion | |
| Captopril | Increases cystine solubility | Children: 1.5–6 mg/kg/d (max 150 mg/d) Adults: 75–150 mg/d TDS | Urine cystine excretion | |
| Lysinuric protein intolerance | Acute management [124] | Reduction in protein and caloric supplementation for preventing protein catabolism | Glucose infusion: 10% glucose (in cases of hyperglycaemia, consider adding insulin) L-arginine: 100–250 mg/kg/d IV Sodium phenylbutyrate: 450–600 mg/kg/d in patients <20 kg, 9.9–13.0 g/m2/d in larger patients Sodium benzoate: 100–250 mg/kg/d PO or IV +/− continuous haemodialysis +/− antibiotics (e.g., neomycin), lactulose, and/or lactobacillus preparation | Blood ammonia, amino acids in blood/urine, blood glucose |
| Dietary: protein restriction, vitamin D, iron, zinc, and calcium supplementation, +/−medical foods, e.g., protein-free drinks | To prevent hyperammonaemia. Zinc, iron, calcium and vitamin D levels tend to be decreased. | Children: 0.8–1.5 g/kg/d protein intake Adults: 0.5–0.8 g/kg/d protein intake [125] | Amino acid (e.g., lysine, arginine, ornithine, glutamine) analysis in blood/urine; 25(OH)D, iron, zinc, calcium levels | |
| L-citrulline [124] | Reduces blood ammonia level, increases in dietary intake, reduction in hepatomegaly | 100 mg/kg/d | Blood ammonia level, amino acids | |
| L-arginine [124] | Reduces blood ammonia level | 120–380 mg/kg/d | Blood ammonia level, amino acids | |
| L-carnitine [124] | Secondary carnitine deficiency | 20–50 mg/kg/d | Blood carnitine level, amino acids | |
| L-lysine [124] | Increases blood lysine levels | 20–50 mg/kg/d | Blood lysine level, amino acids | |
| Nitrogen scavengers [124] | Decreases blood ammonia levels | Sodium phenylbutyrate: 450–600 mg/kg/d in patients weighing < 20 kg and 9.9–13.0 g/m2/d in larger patients Sodium benzoate: 100–250 mg/kg/d | Blood ammonia levels, plasma amino acids, electrolytes (Sodium) | |
| Other treatments [124] | Management of osteoporosis, short stature, hyperlipidaemia, nephritis, pulmonary alveolar proteinosis, and ESRD. | Vitamin D and bisphosphonate, GH injection, statins, ACE inhibitors, corticosteroids, whole lung lavage, GM-CSF, renal transplantation | As per clinical finding | |
| Hartnup disease [126] | Nicotinamide | Management of dermatological and neurological complications. | 50–300 mg PO [127] | |
| High-protein diet | To compensate for amino acid loss | Individualised to patient |
2.11. Lysinuric Protein Intolerance
2.12. Hartnup Disease
2.13. Glutaric Aciduria Type 1
2.14. Hyperprolinaemia Type I and Type II
2.15. Glutamine Synthetase Deficiency
2.16. Asparagine Synthetase Deficiency
3. Discussion
4. Looking Forward: Clinical Innovation and Unmet Needs
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| 5-HIAA | 5-Hydroxyindoleacetic acid |
| AASS | Aminoadipic semialdehyde synthase |
| AAV | Adeno-associated virus |
| AAV8 | Adeno-associated virus serotype 8 |
| ACE | Angiotensin-converting enzyme |
| AEDs | Anti-epileptic drugs |
| AKU | Alkaptonuria |
| AMT | Aminomethyltransferase |
| ASD | Asparagine synthetase deficiency |
| ASNS | Asparagine synthetase gene |
| BCAAs | Branched-chain amino acids |
| BCKD | Branched-chain ketoacid dehydrogenase |
| BCKDC | Branched-chain α-ketoacid dehydrogenase complex |
| BCKDH | Branched-chain ketoacid dehydrogenase |
| BD | Twice daily |
| BH4 | Tetrahydrobiopterin |
| BHMT | Betaine-homocysteine methyltransferase |
| BTMs | Bone turnover markers |
| C3 | Propionylcarnitine |
| CBS | Cystathionine-β-synthase |
| CNS | Central nervous system |
| CSF | Cerebrospinal fluid |
| CT | Computed tomography |
| CYP2D6 | Cytochrome P450 2D6 |
| CYP3A4 | Cytochrome P450 3A4 |
| CYPUGT | Cytochrome P450 / UGT |
| DNAJC12 | DnaJ heat shock protein family (Hsp40) member C12 |
| EC | Enzyme Commission |
| ESPFKU | European Society for Phenylketonuria |
| ESRD | End-stage renal disease |
| FAA | Fumarylacetoacetate |
| FAH | Fumarylacetoacetate hydrolase |
| GA1 | Glutaric aciduria type 1 |
| GCDH | Glutaryl-CoA dehydrogenase |
| GH | Growth hormone |
| GLDC | Glycine decarboxylase |
| GLUL | Glutamate–ammonia ligase (glutamine synthetase) gene |
| GM-CSF | Granulocyte macrophage colony-stimulating factor |
| GSD | Glutamine synthetase deficiency |
| HCU | Homocystinuria |
| HGA | Homogentisic acid |
| HGD | Homogentisate 1,2-dioxygenase |
| HRT | Hormone replacement therapy |
| HRQoL | Health-related quality of life |
| HPD | 4-Hydroxyphenylpyruvate dioxygenase |
| HVA | Homovanillic acid |
| IEAAMs | Inborn errors of amino acid metabolism |
| IV | Intravenous |
| L-DOPA | L-3,4-dihydroxyphenylalanine |
| LNAAs | Large Neutral Amino Acids |
| LP | Lumbar puncture |
| LPI | Lysinuric protein intolerance |
| LRTs | Lysine reduction therapies |
| MAA | Maleylacetoacetate |
| MAAI | Maleylacetoacetate isomerase |
| MAT | Methionine S-adenosyltransferase |
| MMA | Methylmalonic acidaemia |
| MMAA | Methylmalonic acidaemia cblA type |
| MMAB | Methylmalonic acidaemia cblB type |
| MMUT | Methylmalonyl-CoA mutase |
| MS | Methionine synthase |
| MSUD | Maple syrup urine disease |
| MTHFR | Methylenetetrahydrofolate reductase |
| NaCl | Sodium chloride |
| NG | Nasogastric |
| NKH | Nonketotic hyperglycinaemia |
| NMDA | N-methyl-D-aspartate |
| NTBC | Nitisinone (2-(2-nitro-4-trifluoromethylbenzyl)-1,3-cyclohexanedione) |
| OMIM | Online Mendelian Inheritance in Man |
| PAH | Phenylalanine hydroxylase |
| PAL | Phenylalanine ammonia lyase |
| PDE | Pyridoxine-dependent epilepsy |
| Phe | Phenylalanine |
| PKA | Protein kinase A |
| PKC | Protein kinase C |
| PKU | Phenylketonuria |
| PO | By mouth |
| PRODH | Proline dehydrogenase |
| QDS | Four times daily |
| SAH | S-adenosylhomocysteine |
| SAHH | S-adenosylhomocysteine hydrolase |
| SAM | S-adenosyl-L-methionine |
| SC | Subcutaneous |
| TAT | Tyrosine aminotransferase |
| TDS | Three times daily |
| THF | Tetrahydrofolate |
| tHcy | Total homocysteine |
| Tyr | Tyrosine |
| WBC | White blood cell |
References
- Chandel, N.S. Amino Acid Metabolism. Cold Spring Harb. Perspect. Biol. 2021, 13, a040584. [Google Scholar] [CrossRef]
- Tessari, P.; Lante, A.; Mosca, G. Essential amino acids: Master regulators of nutrition and environmental footprint? Sci. Rep. 2016, 6, 26074. [Google Scholar] [CrossRef] [PubMed]
- Ziegler, S.G.; Kim, J.; Ehmsen, J.T.; Vernon, H.J. Inborn errors of amino acid metabolism—From underlying pathophysiology to therapeutic advances. Dis. Model. Mech. 2023, 16, dmm050233. [Google Scholar] [CrossRef]
- Blau, N.; van Spronsen, F.J.; Levy, H.L. Phenylketonuria. Lancet 2010, 376, 1417–1427. [Google Scholar] [CrossRef]
- Blau, N.; Hennermann, J.B.; Langenbeck, U.; Lichter-Konecki, U. Diagnosis, classification, and genetics of phenylketonuria and tetrahydrobiopterin (BH4) deficiencies. Mol. Genet. Metab. 2011, 104, S2–S9. [Google Scholar] [CrossRef]
- van Wegberg, A.M.J.; MacDonald, A.; Ahring, K.; Bélanger-Quintana, A.; Blau, N.; Bosch, A.M.; Burlina, A.; Campistol, J.; Feillet, F.; Giżewska, M.; et al. The complete European guidelines on phenylketonuria: Diagnosis and treatment. Orphanet J. Rare Dis. 2017, 12, 162. [Google Scholar] [CrossRef]
- Boyle, F.; Lynch, G.; Reynolds, C.M.; Green, A.; Parr, G.; Howard, C.; Knerr, I.; Rice, J. Determination of the Protein and Amino Acid Content of Fruit, Vegetables and Starchy Roots for Use in Inherited Metabolic Disorders. Nutrients 2024, 16, 2812. [Google Scholar] [CrossRef]
- Levy, H.L.; Milanowski, A.; Chakrapani, A.; Cleary, M.; Lee, P.; Trefz, F.K.; Whitley, C.B.; Feillet, F.; Feigenbaum, A.S.; Bebchuk, J.D.; et al. Efficacy of sapropterin dihydrochloride (tetrahydrobiopterin, 6R-BH4) for reduction of phenylalanine concentration in patients with phenylketonuria: A phase III randomised placebo-controlled study. Lancet 2007, 370, 504–510. [Google Scholar] [CrossRef]
- Doyle, S.; O’Regan, M.; Stenson, C.; Bracken, J.; Hendroff, U.; Agasarova, A.; Deverell, D.; Treacy, E.P. Extended Experience of Lower Dose Sapropterin in Irish Adults with Mild Phenylketonuria. In JIMD Reports, Volume 40; Morava, E., Baumgartner, M., Patterson, M., Rahman, S., Zschocke, J., Peters, V., Eds.; Springer: Berlin/Heidelberg, Germany, 2018; pp. 71–76. [Google Scholar]
- Thomas, J.; Levy, H.; Amato, S.; Vockley, J.; Zori, R.; Dimmock, D.; Harding, C.O.; Bilder, D.A.; Weng, H.H.; Olbertz, J.; et al. Pegvaliase for the treatment of phenylketonuria: Results of a long-term phase 3 clinical trial program (PRISM). Mol. Genet. Metab. 2018, 124, 27–38. [Google Scholar] [CrossRef]
- Bratkovic, D.; Margvelashvili, L.; Tchan, M.C.; Nisbet, J.; Smith, N. PTC923 (sepiapterin) lowers elevated blood phenylalanine in subjects with phenylketonuria: A phase 2 randomized, multi-center, three-period crossover, open-label, active controlled, all-comers study. Metabolism 2022, 128, 155116. [Google Scholar] [CrossRef] [PubMed]
- Muntau, A.C.; Longo, N.; Ezgu, F.; Schwartz, I.V.D.; Lah, M.; Bratkovic, D.; Margvelashvili, L.; Kiykim, E.; Zori, R.; Campistol Plana, J.; et al. Effects of oral sepiapterin on blood Phe concentration in a broad range of patients with phenylketonuria (APHENITY): Results of an international, phase 3, randomised, double-blind, placebo-controlled trial. Lancet 2024, 404, 1333–1345. [Google Scholar] [CrossRef]
- Ptc Therapeutics, Inc. PTC Therapeutics Announces FDA Approval of Sephience™ (Sepiapterin) for the Treatment of Children and Adults Living with Phenylketonuria (PKU). 28 July 2025. Available online: https://ir.ptcbio.com/news-releases/news-release-details/ptc-therapeutics-announces-fda-approval-sephiencetm-sepiapterin?mobile=1 (accessed on 5 December 2025).
- Wobst, H.J.; Viader, A.; Muncipinto, G.; Hollibaugh, R.; van Kalken, D.; Burkhart, C.T.; Cantin, S.M.; Bates, R.M.; Regimbald-Dumas, Y.; Gross, L.; et al. SLC6A19 inhibition facilitates urinary neutral amino acid excretion and lowers plasma phenylalanine. JCI Insight 2024, 9, e182876. [Google Scholar] [CrossRef]
- A Phase 2 Study of JNT-517 in Adolescent Participants with Phenylketonuria. 2024. Available online: https://www.clinicaltrials.gov/study/NCT06637514 (accessed on 5 December 2025).
- A Phase 1/2, First-In-Human, Multiple Part, Single Ascending and Multiple Dose Study of JNT-517 in Healthy Participants and in Participants with Phenylketonuria. 2023. Available online: https://www.clinicaltrials.gov/study/NCT05781399 (accessed on 5 December 2025).
- An Open-Label, Multiple-Center, Phase I/II Dose Escalation Study for the Safety and Efficacy of NGGT002 in Adults with Classic Phenylketonuria. 2024. Available online: https://clinicaltrials.gov/study/NCT06332807 (accessed on 5 December 2025).
- A Clinical Study for the Safety and Efficacy of IV Infusion of NGGT002 in the Treatment of Phenylketonuria. 2023. Available online: https://clinicaltrials.gov/study/NCT06061614 (accessed on 5 December 2025).
- Song, W.S.; Kim, Y.S.; Bae, Y.S.; Yoon, S.H.; Lim, J.M.; Kim, M.H. GluN2B suppression restores phenylalanine-induced neuroplasticity and cognition impairments in a mouse model of phenylketonuria. J. Clin. Investig. 2025, 135, e184299. [Google Scholar] [CrossRef]
- Choi, J.E.; Park, S.; Song, H.; Hwang, I.T.; Jung, S.-C.; Kim, H.S. Therapeutic effect of histone deacetylase 6 inhibitor for a mouse model of phenylketonuria. Sci. Rep. 2025; online ahead of print. [Google Scholar] [CrossRef]
- MacDonald, A.; van Wegberg, A.M.J.; Ahring, K.; Beblo, S.; Bélanger-Quintana, A.; Burlina, A.; Campistol, J.; Coşkun, T.; Feillet, F.; Giżewska, M.; et al. PKU dietary handbook to accompany PKU guidelines. Orphanet J. Rare Dis. 2020, 15, 171. [Google Scholar] [CrossRef]
- Vernon, H.J.; Koerner, C.B.; Johnson, M.R.; Bergner, A.; Hamosh, A. Introduction of sapropterin dihydrochloride as standard of care in patients with phenylketonuria. Mol. Genet. Metab. 2010, 100, 229–233. [Google Scholar] [CrossRef]
- Sanford, M.; Keating, G.M. Sapropterin. Drugs 2009, 69, 461–476. [Google Scholar] [CrossRef]
- Hollander, S.; Viau, K.; Sacharow, S. Pegvaliase dosing in adults with PKU: Requisite dose for efficacy decreases over time. Mol. Genet. Metab. 2022, 137, 104–106. [Google Scholar] [CrossRef]
- Mahan, K.C.; Gandhi, M.A.; Anand, S. Pegvaliase: A novel treatment option for adults with phenylketonuria. Curr. Med. Res. Opin. 2019, 35, 647–651. [Google Scholar] [CrossRef]
- U.S. Food and Drug Administration. SEPHIENCE (Sepiapterin) Oral Powder Prescribing Information Label; U.S. Food and Drug Administration: Silver Spring, MD, USA, 2025.
- Van Hove, J.L.K.; Coughlin, C., II; Swanson, M.; Hennermann, J.B. Nonketotic Hyperglycinemia. In GeneReviews®; Adam, M.P., Feldman, J., Mirzaa, G.M., Pagon, R.A., Wallace, S.E., Amemiya, A., Eds.; University of Washington: Seattle, WA, USA, 1993. [Google Scholar]
- Bozaci, A.E.; Er, E.; Yazici, H.; Canda, E.; Kalkan Uçar, S.; Güvenc Saka, M.; Eraslan, C.; Onay, H.; Habif, S.; Thöny, B.; et al. Tetrahydrobiopterin deficiencies: Lesson from clinical experience. JIMD Rep. 2021, 59, 42–51. [Google Scholar] [CrossRef]
- Fino, E.; Barbato, A.; Scaturro, G.M.; Procopio, E.; Balestrini, S. DNAJC12 deficiency: Mild hyperphenylalaninemia and neurological impairment in two siblings. Mol. Genet. Metab. Rep. 2023, 37, 101008. [Google Scholar] [CrossRef]
- Abbas, K.; Basit, J.; Rehman, M.E.U. Adequacy of nitisinone for the management of alkaptonuria. Ann. Med. Surg. 2022, 80, 104340. [Google Scholar] [CrossRef]
- Ranganath, L.R.; Milan, A.M.; Hughes, A.T.; Khedr, M.; Norman, B.P.; Alsbou, M.; Imrich, R.; Gornall, M.; Sireau, N.; Gallagher, J.A.; et al. Comparing nitisinone 2 mg and 10 mg in the treatment of alkaptonuria-An approach using statistical modelling. JIMD Rep. 2022, 63, 80–92. [Google Scholar] [CrossRef]
- Ebrahim, I.C.; Hoang, T.D.; Vietor, N.O.; Schacht, J.P.; Shakir, M.K.M. Dilemmas in the diagnosis and management of osteoporosis in a patient with alkaptonuria: Successful treatment with teriparatide. Clin. Case Rep. 2022, 10, e6729. [Google Scholar] [CrossRef]
- Inderjeeth, C.A.; Chan, K.; Glendenning, P. Teriparatide: Its Use in the Treatment of Osteoporosis. Clin. Med. Insights Ther. 2011, 3, 67–80. [Google Scholar] [CrossRef]
- Das, A.M. Clinical utility of nitisinone for the treatment of hereditary tyrosinemia type-1 (HT-1). Appl. Clin. Genet. 2017, 10, 43–48. [Google Scholar] [CrossRef] [PubMed]
- Matthews, R.P. CHAPTER 32—Metabolic Liver Disease: Tyrosinemia, Galactosemia, and Hereditary Fructose Intolerance. In Pediatric Gastroenterology; Liacouras, C.A., Piccoli, D.A., Bell, L.M., Eds.; Mosby: Philadelphia, PA, USA, 2008; pp. 267–275. [Google Scholar]
- Introne, W.J.; Perry, M.; Chen, M. Alkaptonuria. In GeneReviews®; Adam, M.P., Feldman, J., Mirzaa, G.M., Pagon, R.A., Wallace, S.E., Amemiya, A., Eds.; University of Washington: Seattle, WA, USA, 1993. [Google Scholar]
- Sharabi, A.F.; Goudar, R.B. Alkaptonuria. In StatPearls; StatPearls Publishing LLC: Treasure Island, FL, USA, 2025. [Google Scholar]
- Millucci, L.; Braconi, D.; Bernardini, G.; Lupetti, P.; Rovensky, J.; Ranganath, L.; Santucci, A. Amyloidosis in alkaptonuria. J. Inherit. Metab. Dis. 2015, 38, 797–805. [Google Scholar] [CrossRef]
- Teke Kisa, P.; Eroglu Erkmen, S.; Bahceci, H.; Arslan Gulten, Z.; Aydogan, A.; Karalar Pekuz, O.K.; Yuce Inel, T.; Ozturk, T.; Uysal, S.; Arslan, N. Efficacy of Phenylalanine- and Tyrosine-Restricted Diet in Alkaptonuria Patients on Nitisinone Treatment: Case Series and Review of Literature. Ann. Nutr. Metab. 2022, 78, 48–60. [Google Scholar] [CrossRef]
- Trezza, A.; Roncaglia, B.; Visibelli, A.; Barletta, R.; Peruzzi, L.; Marzocchi, B.; Braconi, D.; Spiga, O.; Santucci, A. Integrated Clinomics and Molecular Dynamics Simulation Approaches Reveal the SAA1.1 Allele as a Biomarker in Alkaptonuria Disease Severity. Biomolecules 2025, 15, 194. [Google Scholar] [CrossRef]
- Morrow, G.; Tanguay, R.M. Biochemical and Clinical Aspects of Hereditary Tyrosinemia Type 1. Adv. Exp. Med. Biol. 2017, 959, 9–21. [Google Scholar] [CrossRef] [PubMed]
- van Ginkel, W.G.; Pennings, J.P.; van Spronsen, F.J. Liver Cancer in Tyrosinemia Type 1. Adv. Exp. Med. Biol. 2017, 959, 101–109. [Google Scholar] [CrossRef]
- Chinsky, J.M.; Singh, R.; Ficicioglu, C.; van Karnebeek, C.D.M.; Grompe, M.; Mitchell, G.; Waisbren, S.E.; Gucsavas-Calikoglu, M.; Wasserstein, M.P.; Coakley, K.; et al. Diagnosis and treatment of tyrosinemia type I: A US and Canadian consensus group review and recommendations. Genet. Med. 2017, 19, 1380–1395. [Google Scholar] [CrossRef]
- Nicolas, C.T.; VanLith, C.J.; Hickey, R.D.; Du, Z.; Hillin, L.G.; Guthman, R.M.; Cao, W.J.; Haugo, B.; Lillegard, A.; Roy, D.; et al. In vivo lentiviral vector gene therapy to cure hereditary tyrosinemia type 1 and prevent development of precancerous and cancerous lesions. Nat. Commun. 2022, 13, 5012. [Google Scholar] [CrossRef]
- Chen, T.; Barzi, M.; Furey, N.; Kim, H.R.; Pankowicz, F.P.; Legras, X.; Elsea, S.H.; Hurley, A.E.; Yang, D.; Wheeler, D.A.; et al. CRISPR/Cas9 gene therapy increases the risk of tumorigenesis in the mouse model of hereditary tyrosinemia type I. JHEP Rep. 2025, 7, 101327. [Google Scholar] [CrossRef] [PubMed]
- Gu, P.; Xie, L.; Chen, T.; Yang, Q.; Zhang, X.; Liu, R.; Guo, J.; Wei, R.; Li, D.; Jiang, Y.; et al. An engineered Escherichia coli Nissle strain prevents lethal liver injury in a mouse model of tyrosinemia type 1. J. Hepatol. 2024, 80, 454–466. [Google Scholar] [CrossRef]
- Bouyacoub, Y.; Zribi, H.; Azzouz, H.; Nasrallah, F.; Abdelaziz, R.B.; Kacem, M.; Rekaya, B.; Messaoud, O.; Romdhane, L.; Charfeddine, C.; et al. Novel and recurrent mutations in the TAT gene in Tunisian families affected with Richner-Hanhart syndrome. Gene 2013, 529, 45–49. [Google Scholar] [CrossRef]
- Čulic, V.; Betz, R.C.; Refke, M.; Fumic, K.; Pavelic, J. Tyrosinemia type II (Richner–Hanhart syndrome): A new mutation in the TAT gene. Eur. J. Med. Genet. 2011, 54, 205–208. [Google Scholar] [CrossRef]
- Peña-Quintana, L.; Scherer, G.; Curbelo-Estévez, M.L.; Jiménez-Acosta, F.; Hartmann, B.; La Roche, F.; Meavilla-Olivas, S.; Pérez-Cerdá, C.; García-Segarra, N.; Giguère, Y.; et al. Tyrosinemia type II: Mutation update, 11 novel mutations and description of 5 independent subjects with a novel founder mutation. Clin. Genet. 2017, 92, 306–317. [Google Scholar] [CrossRef] [PubMed]
- Russo, P.A.; Mitchell, G.A.; Tanguay, R.M. Tyrosinemia: A review. Pediatr. Dev. Pathol. 2001, 4, 212–221. [Google Scholar] [CrossRef]
- Kahraman, A.B.; Akar, H.T.; Güleray Lafcı, N.; Yıldız, Y.; Tokatlı, A. Novel Cranial Imaging Findings and a Splice-Site Variant in a Patient with Tyrosinemia Type III, and a Summary of Published Cases. Mol. Syndromol. 2022, 13, 193–199. [Google Scholar] [CrossRef]
- Rüetschi, U.; Cerone, R.; Pérez-Cerda, C.; Schiaffino, M.C.; Standing, S.; Ugarte, M.; Holme, E. Mutations in the 4-hydroxyphenylpyruvate dioxygenase gene (HPD) in patients with tyrosinemia type III. Hum. Genet. 2000, 106, 654–662. [Google Scholar] [CrossRef] [PubMed]
- Szymanska, E.; Sredzinska, M.; Ciara, E.; Piekutowska-Abramczuk, D.; Ploski, R.; Rokicki, D.; Tylki-Szymanska, A. Tyrosinemia type III in an asymptomatic girl. Mol. Genet. Metab. Rep. 2015, 5, 48–50. [Google Scholar] [CrossRef] [PubMed]
- Barroso, F.; Correia, J.; Bandeira, A.; Carmona, C.; Vilarinho, L.; Almeida, M.; Rocha, J.C.; Martins, E. Tyrosinemia type III: A case report of siblings and literature review. Rev. Paul. Pediatr. 2020, 38, e2018158. [Google Scholar] [CrossRef] [PubMed]
- Sellos-Moura, M.; Glavin, F.; Lapidus, D.; Evans, K.; Lew, C.R.; Irwin, D.E. Prevalence, characteristics, and costs of diagnosed homocystinuria, elevated homocysteine, and phenylketonuria in the United States: A retrospective claims-based comparison. BMC Health Serv. Res. 2020, 20, 183. [Google Scholar] [CrossRef]
- Weber Hoss, G.R.; Sperb-Ludwig, F.; Schwartz, I.V.D.; Blom, H.J. Classical homocystinuria: A common inborn error of metabolism? An epidemiological study based on genetic databases. Mol. Genet. Genom. Med. 2020, 8, e1214. [Google Scholar] [CrossRef]
- Gerrard, A.; Dawson, C. Homocystinuria diagnosis and management: It is not all classical. J. Clin. Pathol. 2022, 75, 744–750. [Google Scholar] [CrossRef]
- Aljassim, N.; Alfadhel, M.; Nashabat, M.; Eyaid, W. Clinical presentation of seven patients with Methylenetetrahydrofolate reductase deficiency. Mol. Genet. Metab. Rep. 2020, 25, 100644. [Google Scholar] [CrossRef]
- Morris, A.A.; Kožich, V.; Santra, S.; Andria, G.; Ben-Omran, T.I.; Chakrapani, A.B.; Crushell, E.; Henderson, M.J.; Hochuli, M.; Huemer, M.; et al. Guidelines for the diagnosis and management of cystathionine beta-synthase deficiency. J. Inherit. Metab. Dis. 2017, 40, 49–74. [Google Scholar] [CrossRef]
- Allen, J.; Power, B.; Abedin, A.; Purcell, O.; Knerr, I.; Monavari, A. Plasma methionine concentrations and incidence of hypermethioninemic encephalopathy during infancy in a large cohort of 36 patients with classical homocystinuria in the Republic of Ireland. JIMD Rep. 2019, 47, 41–46. [Google Scholar] [CrossRef]
- Ludolph, A.C.; Masur, H.; Oberwittler, C.; Koch, H.G.; Ullrich, K. Sensory neuropathy and vitamin B6 treatment in homocystinuria. Eur. J. Pediatr. 1993, 152, 271. [Google Scholar] [CrossRef] [PubMed]
- Ficicioglu, C.; Thomas, J.A.; Ganesh, J.; Kudrow, D.; Lah, M.; Smith, W.E.; Güner, J.; McDermott, S.; Vaidya, S.A.; Wilkening, L.; et al. Safety and efficacy of pegtibatinase enzyme replacement therapy in adults with classical homocystinuria in the COMPOSE phase 1/2 randomized trial. Genet. Med. 2025, 27, 101456. [Google Scholar] [CrossRef]
- Travere Therapeutics, Inc. Travere Therapeutics Reports Third Quarter 2025 Financial Results. BioSpace Press Releases, 30 October 2025. [Google Scholar]
- Skvorak, K.; Mitchell, V.; Teadt, L.; Franklin, K.A.; Lee, H.O.; Kruse, N.; Huitt-Roehl, C.; Hang, J.; Du, F.; Galanie, S.; et al. An orally administered enzyme therapeutic for homocystinuria that suppresses homocysteine by metabolizing methionine in the gastrointestinal tract. Mol. Genet. Metab. 2023, 139, 107653. [Google Scholar] [CrossRef]
- Gupta, S.; Lee, H.O.; Wang, L.; Kruger, W.D. Examination of two different proteasome inhibitors in reactivating mutant human cystathionine β-synthase in mice. PLoS ONE 2023, 18, e0286550. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.O.; Salami, C.O.; Sondhi, D.; Kaminsky, S.M.; Crystal, R.G.; Kruger, W.D. Long-term functional correction of cystathionine β-synthase deficiency in mice by adeno-associated viral gene therapy. J. Inherit. Metab. Dis. 2021, 44, 1382–1392. [Google Scholar] [CrossRef]
- Lee, H.O.; Gallego-Villar, L.; Grisch-Chan, H.M.; Häberle, J.; Thöny, B.; Kruger, W.D. Treatment of Cystathionine β-Synthase Deficiency in Mice Using a Minicircle-Based Naked DNA Vector. Hum. Gene Ther. 2019, 30, 1093–1100. [Google Scholar] [CrossRef]
- Strauss, K.A.; Morton, D.H.; Puffenberger, E.G.; Hendrickson, C.; Robinson, D.L.; Wagner, C.; Stabler, S.P.; Allen, R.H.; Chwatko, G.; Jakubowski, H.; et al. Prevention of brain disease from severe 5,10-methylenetetrahydrofolate reductase deficiency. Mol. Genet. Metab. 2007, 91, 165–175. [Google Scholar] [CrossRef]
- Furujo, M.; Kinoshita, M.; Nagao, M.; Kubo, T. S-adenosylmethionine treatment in methionine adenosyltransferase deficiency, a case report. Mol. Genet. Metab. 2012, 105, 516–518. [Google Scholar] [CrossRef]
- Liang, Z.; Wu, J.; Liu, Q.; Qin, D.; Wang, M.; Zhong, X.; Guo, W. Targeting Lysine α-Ketoglutarate Reductase to Treat Pyridoxine-Dependent Epilepsy. J. Neurosci. 2025, 45, e0370252025. [Google Scholar] [CrossRef]
- Huang, Y.; Chang, R.; Abdenur, J.E. The biochemical profile and dietary management in S-adenosylhomocysteine hydrolase deficiency. Mol. Genet. Metab. Rep. 2022, 32, 100885. [Google Scholar] [CrossRef] [PubMed]
- Elmonem, M.A.; Veys, K.R.; Soliman, N.A.; van Dyck, M.; van den Heuvel, L.P.; Levtchenko, E. Cystinosis: A review. Orphanet J. Rare Dis. 2016, 11, 47. [Google Scholar] [CrossRef] [PubMed]
- Head, P.E.; Meier, J.L.; Venditti, C.P. New insights into the pathophysiology of methylmalonic acidemia. J. Inherit. Metab. Dis. 2023, 46, 436–449. [Google Scholar] [CrossRef]
- Shan, S.; Liu, M.; Ma, Y.; Sun, M.; Wang, Y.; Zou, H. Animal models of methylmalonic acidemia: Insights and challenges. Orphanet J. Rare Dis. 2025, 20, 583. [Google Scholar] [CrossRef] [PubMed]
- Forny, P.; Hörster, F.; Ballhausen, D.; Chakrapani, A.; Chapman, K.A.; Dionisi-Vici, C.; Dixon, M.; Grünert, S.C.; Grunewald, S.; Haliloglu, G.; et al. Guidelines for the diagnosis and management of methylmalonic acidaemia and propionic acidaemia: First revision. J. Inherit. Metab. Dis. 2021, 44, 566–592. [Google Scholar] [CrossRef]
- Manoli, I.; Sloan, J.L.; Venditti, C.P. Isolated Methylmalonic Acidemia. In GeneReviews®; Adam, M.P., Feldman, J., Mirzaa, G.M., Pagon, R.A., Wallace, S.E., Amemiya, A., Eds.; University of Washington: Seattle, WA, USA, 1993. [Google Scholar]
- Zhou, X.; Cui, Y.; Han, J. Methylmalonic acidemia: Current status and research priorities. Intractable Rare Dis. Res. 2018, 7, 73–78. [Google Scholar] [CrossRef]
- Sen, K.; Burrage, L.C.; Chapman, K.A.; Ginevic, I.; Mazariegos, G.V.; Graham, B.H. Solid organ transplantation in methylmalonic acidemia and propionic acidemia: A points to consider statement of the American College of Medical Genetics and Genomics (ACMG). Genet. Med. 2023, 25, 100337. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.H.; Bourdages, M.H.; Kurtz, A.; MacLoed, E.; Norman, C.; Ratko, S.; van Calcar, S.C.; Kenneson, A. The efficacy of Carbamylglutamate impacts the nutritional management of patients with N-Acetylglutamate synthase deficiency. Orphanet J. Rare Dis. 2024, 19, 168. [Google Scholar] [CrossRef]
- Dao, M.; Arnoux, J.-B.; Bienaimé, F.; Brassier, A.; Brazier, F.; Benoist, J.-F.; Pontoizeau, C.; Ottolenghi, C.; Krug, P.; Boyer, O.; et al. Long-term renal outcome in methylmalonic acidemia in adolescents and adults. Orphanet J. Rare Dis. 2021, 16, 220. [Google Scholar] [CrossRef]
- Marelli, C.; Fouilhoux, A.; Benoist, J.-F.; De Lonlay, P.; Guffon-Fouilhoux, N.; Brassier, A.; Cano, A.; Chabrol, B.; Pennisi, A.; Schiff, M.; et al. Very long-term outcomes in 23 patients with cblA type methylmalonic acidemia. J. Inherit. Metab. Dis. 2022, 45, 937–951. [Google Scholar] [CrossRef]
- Coughlan, K.A.; Eybye, M.; Henderson, N.; DeAntonis, C.M.; Frassetto, A.; Hanahoe, E.; Ketova, T.; Jacquinet, E.; Presnyak, V.; Jain, R.; et al. Improved therapeutic efficacy in two mouse models of methylmalonic acidemia (MMA) using a second-generation mRNA therapy. Mol. Genet. Metab. 2024, 143, 108560. [Google Scholar] [CrossRef]
- A Global, Phase 1/2, Open-Label, Dose Optimization Study to Evaluate the Safety, Tolerability, Pharmacodynamics, and Pharmacokinetics of mRNA-3705 in Participants with Isolated Methylmalonic Acidemia Due to Methylmalonyl-CoA Mutase Deficiency. 2021. Available online: https://www.clinicaltrials.gov/study/NCT04899310 (accessed on 5 December 2025).
- Morton, D.H.; Strauss, K.A.; Robinson, D.L.; Puffenberger, E.G.; Kelley, R.I. Diagnosis and treatment of maple syrup disease: A study of 36 patients. Pediatrics 2002, 109, 999–1008. [Google Scholar] [CrossRef]
- Frazier, D.M.; Allgeier, C.; Homer, C.; Marriage, B.J.; Ogata, B.; Rohr, F.; Splett, P.L.; Stembridge, A.; Singh, R.H. Nutrition management guideline for maple syrup urine disease: An evidence- and consensus-based approach. Mol. Genet. Metab. 2014, 112, 210–217. [Google Scholar] [CrossRef] [PubMed]
- Ziadlou, M.; MacDonald, A. Alternative sources of valine and isoleucine for prompt reduction of plasma leucine in maple syrup urine disease patients: A case series. JIMD Rep. 2022, 63, 555–562. [Google Scholar] [CrossRef] [PubMed]
- Nyhan, W.L.; Rice-Kelts, M.; Klein, J.; Barshop, B.A. Treatment of the acute crisis in maple syrup urine disease. Arch. Pediatr. Adolesc. Med. 1998, 152, 593–598. [Google Scholar] [CrossRef]
- O’Reilly, D.; Crushell, E.; Hughes, J.; Ryan, S.; Rogers, Y.; Borovickova, I.; Mayne, P.; Riordan, M.; Awan, A.; Carson, K.; et al. Maple syrup urine disease: Clinical outcomes, metabolic control, and genotypes in a screened population after four decades of newborn bloodspot screening in the Republic of Ireland. J. Inherit. Metab. Dis. 2021, 44, 639–655. [Google Scholar] [CrossRef] [PubMed]
- Pontoizeau, C.; Simon-Sola, M.; Gaborit, C.; Nguyen, V.; Rotaru, I.; Tual, N.; Colella, P.; Girard, M.; Biferi, M.G.; Arnoux, J.B.; et al. Neonatal gene therapy achieves sustained disease rescue of maple syrup urine disease in mice. Nat. Commun. 2022, 13, 3278. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Poskitt, L.E.; Gallagher, J.; Puffenberger, E.G.; Wynn, R.M.; Shishodia, G.; Chuang, D.T.; Beever, J.; Hardin, D.L.; Brigatti, K.W.; et al. BCKDHA-BCKDHB digenic gene therapy restores metabolic homeostasis in two mouse models and a calf with classic maple syrup urine disease. Sci. Transl. Med. 2025, 17, eads0539. [Google Scholar] [CrossRef]
- Greig, J.A.; Jennis, M.; Dandekar, A.; Chorazeczewski, J.K.; Param, N.; So, M.; Nayal, M.; Bell, P.; Coughlan, K.; Choi, M.; et al. Lipid Nanoparticle mRNA Therapy Improves Survival and Reduces Serum Branched-Chain Amino Acids in Mouse Models of Maple Syrup Urine Disease. Hum. Gene Ther. 2024, 35, 726–733. [Google Scholar] [CrossRef]
- Blackburn, P.R.; Gass, J.M.; Vairo, F.P.E.; Farnham, K.M.; Atwal, H.K.; Macklin, S.; Klee, E.W.; Atwal, P.S. Maple syrup urine disease: Mechanisms and management. Appl. Clin. Genet. 2017, 10, 57–66. [Google Scholar] [CrossRef]
- Coughlin, C.R., 2nd; Swanson, M.A.; Kronquist, K.; Acquaviva, C.; Hutchin, T.; Rodríguez-Pombo, P.; Väisänen, M.L.; Spector, E.; Creadon-Swindell, G.; Brás-Goldberg, A.M.; et al. The genetic basis of classic nonketotic hyperglycinemia due to mutations in GLDC and AMT. Genet. Med. 2017, 19, 104–111. [Google Scholar] [CrossRef]
- Krawiec, C.; Anastasopoulou, C. Nonketotic Hyperglycinemia. In StatPearls; StatPearls Publishing LLC: Treasure Island, FL, USA, 2025. [Google Scholar]
- Bayrak, H.; Sharafi, P.; Özketen, A.; Kılıç, M. Comparative Small RNA Sequencing Reveals Candidate Functional miRNAs in Nonketotic Hyperglycinemia. Mol. Syndromol. 2025; online ahead of print. [Google Scholar] [CrossRef]
- Hennermann, J.B.; Berger, J.M.; Grieben, U.; Scharer, G.; Van Hove, J.L. Prediction of long-term outcome in glycine encephalopathy: A clinical survey. J. Inherit. Metab. Dis. 2012, 35, 253–261. [Google Scholar] [CrossRef]
- Wiltshire, E.J.; Poplawski, N.K.; Harrison, J.R.; Fletcher, J.M. Treatment of late-onset nonketotic hyperglycinaemia: Effectiveness of imipramine and benzoate. J. Inherit. Metab. Dis. 2000, 23, 15–21. [Google Scholar] [CrossRef]
- Nowak, M.; Chuchra, P.; Paprocka, J. Nonketotic Hyperglycinemia: Insight into Current Therapies. J. Clin. Med. 2022, 11, 3027. [Google Scholar] [CrossRef]
- Van Hove, J.L.K. The role of NMDA-receptor type glutamatergic antagonists dextromethorphan or ketamine in the treatment of nonketotic hyperglycinemia: A critical reassessment. Mol. Genet. Metab. 2024, 143, 108594. [Google Scholar] [CrossRef]
- Forget, P.; le Polain de Waroux, B.; Wallemacq, P.; Gala, J.L. Life-threatening dextromethorphan intoxication associated with interaction with amitriptyline in a poor CYP2D6 metabolizer: A single case re-exposure study. J. Pain Symptom Manag. 2008, 36, 92–96. [Google Scholar] [CrossRef]
- Leung, K.-Y.; Santos, C.; De Castro, S.C.P.; Diaz, D.G.; Copp, A.J.; Waddington, S.; Greene, N.D.E. AAV-mediated expression of mouse or human GLDC normalises metabolic biomarkers in a GLDC-deficient mouse model of Non-Ketotic Hyperglycinemia. Mol. Genet. Metab. 2024, 142, 108496. [Google Scholar] [CrossRef]
- Van Hove, J.L.; Vande Kerckhove, K.; Hennermann, J.B.; Mahieu, V.; Declercq, P.; Mertens, S.; De Becker, M.; Kishnani, P.S.; Jaeken, J. Benzoate treatment and the glycine index in nonketotic hyperglycinaemia. J. Inherit. Metab. Dis. 2005, 28, 651–663. [Google Scholar] [CrossRef]
- Coughlin, C.R., 2nd; Tseng, L.A.; Abdenur, J.E.; Ashmore, C.; Boemer, F.; Bok, L.A.; Boyer, M.; Buhas, D.; Clayton, P.T.; Das, A.; et al. Consensus guidelines for the diagnosis and management of pyridoxine-dependent epilepsy due to α-aminoadipic semialdehyde dehydrogenase deficiency. J. Inherit. Metab. Dis. 2021, 44, 178–192. [Google Scholar] [CrossRef]
- Tabatabaie, L.; Klomp, L.W.; Rubio-Gozalbo, M.E.; Spaapen, L.J.; Haagen, A.A.; Dorland, L.; de Koning, T.J. Expanding the clinical spectrum of 3-phosphoglycerate dehydrogenase deficiency. J. Inherit. Metab. Dis. 2011, 34, 181–184. [Google Scholar] [CrossRef] [PubMed]
- van der Crabben, S.N.; Verhoeven-Duif, N.M.; Brilstra, E.H.; Van Maldergem, L.; Coskun, T.; Rubio-Gozalbo, E.; Berger, R.; de Koning, T.J. An update on serine deficiency disorders. J. Inherit. Metab. Dis. 2013, 36, 613–619. [Google Scholar] [CrossRef] [PubMed]
- Coughlin, C.R., II; Gospe, S.M., Jr. Pyridoxine-dependent epilepsy: Current perspectives and questions for future research. Ann. Child Neurol. Soc. 2023, 1, 24–37. [Google Scholar] [CrossRef] [PubMed]
- Coughlin, C.R., 2nd; Swanson, M.A.; Spector, E.; Meeks, N.J.L.; Kronquist, K.E.; Aslamy, M.; Wempe, M.F.; van Karnebeek, C.D.M.; Gospe, S.M., Jr.; Aziz, V.G.; et al. The genotypic spectrum of ALDH7A1 mutations resulting in pyridoxine dependent epilepsy: A common epileptic encephalopathy. J. Inherit. Metab. Dis. 2019, 42, 353–361. [Google Scholar] [CrossRef]
- van Karnebeek, C.D.M.; Gailus-Durner, V.; Engelke, U.F.; Seisenberger, C.; Marschall, S.; Dragano, N.R.V.; da Silva-Buttkus, P.; Leuchtenberger, S.; Fuchs, H.; Hrabě de Angelis, M.; et al. New treatment for pyridoxine-dependent epilepsy due to ALDH7A1 deficiency: First proof-of-principle of upstream enzyme inhibition in the mouse. Brain Commun. 2025, 7, fcaf397. [Google Scholar] [CrossRef]
- Ferreira, C.R.; van Karnebeek, C.D.M. Inborn errors of metabolism. Handb. Clin. Neurol. 2019, 162, 449–481. [Google Scholar] [CrossRef]
- Hart, C.E.; Race, V.; Achouri, Y.; Wiame, E.; Sharrard, M.; Olpin, S.E.; Watkinson, J.; Bonham, J.R.; Jaeken, J.; Matthijs, G.; et al. Phosphoserine aminotransferase deficiency: A novel disorder of the serine biosynthesis pathway. Am. J. Hum. Genet. 2007, 80, 931–937. [Google Scholar] [CrossRef]
- Eggermann, T.; Venghaus, A.; Zerres, K. Cystinuria: An inborn cause of urolithiasis. Orphanet J. Rare Dis. 2012, 7, 19. [Google Scholar] [CrossRef]
- Feld, R.D. Cystinuria. Crit. Rev. Clin. Lab. Sci. 1988, 26, 243–261. [Google Scholar] [CrossRef]
- Pak, C.Y.; Fuller, C.; Sakhaee, K.; Zerwekh, J.E.; Adams, B.V. Management of cystine nephrolithiasis with alpha-mercaptopropionylglycine. J. Urol. 1986, 136, 1003–1008. [Google Scholar] [CrossRef] [PubMed]
- Gillion, V.; Saussez, T.P.; Van Nieuwenhove, S.; Jadoul, M. Extremely rapid stone formation in cystinuria: Look out for dietary supplements! Clin. Kidney J. 2021, 14, 1694–1696. [Google Scholar] [CrossRef] [PubMed]
- Sahota, A.; Tischfield, J.A.; Goldfarb, D.S.; Ward, M.D.; Hu, L. Cystinuria: Genetic aspects, mouse models, and a new approach to therapy. Urolithiasis 2019, 47, 57–66. [Google Scholar] [CrossRef] [PubMed]
- Ergül, R.B.; Gürlek, I.T.; Özervarlı, M.F.; Özcan, Ş.G.; Pietropaolo, A.; Tsaturyan, A.; Juliebø-Jones, P.; Tzelves, L.; Şener, T.E.; Dönmez, M.İ.; et al. Tiopronin safety in cystinuria: First real-world pharmacovigilance analysis using the FDA Adverse Event Reporting System (FAERS): An EAU YAU and Endourology sections review. Pediatr. Nephrol. 2025; online ahead of print. [Google Scholar] [CrossRef]
- El Hosry, L.; Elias, V.; Chamoun, V.; Halawi, M.; Cayot, P.; Nehme, A.; Bou-Maroun, E. Maillard Reaction: Mechanism, Influencing Parameters, Advantages, Disadvantages, and Food Industrial Applications: A Review. Foods 2025, 14, 1881. [Google Scholar] [CrossRef]
- Sui, W.; Yang, H.; Desai, M.; Chi, T.; Stoller, M. The potential role of Sodium/Glucose Cotransporter 2 inhibitors in the treatment of cystinuria. Urolithiasis 2024, 52, 168. [Google Scholar] [CrossRef]
- Hu, L.; Albanyan, H.; Yang, J.; Wang, Y.; Yang, M.; Tan, X.; Zhong, X.; Ward, M.D.; Sahota, A. 8-l-Cystinyl Bis(1,8-diazaspiro[4.5]decane) as an Orally Bioavailable l-Cystine Crystallization Inhibitor for Cystinuria. ACS Med. Chem. Lett. 2024, 15, 1026–1031. [Google Scholar] [CrossRef]
- Mayayo-Vallverdú, C.; López de Heredia, M.; Prat, E.; González, L.; Espino Guarch, M.; Vilches, C.; Muñoz, L.; Asensi, M.A.; Serra, C.; Llebaria, A.; et al. The antioxidant l-Ergothioneine prevents cystine lithiasis in the Slc7a9−/− mouse model of cystinuria. Redox Biol. 2023, 64, 102801. [Google Scholar] [CrossRef] [PubMed]
- Woodard, L.E.; Welch, R.C.; Menshikh, A.; Luo, W.; Williams, F.M.; Peek, J.L.; Sha, F.; Veach, R.A.; Kaja, A.; Beckermann, T.M.; et al. Partial correction of cystinuria type A in mice via kidney-targeted transposon delivery. Mol. Ther. Nucleic Acids 2025, 36, 102639. [Google Scholar] [CrossRef] [PubMed]
- Liao, D.; Yi, X.; Bai, Y.; Yang, Y.; Ai, J. Adeno-associated virus-based gene therapy for cystinuria. BJU Int. 2024, 134, 742–744. [Google Scholar] [CrossRef]
- Sadiq, S.; Cil, O. Cystinuria: An Overview of Diagnosis and Medical Management. Turk. Arch. Pediatr. 2022, 57, 377–384. [Google Scholar] [CrossRef]
- Noguchi, A.; Takahashi, T. Overview of symptoms and treatment for lysinuric protein intolerance. J. Hum. Genet. 2019, 64, 849–858. [Google Scholar] [CrossRef] [PubMed]
- Sebastio, G.; Sperandeo, M.P.; Andria, G. Lysinuric protein intolerance: Reviewing concepts on a multisystem disease. Am. J. Med. Genet. C Semin. Med. Genet. 2011, 157c, 54–62. [Google Scholar] [CrossRef]
- Hashmi, M.S.; Gupta, V. Hartnup Disease. In StatPearls; StatPearls Publishing LLC: Treasure Island, FL, USA, 2025. [Google Scholar]
- Piña-Garza, J.E.; James, K.C. Ataxia. In Fenichel’s Clinical Pediatric Neurology, 8th ed.; Piña-Garza, J.E., James, K.C., Eds.; Elsevier: Philadelphia, PA, USA, 2019; pp. 218–237. [Google Scholar]
- Mauhin, W.; Habarou, F.; Gobin, S.; Servais, A.; Brassier, A.; Grisel, C.; Roda, C.; Pinto, G.; Moshous, D.; Ghalim, F.; et al. Update on Lysinuric Protein Intolerance, a Multi-faceted Disease Retrospective cohort analysis from birth to adulthood. Orphanet J. Rare Dis. 2017, 12, 3. [Google Scholar] [CrossRef]
- Nunes, V.; Niinikoski, H. Lysinuric Protein Intolerance. In GeneReviews®; Adam, M.P., Bick, S., Mirzaa, G.M., Pagon, R.A., Wallace, S.E., Amemiya, A., Eds.; University of Washington: Seattle, WA, USA, 1993. [Google Scholar]
- Rautanen, T.; Ahopelto, K.; Niinikoski, H.; Karppinen, S.; Lempinen, M.; Ortiz, F.; Helanterä, I. Outcomes of kidney transplantation in patients with lysinuric protein intolerance. Clin. Kidney J. 2025, 18, sfae373. [Google Scholar] [CrossRef]
- Giroud-Gerbetant, J.; Sotillo, F.; Hernández, G.; Ruano, I.; Sebastián, D.; Fort, J.; Sánchez, M.; Weiss, G.; Prats, N.; Zorzano, A.; et al. Defective Slc7a7 transport reduces erythropoietin compromising erythropoiesis. Mol. Med. 2025, 31, 29. [Google Scholar] [CrossRef] [PubMed]
- Valimahamed-Mitha, S.; Berteloot, L.; Ducoin, H.; Ottolenghi, C.; de Lonlay, P.; de Blic, J. Lung involvement in children with lysinuric protein intolerance. J. Inherit. Metab. Dis. 2015, 38, 257–263. [Google Scholar] [CrossRef]
- Rajantie, J.; Simell, O.; Rapola, J.; Perheentupa, J. Lysinuric protein intolerance: A two-year trial of dietary supplementation therapy with citrulline and lysine. J. Pediatr. 1980, 97, 927–932. [Google Scholar] [CrossRef]
- Kleta, R.; Romeo, E.; Ristic, Z.; Ohura, T.; Stuart, C.; Arcos-Burgos, M.; Dave, M.H.; Wagner, C.A.; Camargo, S.R.M.; Inoue, S.; et al. Mutations in SLC6A19, encoding B0AT1, cause Hartnup disorder. Nat. Genet. 2004, 36, 999–1002. [Google Scholar] [CrossRef]
- Kravetz, Z.; Schmidt-Kastner, R. New aspects for the brain in Hartnup disease based on mining of high-resolution cellular mRNA expression data for SLC6A19. IBRO Neurosci. Rep. 2023, 14, 393–397. [Google Scholar] [CrossRef]
- Patel, A.B.; Prabhu, A.S. Hartnup disease. Indian J. Dermatol. 2008, 53, 31–32. [Google Scholar] [CrossRef]
- Bachmann, T.; Faust, H.; Abou Jamra, R.; Pott, C.; Kluge, M.; Rumpf, J.J.; Then Bergh, F.; Beblo, S. Adult Neuropsychiatric Manifestation of Hartnup Disease With a Novel SLCA6A19 Variant: A Case Report. Neurol. Genet. 2024, 10, e200195. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Chen, Y.; Zhang, Y.; Shen, Y.; Xu, K.; Liu, Y.; Wang, Z.; Yan, R. Structural insight into the substrate recognition and transport mechanism of amino acid transporter complex ACE2-B0AT1 and ACE2-SIT1. Cell Discov. 2023, 9, 93. [Google Scholar] [CrossRef]
- Alkhofash, N.F.; Ali, B.R. Hartnup disease-causing SLC6A19 mutations lead to B0AT1 aberrant trafficking and ACE2 mis-localisation implicating the endoplasmic reticulum protein quality control. Front. Cell Dev. Biol. 2025, 13, 1589534. [Google Scholar] [CrossRef]
- Kölker, S.; Christensen, E.; Leonard, J.V.; Greenberg, C.R.; Boneh, A.; Burlina, A.B.; Burlina, A.P.; Dixon, M.; Duran, M.; García Cazorla, A.; et al. Diagnosis and management of glutaric aciduria type I--revised recommendations. J. Inherit. Metab. Dis. 2011, 34, 677–694. [Google Scholar] [CrossRef] [PubMed]
- Rai, S.P. Glutaric aciduria type1: CT diagnosis. J. Pediatr. Neurosci. 2009, 4, 143. [Google Scholar] [CrossRef] [PubMed]
- Vester, M.E.M.; Bilo, R.A.C.; Karst, W.A.; Daams, J.G.; Duijst, W.L.J.M.; van Rijn, R.R. Subdural hematomas: Glutaric aciduria type 1 or abusive head trauma? A systematic review. Forensic Sci. Med. Pathol. 2015, 11, 405–415. [Google Scholar] [CrossRef] [PubMed]
- Sen, A.; Pillay, R.S. Striatal necrosis in type 1 glutaric aciduria: Different stages in two siblings. J. Pediatr. Neurosci. 2011, 6, 146–148. [Google Scholar] [CrossRef]
- Lipkin, P.H.; Roe, C.R.; Goodman, S.I.; Batshaw, M.L. A case of glutaric acidemia type I: Effect of riboflavin and carnitine. J. Pediatr. 1988, 112, 62–65. [Google Scholar] [CrossRef]
- Seccombe, D.W.; James, L.; Booth, F. L-Carnitine treatment in glutaric aciduria type I. Neurology 1986, 36, 264. [Google Scholar] [CrossRef]
- Kölker, S.; Garbade, S.F.; Boy, N.; Maier, E.M.; Meissner, T.; Mühlhausen, C.; Hennermann, J.B.; Lücke, T.; Häberle, J.; Baumkötter, J.; et al. Decline of Acute Encephalopathic Crises in Children with Glutaryl-CoA Dehydrogenase Deficiency Identified by Newborn Screening in Germany. Pediatr. Res. 2007, 62, 357–363. [Google Scholar] [CrossRef] [PubMed]
- Strauss, K.A.; Puffenberger, E.G.; Robinson, D.L.; Morton, D.H. Type I glutaric aciduria, part 1: Natural history of 77 patients. Am. J. Med. Genet. Part C Semin. Med. Genet. 2003, 121C, 38–52. [Google Scholar] [CrossRef]
- Boy, N.; Mühlhausen, C.; Maier, E.M.; Ballhausen, D.; Baumgartner, M.R.; Beblo, S.; Burgard, P.; Chapman, K.A.; Dobbelaere, D.; Heringer-Seifert, J.; et al. Recommendations for diagnosing and managing individuals with glutaric aciduria type 1: Third revision. J. Inherit. Metab. Dis. 2023, 46, 482–519. [Google Scholar] [CrossRef]
- Foran, J.; Moore, M.; Crushell, E.; Knerr, I.; McSweeney, N. Low excretor glutaric aciduria type 1 of insidious onset with dystonia and atypical clinical features, a diagnostic dilemma. JIMD Rep. 2021, 58, 12–20. [Google Scholar] [CrossRef]
- Segur-Bailach, E.; Mateu-Bosch, A.; Bofill-De Ros, X.; Parés, M.; da Silva Buttkus, P.; Rathkolb, B.; Gailus-Durner, V.; Hrabě de Angelis, M.; Moeini, P.; Gonzalez-Aseguinolaza, G.; et al. Therapeutic AASS inhibition by AAV-miRNA rescues glutaric aciduria type I severe phenotype in mice. Mol. Ther. 2025, 33, 4820–4833. [Google Scholar] [CrossRef]
- Mateu-Bosch, A.; Segur-Bailach, E.; Muñoz-Moreno, E.; Barallobre, M.J.; Arbonés, M.L.; Gea-Sorlí, S.; Tort, F.; Ribes, A.; García-Villoria, J.; Fillat, C. Systemic delivery of AAV-GCDH ameliorates HLD-induced phenotype in a glutaric aciduria type I mouse model. Mol. Ther. Methods Clin. Dev. 2024, 32, 101276. [Google Scholar] [CrossRef] [PubMed]
- An Open-Label, Dose-Escalation and Dose-Expansion Phase I Clinical Study to Evaluate the Tolerability, Safety and Efficacy of VGM-R02b in Patients with Glutaric Acidemia Type I. 2023. Available online: https://www.clinicaltrials.gov/study/NCT06217861 (accessed on 5 December 2025).
- Tondo, M.; Calpena, E.; Arriola, G.; Sanz, P.; Martorell, L.; Ormazabal, A.; Castejon, E.; Palacin, M.; Ugarte, M.; Espinos, C.; et al. Clinical, biochemical, molecular and therapeutic aspects of 2 new cases of 2-aminoadipic semialdehyde synthase deficiency. Mol. Genet. Metab. 2013, 110, 231–236. [Google Scholar] [CrossRef]
- Houten, S.M.; te Brinke, H.; Denis, S.; Ruiter, J.P.N.; Knegt, A.C.; de Klerk, J.B.C.; Augoustides-Savvopoulou, P.; Häberle, J.; Baumgartner, M.R.; Coşkun, T.; et al. Genetic basis of hyperlysinemia. Orphanet J. Rare Dis. 2013, 8, 57. [Google Scholar] [CrossRef]
- Marinella, G.; Pascarella, F.; Vetro, A.; Bonuccelli, A.; Pochiero, F.; Santangelo, A.; Alessandrì, M.G.; Pasquariello, R.; Orsini, A.; Battini, R. Hyperlysinemia, an ultrarare inborn error of metabolism: Review and update. Seizure-Eur. J. Epilepsy 2024, 120, 135–141. [Google Scholar] [CrossRef]
- Guilmatre, A.; Legallic, S.; Steel, G.; Willis, A.; Di Rosa, G.; Goldenberg, A.; Drouin-Garraud, V.; Guet, A.; Mignot, C.; Des Portes, V.; et al. Type I hyperprolinemia: Genotype/phenotype correlations. Hum. Mutat. 2010, 31, 961–965. [Google Scholar] [CrossRef] [PubMed]
- Bender, H.U.; Almashanu, S.; Steel, G.; Hu, C.A.; Lin, W.W.; Willis, A.; Pulver, A.; Valle, D. Functional consequences of PRODH missense mutations. Am. J. Hum. Genet. 2005, 76, 409–420. [Google Scholar] [CrossRef]
- Clelland, C.L.; Read, L.L.; Baraldi, A.N.; Bart, C.P.; Pappas, C.A.; Panek, L.J.; Nadrich, R.H.; Clelland, J.D. Evidence for association of hyperprolinemia with schizophrenia and a measure of clinical outcome. Schizophr. Res. 2011, 131, 139–145. [Google Scholar] [CrossRef] [PubMed]
- Mitsubuchi, H.; Nakamura, K.; Matsumoto, S.; Endo, F. Biochemical and clinical features of hereditary hyperprolinemia. Pediatr. Int. 2014, 56, 492–496. [Google Scholar] [CrossRef]
- Namavar, Y.; Duineveld, D.J.; Both, G.I.A.; Fiksinski, A.M.; Vorstman, J.A.S.; Verhoeven-Duif, N.M.; Zinkstok, J.R. Psychiatric phenotypes associated with hyperprolinemia: A systematic review. Am. J. Med. Genet. Part B Neuropsychiatr. Genet. 2021, 186, 289–317. [Google Scholar] [CrossRef]
- Martinelli, D.; Häberle, J.; Rubio, V.; Giunta, C.; Hausser, I.; Carrozzo, R.; Gougeard, N.; Marco-Marín, C.; Goffredo, B.M.; Meschini, M.C.; et al. Understanding pyrroline-5-carboxylate synthetase deficiency: Clinical, molecular, functional, and expression studies, structure-based analysis, and novel therapy with arginine. J. Inherit. Metab. Dis. 2012, 35, 761–776. [Google Scholar] [CrossRef]
- van de Ven, S.; Gardeitchik, T.; Kouwenberg, D.; Kluijtmans, L.; Wevers, R.; Morava, E. Long-term clinical outcome, therapy and mild mitochondrial dysfunction in hyperprolinemia. J. Inherit. Metab. Dis. 2014, 37, 383–390. [Google Scholar] [CrossRef]
- Balfoort, B.M.; Buijs, M.J.N.; ten Asbroek, A.L.M.A.; Bergen, A.A.B.; Boon, C.J.F.; Ferreira, E.A.; Houtkooper, R.H.; Wagenmakers, M.A.E.M.; Wanders, R.J.A.; Waterham, H.R.; et al. A review of treatment modalities in gyrate atrophy of the choroid and retina (GACR). Mol. Genet. Metab. 2021, 134, 96–116. [Google Scholar] [CrossRef]
- Häberle, J.; Boddaert, N.; Burlina, A.; Chakrapani, A.; Dixon, M.; Huemer, M.; Karall, D.; Martinelli, D.; Crespo, P.S.; Santer, R.; et al. Suggested guidelines for the diagnosis and management of urea cycle disorders. Orphanet J. Rare Dis. 2012, 7, 32. [Google Scholar] [CrossRef]
- Camacho, J.; Rioseco-Camacho, N. Hyperornithinemia-Hyperammonemia-Homocitrullinuria Syndrome. In GeneReviews®; Adam, M.P., Bick, S., Mirzaa, G.M., Pagon, R.A., Wallace, S.E., Amemiya, A., Eds.; University of Washington: Seattle, WA, USA, 1993. [Google Scholar]
- Häberle, J.; Shahbeck, N.; Ibrahim, K.; Hoffmann, G.F.; Ben-Omran, T. Natural course of glutamine synthetase deficiency in a 3year old patient. Mol. Genet. Metab. 2011, 103, 89–91. [Google Scholar] [CrossRef] [PubMed]
- Spodenkiewicz, M.; Diez-Fernandez, C.; Rüfenacht, V.; Gemperle-Britschgi, C.; Häberle, J. Minireview on Glutamine Synthetase Deficiency, an Ultra-Rare Inborn Error of Amino Acid Biosynthesis. Biology 2016, 5, 40. [Google Scholar] [CrossRef] [PubMed]
- Häberle, J.; Shahbeck, N.; Ibrahim, K.; Schmitt, B.; Scheer, I.; O’Gorman, R.; Chaudhry, F.A.; Ben-Omran, T. Glutamine supplementation in a child with inherited GS deficiency improves the clinical status and partially corrects the peripheral and central amino acid imbalance. Orphanet J. Rare Dis. 2012, 7, 48. [Google Scholar] [CrossRef] [PubMed]
- Espíritu-Ramírez, P.; Ortega-Balderas, N.Y.; Sevilla-Tapia, L.; Montiel-Martínez, A.G.; Pastor-Flores, A.R.; Palomares, L.A.; Torres-Vega, M.A. Gene Therapy for Treatment of Chronic Hyperammonemia in a Rat Model of Hepatic Encephalopathy. Ann. Hepatol. 2018, 17, 1026–1034. [Google Scholar] [CrossRef] [PubMed]
- Alfadhel, M.; Alrifai, M.T.; Trujillano, D.; Alshaalan, H.; Al Othaim, A.; Al Rasheed, S.; Assiri, H.; Alqahtani, A.A.; Alaamery, M.; Rolfs, A.; et al. Asparagine Synthetase Deficiency: New Inborn Errors of Metabolism. JIMD Rep. 2015, 22, 11–16. [Google Scholar] [CrossRef]
- Alfadhel, M.; El-Hattab, A.W. Asparagine Synthetase Deficiency. In GeneReviews®; Adam, M.P., Bick, S., Mirzaa, G.M., Pagon, R.A., Wallace, S.E., Amemiya, A., Eds.; University of Washington: Seattle, WA, USA, 1993. [Google Scholar]
- Sacharow, S.J.; Dudenhausen, E.E.; Lomelino, C.L.; Rodan, L.; El Achkar, C.M.; Olson, H.E.; Genetti, C.A.; Agrawal, P.B.; McKenna, R.; Kilberg, M.S. Characterization of a novel variant in siblings with Asparagine Synthetase Deficiency. Mol. Genet. Metab. 2018, 123, 317–325. [Google Scholar] [CrossRef]
- van Wegberg, A.M.J.; MacDonald, A.; Ahring, K.; Bélanger-Quintana, A.; Beblo, S.; Blau, N.; Bosch, A.M.; Burlina, A.; Campistol, J.; Coşkun, T.; et al. European guidelines on diagnosis and treatment of phenylketonuria: First revision. Mol. Genet. Metab. 2025, 145, 109125. [Google Scholar] [CrossRef]
- Altman, G.; Hussain, K.; Green, D.; Strauss, B.J.G.; Wilcox, G. Mental health diagnoses in adults with phenylketonuria: A retrospective systematic audit in a large UK single centre. Orphanet J. Rare Dis. 2021, 16, 520. [Google Scholar] [CrossRef]
- ten Hoedt, A.E.; de Sonneville, L.M.; Francois, B.; ter Horst, N.M.; Janssen, M.C.; Rubio-Gozalbo, M.E.; Wijburg, F.A.; Hollak, C.E.; Bosch, A.M. High phenylalanine levels directly affect mood and sustained attention in adults with phenylketonuria: A randomised, double-blind, placebo-controlled, crossover trial. J. Inherit. Metab. Dis. 2011, 34, 165–171. [Google Scholar] [CrossRef]
- Gersting, S.W.; Staudigl, M.; Truger, M.S.; Messing, D.D.; Danecka, M.K.; Sommerhoff, C.P.; Kemter, K.F.; Muntau, A.C. Activation of phenylalanine hydroxylase induces positive cooperativity toward the natural cofactor. J. Biol. Chem. 2010, 285, 30686–30697. [Google Scholar] [CrossRef] [PubMed]
- Trefz, F.K.; Burton, B.K.; Longo, N.; Casanova, M.M.; Gruskin, D.J.; Dorenbaum, A.; Kakkis, E.D.; Crombez, E.A.; Grange, D.K.; Harmatz, P.; et al. Efficacy of sapropterin dihydrochloride in increasing phenylalanine tolerance in children with phenylketonuria: A phase III, randomized, double-blind, placebo-controlled study. J. Pediatr. 2009, 154, 700–707. [Google Scholar] [CrossRef]
- Camp, K.M.; Parisi, M.A.; Acosta, P.B.; Berry, G.T.; Bilder, D.A.; Blau, N.; Bodamer, O.A.; Brosco, J.P.; Brown, C.S.; Burlina, A.B.; et al. Phenylketonuria Scientific Review Conference: State of the science and future research needs. Mol. Genet. Metab. 2014, 112, 87–122. [Google Scholar] [CrossRef]
- Burton, B.K.; Grange, D.K.; Milanowski, A.; Vockley, G.; Feillet, F.; Crombez, E.A.; Abadie, V.; Harding, C.O.; Cederbaum, S.; Dobbelaere, D.; et al. The response of patients with phenylketonuria and elevated serum phenylalanine to treatment with oral sapropterin dihydrochloride (6R-tetrahydrobiopterin): A phase II, multicentre, open-label, screening study. J. Inherit. Metab. Dis. 2007, 30, 700–707. [Google Scholar] [CrossRef]
- Hennermann, J.B.; Roloff, S.; Gebauer, C.; Vetter, B.; von Arnim-Baas, A.; Mönch, E. Long-term treatment with tetrahydrobiopterin in phenylketonuria: Treatment strategies and prediction of long-term responders. Mol. Genet. Metab. 2012, 107, 294–301. [Google Scholar] [CrossRef] [PubMed]
- Scala, I.; Concolino, D.; Casa, R.D.; Nastasi, A.; Ungaro, C.; Paladino, S.; Capaldo, B.; Ruoppolo, M.; Daniele, A.; Bonapace, G.; et al. Long-term follow-up of patients with phenylketonuria treated with tetrahydrobiopterin: A seven years experience. Orphanet J. Rare Dis. 2015, 10, 14. [Google Scholar] [CrossRef]
- Hydery, T.; Coppenrath, V.A. A Comprehensive Review of Pegvaliase, an Enzyme Substitution Therapy for the Treatment of Phenylketonuria. Drug Target Insights 2019, 13, 1177392819857089. [Google Scholar] [CrossRef]
- Hausmann, O.; Daha, M.; Longo, N.; Knol, E.; Müller, I.; Northrup, H.; Brockow, K. Pegvaliase: Immunological profile and recommendations for the clinical management of hypersensitivity reactions in patients with phenylketonuria treated with this enzyme substitution therapy. Mol. Genet. Metab. 2019, 128, 84–91. [Google Scholar] [CrossRef] [PubMed]
- Harding, C.O.; Amato, R.S.; Stuy, M.; Longo, N.; Burton, B.K.; Posner, J.; Weng, H.H.; Merilainen, M.; Gu, Z.; Jiang, J.; et al. Pegvaliase for the treatment of phenylketonuria: A pivotal, double-blind randomized discontinuation Phase 3 clinical trial. Mol. Genet. Metab. 2018, 124, 20–26. [Google Scholar] [CrossRef] [PubMed]
- Cunningham, A.; Rohr, F.; Splett, P.; Mofidi, S.; Bausell, H.; Stembridge, A.; Kenneson, A.; Singh, R.H. Nutrition management of PKU with pegvaliase therapy: Update of the web-based PKU nutrition management guideline recommendations. Orphanet J. Rare Dis. 2023, 18, 155. [Google Scholar] [CrossRef] [PubMed]
- Bosch, A.M.; Burlina, A.; Cunningham, A.; Bettiol, E.; Moreau-Stucker, F.; Koledova, E.; Benmedjahed, K.; Regnault, A. Assessment of the impact of phenylketonuria and its treatment on quality of life of patients and parents from seven European countries. Orphanet J. Rare Dis. 2015, 10, 80. [Google Scholar] [CrossRef]
- Bik-Multanowski, M.; Didycz, B.; Mozrzymas, R.; Nowacka, M.; Kaluzny, L.; Cichy, W.; Schneiberg, B.; Amilkiewicz, J.; Bilar, A.; Gizewska, M.; et al. Quality of life in noncompliant adults with phenylketonuria after resumption of the diet. J. Inherit. Metab. Dis. 2008, 31, S415–S418. [Google Scholar] [CrossRef]
- Dawson, C.; Murphy, E.; Maritz, C.; Chan, H.; Ellerton, C.; Carpenter, R.H.S.; Lachmann, R.H. Dietary treatment of phenylketonuria: The effect of phenylalanine on reaction time. J. Inherit. Metab. Dis. 2011, 34, 449–454. [Google Scholar] [CrossRef]
- Fernstrom, J.D.; Fernstrom, M.H. Tyrosine, Phenylalanine, and Catecholamine Synthesis and Function in the Brain123. J. Nutr. 2007, 137, 1539S–1547S. [Google Scholar] [CrossRef] [PubMed]
- Sara, G.-L.; Alejandra, L.-M.L.; Isabel, I.-G.; Marcela, V.-A. Conventional Phenylketonuria Treatment. J. Inborn Errors Metab. Screen. 2016, 4, 2326409816685733. [Google Scholar] [CrossRef]
- Remmington, T.; Smith, S. Tyrosine supplementation for phenylketonuria. Cochrane Database Syst. Rev. 2021, 2021, CD001507. [Google Scholar] [CrossRef]
- Orzincolo, C.; Castaldi, G.; Scutellari, P.N.; Cicognani, P.; Bariani, L.; Feggi, L. Ochronotic arthropathy in alkaptonuria. Radiological manifestations and physiopathological signs. Radiol. Med. 1988, 75, 476–481. [Google Scholar]
- Ranga, U.; Aiyappan, S.K.; Shanmugam, N.; Veeraiyan, S. Ochronotic spondyloarthropathy. J. Clin. Diagn. Res. 2013, 7, 403–404. [Google Scholar] [CrossRef]
- Wolff, J.A.; Barshop, B.; Nyhan, W.L.; Leslie, J.; Seegmiller, J.E.; Gruber, H.; Garst, M.; Winter, S.; Michals, K.; Matalon, R. Effects of Ascorbic Acid in Alkaptonuria: Alterations in Benzoquinone Acetic Acid and an Ontogenic Effect in Infancy. Pediatr. Res. 1989, 26, 140–144. [Google Scholar] [CrossRef]
- Feier, F.; Schwartz, I.V.; Benkert, A.R.; Seda Neto, J.; Miura, I.; Chapchap, P.; da Fonseca, E.A.; Vieira, S.; Zanotelli, M.L.; Pinto e Vairo, F.; et al. Living related versus deceased donor liver transplantation for maple syrup urine disease. Mol. Genet. Metab. 2016, 117, 336–343. [Google Scholar] [CrossRef] [PubMed]
- Walther, F.; Radke, M.; KrÜGer, G.; Hobusch, D.; Uhlemann, M.; Tittelbach-Helmrich, W.; Stolpe, H.J. Response to sodium benzoate treatment in non-ketotic hyperglycinaemia. Pediatr. Int. 1994, 36, 75–79. [Google Scholar] [CrossRef] [PubMed]
- Shelkowitz, E.; Saneto, R.P.; Al-Hertani, W.; Lubout, C.M.A.; Stence, N.V.; Brown, M.S.; Long, P.; Walleigh, D.; Nelson, J.A.; Perez, F.E.; et al. Ketogenic diet as a glycine lowering therapy in nonketotic hyperglycinemia and impact on brain glycine levels. Orphanet J. Rare Dis. 2022, 17, 423. [Google Scholar] [CrossRef] [PubMed]
- Bjoraker, K.J.; Swanson, M.A.; Coughlin, C.R., 2nd; Christodoulou, J.; Tan, E.S.; Fergeson, M.; Dyack, S.; Ahmad, A.; Friederich, M.W.; Spector, E.B.; et al. Neurodevelopmental Outcome and Treatment Efficacy of Benzoate and Dextromethorphan in Siblings with Attenuated Nonketotic Hyperglycinemia. J. Pediatr. 2016, 170, 234–239. [Google Scholar] [CrossRef]
- Lee, N.; Kim, D. Toxic Metabolites and Inborn Errors of Amino Acid Metabolism: What One Informs about the Other. Metabolites 2022, 12, 527. [Google Scholar] [CrossRef]
- van Karnebeek, C.D.; Hartmann, H.; Jaggumantri, S.; Bok, L.A.; Cheng, B.; Connolly, M.; Coughlin, C.R., 2nd; Das, A.M.; Gospe, S.M., Jr.; Jakobs, C.; et al. Lysine restricted diet for pyridoxine-dependent epilepsy: First evidence and future trials. Mol. Genet. Metab. 2012, 107, 335–344. [Google Scholar] [CrossRef]
- Coughlin, C.R.; Tseng, L.A.; Bok, L.A.; Hartmann, H.; Footitt, E.; Striano, P.; Tabarki, B.M.; Lunsing, R.J.; Stockler-Ipsiroglu, S.; Gordon, S.; et al. Association Between Lysine Reduction Therapies and Cognitive Outcomes in Patients With Pyridoxine-Dependent Epilepsy. Neurology 2022, 99, e2627–e2636. [Google Scholar] [CrossRef]
- Kölker, S.; Boy, S.P.N.; Heringer, J.; Müller, E.; Maier, E.M.; Ensenauer, R.; Mühlhausen, C.; Schlune, A.; Greenberg, C.R.; Koeller, D.M.; et al. Complementary dietary treatment using lysine-free, arginine-fortified amino acid supplements in glutaric aciduria type I—A decade of experience. Mol. Genet. Metab. 2012, 107, 72–80. [Google Scholar] [CrossRef]

| Disorder | OMIM | Incidence/Prevalence | Affected Gene | Main Organs/Systems Involved | On-Label FDA-Approved Drugs | Ongoing Clinical Trials for Novel Drugs | Recent Preclinical Research |
|---|---|---|---|---|---|---|---|
| Phenylketonuria | 261600 | 1 in 16,000 live births in USA; higher in Turkey and Ireland | PAH | CNS, skin | Sapropterin (2007) Pegvaliase (2018) Sepiapterin (2025) | JNT-517 (NCT06637514, NCT05781399) NGGT002 gene therapy (NCT06332807, NCT06061614) | HDAC6i reduces plasma Phe; GluN2B inhibition limits cognitive impairment. |
| Alkaptonuria | 203500 | Global prevalence of 1:250,000–1:1,000,000. | HGD | Musculoskeletal, heart, kidneys, sclera | Nitisinone (2025) | None | SAA1.1 allele as potential amyloidosis biomarker. |
| Tyrosinaemia type I | 276700 | 1 in 100,000 live births; higher in Scandinavia and Québec | FAH | Liver, kidneys | Nitisinone (2002) | None | Lentiviral FAH expression in pigs; CRISPR deletion of HPD results in metabolic correction but increases HCC in mice. Genetically engineered Tyr-degrading E. coli. |
| Tyrosinaemia type II | 276600 | Incidence less than 1 in 250,000 | TAT | Eyes, skin, CNS | None | None | None in scope of this article. |
| Tyrosinaemia type III | 276710 | Less than 20 cases reported | HPD | CNS | None | None | None in scope of this article. |
| Homocystinuria | 236200 236250 | Worldwide prevalence 0.8/100,000 | CBS MTHFR | CNS, eyes, cardiovascular | Betaine (1996) | Pegtibatinase (HARMONY trial paused, may resume 2026) | SYNT-202 (methionine gamma-lyase). AAVrh.10-CBS gene therapy. Minicircle-based gene therapy. |
| Methylmalonic acidaemia | 251000 251100 251110 | 1:50,000 to 1:360,000 live births | MMA MMUT MMAB | CNS, kidneys | Carglumic acid (2021) | mRNA-3705 (NCT04899310) | None in scope of article. |
| Maple syrup urine disease | 608348 620698 620699 | Incidence 1 in 185,000 | BCKDHA BCKDHB DBT | CNS, liver, musculoskeletal | None | None | AAV8 gene therapy. AAV9 dual gene therapy. Lipid nanoparticles encapsulating mRNA. |
| Nonketotic hyperglycinaemia | 605899 | Incidence 1 in 76,000 | GLDC AMT | CNS | None | None | AAV9-GLDC gene therapy. |
| Pyridoxine-dependent epilepsy | 266100 | Incidence 1 in 65,000 | ALDH7A1 PNPO PLPBP | CNS | None | None | Targeting upstream AASS or lysine α-ketoglutarate reductase. |
| Cystinuria | 220100 | Incidence 1 in 7000 | SLC3A1 SLC7A9 | Kidneys | D-Penicillamine (1970) Tiopronin (1988) | None | SGLT2i through Maillard reaction. 8-l-Cystinyl Bis(1,8-diazaspiro[4.5]decane). l-Ergothioneine. Transposon-mediated gene therapy. AAV9 gene therapy. |
| Lysinuric protein intolerance | 222700 | Incidence 1:60,000 in Finland and 1:57,000 in Japan | SLC7A7 | Kidneys, respiratory | None | None | Anaemia partly due to EPO deficiency. |
| Hartnup disease | 234500 | Incidence 1 in 15,000 | SLC6A19 | Skin, CNS | None | None | SLC6A19 variants may result in ER retention of B0AT1 and ACE2. |
| Glutaric aciduria type I | 231670 | Incidence 1 in 100,000; higher in Oji Cree, Amish, and Irish Travellers | GCDH | CNS | None | VGM-R02b (NCT06217861) | AAV-microRNA targeting AASS, AAV9-GCDH. |
| Serine deficiency | 601815 610992 | Not known; >50 reported cases | 3-PGDH PSAT | CNS | None | None | None in scope of article. |
| Hyperprolinaemia type I | 239500 | Unknown | POX | Not adequately established, evidence for CNS | None | None | None in scope of article. |
| Hyperprolinaemia type II | 239510 | Unknown | ALDH4A1 | CNS | None | None | None in scope of article. |
| Glutamine synthetase deficiency | 610015 | Unknown | GLUL | CNS | None | None | None specifically for GSD, but GS gene therapy has been successful in mice to reduce hyperammonaemia in liver disease. |
| Asparagine synthetase deficiency | 615574 | More than 20 cases reported | ASNS | CNS | None | None | None in scope of article. |
| Disorder | Treatment | Rationale/Mechanism | Dose | Monitoring |
|---|---|---|---|---|
| Δ1-Pyrroline-5-carboxylate synthetase deficiency | Arginine | Increases arginine availability to brain; improvement of neurodevelopmental and metabolic parameters | 150 mg/kg/d [161] | Amino acid analysis (proline, ornithine, arginine, citrulline), and ammonia levels |
| Hyperprolinaemia type I | Anti-epileptic medication and schizophrenia medication if required | |||
| Avoid protein excess | Reduce accumulation of proline or P5C | Plasma amino acids (proline) | ||
| Hyperprolinaemia type II | B6 supplementation | Avoid deficiency | e.g., 50–100 mg/day [162] | B6 levels |
| Avoid protein excess | Reduce accumulation of proline or P5C | Plasma amino acids, urine organic acids | ||
| Anti-epileptic medication and schizophrenia medication if required | ||||
| Ornithine δ-aminotransferase deficiency (gyrate atrophy) | Arginine-restricted diet with synthetic amino acid supplementation | Aim to decrease plasma ornithine levels and slow disease progression | 10–35 g/d protein intake [163] | Ornithine and arginine levels |
| Trial of B6, lysine, and creatine supplementation | B6—aims to stimulate residual enzyme activity; lysine—may increase kidney excretion of ornithine and arginine; creatine and precursors—to treat secondary creatine deficiency | B6: 100–1000 mg/d [163] | B6 and plasma amino acids; blood/urine creatine | |
| Hyperornithinaemia–hyperammonaemia– homocitrullinuria | Acute management | Stop protein intake for 24 h and commence IV 10% glucose (plus electrolytes); arginine +/− citrulline supplementation; Ammonia scavengers (sodium benzoate and sodium phenylbutyrate); +/−haemodialysis (if neurological status is deteriorating) | Glucose dose at appropriate dose to prevent catabolism; sodium benzoate: 250 mg/kg bolus (90–120 min), then maintenance 250–500 mg/kg/d (>20 kg, 5.5 g/m2/d); sodium phenylbutyrate: 250 mg/kg bolus (90–120 min), then 250–500 mg/kg/d as maintenance [164] | Blood ammonia levels, blood glucose |
| Long-term management | Protein-restricted diet with citrulline or arginine (+/−sodium benzoate or sodium phenylbutyrate) | Protein restriction individualised to patient Sodium benzoate: ≤250 mg/kg/d Sodium phenylbutyrate: <20 kg ≤250 mg/kg/d, >20 kg 5 g/m2/d L-citrulline: 100–200 mg/kg/d L-arginine: <20 kg 100–200 mg/kg/d, >20 kg 2.5–6 g/m2/d [164] | Blood ammonia levels, plasma amino acids, urinary orotic acid | |
| Creatine supplementation [165] (if plasma creatine levels low) | To treat secondary creatine deficiency | Dosed according to degree of creatine deficiency | Plasma creatinine levels, blood/urine creatine |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shakerdi, A.L.; Nerney, D.; Molloy, E.J.; Knerr, I. Inborn Errors of Amino Acid Metabolism Revisited: Clinical Implications and Insights into Current Therapies. J. Clin. Med. 2025, 14, 8749. https://doi.org/10.3390/jcm14248749
Shakerdi AL, Nerney D, Molloy EJ, Knerr I. Inborn Errors of Amino Acid Metabolism Revisited: Clinical Implications and Insights into Current Therapies. Journal of Clinical Medicine. 2025; 14(24):8749. https://doi.org/10.3390/jcm14248749
Chicago/Turabian StyleShakerdi, Abdul L., Darragh Nerney, Eleanor J. Molloy, and Ina Knerr. 2025. "Inborn Errors of Amino Acid Metabolism Revisited: Clinical Implications and Insights into Current Therapies" Journal of Clinical Medicine 14, no. 24: 8749. https://doi.org/10.3390/jcm14248749
APA StyleShakerdi, A. L., Nerney, D., Molloy, E. J., & Knerr, I. (2025). Inborn Errors of Amino Acid Metabolism Revisited: Clinical Implications and Insights into Current Therapies. Journal of Clinical Medicine, 14(24), 8749. https://doi.org/10.3390/jcm14248749






