Salivary Flow Rate in Patients with Kidney Failure on Hemodialysis—A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Methods
2.1. Search Strategy
2.2. Inclusion and Exclusion Criteria
2.3. Data Collection
2.4. Quality Assessment
2.5. Meta-Analysis
3. Results
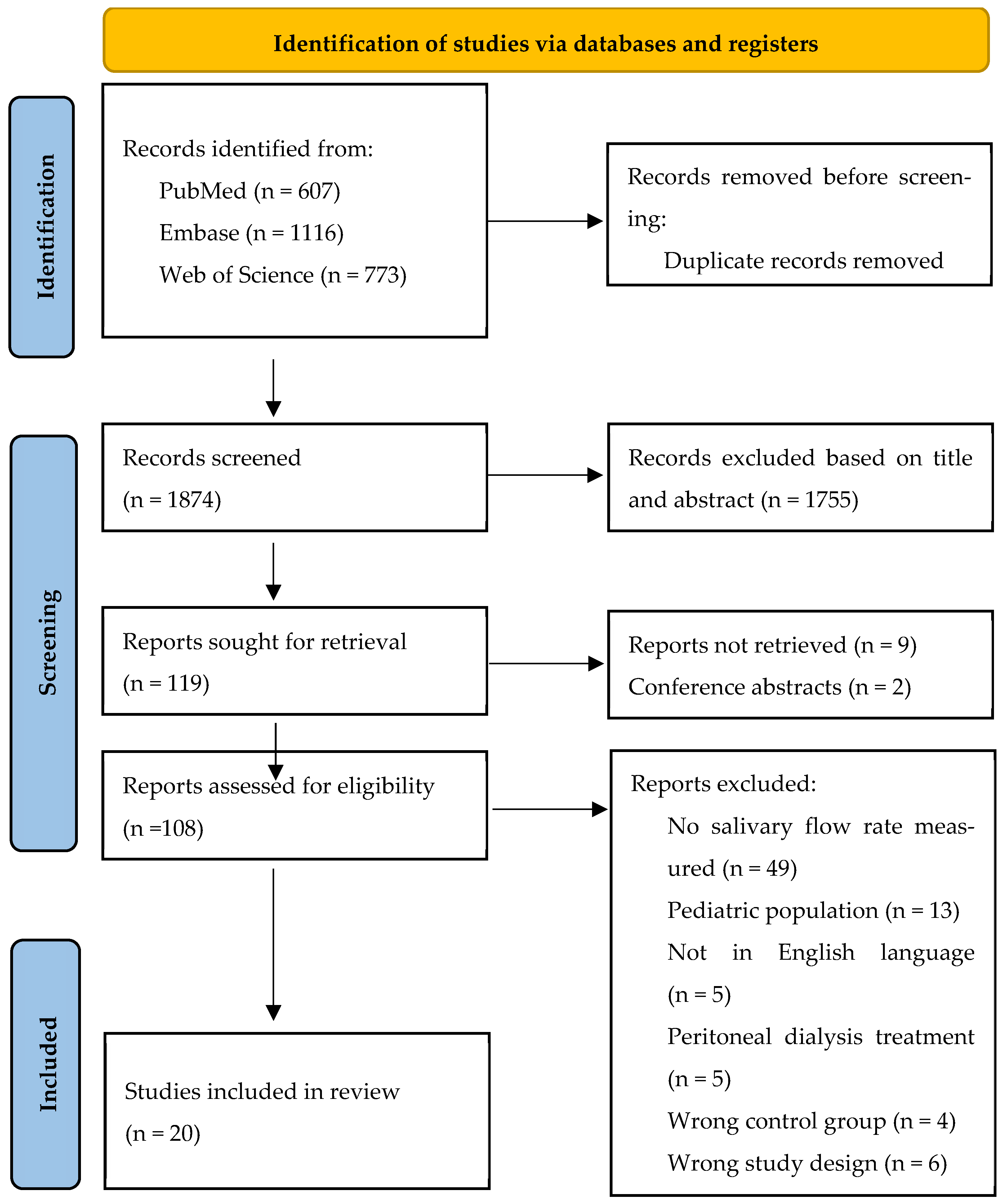
3.1. Study Selection
3.2. Quality Assessment
3.3. Study Characteristics
3.4. Study Population
3.5. Participant Age and Sex
3.6. Treatment Details
3.7. Saliva Collection Details
3.7.1. Unstimulated Whole Saliva
3.7.2. Stimulated Whole Saliva
3.7.3. Stimulated Parotid Saliva
3.7.4. Unstimulated Parotid Saliva
3.7.5. Submandibular Saliva
3.8. Clinical Findings
3.8.1. Unstimulated Whole Saliva
3.8.2. Stimulated Whole Saliva
3.8.3. Stimulated Parotid Saliva
3.8.4. Unstimulated Parotid Saliva
3.8.5. Submandibular Saliva
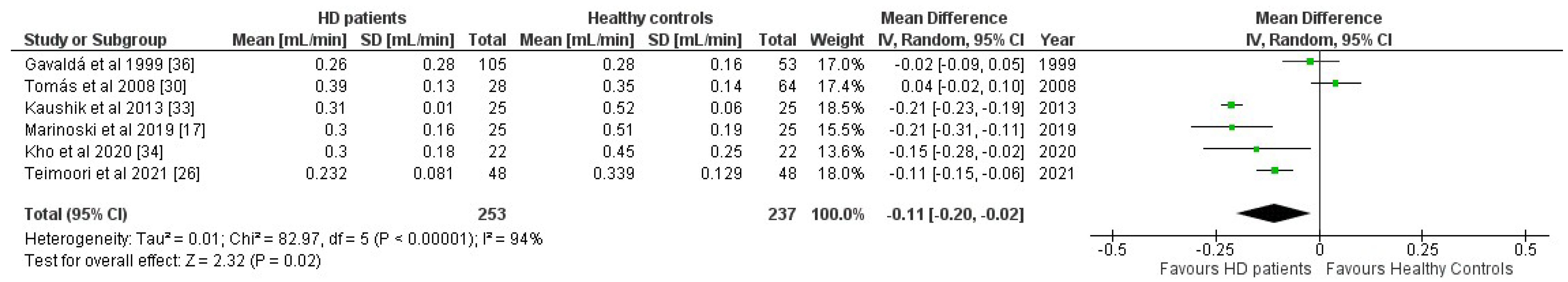
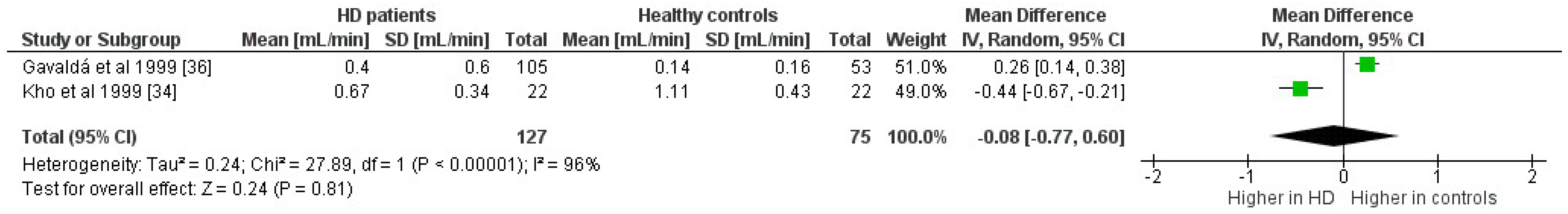
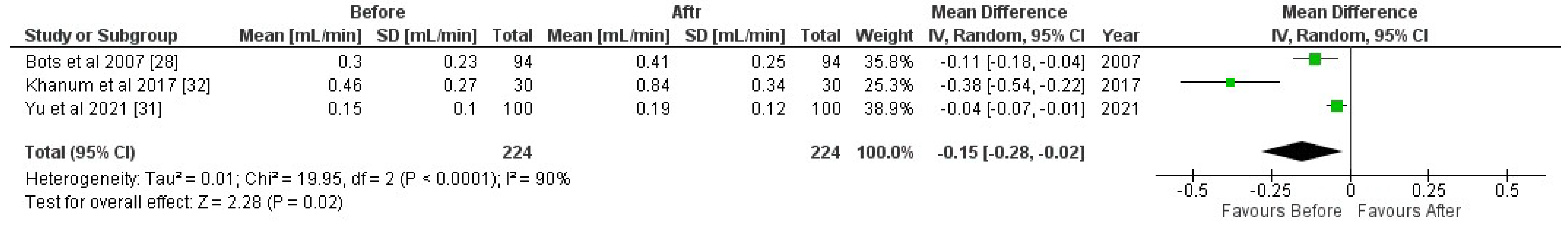
3.9. Meta-Analysis
3.10. Comparison Between Hemodialysis Patients and Healthy Controls
3.10.1. Unstimulated Whole Saliva
3.10.2. Stimulated Whole Saliva
3.10.3. Stimulated Parotid Saliva
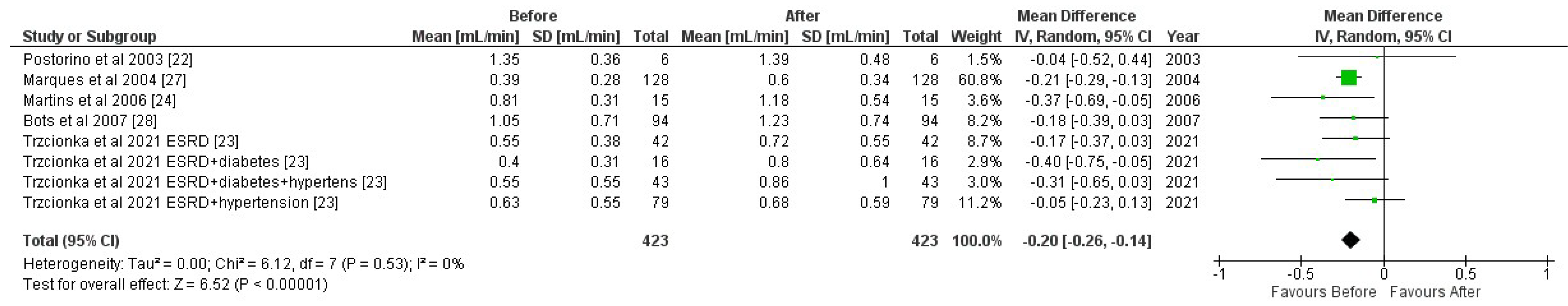
3.11. Before-After Comparison in Hemodialysis Patients
3.11.1. Unstimulated Whole Saliva
3.11.2. Stimulated Whole Saliva
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
Appendix A.1. Search Strategy Details
Appendix A.1.1. Pubmed Search
Appendix A.1.2. Embase Search
Appendix A.1.3. Web of Science Search
References
- Ammirati, A.L. Chronic kidney disease. Rev. Assoc. Med. Bras. 2020, 66, S3–S9. [Google Scholar] [CrossRef]
- Hill, N.R.; Fatoba, S.T.; Oke, J.L.; Hirst, J.A.; O’Callaghan, C.A.; Lasserson, D.S.; Hobbs, F.D.R. Global prevalence of chronic kidney disease—A systematic review and meta-analysis. PLoS ONE 2016, 11, e0158765. [Google Scholar] [CrossRef]
- Wang, H.; Naghavi, M.; Allen, C.; Barber, R.M.; Bhutta, Z.A.; Carter, A.; Casey, D.C.; Charlson, F.J.; Chen, A.Z.; Coates, M.M.; et al. Global, regional, and national life expectancy, all-cause mortality, and cause-specific mortality for 249 causes of death, 1980–2015. Lancet 2016, 388, 1459–1544. [Google Scholar] [CrossRef] [PubMed]
- Vaidya, S.; Narothama, A. Chronic kidney disease. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2024. [Google Scholar]
- Khor, C.-S.; Wang, W.-J. The role of acute kidney injury duration in clinical practice. Ann. Transl. Med. 2019, 7, S88. [Google Scholar] [CrossRef] [PubMed]
- Bots, C.P.; Brand, H.S.; Veerman, E.C.I.; Korevaar, J.C.; Valentijn-Benz, M.; Bezemer, P.D.; Valentijn, R.M.; Vos, P.F.; Bijlsma, J.A.; ter Wee, P.M.; et al. Chewing gum and a saliva substitute alleviate thirst and xerostomia in patients on haemodialysis. Nephrol. Dial. Transplant. 2005, 20, 578–584. [Google Scholar] [CrossRef]
- Bossola, M.; Tazza, L. Xerostomia in patients on chronic hemodialysis. Nat. Rev. Nephrol. 2012, 8, 176–182. [Google Scholar] [CrossRef]
- Rodrigues, R.P.C.B.; Vidigal, M.T.C.; Vieira, W.A.; Nascimento, G.G.; Sabino-Silva, R.; Blumenberg, C.; Siqueira, M.F.; Siqueira, W.L.; Paranhos, L.R. Salivary changes in chronic kidney disease and in patients undergoing hemodialysis: A systematic review and meta-analysis. J. Nephrol. 2022, 35, 1339–1367. [Google Scholar] [CrossRef]
- Humphrey, S.P.; Williamson, R.T. A review of saliva: Normal composition, flow, and function. J. Prosthet. Dent. 2001, 85, 162–169. [Google Scholar] [CrossRef]
- Mese, H.; Matsuo, R. Salivary secretion, taste and hyposalivation. J. Oral Rehabil. 2007, 34, 711–723. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. Syst. Rev. 2021, 10, 89. [Google Scholar] [CrossRef]
- Morgan, R.L.; Whaley, P.; Thayer, K.A.; Schünemann, H.J. Identifying the PECO: A framework for formulating good questions. Environ. Int. 2018, 121, 1027–1031. [Google Scholar] [CrossRef]
- National Heart, Lung, and Blood Institute. Study Quality Assessment Tools. 2021. Available online: https://www.nhlbi.nih.gov/health-topics/study-quality-assessment-tools (accessed on 30 June 2025).
- Schwendinger, F.; Infanger, D.; Pocecco, E.; Gander, J.; Hinrichs, T.; Schmidt-Trucksäss, A. Protocol for a systematic review of observational studies on COVID-19 safety measures and physical activity. Syst. Rev. 2021, 10, 281. [Google Scholar] [CrossRef]
- Christenson, E.C.; Cronk, R.; Atkinson, H.; Bhatt, A.; Berdiel, E.; Cawley, M.; Cho, G.; Coleman, C.K.; Harrington, C.; Heilferty, K.; et al. Disinfection efficacy on environmental surfaces in healthcare facilities. Int. J. Environ. Res. Public Health 2021, 18, 11100. [Google Scholar] [CrossRef] [PubMed]
- Khella, C.M.; Asgarian, R.; Horvath, J.M.; Rolauffs, B.; Hart, M.L. Human knee post-traumatic osteoarthritis: Timeline and markers. Int. J. Mol. Sci. 2021, 22, 1996. [Google Scholar] [CrossRef]
- Marinoski, J.; Bokor-Bratić, M.; Mitić, I.; Ćanković, M. Oral mucosa and salivary findings in non-diabetic CKD patients. Arch. Oral Biol. 2019, 102, 205–211. [Google Scholar] [CrossRef]
- Limeres, J.; Garcez, J.F.; Marinho, J.S.; Loureiro, A.; Diniz, M.; Diz, P. Early tooth loss in ESRD patients on haemodialysis. Oral Dis. 2016, 22, 530–535. [Google Scholar] [CrossRef]
- Shannon, I.L.; Feller, R.P.; Eknoyan, G.; Suddick, R.P. Human parotid saliva urea in renal failure and during dialysis. Arch. Oral Biol. 1977, 22, 83–86. [Google Scholar] [CrossRef]
- Epstein, S.R.; Mandel, I.; Scopp, I.W. Salivary composition and calculus formation in hemodialysis patients. J. Periodontol. 1980, 51, 336–338. [Google Scholar] [CrossRef]
- Kumar, T.; Kishore, J.; Kumari, M.; Rai, A.; Rai, S.; Jha, A. Salivary flow rate, pH and buffer capacities in ESRD versus control. J. Fam. Med. Prim. Care 2020, 9, 2985–2989. [Google Scholar] [CrossRef]
- Bayraktar, G.; Kazancioglu, R.; Bozfakioglu, S.; Yildiz, A.; Ark, E. Salivary parameters and dental status in hemodialysis. Clin. Nephrol. 2004, 62, 380–383. [Google Scholar] [CrossRef]
- Bayraktar, G.; Kurtulus, I.; Kazancioglu, R.; Bayramgurler, I.; Cintan, S.; Bural, C.; Bozfakioglu, S.; Issever, H.; Yildiz, A. Oral health and inflammation in ESRF. Perit. Dial. Int. 2009, 29, 472–479. [Google Scholar] [CrossRef]
- Gavaldá, C.; Bagán, J.; Scully, C.; Silvestre, F.; Milián, M.; Jiménez, Y. Oral, salivary and periodontal findings in hemodialysis. Oral Dis. 1999, 5, 299–302. [Google Scholar] [CrossRef]
- Kaushik, A.; Reddy, S.S.; Umesh, L.; Devi, B.K.Y.; Santana, N.; Rakesh, N. Oral and salivary changes in renal patients on hemodialysis. Indian J. Nephrol. 2013, 23, 125–129. [Google Scholar] [CrossRef]
- Kho, H.S.; Lee, S.W.; Chung, S.C.; Kim, Y.K. Oral manifestations and saliva in ESRD on hemodialysis. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 1999, 88, 316–319. [Google Scholar] [CrossRef]
- Martins, C.; Siqueira, W.L.; de Oliveira, E.; Primo, L.S.G.; Nicolau, J. Salivary analysis in chronic renal failure on hemodialysis. Spec. Care Dentist 2006, 26, 205–208. [Google Scholar] [CrossRef]
- Postorino, M.; Catalano, C.; Martorano, C.; Cutrupi, S.; Marino, C.; Cozzupoli, P.; Scudo, P.; Zoccali, C. Salivary and lacrimal secretion is reduced in ESRD. Am. J. Kidney Dis. 2003, 42, 722–728. [Google Scholar] [CrossRef]
- Shetty, P.; Hegde, M.N.; Eraly, S.M. Salivary parameters and dental status in adult hemodialysis patients. J. Clin. Exp. Dent. 2018, 10, e419–e424. [Google Scholar] [CrossRef]
- Teimoori, F.; Hajilooei, M.; Abdolsamadi, H.; Eslami, K.; Moghimbeigi, A.; Motamayel, F.A. Salivary Helicobacter pylori, calcium, urea, pH and flow in hemodialysis. Infect. Disord. Drug Targets 2021, 21, e160921189262. [Google Scholar] [CrossRef] [PubMed]
- Tomás, I.; Marinho, J.S.; Limeres, J.; Santos, M.; Araújo, L.; Diz, P. Salivary composition in renal failure. Arch. Oral Biol. 2008, 53, 528–532. [Google Scholar] [CrossRef] [PubMed]
- Trzcionka, A.; Twardawa, H.; Mocny-Pachońska, K.; Korkosz, R.; Tanasiewicz, M. Oral mucosa status in end-stage CKD. Int. J. Environ. Res. Public Health 2021, 18, 12515. [Google Scholar] [CrossRef]
- Bots, C.P.; Brand, H.S.; Veerman, E.C.I.; Valentijn-Benz, M.; Henskens, Y.M.C.; Valentijn, R.M.; Vos, P.F.; Bijlsma, J.A.; Ter Wee, P.M.; Van Amerongen, B.M.; et al. Acute effects of hemodialysis on salivary flow and composition. Clin. Nephrol. 2007, 67, 25–31. [Google Scholar] [CrossRef]
- Khanum, N.; Mysore-Shivalingu, M.; Basappa, S.; Patil, A.; Kanwar, S. Salivary changes before and after hemodialysis. J. Clin. Exp. Dent. 2017, 9, e1340–e1345. [Google Scholar] [CrossRef]
- Marques, P.L.P.; Libório, A.B.; Saintrain, M.V.L. Hemodialysis-specific factors and salivary flow. Artif. Organs 2015, 39, 181–186. [Google Scholar] [CrossRef]
- Yu, I.-C.; Liu, C.-Y.; Fang, J.-T. Hemodialysis effects on saliva flow and composition. Ren. Fail. 2021, 43, 71–78. [Google Scholar] [CrossRef]
- Bossola, M.; Calvani, R.; Marzetti, E.; Picca, A.; Antocicco, E. Thirst in chronic hemodialysis: Current knowledge. Int. Urol. Nephrol. 2020, 52, 697–711. [Google Scholar] [CrossRef]
- Mehta, A.R. Why does plasma urea rise in acute dehydration? Adv. Physiol. Educ. 2008, 32, 336. [Google Scholar] [CrossRef] [PubMed]
- Himmelfarb, J.; Ikizler, T.A. Hemodialysis. N. Engl. J. Med. 2010, 363, 1833–1845. [Google Scholar] [CrossRef]
- Pandya, D.; Nagrajappa, A.K.; Ravi, K.S. Urea and creatinine in saliva and serum. J. Clin. Diagn. Res. 2016, 10, ZC58–ZC62. [Google Scholar] [CrossRef] [PubMed]
- Oliver, S.J.; Laing, S.J.; Wilson, S.; Bilzon, J.L.; Walsh, N.P. Saliva indices track hypohydration. Arch. Oral Biol. 2008, 53, 975–980. [Google Scholar] [CrossRef]
- Walsh, N.; Laing, S.; Oliver, S.; Montague, J.C.; Walters, R.; Bilzon, J.L.J. Saliva parameters in acute dehydration. Med. Sci. Sports Exerc. 2004, 36, 1535–1542. [Google Scholar] [CrossRef]
- Aps, J.K.M.; Martens, L.C. Physiology of saliva and drug transfer. Forensic Sci. Int. 2005, 150, 119–131. [Google Scholar] [CrossRef]
- Romero, A.C.; Bergamaschi, C.T.; de Souza, D.N.; Nogueira, F.N. Salivary alterations in rats with CKD. PLoS ONE 2016, 11, e0148742. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Ye, Y.; Wu, Y.; Li, L.; Hu, J.; Luo, D.; Li, Y.; Yang, J.; Gao, Y.; Hai, W.; et al. Aging salivary gland histology and inflammation. iScience 2023, 26, 106571. [Google Scholar] [CrossRef] [PubMed]
- Affoo, R.H.; Foley, N.; Garrick, R.; Siqueira, W.L.; Martin, R.E. Meta-analysis of salivary flow rates. J. Am. Geriatr. Soc. 2015, 63, 2142–2151. [Google Scholar] [CrossRef]
- López-Pintor, R.M.; Casañas, E.; González-Serrano, J.; Serrano, J.; Ramírez, L.; de Arriba, L.; Hernández, G. Xerostomia and salivary flow in diabetes. J. Diabetes Res. 2016, 2016, 4372852. [Google Scholar] [CrossRef]
- Meyer, T.W.; Hostetter, T.H. Uremia. N. Engl. J. Med. 2007, 357, 1316–1325. [Google Scholar] [CrossRef]
- Bruzda-Zwiech, A.; Szczepańska, J.; Zwiech, R. Sodium gradient, xerostomia and weight gain in hemodialysis. Int. Urol. Nephrol. 2014, 46, 1411–1417. [Google Scholar] [CrossRef][Green Version]
- Teratani, G.; Awano, S.; Soh, I.; Yoshida, A.; Kinoshita, N.; Hamasaki, T.; Takata, Y.; Sonoki, K.; Nakamura, H.; Ansai, T. Oral health in haemodialysis for diabetic nephropathy. Clin. Oral Investig. 2013, 17, 483–489. [Google Scholar] [CrossRef]
- Dobrescu, A.I.; Nussbaumer-Streit, B.; Klerings, I.; Wagner, G.; Persad, E.; Sommer, I.; Herkner, H.; Gartlehner, G. Restricting evidence syntheses of interventions to English-language publications is a viable methodological shortcut for most medical topics: A systematic review. J. Clin. Epidemiol. 2021, 137, 209–217. [Google Scholar] [CrossRef]
- Navazesh, M.; Christensen, C.M. A comparison of whole mouth resting and stimulated salivary measurement procedures. J. Dent. Res. 1982, 61, 1158–1162. [Google Scholar] [CrossRef]
- Walsh, L. Clinical aspects of salivary biology. Int. Dent. S. Afr. 2007, 2, 16–30. [Google Scholar]
- Inoue, H.; Ono, K.; Masuda, W.; Morimoto, Y.; Tanaka, T.; Yokota, M.; Inenaga, K. Gender difference in unstimulated whole saliva flow rate. Arch. Oral Biol. 2006, 51, 1055–1060. [Google Scholar] [CrossRef]
- Bossola, M.; Mariani, I.; Strizzi, C.T.; Piccinni, C.P.; Di Stasio, E. Limiting interdialytic weight gain on hemodialysis. J. Clin. Med. 2025, 14, 1846. [Google Scholar] [CrossRef]
- Melo, J.L.M.A.; Coelho, C.P.S.; Nunes, F.P.S.; Heller, D.; Grisi, D.C.; Guimarães, M.D.C.M.; Dame-Teixeira, N. Hyposalivation and systemic conditions: Scoping review. BMC Oral Health 2023, 23, 505. [Google Scholar] [CrossRef]
- Sung, J.-M.; Kuo, S.-C.; Guo, H.-R.; Chuang, S.-F.; Lee, S.-Y.; Huang, J.-J. Decreased salivary flow as a dipsogenic factor in hemodialysis. J. Am. Soc. Nephrol. 2005, 16, 3418–3429. [Google Scholar] [CrossRef] [PubMed]
- Perez, L.M.; Fang, H.; Ashrafi, S.; Burrows, B.T.; King, A.C.; Larsen, R.J.; Sutton, B.P.; Wilund, K.R. Low-sodium meals to reduce interdialytic weight gain. Hemodial. Int. 2021, 25, 265–274. [Google Scholar] [CrossRef] [PubMed]
- Laheij, A.; Rooijers, W.; Bidar, L.; Haidari, L.; Neradova, A.; de Vries, R.; Rozema, F. Oral health in end-stage renal disease: Scoping review. Clin. Exp. Dent. Res. 2022, 8, 54–67. [Google Scholar] [CrossRef]

| Criteria | Bayraktar (2004) [22] | Bayraktar (2009) [23] | Epstein (1980) [20] | Gavaldá (1999) [24] | Kaushik (2013) [25] | Kho (1999) [26] | Kumar (2020) [21] | Limeres (2016) [18] | Marinoski (2019) [17] | Martins (2016) [27] | Postorino (2003) [28] | Shetty (2018) [29] | Teimoori (2021) [30] | Tomás (2008) [31] | Trzcionka (2021) [32] |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1. Was the research question or objective in this paper clearly stated? | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| 2. Was the study population clearly specified and defined? | No | Yes | No | Yes | No | Yes | Yes | Yes | Yes | Yes | Yes | No | Yes | Yes | Yes |
| 3. Was the participation rate of eligible persons at least 50%? | NR | NR | NR | NR | NR | NR | NR | Yes | NR | NR | Yes | NR | NR | NR | NR |
| 4. Were all the subjects selected or recruited from the same or similar populations (including the same time period)? Were inclusion and exclusion criteria for being in the study prespecified and applied uniformly to all participants? | NR | No | NR | NR | NR | Yes | Yes | Yes | Yes | Yes | NR | NR | No | Yes | Yes |
| 5. Was a sample size justification, power description, or variance and effect estimate provided? | No | No | No | No | No | No | No | Yes | Yes | No | No | No | No | No | No |
| 6. For the analyses in this paper, were the exposure(s) of interest measured prior to the outcome(s) being measured? | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| 7. Was the timeframe sufficient so that one could reasonably expect to see an association between exposure and outcome if it existed? | No | No | No | No | No | No | No | No | No | Yes | No | No | No | No | Yes |
| 8. For exposures that can vary in amount or level, did the study examine different levels of the exposure as related to the outcome (e.g., categories of exposure or exposure measured as a continuous variable)? | No | No | No | No | No | No | No | No | No | No | No | Yes | No | No | Yes |
| 9. Were the exposure measures (independent variables) clearly defined, valid, reliable, and implemented consistently across all study participants? | Yes | Yes | Yes | Yes | Yes | Yes | No | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| 10. Was the exposure(s) assessed more than once over time? | NA | NA | No | NA | NA | NA | NA | NA | NA | Yes | Yes | NA | NA | Yes | Yes |
| 11. Were the outcome measures (dependent variables) clearly defined, valid, reliable, and implemented consistently across all study participants? | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| 12. Were the outcome assessors blinded to the exposure status of participants? | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | No |
| 13. Was loss to follow-up after baseline 20% or less? | NA | NA | NA | NA | NA | NA | NA | NA | NA | NR | Yes | NA | NA | NA | NR |
| 14. Were key potential confounding variables measured and adjusted statistically for their impact on the relationship between exposure(s) and outcome(s)? | No | Yes | No | No | No | No | No | Yes | Yes | No | No | No | No | Yes | No |
| Total score | 6 | 8 | 5 | 7 | 6 | 8 | 7 | 11 | 10 | 8 | 8 | 7 | 7 | 9 | 9 |
| Quality rating | Fair | Fair | Poor | Fair | Fair | Fair | Fair | Good | Good | Fair | Fair | Fair | Fair | Fair | Fair |
| Criteria | Bots (2007) [33] | Khanum (2017) [34] | Marques (2015) [35] | Shannon (1977) [19] | Yu (2021) [36] |
|---|---|---|---|---|---|
| 1. Was the study question or objective clearly stated? | Yes | Yes | Yes | No | Yes |
| 2. Were eligibility/selection criteria for the study population prespecified and clearly described? | Yes | Yes | Yes | No | Yes |
| 3. Were the participants in the study representative of those who would be eligible for the test/service/intervention in the general or clinical population of interest? | Yes | Yes | Yes | NR | No |
| 4. Were all eligible participants that met the prespecified entry criteria enrolled? | NR | No | NR | NR | Yes |
| 5. Was the sample size sufficiently large to provide confidence in the findings? | Yes | No | Yes | No | Yes |
| 6. Was the test/service/intervention clearly described and delivered consistently across the study population? | Yes | Yes | Yes | Yes | Yes |
| 7. Were the outcome measures prespecified, clearly defined, valid, reliable, and assessed consistently across all study participants? | Yes | Yes | Yes | Yes | Yes |
| 8. Were the people assessing the outcomes blinded to the participants’ exposures/interventions? | No | NR | NR | NR | No |
| 9. Was the loss to follow-up after baseline 20% or less? Were those lost to follow-up accounted for in the analysis? | NR | NR | NR | NR | NR |
| 10. Did the statistical methods examine changes in outcome measures from before to after the intervention? Were statistical tests conducted that provided p-values for the pre-to-post changes? | Yes | Yes | Yes | NR | Yes |
| 11. Were outcome measures of interest taken multiple times before the intervention and multiple times after the intervention (i.e., did they use an interrupted time-series design)? | No | No | No | No | No |
| 12. If the intervention was conducted at a group level (e.g., a whole hospital, a community, etc.), did the statistical analysis take into account the use of individual-level data to determine effects at the group level? | NA | NA | NA | NA | NA |
| Total score | 8 | 7 | 8 | 3 | 8 |
| Quality rating | Fair | Fair | Fair | Poor | Fair |
| Author, Year | Country | Groups (n), Mean Age and Sex | SFR HD | SFR C | Findings |
|---|---|---|---|---|---|
| Gavaldá et al. (1999) [24] | Spain | HD(105), 58.9 ± 14.9 y (50% m, 50% f) C (53), 55.7 ± 10.7 y (55% m, 45% f) | 0.26 ± 0.28 mL/min | 0.28 ± 0.16 mL/min | No significant difference in unstimulated whole salivary flow rates (p = 0.5) |
| Kaushik et al. (2013) [25] | India | HD(25), 44.4 ± 7.5 y (61% m, 39% f) C (25), not reported | 0.31 ± 0.01 mL/min | 0.52 ± 0.06 mL/min | Significantly lower unstimulated whole salivary flow rates in HD patients (p < 0.001) |
| Kho et al. (1999) [26] | Republic of Korea | HD (22), 34.7 ± 10.8 y (64% m, 36% f) C (22), 30.5 ± 7.9 y (sex not reported) | 0.30 ± 0.18 mL/min | 0.45 ± 0.25 mL/min | Significantly lower unstimulated whole salivary flow rates in HD patients (p < 0.05) |
| Marinoski et al. (2019) [17] | Serbia | HD(25), 54.9 ± 13.6 y (72% m, 28% f) C(25), 54.2 ± 12.7 y (64% m, 36% f) | 0.30 ± 0.16 mL/min | 0.51 ± 0.19 mL/min | Significantly lower unstimulated whole salivary flow rates in HD patients (p < 0.001) |
| Teimoori et al. (2021) [30] | Iran | HD (48), 53.5 ± 1.9 y (58% m, 42% f) C (48), 59.3 ± 1.6 y (58% m, 42% f) | 0.232 ± 0.081 mL/min | 0.339 ± 0.129 mL/min | Significantly lower unstimulated whole salivary flow rates in HD patients (p = 0.000) |
| Tomás et al. (2008) [31] | Portugal | HD (28), 64 ± 11 y (46% m, 54% f) C (64), 60 ± 11 (43% m, 57% f) | 0.39 ± 0.13 mL/min | 0.35 ± 0.14 mL/min | No significant difference in unstimulated whole salivary flow rates (p > 0.05) |
| Author, Year | Country | Groups (n), Mean Age, and Sex | SFR BHD | SFR AHD | Findings |
|---|---|---|---|---|---|
| Bots et al. (2007) [33] | The Netherlands | Pre, Post HD (94), 56.4 ± 16.7 y (68% m, 32% f) | 0.30 ± 0.23 mL/min | 0.41 ± 0.25 mL/min | Significantly higher unstimulated whole salivary flow rates post-dialysis (p < 0.05) |
| Khanum et al. (2017) [34] | India | Pre, Post HD (30), not reported | 0.46 ± 0.27 mL/min | 0.84 ± 0.34 mL/min | Significantly higher unstimulated whole salivary flow rates post-dialysis (p = 0.000) |
| Yu et al. (2021) [36] | Taiwan | Pre, Post HD (100), 62.45 ± 11.09 y (66% m, 34% f) | 0.15 ± 0.10 mL/min | 0.19 ± 0.12 mL/min | Significantly higher unstimulated whole salivary flow rates post-dialysis (p = 0.006) |
| Author, Year | Country | Groups (n), Mean Age, and Sex | SFR HD | SFR C | Findings |
|---|---|---|---|---|---|
| Postorino et al. (2003) [28] | Italy | HD(63), 50.2 ± 13.8 y (60% m, 40% f) C (23), 46.0 ± 13.2 y (66% m, 34% f) | 1.65 ± 0.66 mL/min | 2.05 ± 0.66 mL/min | Significantly lower stimulated whole salivary flow rates in HD patients (p < 0.02) |
| Bayraktar et al. (2009) [23] | Turkey | HD (100), 46 ± 14 y (56% m, 44% f) C (111), 45 ± 18 y (41% m, 59% f) | 0.70 ± 0.32 mL/min | 1.64 ± 0.45 mL/min | Significantly lower stimulated whole salivary flow rates in HD patients (p < 0.001) |
| Gavaldá et al. (1999) [24] | Spain | HD (105), 58.9 ± 14.9 y (50% m, 50% f) C(53), 55.7 ± 10.7 y (55% m, 45% f) | 0.76 ± 0.38 mL/min | 1.26 ± 0.76 mL/min | Significantly lower stimulated whole salivary flow rates in HD patients (p < 0.001) |
| Kaushik et al. (2013) [25] | India | HD (25), 44.42 ± 7.53 y (61% m, 39% f) C (25), not reported | 0.66 ± 0.02 mL/min | 1.16 ± 0.11 mL/min | Significantly lower stimulated whole salivary flow rates in HD patients (p < 0.001) |
| Limeres et al. (2016) [18] | Portugal | HD (44), 69.8 ± 10.0 y (41% m, 59% f) C (44), 69.3 ± 10.0 y (sex not reported) | 1.09 ± 0.90 mL/min | 1.87 ± 0.78 mL/min | Significantly lower stimulated whole salivary flow rates in HD patients (p < 0.001) |
| Martins et al. (2006) [27] | Brazil | HD (15), 47.4 ± 9.73 y (67% m, 33% f) C (15), 39.8 ± 9.97 y (27% m, 73% f) | 0.81 ± 0.31 mL/min | 1.12 ± 0.29 mL/min | Significantly lower stimulated whole salivary flow rates in HD patients (p < 0.05) |
| Shetty et al. (2018) [29] | India | HD [dd 0.5–2 y] (20), not reported C (20), not reported | 0.99 ± 0.32 mL/min | 1.84 ± 0.34 mL/min | Significantly lower stimulated whole salivary flow rates in all subgroups of HD patients (p < 0.001) compared to controls. SFR HD [>2 y] < HD [0.5–2 y] (p < 0.05) |
| HD [dd > 2 y] (20), not reported C (20), not reported | 0.726 ± 0.264 mL/min | 1.84 ± 0.34 mL/min | |||
| Trzcionka et al. (2021) [32] | Poland | HD [R] (42), 67.21 y (60% m, 40% f) C (48), 52.71 y (31% m, 69% f) | 0.55 ± 0.38 mL/min | 1.55 ± 0.86 mL/min | Significantly lower stimulated whole salivary flow rates in all subgroups of HD patients (p < 0.001). |
| HD [R + H] (79), 62.54 y (61% m, 40% f) C (48), 52.71 y (31% m, 69% f) | 0.63 ± 0.55 mL/min | 1.55 ± 0.86 mL/min | |||
| HD [R + D] (16), 70.16 y (63% m, 37% f) C (48), 52.71 y (31% m, 69% f) | 0.40 ± 0.31 mL/min | 1.55 ± 0.86 mL/min | |||
| HD [R + D + H] (43), 72.86 y (63% m, 37% f) C (48), 52.71 y (31% m, 69% f) | 0.55 ± 0.55 mL/min | 1.55 ± 0.86 mL/min | |||
| Bayraktar et al. (2004) [22] | Turkey | HD (72), 45.05 ± 14.15 y (53% m, 47% f) C (50), 43.92 ± 18.80 y (48% m, 52% f) | 0.69 ± 0.31 mL/min | 1.64 ± 0.44 mL/min | Significantly lower stimulated whole salivary flow rates in HD patients (p < 0.001) |
| Author, Year | Country | Groups (n), Mean Age, and Sex | SFR HD | SFR C | Findings |
|---|---|---|---|---|---|
| Bots et al. (2007) [33] | The Netherlands | Pre, Post HD (94), 56.4 ± 16.7 y (68% m, 32% f) | 1.05 ± 0.71 mL/min | 1.23 ± 0.74 mL/min | Significantly higher stimulated whole salivary flow rates post-dialysis (p < 0.05) |
| Marques et al. (2015) [35] | Brazil | Pre, Post HD (128), 56.2 ± 17.1 y (52% m, 48% f) | 0.39 ± 0.28 mL/min | 0.60 ± 0.34 mL/min | Significantly higher stimulated whole salivary flow rates post-dialysis (p < 0.001) |
| Martins et al. (2006) [27] | Brazil | Pre, Post HD (15), 47.4 ± 9.73 y (67% m, 33% f) | 0.81 ± 0.31 mL/min | 1.18 ± 0.54 mL/min | Significantly higher stimulated whole salivary flow rates post-dialysis (p < 0.05) |
| Postorino et al. (2003) [28] | Italy | Pre, Post HD (6), not reported | 1.35 ± 0.36 mL/min | 1.39 ± 0.48 mL/min | No significant difference in stimulated whole salivary flow rates before or after dialysis |
| Trzcionka et al. (2021) [32] | Poland | Pre, Post HD [R] (42), 67.21 y (60% m, 40% f) | 0.55 ± 0.38 mL/min | 0.72 ± 0.55 mL/min | Significantly higher stimulated whole salivary flow rates post-dialysis in all subgroups of HD patients (p < 0.001). |
| Pre, Post HD [R + H] (79), 62.54 y (61% m, 40% f) | 0.63 ± 0.55 mL/min | 0.68 ± 0.59 mL/min | |||
| Pre, Post HD [R + D] (16), 70.16 y (63% m, 37% f) | 0.40 ± 0.31 mL/min | 0.80 ± 0.64 mL/min | |||
| Pre, Post HD [R + D + H] (43), 72.86 y (63% m, 37% f) | 0.55 ± 0.55 mL/min | 0.86 ± 1.00 mL/min |
| Author, Year | Country | Groups (n), Mean Age, and Sex | SFR HD | SFR C | Findings |
|---|---|---|---|---|---|
| Gavaldá et al. (1999) [24] | Spain | HD (105), 58.9 ± 14.9 y (50% m, 50% f) C (53), 55.7 ± 10.7 y (55% m, 45% f) | 0.4 ± 0.6 mL/min | 0.14 ± 0.16 mL/min | Significantly lower stimulated parotid salivary flow rates in HD patients (p = 0.01). |
| Kho et al. (1999) [26] | Republic of Korea | HD (22), 34.7 ± 10.8 y (64% m, 36% f) C (22), 30.5 ± 7.9 y (sex not reported) | 0.67 ± 0.34 mL/min | 1.11 ± 0.43 mL/min | Significantly lower stimulated parotid salivary flow rates in HD patients (p < 0.001). |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mohammadi, P.; Bots, C.P.; Brand, H.S. Salivary Flow Rate in Patients with Kidney Failure on Hemodialysis—A Systematic Review and Meta-Analysis. J. Clin. Med. 2025, 14, 6108. https://doi.org/10.3390/jcm14176108
Mohammadi P, Bots CP, Brand HS. Salivary Flow Rate in Patients with Kidney Failure on Hemodialysis—A Systematic Review and Meta-Analysis. Journal of Clinical Medicine. 2025; 14(17):6108. https://doi.org/10.3390/jcm14176108
Chicago/Turabian StyleMohammadi, Parinaz, Casper P. Bots, and Henk S. Brand. 2025. "Salivary Flow Rate in Patients with Kidney Failure on Hemodialysis—A Systematic Review and Meta-Analysis" Journal of Clinical Medicine 14, no. 17: 6108. https://doi.org/10.3390/jcm14176108
APA StyleMohammadi, P., Bots, C. P., & Brand, H. S. (2025). Salivary Flow Rate in Patients with Kidney Failure on Hemodialysis—A Systematic Review and Meta-Analysis. Journal of Clinical Medicine, 14(17), 6108. https://doi.org/10.3390/jcm14176108











