Carbon-Ion Radiotherapy for Hepatocellular Carcinoma: Current Status and Future Prospects: A Narrative Review
Abstract
1. Introduction
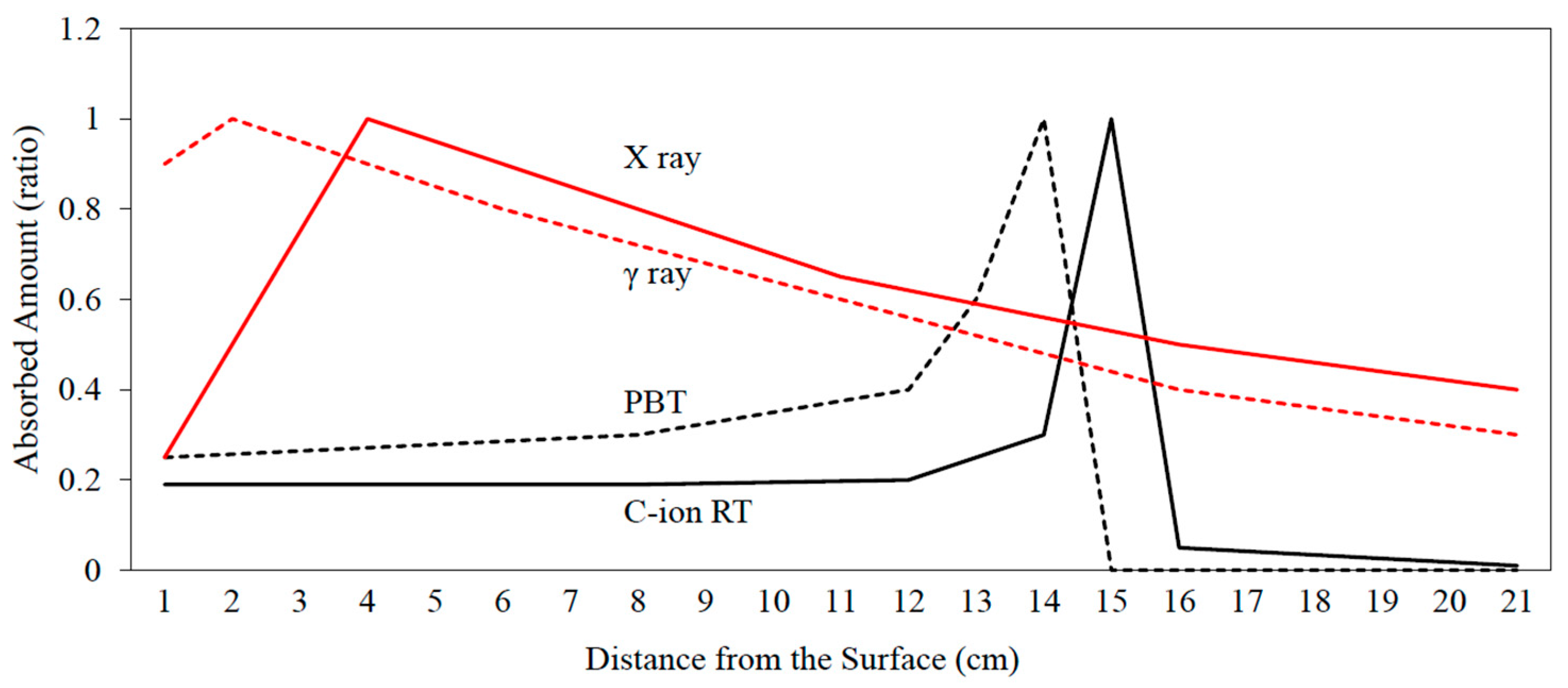
2. Carbon-Ion Radiotherapy (C-Ion RT)
3. Radiation-Induced Liver Disease (RILD) in Liver Radiotherapy for Hepatocellular Carcinoma (HCC)
4. Clinical Outcomes of Carbon-Ion Radiotherapy (C-Ion RT) for Hepatocellular Carcinoma (HCC)
5. Comparison of Carbon-Ion Radiotherapy (C-Ion RT)and Other Therapies for Hepatocellular Carcinoma (HCC)
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- London, W.T.; Petrick, J.L.; McGlynn, K.A. Liver Cancer. In Cancer Epidemiology and Prevention, 4th ed.; Thun, M.J., Linet, M.S., Cerhan, J.R., Haiman, C.A., Schottenfeld, D., Eds.; Oxford University Press: New York, NY, USA, 2018; pp. 635–660. [Google Scholar]
- Schneider, C.V.; Schneider, K.M.; Raptis, A.; Huang, H.; Trautwein, C.; Loomba, R. Prevalence of at-risk MASH, MetALD and alcohol-associated steatotic liver disease in the general population. Aliment. Pharmacol. Ther. 2024, 59, 1271–1281. [Google Scholar] [CrossRef] [PubMed]
- Petrick, J.L.; Florio, A.A.; Znaor, A.; Ruggieri, D.; Laversanne, M.; Alvarez, C.S.; Ferlay, J.; Valery, P.C.; Bray, F.; McGlynn, K.A. International trends in hepatocellular carcinoma incidence, 1978–2012. Int. J. Cancer 2020, 147, 317–330. [Google Scholar] [CrossRef] [PubMed]
- Sarveazad, A.; Agah, S.; Babahajian, A.; Amini, N.; Bahardoust, M. Predictors of 5 year survival rate in hepatocellular carcinoma patients. J. Res. Med. Sci. 2019, 24, 86. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Ren, X.; Zhang, Q. Incidence, risk factors, and prognosis in patients with primary hepatocellular carcinoma and lung metastasis: A population-based study. Cancer Manag. Res. 2019, 11, 2759–2768. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Gu, X.; Thrift, A.P. Factors associated with favorable survival outcomes for Asians with hepatocellular carcinoma: A sequential matching cohort study. PLoS ONE 2019, 14, e0214721. [Google Scholar] [CrossRef] [PubMed]
- Singal, A.G.; Llovet, J.M.; Yarchoan, M.; Mehta, N.; Heimbach, J.K.; Dawson, L.A.; Jou, J.H.; Kulik, L.M.; Agopian, V.G.; Marrero, J.A.; et al. AASLD Practice Guidance on prevention, diagnosis, and treatment of hepatocellular carcinoma. Hepatology 2023, 78, 1922–1965. [Google Scholar] [CrossRef] [PubMed]
- European Association for the Study of the Liver. EASL Clinical Practice Guidelines: Management of hepatocellular carcinoma. J. Hepatol. 2018, 69, 182–236. [Google Scholar] [CrossRef] [PubMed]
- Omata, M.; Cheng, A.L.; Kokudo, N.; Kudo, M.; Lee, J.M.; Jia, J.; Tateishi, R.; Han, K.H.; Chawla, Y.K.; Shiina, S.; et al. Asia-Pacific clinical practice guidelines on the management of hepatocellular carcinoma: A 2017 update. Hepatol. Int. 2017, 11, 317–370. [Google Scholar] [CrossRef] [PubMed]
- Hasegawa, K.; Takemura, N.; Yamashita, T.; Watadani, T.; Kaibori, M.; Kubo, S.; Shimada, M.; Nagano, H.; Hatano, E.; Aikata, H.; et al. Clinical Practice Guidelines for Hepatocellular Carcinoma: The Japan Society of Hepatology 2021 version (5th JSH-HCC Guidelines). Hepatol. Res. 2023, 53, 383–390. [Google Scholar] [CrossRef] [PubMed]
- Nagasue, N.; Uchida, M.; Makino, Y.; Takemoto, Y.; Yamanoi, A.; Hayashi, T.; Chang, Y.C.; Kohno, H.; Nakamura, T.; Yukaya, H. Incidence and factors associated with intrahepatic recurrence following resection of hepatocellular carcinoma. Gastroenterology 1993, 105, 488–494. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.; Zhu, W.; Zhang, J.; Fan, C.; Zhang, J. Cancer-associated fibroblasts in hepatocellular carcinoma: Origins, heterogeneity, and therapeutic implications. Front. Immunol. 2025, 16, 1620075. [Google Scholar] [CrossRef] [PubMed]
- Manfredi, G.F.; Fulgenzi, C.A.M.; Celsa, C.; Stefanini, B.; D’Alessio, A.; Pinter, M.; Scheiner, B.; Awosika, N.; Brunetti, L.; Lombardi, P.; et al. Efficacy of atezolizumab plus bevacizumab for unresectable HCC: Systematic review and meta-analysis of real-world evidence. JHEP Rep. 2025, 7, 101431. [Google Scholar] [CrossRef] [PubMed]
- Lau, G.; Sangro, B.; Cheng, A.L.; Kudo, M.; Kelley, R.K.; Tak, W.Y.; Gasbarrini, A.; Reig, M.; Lim, H.Y.; Tougeron, D.; et al. Immune-mediated adverse events and overall survival with tremelimumab plus durvalumab and durvalumab monotherapy in unresectable HCC: HIMALAYA phase III randomized clinical trial. Hepatology, 2025; Online ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Nguyen-Khac, E.; Merle, P.; Ben Khadhra, H.; Giuliana, A.; Decaens, T.; Uguen, T.; Blanc, J.F.; Ganne-Carrie, N.; Bouattour, M.; Cattan, S.; et al. Real-life data on hepatocellular carcinoma in the French prospective CHIEF cohort in the era of immunotherapy. Eur. J. Gastroenterol. Hepatol. 2025, 37, 1063–1077. [Google Scholar] [CrossRef] [PubMed]
- Caillet, V.; Booth, J.T.; Keall, P. IGRT and motion management during lung SBRT delivery. Phys. Med. 2017, 44, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Seo, Y.S.; Kim, M.S.; Yoo, H.J.; Jang, W.I.; Paik, E.K.; Han, C.J.; Lee, B.H. Radiofrequency ablation versus stereotactic body radiotherapy for small hepatocellular carcinoma: A Markov model-based analysis. Cancer Med. 2016, 5, 3094–3101. [Google Scholar] [CrossRef] [PubMed]
- Collings, E.W.; Lu, L.; Gupta, N.; Sumption, M.D. Accelerators, Gantries, Magnets and Imaging Systems for Particle Beam Therapy: Recent Status and Prospects for Improvement. Front. Oncol. 2022, 11, 737837. [Google Scholar] [CrossRef] [PubMed]
- Wilson, R.R. Radiological use of fast protons. Radiology 1946, 47, 487–491. [Google Scholar] [CrossRef] [PubMed]
- University of Tsukuba. Proton Medical Research Center. Available online: https://www.pmrc.tsukuba.ac.jp/research/en/outline (accessed on 28 May 2025).
- Schaub, L.; Harrabi, S.B.; Debus, J. Particle therapy in the future of precision therapy. Br. J. Radiol. 2020, 93, 20200183. [Google Scholar] [CrossRef] [PubMed]
- Gungor Price, G.M.; Sarigul, N. The effect of voxelization in Monte Carlo simulation to validate Bragg peak characteristics for a pencil proton beam. Rep. Pract. Oncol. Radiother. 2023, 28, 102–113. [Google Scholar] [CrossRef] [PubMed]
- Kirkby, K.J.; Kirkby, N.F.; Burnet, N.G.; Owen, H.; Mackay, R.I.; Crellin, A.; Green, S. Heavy charged particle beam therapy and related new radiotherapy technologies: The clinical potential, physics and technical developments required to deliver benefit for patients with cancer. Br. J. Radiol. 2020, 93, 20200247. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Li, X.; Yang, J.; Wang, S.; Tang, M.; Xia, J.; Gao, Y. Flourish of Proton and Carbon Ion Radiotherapy in China. Front. Oncol. 2022, 12, 819905. [Google Scholar] [CrossRef] [PubMed]
- Kanemoto, A.; Hirayama, R.; Moritake, T.; Furusawa, Y.; Sun, L.; Sakae, T.; Kuno, A.; Terunuma, T.; Yasuoka, K.; Mori, Y.; et al. RBE and OER within the spread-out Bragg peak for proton beam therapy: In vitro study at the Proton Medical Research Center at the University of Tsukuba. J. Radiat. Res. 2014, 55, 1028–1032. [Google Scholar] [CrossRef] [PubMed]
- Ebrahimi Loushab, M.; Mowlavi, A.A.; Hadizadeh, M.H.; Izadi, R.; Jia, S.B. Impact of Various Beam Parameters on Lateral Scattering in Proton and Carbon-ion Therapy. J. Biomed. Phys. Eng. 2015, 5, 169–176. [Google Scholar] [PubMed]
- Paganetti, H. Relative biological effectiveness (RBE) values for proton beam therapy. Variations as a function of biological endpoint, dose, and linear energy transfer. Phys. Med. Biol. 2014, 59, R419-72. [Google Scholar] [CrossRef] [PubMed]
- Byun, H.K.; Kim, C.; Seong, J. Carbon Ion Radiotherapy in the Treatment of Hepatocellular Carcinoma. Clin. Mol. Hepatol. 2023, 29, 945–957. [Google Scholar] [CrossRef] [PubMed]
- Schulz-Ertner, D.; Jäkel, O.; Schlegel, W. Radiation therapy with charged particles. Semin. Radiat. Oncol. 2006, 16, 249–259. [Google Scholar] [CrossRef] [PubMed]
- Ward, J.F. The complexity of DNA damage: Relevance to biological consequences. Int. J. Radiat. Biol. 1994, 66, 427–432. [Google Scholar] [CrossRef] [PubMed]
- Sage, E.; Harrison, L. Clustered DNA lesion repair in eukaryotes: Relevance to mutagenesis and cell survival. Mutat. Res. 2011, 711, 123–133. [Google Scholar] [CrossRef] [PubMed]
- Souli, M.P.; Nikitaki, Z.; Puchalska, M.; Brabcová, K.P.; Spyratou, E.; Kote, P.; Efstathopoulos, E.P.; Hada, M.; Georgakilas, A.G.; Sihver, L. Clustered DNA Damage Patterns after Proton Therapy Beam Irradiation Using Plasmid DNA. Int. J. Mol. Sci. 2022, 23, 15606. [Google Scholar] [CrossRef] [PubMed]
- Fontana, A.O.; Augsburger, M.A.; Grosse, N.; Guckenberger, M.; Lomax, A.J.; Sartori, A.A.; Pruschy, M.N. Differential DNA repair pathway choice in cancer cells after proton- and photon-irradiation. Radiother. Oncol. 2015, 116, 374–380. [Google Scholar] [CrossRef] [PubMed]
- Grosse, N.; Fontana, A.O.; Hug, E.B.; Lomax, A.; Coray, A.; Augsburger, M.; Paganetti, H.; Sartori, A.A.; Pruschy, M. Deficiency in homologous recombination renders Mammalian cells more sensitive to proton versus photon irradiation. Int. J. Radiat. Oncol. Biol. Phys. 2014, 88, 175–181. [Google Scholar] [CrossRef] [PubMed]
- Jeggo, P.A.; Geuting, V.; Löbrich, M. The role of homologous recombination in radiation-induced double-strand break repair. Radiother. Oncol. 2011, 101, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Quintiliani, M. The oxygen effect in radiation inactivation of DNA and enzymes. Int. J. Radiat. Biol. Relat. Stud. Phys. Chem. Med. 1986, 50, 573–594. [Google Scholar] [CrossRef] [PubMed]
- Klein, C.; Dokic, I.; Mairani, A.; Mein, S.; Brons, S.; Häring, P.; Haberer, T.; Jäkel, O.; Zimmermann, A.; Zenke, F.; et al. Overcoming hypoxia-induced tumor radioresistance in non-small cell lung cancer by targeting DNA-dependent protein kinase in combination with carbon ion irradiation. Radiat. Oncol. 2017, 12, 208. [Google Scholar] [CrossRef] [PubMed]
- Narayanan, D.; Ma, S.; Özcelik, D. Targeting the Redox Landscape in Cancer Therapy. Cancers 2020, 12, 1706. [Google Scholar] [CrossRef] [PubMed]
- Tinganelli, W.; Ma, N.Y.; Von Neubeck, C.; Maier, A.; Schicker, C.; Kraft-Weyrather, W.; Durante, M. Influence of acute hypoxia and radiation quality on cell survival. J. Radiat. Res. 2013, 54 (Suppl. 1), i23–i30. [Google Scholar] [CrossRef] [PubMed]
- Fukawa, T.; Takematsu, K.; Oka, K.; Koike, S.; Ando, K.; Kobayashi, H.; Tanishita, K. Differences in pO2 peaks of a murine fibrosarcoma between carbon-ion and X-ray irradiation. J. Radiat. Res. 2004, 45, 303–308. [Google Scholar] [CrossRef] [PubMed]
- Nakano, T.; Suzuki, Y.; Ohno, T.; Kato, S.; Suzuki, M.; Morita, S.; Sato, S.; Oka, K.; Tsujii, H. Carbon beam therapy overcomes the radiation resistance of uterine cervical cancer originating from hypoxia. Clin. Cancer Res. 2006, 12, 2185–2190. [Google Scholar] [CrossRef] [PubMed]
- Shiba, S.; Abe, T.; Shibuya, K.; Katoh, H.; Koyama, Y.; Shimada, H.; Kakizaki, S.; Shirabe, K.; Kuwano, H.; Ohno, T.; et al. Carbon ion radiotherapy for 80 years or older patients with hepatocellular carcinoma. BMC Cancer 2017, 17, 721. [Google Scholar] [CrossRef] [PubMed]
- Park, H.C.; Seong, J.; Han, K.H.; Chon, C.Y.; Moon, Y.M.; Suh, C.O. Dose-response relationship in local radiotherapy for hepatocellular carcinoma. Int. J. Radiat. Oncol. Biol. Phys. 2002, 54, 150–155. [Google Scholar] [CrossRef] [PubMed]
- Ingold, J.A.; Reed, G.B.; Kaplan, H.S.; Bagshaw, M.A. Radiation Hepatitis. Am. J. Roentgenol. Radium. Ther. Nucl. Med. 1965, 93, 200–208. [Google Scholar] [PubMed]
- Lawrence, T.S.; Ten Haken, R.K.; Kessler, M.L.; Robertson, J.M.; Lyman, J.T.; Lavigne, M.L.; Brown, M.B.; DuRoss, D.J.; Andrews, J.C.; Ensminger, W.D.; et al. The use of 3-D dose volume analysis to predict radiation hepatitis. Int. J. Radiat. Oncol. Biol. Phys. 1992, 23, 781–788. [Google Scholar] [CrossRef] [PubMed]
- Liang, S.X.; Zhu, X.D.; Xu, Z.Y.; Zhu, J.; Zhao, J.D.; Lu, H.J.; Yang, Y.L.; Chen, L.; Wang, A.Y.; Fu, X.L.; et al. Radiation-induced liver disease in three-dimensional conformal radiation therapy for primary liver carcinoma: The risk factors and hepatic radiation tolerance. Int. J. Radiat. Oncol. Biol. Phys. 2006, 65, 426–434. [Google Scholar] [CrossRef] [PubMed]
- Fajardo, L.F.; Colby, T.V. Pathogenesis of veno-occlusive liver disease after radiation. Arch. Pathol. Lab. Med. 1980, 104, 584–588. [Google Scholar] [PubMed]
- Pan, C.C.; Kavanagh, B.D.; Dawson, L.A.; Li, X.A.; Das, S.K.; Miften, M.; Ten Haken, R.K. Radiation-associated liver injury. Int. J. Radiat. Oncol. Biol. Phys. 2010, 76, S94–S100. [Google Scholar] [CrossRef] [PubMed]
- Dawson, L.A.; Normolle, D.; Balter, J.M.; McGinn, C.J.; Lawrence, T.S.; Ten Haken, R.K. Analysis of radiation-induced liver disease using the Lyman NTCP model. Int. J. Radiat. Oncol. Biol. Phys. 2002, 53, 810–821. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.C.; Wu, J.K.; Huang, C.M.; Liu, H.S.; Huang, D.Y.; Cheng, S.H.; Tsai, S.Y.; Jian, J.J.; Lin, Y.M.; Cheng, T.I.; et al. Radiation-induced liver disease after three-dimensional conformal radiotherapy for patients with hepatocellular carcinoma: Dosimetric analysis and implication. Int. J. Radiat. Oncol. Biol. Phys. 2002, 54, 156–162. [Google Scholar] [CrossRef] [PubMed]
- Bujold, A.; Massey, C.A.; Kim, J.J.; Brierley, J.; Cho, C.; Wong, R.K.; Dinniwell, R.E.; Kassam, Z.; Ringash, J.; Cummings, B.; et al. Sequential phase I and II trials of stereotactic body radiotherapy for locally advanced hepatocellular carcinoma. J. Clin. Oncol. 2013, 31, 1631–1639. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Wang, Z.; Sheng, Y.; Ming, X.; Jiang, G.L.; Wang, W. Indications of IMRT, PRT and CIRT for HCC from comparisons of dosimetry and normal tissue complication possibility. Strahlenther. Onkol. 2022, 198, 361–369. [Google Scholar] [CrossRef] [PubMed]
- Kato, H.; Tsujii, H.; Miyamoto, T.; Mizoe, J.E.; Kamada, T.; Tsuji, H.; Yamada, S.; Kandatsu, S.; Yoshikawa, K.; Obata, T.; et al. Results of the first prospective study of carbon ion radiotherapy for hepatocellular carcinoma with liver cirrhosis. Int. J. Radiat. Oncol. Biol. Phys. 2004, 59, 1468–1476. [Google Scholar] [CrossRef] [PubMed]
- Kasuya, G.; Kato, H.; Yasuda, S.; Tsuji, H.; Yamada, S.; Haruyama, Y.; Kobashi, G.; Ebner, D.K.; Okada, N.N.; Makishima, H.; et al. Progressive hypofractionated carbon-ion radiotherapy for hepatocellular carcinoma: Combined analyses of 2 prospective trials. Cancer 2017, 123, 3955–3965. [Google Scholar] [CrossRef] [PubMed]
- Shiba, S.; Shibuya, K.; Katoh, H.; Koyama, Y.; Okamoto, M.; Abe, T.; Ohno, T.; Nakano, T. No Deterioration in Clinical Outcomes of Carbon Ion Radiotherapy for Sarcopenia Patients with Hepatocellular Carcinoma. Anticancer Res. 2018, 38, 3579–3586. [Google Scholar] [CrossRef] [PubMed]
- Shibuya, K.; Ohno, T.; Katoh, H.; Okamoto, M.; Shiba, S.; Koyama, Y.; Kakizaki, S.; Shirabe, K.; Nakano, T. A feasibility study of high-dose hypofractionated carbon ion radiation therapy using four fractions for localized hepatocellular carcinoma measuring 3 cm or larger. Radiother. Oncol. 2019, 132, 230–235. [Google Scholar] [CrossRef] [PubMed]
- Yasuda, S.; Kato, H.; Imada, H.; Isozaki, Y.; Kasuya, G.; Makishima, H.; Tsuji, H.; Ebner, D.K.; Yamada, S.; Kamada, T.; et al. Long-Term Results of High-Dose 2-Fraction Carbon Ion Radiation Therapy for Hepatocellular Carcinoma. Adv. Radiat. Oncol. 2019, 5, 196–203. [Google Scholar] [CrossRef] [PubMed]
- Shiba, S.; Shibuya, K.; Okamoto, M.; Okazaki, S.; Komatsu, S.; Kubota, Y.; Nakano, T.; Ohno, T. Clinical impact of Hypofractionated carbon ion radiotherapy on locally advanced hepatocellular carcinoma. Radiat. Oncol. 2020, 15, 195. [Google Scholar] [CrossRef] [PubMed]
- Shibuya, K.; Katoh, H.; Koyama, Y.; Shiba, S.; Okamoto, M.; Okazaki, S.; Araki, K.; Kakizaki, S.; Shirabe, K.; Ohno, T. Efficacy and Safety of 4 Fractions of Carbon-Ion Radiation Therapy for Hepatocellular Carcinoma: A Prospective Study. Liver Cancer 2021, 11, 61–74. [Google Scholar] [CrossRef] [PubMed]
- Hong, Z.; Zhang, W.; Cai, X.; Yu, Z.; Sun, J.; Wang, W.; Lin, L.; Zhao, J.; Cheng, J.; Zhang, G.; et al. Carbon ion radiotherapy with pencil beam scanning for hepatocellular carcinoma: Long-term outcomes from a phase I trial. Cancer Sci. 2023, 114, 976–983. [Google Scholar] [CrossRef] [PubMed]
- Hiroshima, Y.; Wakatsuki, M.; Kaneko, T.; Makishima, H.; Okada, N.N.; Yasuda, S.; Ishikawa, H.; Tsuji, H. Clinical impact of carbon-ion radiotherapy on hepatocellular carcinoma with Child-Pugh B cirrhosis. Cancer Med. 2023, 12, 14004–14014. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Cai, X.; Sun, J.; Wang, W.; Zhao, J.; Zhang, Q.; Jiang, G.; Wang, Z. Pencil Beam Scanning Carbon Ion Radiotherapy for Hepatocellular Carcinoma. J. Hepatocell. Carcinoma. 2023, 10, 2397–2409. [Google Scholar] [CrossRef] [PubMed]
- Kaneko, T.; Makishima, H.; Wakatsuki, M.; Hiroshima, Y.; Matsui, T.; Yasuda, S.; Okada, N.N.; Nemoto, K.; Tsuji, H.; Yamada, S.; et al. Carbon-ion radiotherapy for hepatocellular carcinoma with major vascular invasion: A retrospective cohort study. BMC Cancer 2024, 24, 383. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, K.; Suzuki, O.; Wakisaka, Y.; Ichise, K.; Uchida, H.; Anzai, M.; Hasegawa, A.; Seo, Y.; Shimizu, S.; Ishii, T.; et al. Prognostic analysis of radiation-induced liver damage following carbon-ion radiotherapy for hepatocellular carcinoma. Radiat. Oncol. 2024, 19, 51. [Google Scholar] [CrossRef] [PubMed]
- Hoegen-Saßmannshausen, P.; Naumann, P.; Hoffmeister-Wittmann, P.; Ben Harrabi, S.; Seidensaal, K.; Weykamp, F.; Mielke, T.; Ellerbrock, M.; Habermehl, D.; Springfeld, C.; et al. Carbon ion radiotherapy of hepatocellular carcinoma provides excellent local control: The prospective phase I PROMETHEUS trial. JHEP Rep. 2024, 6, 101063. [Google Scholar] [CrossRef] [PubMed]
- Lan, T.L.; Shiau, C.Y.; Wang, L.W.; Liu, Y.M.; Chen, Y.W.; Huang, P.I.; Hu, Y.W.; Lai, I.C.; Wu, Y.H.; Lai, T.Y.; et al. Feasibility and safety of Taipei Veterans General Hospital Heavy Ion Therapy Center: The first carbon-ion irradiation facility in Taiwan. J. Chin. Med. Assoc. 2025, 88, 196–204. [Google Scholar] [CrossRef] [PubMed]
- Jackson, W.C.; Tang, M.; Maurino, C.; Mendiratta-Lala, M.; Parikh, N.D.; Matuszak, M.M.; Dow, J.S.; Cao, Y.; Mayo, C.S.; Ten Haken, R.K.; et al. Individualized Adaptive Radiation Therapy Allows for Safe Treatment of Hepatocellular Carcinoma in Patients With Child-Turcotte-Pugh B Liver Disease. Int. J. Radiat. Oncol. Biol. Phys. 2021, 109, 212–219. [Google Scholar] [CrossRef] [PubMed]
- Voron, T.; Tselikas, L.; Pietrasz, D.; Pigneur, F.; Laurent, A.; Compagnon, P.; Salloum, C.; Luciani, A.; Azoulay, D. Sarcopenia Impacts on Short- and Long-term Results of Hepatectomy for Hepatocellular Carcinoma. Ann. Surg. 2015, 261, 1173–1183. [Google Scholar] [CrossRef] [PubMed]
- Harimoto, N.; Shirabe, K.; Yamashita, Y.I.; Ikegami, T.; Yoshizumi, T.; Soejima, Y.; Ikeda, T.; Maehara, Y.; Nishie, A.; Yamanaka, T. Sarcopenia as a predictor of prognosis in patients following hepatectomy for hepatocellular carcinoma. Br. J. Surg. 2013, 100, 1523–1530. [Google Scholar] [CrossRef] [PubMed]
- Bruix, J.; Raoul, J.L.; Sherman, M.; Mazzaferro, V.; Bolondi, L.; Craxi, A.; Galle, P.R.; Santoro, A.; Beaugrand, M.; Sangiovanni, A.; et al. Efficacy and safety of sorafenib in patients with advanced hepatocellular carcinoma: Subanalyses of a phase III trial. J. Hepatol. 2012, 57, 821–829. [Google Scholar] [CrossRef] [PubMed]
- Yoon, S.M.; Ryoo, B.Y.; Lee, S.J.; Kim, J.H.; Shin, J.H.; An, J.H.; Lee, H.C.; Lim, Y.S. Efficacy and Safety of Transarterial Chemoembolization Plus External Beam Radiotherapy vs Sorafenib in Hepatocellular Carcinoma With Macroscopic Vascular Invasion: A Randomized Clinical Trial. JAMA Oncol. 2018, 4, 661–669. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; Lai, E.C.; Li, N.; Guo, W.X.; Xue, J.; Lau, W.Y.; Wu, M.C.; Cheng, S.Q. Surgical treatment of hepatocellular carcinoma with portal vein tumor thrombus. Ann. Surg. Oncol. 2010, 17, 2073–2080. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.H.; Lee, Y.H.; Hsia, C.Y.; Hsu, C.Y.; Huang, Y.H.; Chiou, Y.Y.; Lin, H.C.; Huo, T.I. Surgical resection versus transarterial chemoembolization for hepatocellular carcinoma with portal vein tumor thrombosis: A propensity score analysis. Ann. Surg. Oncol. 2014, 21, 1825–1833. [Google Scholar] [CrossRef] [PubMed]
- Takeda, A.; Sanuki, N.; Tsurugai, Y.; Iwabuchi, S.; Matsunaga, K.; Ebinuma, H.; Imajo, K.; Aoki, Y.; Saito, H.; Kunieda, E. Phase 2 study of stereotactic body radiotherapy and optional transarterial chemoembolization for solitary hepatocellular carcinoma not amenable to resection and radiofrequency ablation. Cancer 2016, 122, 2041–2049. [Google Scholar] [CrossRef] [PubMed]
- Chiba, T.; Tokuuye, K.; Matsuzaki, Y.; Sugahara, S.; Chuganji, Y.; Kagei, K.; Shoda, J.; Hata, M.; Abei, M.; Igaki, H.; et al. Proton beam therapy for hepatocellular carcinoma: A retrospective review of 162 patients. Clin. Cancer Res. 2005, 11, 3799–3805. [Google Scholar] [CrossRef] [PubMed]
- Leksell, L. The stereotaxic method and radiosurgery of the brain. Acta Chir. Scand. 1951, 102, 316–319. [Google Scholar] [PubMed]
- Leksell, L. Stereotactic radiosurgery. J. Neurol. Neurosurg. Psychiatry 1983, 46, 797–803. [Google Scholar] [CrossRef] [PubMed]
- Pow, E.H.; Kwong, D.L.; McMillan, A.S.; Wong, M.C.; Sham, J.S.; Leung, L.H.; Leung, W.K. Xerostomia and quality of life after intensity-modulated radiotherapy vs. conventional radiotherapy for early-stage nasopharyngeal carcinoma: Initial report on a randomized controlled clinical trial. Int. J. Radiat. Oncol. Biol. Phys. 2006, 66, 981–991. [Google Scholar] [CrossRef] [PubMed]
- Vedam, S.; Archambault, L.; Starkschall, G.; Mohan, R.; Beddar, S. Determination of prospective displacement-based gate threshold for respiratory-gated radiation delivery from retrospective phase-based gate threshold selected at 4D CT simulation. Med. Phys. 2007, 34, 4247–4255. [Google Scholar] [CrossRef] [PubMed]
- Santoro, J.P.; Yorke, E.; Goodman, K.A.; Mageras, G.S. From phase-based to displacement-based gating: A software tool to facilitate respiration-gated radiation treatment. J. Appl. Clin. Med. Phys. 2009, 10, 132–141. [Google Scholar] [CrossRef] [PubMed]
- Abe, T.; Saitoh, J.; Kobayashi, D.; Shibuya, K.; Koyama, Y.; Shimada, H.; Shirai, K.; Ohno, T.; Nakano, T. Dosimetric comparison of carbon ion radiotherapy and stereotactic body radiotherapy with photon beams for the treatment of hepatocellular carcinoma. Radiat. Oncol. 2015, 10, 187. [Google Scholar] [CrossRef] [PubMed]
- Shiba, S.; Shibuya, K.; Kawashima, M.; Okano, N.; Kaminuma, T.; Okamoto, M.; Kubota, Y.; Nakano, T.; Ohno, T. Comparison of Dose Distributions When Using Carbon Ion Radiotherapy Versus Intensity-modulated Radiotherapy for Hepatocellular Carcinoma With Macroscopic Vascular Invasion: A Retrospective Analysis. Anticancer Res. 2020, 40, 459–464. [Google Scholar] [CrossRef] [PubMed]
- Ronot, M.; Loffroy, R.; Arnold, D.; Greget, M.; Sengel, C.; Pinaquy, J.B.; Pellerin, O.; Maleux, G.; Peynircioglu, B.; Pelage, J.P.; et al. Transarterial Radioembolisation with Y90 Resin Microspheres and the Effect of Reimbursement Criteria in France: Final Results of the CIRT-FR Prospective Observational Study. Cardiovasc. Intervent. Radiol. 2025, 48, 205–220. [Google Scholar] [CrossRef] [PubMed]
- Bonù, M.L.; Nicosia, L.; Turkaj, A.; Pastorello, E.; Vitali, P.; Frassine, F.; Toraci, C.; Spiazzi, L.; Lechiara, M.; Frittoli, B.; et al. High dose proton and photon-based radiation therapy for 213 liver lesions: A multi-institutional dosimetric comparison with a clinical perspective. Radiol. Med. 2024, 129, 497–506. [Google Scholar] [CrossRef] [PubMed]
- Kolligs, F.; Arnold, D.; Golfieri, R.; Pech, M.; Peynircioglu, B.; Pfammatter, T.; Ronot, M.; Sangro, B.; Schaefer, N.; Maleux, G.; et al. Factors impacting survival after transarterial radioembolization in patients with hepatocellular carcinoma: Results from the prospective CIRT study. JHEP Rep. 2022, 5, 100633. [Google Scholar] [CrossRef] [PubMed]
- Thonglert, K.; Greer, M.D.; Schaub, S.K.; Bowen, S.R.; Menghini, A.M.; Nyflot, M.J.; Grassberger, C.; Tsai, J.; Zaki, P.; Kim, E.Y.; et al. Simultaneous integrated boost and protection proton beam therapy approach for hepatocellular carcinoma. Clin. Transl. Radiat. Oncol. 2025, 54, 101008. [Google Scholar] [CrossRef] [PubMed]
- Chok, K.S.H.; Joeng, T.Y.T.; Poon, D.M.C. Proton beam therapy in the management of hepatocellular carcinoma. Expert Rev. Gastroenterol. Hepatol. 2025, 19, 495–504. [Google Scholar] [CrossRef] [PubMed]
- Yang, A.H.; Urrunaga, N.H.; Siddiqui, O.; Wu, A.; Schliep, M.; Mossahebi, S.; Shetty, K.; Regine, W.F.; Molitoris, J.K.; Lominadze, Z. Proton beam stereotactic body radiotherapy and hypofractionated therapy with pencil beam scanning is safe and effective for advanced hepatocellular carcinoma and intrahepatic cholangiocarcinoma: A single center experience. J. Radiosurg. SBRT 2023, 9, 43–52. [Google Scholar] [PubMed]
- Bush, D.A.; Volk, M.; Smith, J.C.; Reeves, M.E.; Sanghvi, S.; Slater, J.D.; deVera, M. Proton beam radiotherapy versus transarterial chemoembolization for hepatocellular carcinoma: Results of a randomized clinical trial. Cancer 2023, 129, 3554–3563. [Google Scholar] [CrossRef] [PubMed]
- Abousaida, B.; Seneviratne, D.; Hoppe, B.S.; Ko, S.J.; Asaithamby, A.; Cucinotta, F.A.; Kirwan, J.M.; Mody, K.; Toskich, B.; Ashman, J.B.; et al. Carbon Ion Radiotherapy in the Management of Hepatocellular Carcinoma. J. Hepatocell. Carcinoma 2021, 8, 1169–1179. [Google Scholar] [CrossRef] [PubMed]
- Fujita, N.; Kanogawa, N.; Makishima, H.; Ogasawara, S.; Maruta, S.; Iino, Y.; Shiko, Y.; Kanzaki, H.; Koroki, K.; Kobayashi, K.; et al. Carbon-ion radiotherapy versus radiofrequency ablation as initial treatment for early-stage hepatocellular carcinoma. Hepatol. Res. 2022, 52, 1060–1071. [Google Scholar] [CrossRef] [PubMed]
- Shiba, S.; Shibuya, K.; Katoh, H.; Kaminuma, T.; Miyazaki, M.; Kakizaki, S.; Shirabe, K.; Ohno, T.; Nakano, T. A comparison of carbon ion radiotherapy and transarterial chemoembolization treatment outcomes for single hepatocellular carcinoma: A propensity score matching study. Radiat. Oncol. 2019, 14, 137. [Google Scholar] [CrossRef] [PubMed]
- Okazaki, S.; Shibuya, K.; Shiba, S.; Takura, T.; Ohno, T. Cost-Effectiveness Comparison of Carbon-Ion Radiation Therapy and Transarterial Chemoembolization for Hepatocellular Carcinoma. Adv. Radiat. Oncol. 2024, 9, 101441. [Google Scholar] [CrossRef] [PubMed]
- Terzi, E.; Piscaglia, F.; Forlani, L.; Mosconi, C.; Renzulli, M.; Bolondi, L.; Golfieri, R.; BLOG-Bologna Liver Oncology Group, S. Orsola-Malpighi Hospital, University of Bologna, Bologna, Italy. TACE performed in patients with a single nodule of hepatocellular carcinoma. BMC Cancer 2014, 14, 601. [Google Scholar] [CrossRef] [PubMed]
- Kudo, M.; Izumi, N.; Ichida, T.; Ku, Y.; Kokudo, N.; Sakamoto, M.; Takayama, T.; Nakashima, O.; Matsui, O.; Matsuyama, Y. Report of the 19th follow-up survey of primary liver cancer in Japan. Hepatol. Res. 2016, 46, 372–390. [Google Scholar] [CrossRef] [PubMed]
- Fukushima, T.; Kobayashi, S.; Katoh, H.; Hamaguchi, T.; Tozuka, Y.; Asai, Y.; Tezuka, S.; Ueno, M.; Morimoto, M.; Furuse, J.; et al. Comparison of carbon ion radiotherapy and transarterial chemoembolization for unresectable solitary hepatocellular carcinoma >3 cm: A propensity score-matched analysis. J. Radiat. Res. 2025, 66, 306–317. [Google Scholar] [CrossRef] [PubMed]
- Hoffmeister-Wittmann, P.; Hoegen-Saßmannshausen, P.; Wicklein, L.; Weykamp, F.; Seidensaal, K.; Springfeld, C.; Dill, M.T.; Longerich, T.; Schirmacher, P.; Mehrabi, A.; et al. Stereotactic body radiotherapy with carbon ions as local ablative treatment in patients with primary liver cancer. Radiat. Oncol. 2025, 20, 23. [Google Scholar] [CrossRef] [PubMed]
- Kurebayashi, Y.; Sugimoto, K.; Tsujikawa, H.; Matsuda, K.; Nomura, R.; Ueno, A.; Masugi, Y.; Yamazaki, K.; Effendi, K.; Takeuchi, H.; et al. Spatial Dynamics of T- and B-Cell Responses Predicts Clinical Outcome of Resectable and Unresectable Hepatocellular Carcinoma. Clin. Cancer Res. 2024, 30, 5666–5680. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Yang, A.; Quan, C.; Pan, Y.; Zhang, H.; Li, Y.; Gao, C.; Lu, H.; Wang, X.; Cao, P.; et al. A single-cell atlas of the multicellular ecosystem of primary and metastatic hepatocellular carcinoma. Nat. Commun. 2022, 13, 4594. [Google Scholar] [CrossRef] [PubMed]
- Ma, C.; Yu, X.; Zhang, X.; Su, L.; Jiang, O.; Cui, R. Combination of radiotherapy and ICIs in advanced hepatocellular carcinoma: A systematic review of current evidence and future prospects (Review). Oncol. Lett. 2025, 30, 342. [Google Scholar] [CrossRef] [PubMed]
- Chiu, D.K.; Zhang, X.; Cheng, B.Y.; Liu, Q.; Hayashi, K.; Yu, B.; Lee, R.; Zhang, C.; An, X.; Rajadas, J.; et al. Tumor-derived erythropoietin acts as an immunosuppressive switch in cancer immunity. Science 2025, 388, eadr3026. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, R.; Koroki, K.; Makishima, H.; Ogasawara, S.; Ishino, T.; Ogawa, K.; Nakagawa, M.; Fujiwara, K.; Unozawa, H.; Iwanaga, T.; et al. Controlling Major Portal Vein Invasion Progression during Lenvatinib Treatment by Carbon-Ion Radiotherapy in Patients with Advanced Hepatocellular Carcinoma. Case. Rep. Oncol. 2021, 14, 1103–1110. [Google Scholar] [CrossRef] [PubMed]
- Ogasawara, S.; Koroki, K.; Makishima, H.; Wakatsuki, M.; Takahashi, A.; Yumita, S.; Nakagawa, M.; Ishino, T.; Ogawa, K.; Fujiwara, K.; et al. Durvalumab with or without tremelimumab combined with particle therapy for advanced hepatocellular carcinoma with macrovascular invasion: Protocol for the DEPARTURE phase Ib trial. BMJ Open 2022, 12, e059779. [Google Scholar] [CrossRef] [PubMed]
| Photon Beam Therapies | Particle Beam Therapies |
|---|---|
| X-raysand gamma rays | Beams of protons and heavy ions |
| General high-energy radiation | Carbon-ion radiotherapy (C-ion RT) |
| Three-dimensional conformal radiation therapy | Proton beam therapy (PBT) |
| Intensity-modulated radiation therapy (IMRT) | |
| Volumetric-modulated arc therapy (VMAT) | |
| Image-guided radiotherapy (IGRT) | |
| Stereotactic body radiation therapy (SBRT) |
| City, Country | Year | Study | Participants | Results, Efficacy | Adverse Events |
|---|---|---|---|---|---|
| Chiba, Japan [55] | 2017 | Retrospective study [Phase I and II studies] | N = 124 (68 (37–84) years, 71% male/69 (46–84) years, 68% male); Single HCC (77%/74%); Child-Pugh Grade A, B (77%, 23%/80%, 20%); BCLC stage A, B, C (27%, 13%, 60%/20%, 14%, 66%); Main tumor size (mm) 40 (10–120)/37 (12–86); PS 0–1, 2 (87%, 13%/89%, 11%); Treatment-naïve (52%/64%) | Prescription doses, 69.6, 58.0, and 52.8 Gy (RBE) in 12, 8, and 4 fractions/52.8 Gy (RBE) in 4 fractions; Follow-up duration, 27.1 (0.9–154.8) months; OS rates, 1, 3 and 5 years: 90.3%, 50.0% and 25.0%; LC rates, 1, 3 and 5 years: 94.7%, 91.4% and 90.0% | Major complications, 0%; White blood cells, G3, 1%; Hemoglobin, G3, 1%; Platelets, G3, 10%; Skin, G3, 2%; Skin, G2, 7%; Gastrointestinal tract, G2, 2%; Child-Pugh Score, +2, 3%; Child-Pugh Score, +3, 1% |
| Maebashi, Japan [43] | 2017 | Retrospective study | N = 31 (83 (80–95) years, 71% male); No intrahepatic metastasis or distant metastasis; Child-Pugh Grade A, B (87%,13%); BCLC stage A, B, C (42%, 3%, 55%); Main tumor size (mm) 45 (15–93); PS 0, 1, 2 (55%, 39%, 6%); Prior therapy: TACE (10), RFA with TACE (2), TAI (1) | Prescription doses, 52.8 Gy (RBE) in 4 fractions or 60.0 Gy (RBE) in 4 fractions, 60.0 Gy (RBE) in 12 fractions for close-to-gastrointestinal tract cases, which were defined as a distance of < 1 cm between tumor and gastrointestinal tract; Follow-up duration, 23.2 (8.4–55.3) months; OS rates, 2 years: 82.3%; LC rates, 2 years: 89.2% | Major complications, 0%; Dermatitis, G1, 94%; Pneumonitis, G1, 26%, Encephalopathy, G1, 3%; Child-Pugh Score, +2, 3% |
| Maebashi, Japan [56] | 2018 | Retrospective study | N = 68 (sarcopenia/nonsarcopenia, 22/46, 77 (57–95) years/74 (45–90) years, 50% male/65% male); Single HCC and no direct infiltration of the gastrointestinal tract, any intrahepatic metastasis, or distant metastasis; Main tumor size (mm) 30 (12–90)/36 (9–77); PS 0, 1, 2 (55%, 45%, 0%)/(72%/24%/4%) | Prescription doses, 52.8 Gy (RBE) in 4 fraction or 60.0 Gy (RBE) in 4 fractions; Follow-up duration, 33.5 (3.9–83.1) months; OS rates, 3 years: 66% in sarcopenia/77% in nonsarcopenia; LC rates, 3 years: 81% in sarcopenia/72% in nonsarcopenia | Major complications, 0%; ALT G2, 5%; ALT G1 18%; Dermatitis G1, 86% (in sarcopenia group)/ALT G2, 2%; ALT G1 2%; Dermatitis G1, 87% (in nonsarcopenia group) |
| Maebashi, Japan [57] | 2019 | Phase I trial and the follow-up cohort | N = 21 (76 (58–88) years, 67% male); Single HCC (95%), 3 cm ≤ All HCC < 10 cm; Child-Pugh Grade A, 100%; UICC stage I/II/IIIa/IIIb, 71%/14%/5%/10%; Main tumor size (mm) 48 (30–78); PS 0, 1, 2 (62%, 29%, 9%); Treatment-naïve, 67% | Prescription doses, 60 Gy in 4 fractions; Follow-up duration, 24.2 (6.3–43.7) months; OS rates, 1 and 2 years: 90.5% and 80.0%; LC rates, 1 and 2 years: 100% and 92.3% | Major complications, 0%; Gastrointestinal G2, 5%; Investigation G2, 14% |
| Chiba, Japan [58] | 2019 | Retrospective study | N = 57 (75 (49–89) years, 58% male); Single HCC (98%); Child-Pugh Grade A, B (89%, 11%); Main tumor size (mm) 33 (13–95); PS 0, 1, 2 (68%, 30%, 2%); Treatment-naïve, 72% | Prescription doses, 45 Gy in 2 fraction; Follow-up duration, 54 (7–103) months; OS rates, 1, 3 and 5 years: 97%, 67% and 45%; LC rates, 1, 3 and 5 years: 98%, 91% and 91% | Major complications, 0%; Skin G3, 4%; Skin G2, 7%; Liver G2, 9%; Skin G1, 86%; Liver G1, 28%; Lung G1, 21% |
| Maebashi, Japan [59] | 2020 | Retrospective study | N = 11 (76 (57–86) years, 82% male); Child-Pugh Grade A, B (91%, 9%); BCLC stage A, B, C (18%, 0%, 82%); Main tumor size (mm) 53 (27–119); PS = <2 (100%) | Prescription doses, 52.8 Gy in 4 fractions or 60.0 Gy in 4 fractions, 60.0 Gy/in 12 fractions for close-to-gastrointestinal tract cases; Follow-up duration, 36.4 (4.3–86.2) months; OS rates, 3 years: 64%; LC rates, 3 years: 78% | Major complications, 0%; Dermatitis G1, 82%; Pneumonitis G1, 28%; Acsitis G1, 18% |
| Maebashi, Japan [60] | 2021 | Prospective study | N = 35 (75 (57–85) years, 51% male); All HCC ≤ 10 cm; Child-Pugh Grade A, B 83%, 17%; ALBI 1/2a/2b 26%/31%/43%; Main tumor size (mm) 35 (12–77); PS 0, 1, 2 (68%, 29%, 3%); Treatment-naïve, 57% | Prescription doses, 52.8 Gy in 4 fractions (49%) or 60 Gy in 4 fractions (51%); Follow-up duration, 49 (4–62.4) months; OS rates, 2 years: 82.8%; LC rates, 2 years: 92.6% | Major complications, 0%; Ascites G2, 3%; AST G2, 3%; Hypoalbuminemia G2, 3% |
| Shanghai, China [61] | 2023 | Phase I study | N = 23 (57 (28–76) years, 87% male); All HCC ≤ 12 cm; Child-Pugh Grade A 100%, 17%; BCLC stage 0, A, B, C (4%, 0%, 57%, 43%); Main tumor size (mm) 43 (17–85); PS 0, 1, 2 (52%, 48%, 0%) | Prescription doses, 55 Gy in 10 fractions, 60 Gy in 10 fractions, 65 in 10 fractions, or 70 Gy in 10 fractions, 22%/26%/35%/17%; Follow-up duration, 56.1 (5.7–74.4) months; OS rates, 1, 3 and 5 years: 91.3%, 81.9%, and 67.1%; LC rates, 1, 3 and 5 years: 100%, 94.4%, and 73.6% | Major complications, 0%; Leukocytopenia G3, 9%; Leukocytopenia G2, 9%; Neutrocytopenia G2, 26%; Thrombocytopenia G2, 9%; Bilirubin increase G2, 4.3%; Albumin decrease G2, 4.3%; Skin injury G1, 44%; Abdominal pain G1,17%; Leukocytopenia G1, 17%; Neutrocytopenia G1, 13%; Thrombocytopenia G1, 9%; ALP G1, 9% increase; Albumin decrease G1, 9% |
| Chiba, Japan [62] | 2023 | Retrospective study | N = 58 (57 (28–76) years, 87% male); 1~4, HCC ≤ 3 irradiation fields; Child-Pugh Grade B, 100%, score 7/8/9, 72%/22%/6%; ALBI 1/2a/2b/3 2%/12%/79%/7%; Main tumor size (mm) 32 (7–135); PS 0, 1, 2 (74%, 21%, 5%); Treatment-naïve, 24% | Prescription doses, 45 or 48 Gy in 2 fractions, 52.8 or 60 Gy in 4 fractions (when some organ at risk, such as the gastrointestinal (GI) tracts); Follow-up duration, 20.5 (2.7–108) months; OS rates, 1 and 2 years: 80.4% and 46.0%; LC rates, 1 and 2 years: 96.4% and 96.4% | Major complications, 0%. Child-Pugh Score increased more than 2 points in the acute and late phase were 3 and 2 patients, respectively. |
| Shanghai, China [63] | 2023 | Retrospective study | N = 90 (58.5 (28–87) years, 84% male); Single HCC (74%); Child-Pugh Grade A/B, 99%/1%; BCLC stage 0, A, B, C (2%, 7%, 46%, 46%); Main tumor size (mm) 46 (1.6–15.5); PS 0, 1, 2 (52%, 48%, 0%); PS 0, 1, 2 (68%, 32%, 0%) | Prescription doses, 50–70 Gy in 10 fractions (64%), 60–67.5 Gy in 15 fractions (36%); Follow-up duration, 28.6 (5.7–74.6) months; OS rates, 1, 2 and 3 years: 97.8%, 83.3% and 75.4%; LC rates, 1, 2 and 3 years: 96.4%, 96.4% and 93.1% | Death, 2; Leucopenia G3–4, 6.7%; Thrombocytopenia G3–4, 3.3%; Leucopenia G1–2, 38.9%; Thrombocytopenia G1–2, 25.6%; Hyperbilirubinemia G1–2, 2.2%; Hypoalbuminemia G1–2, 10%; γ-glutamyltransferase increase G1–2, 2.2%; Alkaline phosphatase increases G1–2, 6.7%; Dysphagia G1–2, 2.2%; Dermatitis radiation G1–2, 35.6%; Abdominal pain G1–2, 20% |
| Chiba, Japan [64] | 2024 | Retrospective cohort study | N = 76 (71 (45–86) years, 76% male); 1~4 HCC; Child-Pugh Grade A/B, 89%/11%; ALBI 1/2 55%/45%; Main tumor size (mm) 46 (15–130); PS 0, 1, 2 (72%, 23%, 4%); Treatment-naïve, 52.6% | Prescription doses, 45.0~48.0 Gy in 2 fractions, 52.8~60.0 Gy in 4 fractions; Follow-up duration, 27.9 (1.5–180.4) months; OS rates, 2 and 3 years: 70.0% and 50.2%; Local recurrence rates, 2 and 3 years: 8.9% and 10.7% | Major complications, 0%; Dermatitis G3, 3%; Hepatobiliary disorder G3, 3%; Dermatitis G1, 1%; Hepatobiliary disorder G1, 4% |
| Osaka, Japan [65] | 2024 | Retrospective study | N = 108 (76 (47–95) years, 71% male); 1~5 HCC; Child-Pugh Grade A/B, 91%/9%; Main tumor size (mm) 41 (5–160); PS 0, 1, 2 (79%, 17%, 5%); Treatment-naïve, 66.6% | Prescription doses, 60 Gy in 4 fractions; Follow-up duration, 9.7 (2.3–41.1) months; OS rates, N/A; LC rates, N/A | Major complications, 0%. The pretreatment Child-Pugh Grade B (HR = 6.90; p = 0.003) and normal liver volume spared from <30 Gy RBE (HR = 5.22; p = 0.009) were significant risk factors for RILD. |
| Heidelberg, Germany [66] | 2024 | Phase I study | N = 20 (74.7 (55.7–83.6) years, 75% male); 1~5 HCC; Child-Pugh Grade A/B, 70%/10%; BCLC stage 0, A, B, C (15%, 30%, 55%, 0%); PS 0, 1, 2 (45%, 50%, 5%; Treatment-naïve, 66.6% | Prescription doses, 32.0~42.0 Gy in 4 fractions; Follow-up duration, 23.0 months; OS rates, 1, 2, 3, and 4 years: 75%, 64%, 22%, and 15%; LC rates, 1, 2, and 3 years: 59%, 43%, and 43% | Major complications, 0%; No toxicity |
| Taipei, Taiwan [67] | 2025 | Phase I study | N = 1 (72-year-old man); 25 × 23 mm HCC in the right hepatic lobe; Clinical staging, T1bN0M0 | Prescription doses, 52.8 Gy (RBE) in 4 fractions; Follow-up duration, 3 months; Complete response | Major complications, 0%; No adverse events |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sasaki-Tanaka, R.; Abe, H.; Yoshida, T.; Watanabe, Y.; Kimura, N.; Yokoo, T.; Sakamaki, A.; Kamimura, H.; Kamimura, K.; Kanda, T.; et al. Carbon-Ion Radiotherapy for Hepatocellular Carcinoma: Current Status and Future Prospects: A Narrative Review. J. Clin. Med. 2025, 14, 6107. https://doi.org/10.3390/jcm14176107
Sasaki-Tanaka R, Abe H, Yoshida T, Watanabe Y, Kimura N, Yokoo T, Sakamaki A, Kamimura H, Kamimura K, Kanda T, et al. Carbon-Ion Radiotherapy for Hepatocellular Carcinoma: Current Status and Future Prospects: A Narrative Review. Journal of Clinical Medicine. 2025; 14(17):6107. https://doi.org/10.3390/jcm14176107
Chicago/Turabian StyleSasaki-Tanaka, Reina, Hiroyuki Abe, Tomoaki Yoshida, Yusuke Watanabe, Naruhiro Kimura, Takeshi Yokoo, Akira Sakamaki, Hiroteru Kamimura, Kenya Kamimura, Tatsuo Kanda, and et al. 2025. "Carbon-Ion Radiotherapy for Hepatocellular Carcinoma: Current Status and Future Prospects: A Narrative Review" Journal of Clinical Medicine 14, no. 17: 6107. https://doi.org/10.3390/jcm14176107
APA StyleSasaki-Tanaka, R., Abe, H., Yoshida, T., Watanabe, Y., Kimura, N., Yokoo, T., Sakamaki, A., Kamimura, H., Kamimura, K., Kanda, T., & Terai, S. (2025). Carbon-Ion Radiotherapy for Hepatocellular Carcinoma: Current Status and Future Prospects: A Narrative Review. Journal of Clinical Medicine, 14(17), 6107. https://doi.org/10.3390/jcm14176107







