Panretinal Congenital Hypertrophy of the RPE in an 8-Year-Old Girl with an X-Linked STAG2 Mutation
Abstract
1. Introduction
2. Methods
3. Background
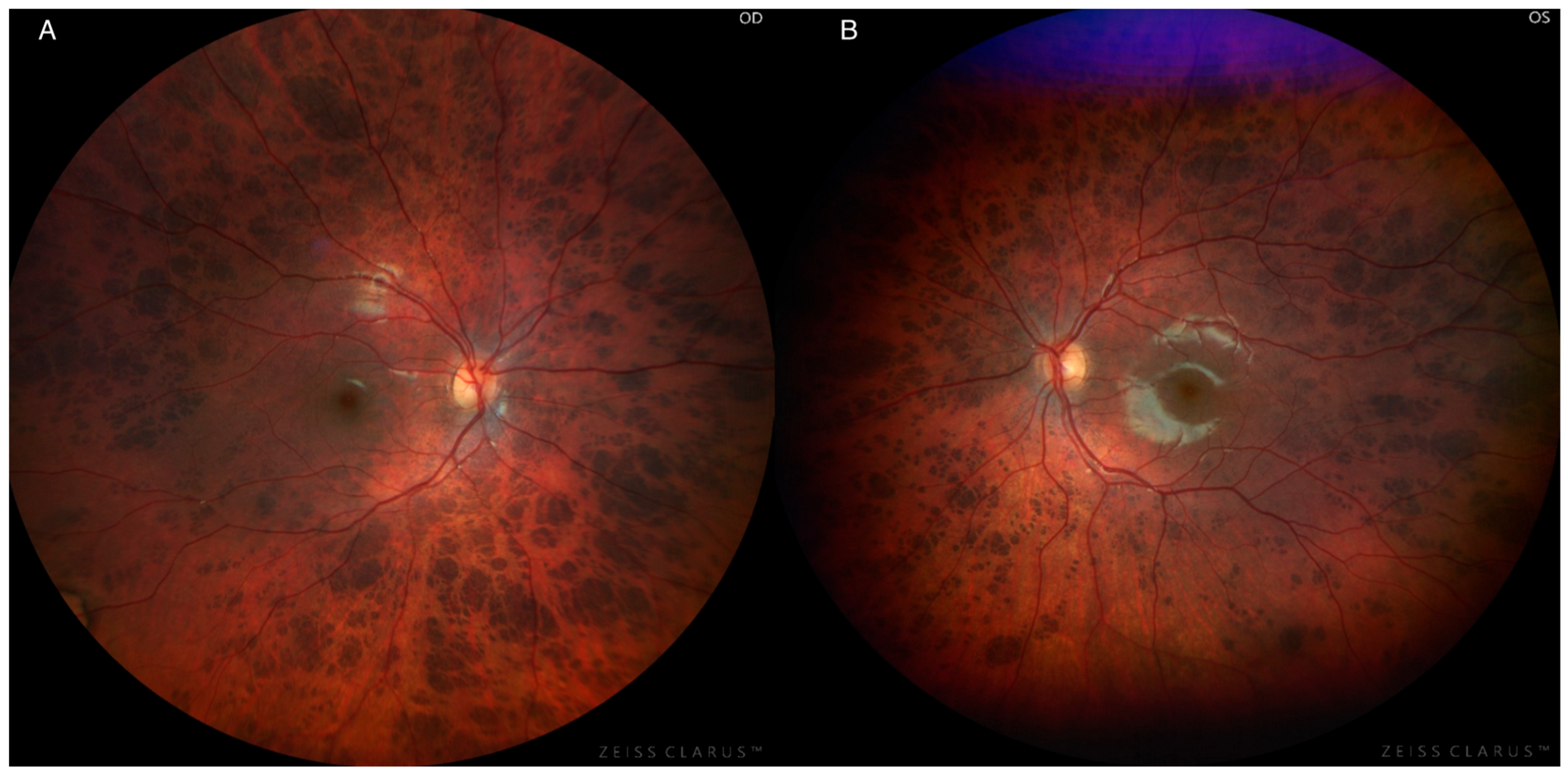
4. Clinical Findings
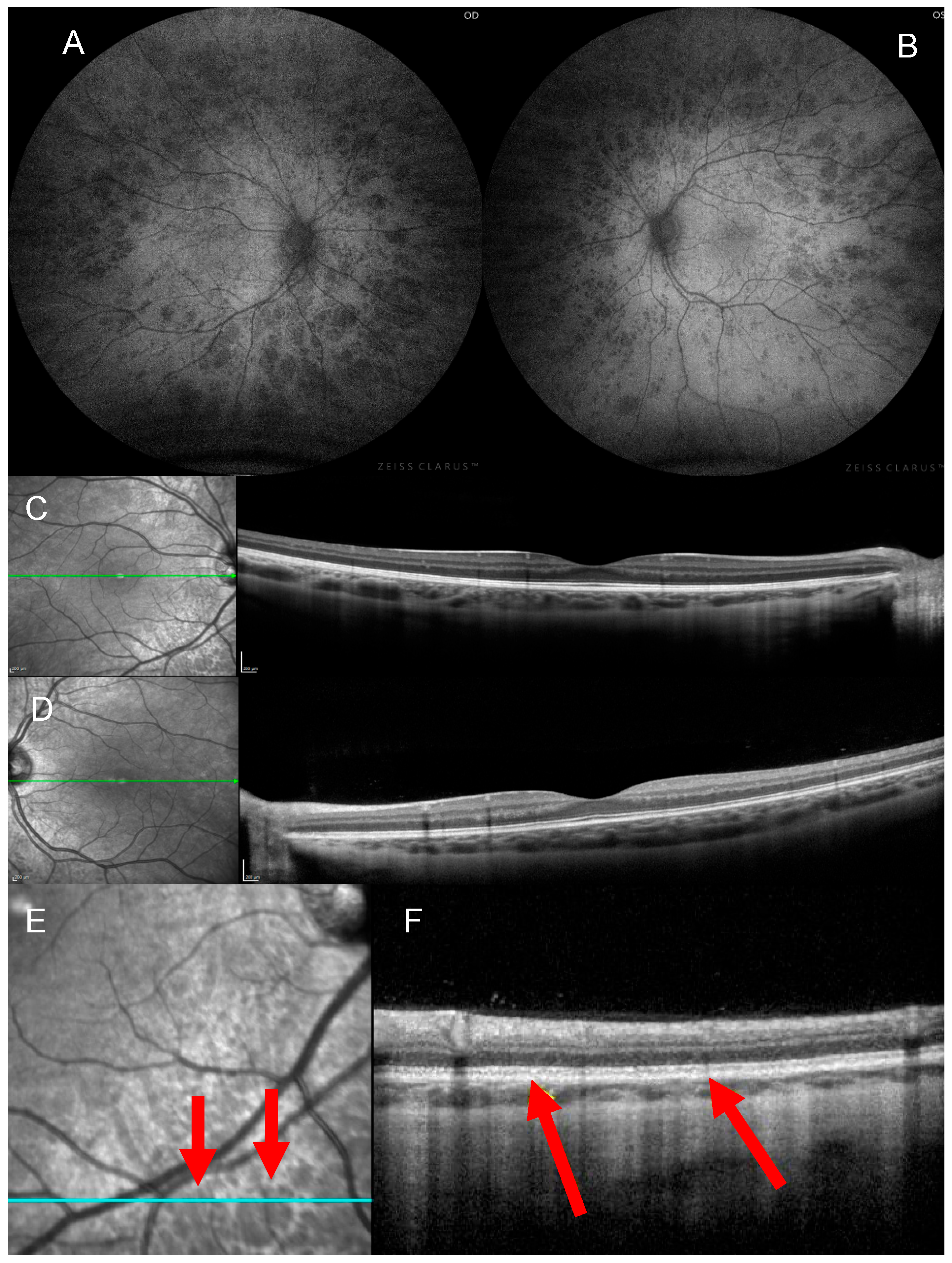
5. Investigations
6. Outcome and Follow-Up
7. Discussion
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Braga, C.S.; Ricardo, O.M.P.; Cordeiro, F.M.; Vieira, J.M.; Nogueira, F.B. Suspect asymptomatic lesions: Congenital hypertrophy of the Retinal Pigment Epithelium (CHRPE). Rom. J. Ophthalmol. 2021, 65, 275–278. [Google Scholar] [CrossRef] [PubMed]
- Deibert, B.; Ferris, L.; Sanchez, N.; Weishaar, P. The link between colon cancer and congenital hypertrophy of the retinal pigment epithelium (CHRPE). Am. J. Ophthalmol. Case Rep. 2019, 15, 100524. [Google Scholar] [CrossRef] [PubMed]
- Coleman, P.; Barnard, N.A. Congenital hypertrophy of the retinal pigment epithelium: Prevalence and ocular features in the optometric population. Ophthalmic Physiol. Opt. 2007, 27, 547–555. [Google Scholar] [CrossRef] [PubMed]
- Regillo, C.D.; Eagle, R.C., Jr.; Shields, J.A.; Shields, C.L.; Arbizo, V.V. Histopathologic findings in congenital grouped pigmentation of the retina. Ophthalmology 1993, 100, 400–405. [Google Scholar] [CrossRef]
- Traboulsi, E.I.; Murphy, S.F.; de la Cruz, Z.C.; Maumenee, I.H.; Green, W.R. A clinicopathologic study of the eyes in familial adenomatous polyposis with extracolonic manifestations (Gardner’s syndrome). Am. J. Ophthalmol. 1990, 110, 550–561. [Google Scholar] [CrossRef]
- Wallis, Y.L.; Macdonald, F.; Hulten, M.; Morton, J.E.; McKeown, C.M.; Neoptolemos, J.P.; Keighley, M.; Morton, D.G. Genotype-phenotype correlation between position of constitutional APC gene mutation and CHRPE expression in familial adenomatous polyposis. Hum. Genet. 1994, 94, 543–548. [Google Scholar] [CrossRef]
- Kruszka, P.; Berger, S.I.; Casa, V.; Dekker, M.R.; Gaesser, J.; Weiss, K.; Martinez, A.F.; Murdock, D.R.; Louie, R.J.; Prijoles, E.J.; et al. Cohesin complex-associated holoprosencephaly. Brain 2019, 142, 2631–2643. [Google Scholar] [CrossRef]
- Brooker, A.S.; Berkowitz, K.M. The roles of cohesins in mitosis, meiosis, and human health and disease. Methods Mol. Biol. 2014, 1170, 229–266. [Google Scholar]
- Kumar, R.; Corbett, M.A.; Van Bon, B.W.; Gardner, A.; Woenig, J.A.; Jolly, L.A.; Douglas, E.; Friend, K.; Tan, C.; Van Esch, H.; et al. Increased STAG2 dosage defines a novel cohesinopathy with intellectual disability and behavioral problems. Hum. Mol. Genet. 2015, 24, 7171–7181. [Google Scholar] [CrossRef]
- Robson, A.G.; Nilsson, J.; Li, S.; Jalali, S.; Fulton, A.B.; Tormene, A.P.; Holder, G.E.; Brodie, S.E. ISCEV guide to visual electrodiagnostic procedures. Doc. Ophthalmol. 2018, 136, 1–26. [Google Scholar] [CrossRef]
- Andreasson, S.O.; Sandberg, M.A.; Berson, E.L. Narrow-band filtering for monitoring low-amplitude cone electroretinograms in retinitis pigmentosa. Am. J. Ophthalmol. 1988, 105, 500–503. [Google Scholar] [CrossRef]
- Berson, E.L.; Rosner, B.; Sandberg, M.A.; Hayes, K.C.; Nicholson, B.W.; Weigel-DiFranco, C.; Willett, W. A randomized trial of vitamin A and vitamin E supplementation for retinitis pigmentosa. Arch. Ophthalmol. 1993, 111, 761–772. [Google Scholar] [CrossRef]
- Richards, S.; Aziz, N.; Bale, S.; Bick, D.; Das, S.; Gastier-Foster, J.; Grody, W.W.; Hegde, M.; Lyon, E.; Spector, E.; et al. Standards and guidelines for the interpretation of sequence variants: A joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet. Med. 2015, 17, 405–424. [Google Scholar] [CrossRef]
- Gudmundsson, S.; Singer-Berk, M.; Watts, N.A.; Phu, W.; Goodrich, J.K.; Solomonson, M.; Genome Aggregation Database, C.; Rehm, H.L.; MacArthur, D.G.; O’Donnell-Luria, A. Variant interpretation using population databases: Lessons from gnomAD. Hum. Mutat. 2022, 43, 1012–1030. [Google Scholar] [CrossRef] [PubMed]
- Schubach, M.; Maass, T.; Nazaretyan, L.; Roner, S.; Kircher, M. CADD v1.7: Using protein language models, regulatory CNNs and other nucleotide-level scores to improve genome-wide variant predictions. Nucleic Acids Res. 2024, 52, D1143–D1154. [Google Scholar] [CrossRef] [PubMed]
- Mullegama, S.V.; Klein, S.D.; Mulatinho, M.V.; Senaratne, T.N.; Singh, K.; Center, U.C.G.; Nguyen, D.C.; Gallant, N.M.; Strom, S.P.; Ghahremani, S.; et al. De novo loss-of-function variants in STAG2 are associated with developmental delay, microcephaly, and congenital anomalies. Am. J. Med. Genet. A 2017, 173, 1319–1327. [Google Scholar] [CrossRef]
- Yuan, B.; Neira, J.; Pehlivan, D.; Santiago-Sim, T.; Song, X.; Rosenfeld, J.; Posey, J.E.; Patel, V.; Jin, W.; Adam, M.P.; et al. Clinical exome sequencing reveals locus heterogeneity and phenotypic variability of cohesinopathies. Genet. Med. 2019, 21, 663–675. [Google Scholar] [CrossRef]
- Aoi, H.; Lei, M.; Mizuguchi, T.; Nishioka, N.; Goto, T.; Miyama, S.; Suzuki, T.; Iwama, K.; Uchiyama, Y.; Mitsuhashi, S.; et al. Nonsense variants of STAG2 result in distinct congenital anomalies. Hum. Genome Var. 2020, 7, 26. [Google Scholar] [CrossRef] [PubMed]
- Rinaldi, B.; Race, V.; Corveleyn, A.; Van Hoof, E.; Bauters, M.; Van Den Bogaert, K.; Denayer, E.; de Ravel, T.; Legius, E.; Baldewijns, M.; et al. Next-generation sequencing in prenatal setting: Some examples of unexpected variant association. Eur. J. Med. Genet. 2020, 63, 103875. [Google Scholar] [CrossRef]
- Freyberger, F.; Kokotovic, T.; Krnjak, G.; Frkovic, S.H.; Nagy, V. Expanding the known phenotype of Mullegama-Klein-Martinez syndrome in male patients. Hum. Genome Var. 2021, 8, 37. [Google Scholar] [CrossRef]
- Schmidt, J.; Dreha-Kulaczewski, S.; Zafeiriou, M.P.; Schreiber, M.K.; Wilken, B.; Funke, R.; Neuhofer, C.M.; Altmuller, J.; Thiele, H.; Nurnberg, P.; et al. Somatic mosaicism in STAG2-associated cohesinopathies: Expansion of the genotypic and phenotypic spectrum. Front. Cell Dev. Biol. 2022, 10, 1025332. [Google Scholar] [CrossRef] [PubMed]
- Comaratta, M.; Bakall, B. Extensive Grouped Congenital Hypertrophy of the Retinal Pigment Epithelium. Ophthalmol. Retina 2018, 2, 224. [Google Scholar] [CrossRef] [PubMed]
- Li, C.J.; Yaghy, A.; Shields, C.L. Pigmented Ocular Fundus Lesions Associated with Familial Adenomatous Polyposis. Ophthalmic Surg. Lasers Imaging Retin. 2020, 51, 124. [Google Scholar] [CrossRef]
- Ly, A.; Nivison-Smith, L.; Hennessy, M.; Kalloniatis, M. Pigmented Lesions of the Retinal Pigment Epithelium. Optom. Vis. Sci. 2015, 92, 844–857. [Google Scholar] [CrossRef]
- Traboulsi, E.I. Ocular manifestations of familial adenomatous polyposis (Gardner syndrome). Ophthalmol. Clin. N. Am. 2005, 18, 163–166. [Google Scholar] [CrossRef]
- Shields, C.L.; Mashayekhi, A.; Ho, T.; Cater, J.; Shields, J.A. Solitary congenital hypertrophy of the retinal pigment epithelium: Clinical features and frequency of enlargement in 330 patients. Ophthalmology 2003, 110, 1968–1976. [Google Scholar] [CrossRef]
- Santos, A.; Morales, L.; Hernandez-Quintela, E.; Jimenez-Sierra, J.M.; Villalobos, J.J.; Panduro, A. Congenital hypertrophy of the retinal pigment epithelium associated with familial adenomatous polyposis. Retina 1994, 14, 6–9. [Google Scholar] [CrossRef]
- Shields, C.L.; Materin, M.A.; Walker, C.; Marr, B.P.; Shields, J.A. Photoreceptor loss overlying congenital hypertrophy of the retinal pigment epithelium by optical coherence tomography. Ophthalmology 2006, 113, 661–665. [Google Scholar] [CrossRef]
- Marmoy, O.R.; Blackwell, C.; Cornelius, S.; Thompson, D.A.; Henderson, R.H. Diffuse bear-track retina: Profound, bilateral, grouped congenital pigmentation of the retinal pigment epithelium in an infant. J. AAPOS 2020, 24, 384–386. [Google Scholar] [CrossRef]
- Piche, J.; Van Vliet, P.P.; Puceat, M.; Andelfinger, G. The expanding phenotypes of cohesinopathies: One ring to rule them all! Cell Cycle 2019, 18, 2828–2848. [Google Scholar] [CrossRef] [PubMed]
- Kline, A.D.; Moss, J.F.; Selicorni, A.; Bisgaard, A.M.; Deardorff, M.A.; Gillett, P.M.; Ishman, S.L.; Kerr, L.M.; Levin, A.V.; Mulder, P.A.; et al. Diagnosis and management of Cornelia de Lange syndrome: First international consensus statement. Nat. Rev. Genet. 2018, 19, 649–666. [Google Scholar] [CrossRef] [PubMed]
- Dorsett, D.; Krantz, I.D. On the molecular etiology of Cornelia de Lange syndrome. Ann. N. Y. Acad. Sci. 2009, 1151, 22–37. [Google Scholar] [CrossRef] [PubMed]
- Gibellato, E.; Cianci, P.; Mariani, M.; Parma, B.; Huisman, S.; Smigiel, R.; Bisgaard, A.M.; Massa, V.; Gervasini, C.; Moretti, A.; et al. SMC1A epilepsy syndrome: Clinical data from a large international cohort. Am. J. Med. Genet. A 2024, 194, e63577. [Google Scholar] [CrossRef] [PubMed]

| Reported Variant | Sex | Summary of Systemic Findings | Ocular Findings | |
|---|---|---|---|---|
| Present Study | c.3222dup, p.Ser1075IlefsTer12 | F | Feeding problems, speech delay | Optic nerve pallor, congenital hypertrophy of the RPE |
| Mullegama et al., 2017 [16] | c.205C > T, p.Arg69Ter | F | Dysgenesis of splenium and corpus callosum, speech delay, cleft palate, bilateral microtia, hearing loss, hemivertebrae, butterfly vertebrae, fifth finger clinodactyly, VSD | Strabismus |
| Kruszka et al., 2019 [7] | c.3034C > T, p.Arg1012Ter | F | Alobar brain, microcephaly, midline cleft palate, low-set ears, lumbar spina bifida, GERD | NR |
| c.205C > T, p.Arg69Ter | F | Semi-lobar brain, global developmental delay, growth delay, microcephaly, cleft palate, micrognathia, PFO, PDA | NR | |
| c.436C > T, p.Arg146Ter | F | Alobar brain, growth delay, microcephaly, absent nose, hypognathia, hypoplastic ear, hemivertebrae, VSD, duodenal atresia | Cyclopia | |
| c.2533 + 1G > A (intronic) | F | Semi-lobar brain, microcephaly, hypoplastic left heart, double outlet right ventricle | NR | |
| c.2898_2899del, p.Glu968SerfsTer15 | F | Microform brain, developmental delay, growth delay, microcephaly | NR | |
| c.775C > T, p.Arg259Ter | F | Septo-optic dysplasia, intellectual disability, motor delay, left hip dysplasia, VSD | Bilateral optic nerve hypoplasia | |
| Yuan et al., 2019 [17] | c.418C > T, p.Gln140Ter | F | Seizure disorder, motor and speech delay, dysmorphic ears, vertebral clefts, hypoplastic left heart | NR |
| c.1605T > A, p.Cys535Ter | F | Intellectual disability, motor and speech delay, growth delay, microcephaly, micrognathia, microtia, hearing loss, fifth finger clinodactyly | Strabismus | |
| c.1811G > A, p.Arg604Gln | F | Intellectual disability, growth delay, microcephaly, micrognathia, hypotonia, vertebral clefts, CDH, pulmonary hypoplasia, GERD | NR | |
| c.1658_1660delinsT, p.Lys533IlefsTer6 | F | Microform brain with agenesis of the corpus callosum, seizure disorder, intellectual disability, motor and speech delay, growth delay, microcephaly with colpocephaly, single central incisor, micrognathia, dysmorphic ears, vertebral anomalies | NR | |
| c.476A > G, p.Tyr159Cys | M | Ectopic posterior pituitary, intellectual disability, growth delay, cleft palate, low-set ears, hypotonia, scoliosis, single kidney | None | |
| Aoi et al., 2020 [18] | c.3097C > T, p.Arg1033Ter | M | Holoprosencephaly, cleft palate, hypoplastic left heart | NR |
| c.2229C > T, p.Trp743Ter | F | White matter hypoplasia, seizure disorder, intellectual disability, growth delay, cleft palate, hearing loss, thoracic hemivertebrae | NR | |
| Rinaldi et al., 2020 [19] | c.3724C > T, p.Arg1242Ter | F | Holoprosencephaly, low-set ears, CDH, ambiguous genitalia | Unspecified eye abnormalities |
| Freyberger et al., 2021 [20] | c.475T > C, p.Tyr159His | M | Polymicrogyria with hypotrophic corpus callosum, dolichocephaly, high arched palate, hypotonia | Atrophic retinal and uveal scar |
| Schmidt et al., 2022 [21] | c. 2184G > T, p.Gln728His | F | Hypotrophic right hemisphere with dysplastic corpus callosum and white matter hypoplasia, microcephaly, low-set ears, hearing loss, hemivertebrae, butterfly vertebrae, hemangioma | Limbal dermoid |
| c.1412_1416 + 9del (intronic) | F | Holoprosencephaly, speech and motor delay, microcephaly, low-set ears, retrognathia, dystonia, scoliosis | Strabismus |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kong, M.D.; Sylla, M.M.; Oh, J.K.; Dedania, V.S.; Soucy, M.; Demirkol, A.; Brodie, S.E.; Maumenee, I.H.; Tsang, S.H. Panretinal Congenital Hypertrophy of the RPE in an 8-Year-Old Girl with an X-Linked STAG2 Mutation. J. Clin. Med. 2025, 14, 6110. https://doi.org/10.3390/jcm14176110
Kong MD, Sylla MM, Oh JK, Dedania VS, Soucy M, Demirkol A, Brodie SE, Maumenee IH, Tsang SH. Panretinal Congenital Hypertrophy of the RPE in an 8-Year-Old Girl with an X-Linked STAG2 Mutation. Journal of Clinical Medicine. 2025; 14(17):6110. https://doi.org/10.3390/jcm14176110
Chicago/Turabian StyleKong, Maximilian D., Mohamed M. Sylla, Jin Kyun Oh, Vaidehi S. Dedania, Megan Soucy, Aykut Demirkol, Scott E. Brodie, Irene H. Maumenee, and Stephen H. Tsang. 2025. "Panretinal Congenital Hypertrophy of the RPE in an 8-Year-Old Girl with an X-Linked STAG2 Mutation" Journal of Clinical Medicine 14, no. 17: 6110. https://doi.org/10.3390/jcm14176110
APA StyleKong, M. D., Sylla, M. M., Oh, J. K., Dedania, V. S., Soucy, M., Demirkol, A., Brodie, S. E., Maumenee, I. H., & Tsang, S. H. (2025). Panretinal Congenital Hypertrophy of the RPE in an 8-Year-Old Girl with an X-Linked STAG2 Mutation. Journal of Clinical Medicine, 14(17), 6110. https://doi.org/10.3390/jcm14176110






