Orally Administered CBD/CBG Hemp Extract Reduces Severity of Ulcerative Colitis and Pain in a Murine Model
Abstract
1. Introduction
2. Materials and Methods
2.1. Administration of Hemp Extract (HE)
2.2. Animals and Induction of Colitis
2.3. Mass Spectroscopy
2.4. Disease Activity Index (DAI) Scoring
2.5. Tissue Collection and Processing
2.6. Von Frey Assay
2.7. Alcian Blue Staining
2.8. Statistical Analysis
3. Results
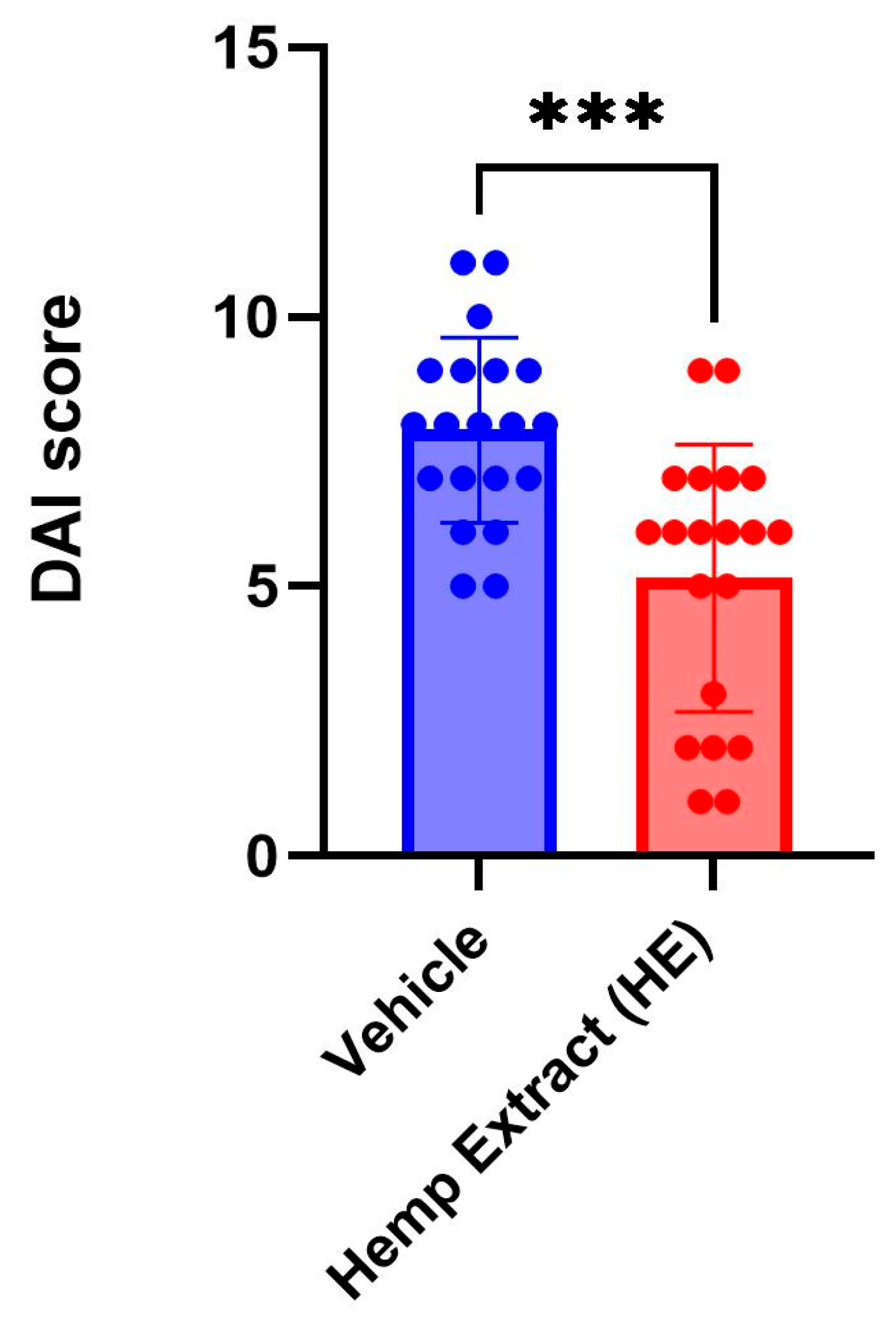
3.1. Oral Administration of CBD/CBG Hemp Extract Reduces Colitis Severity
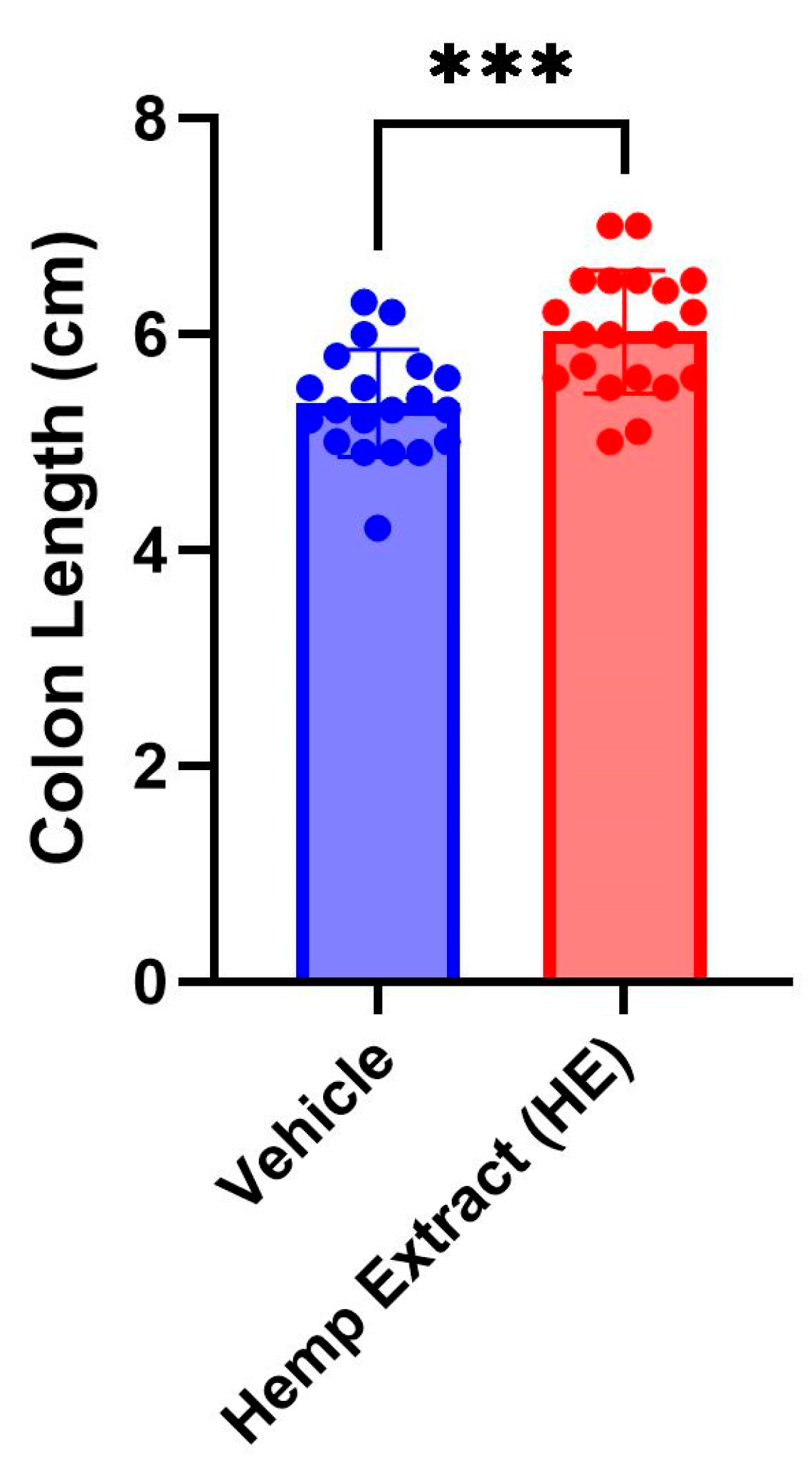
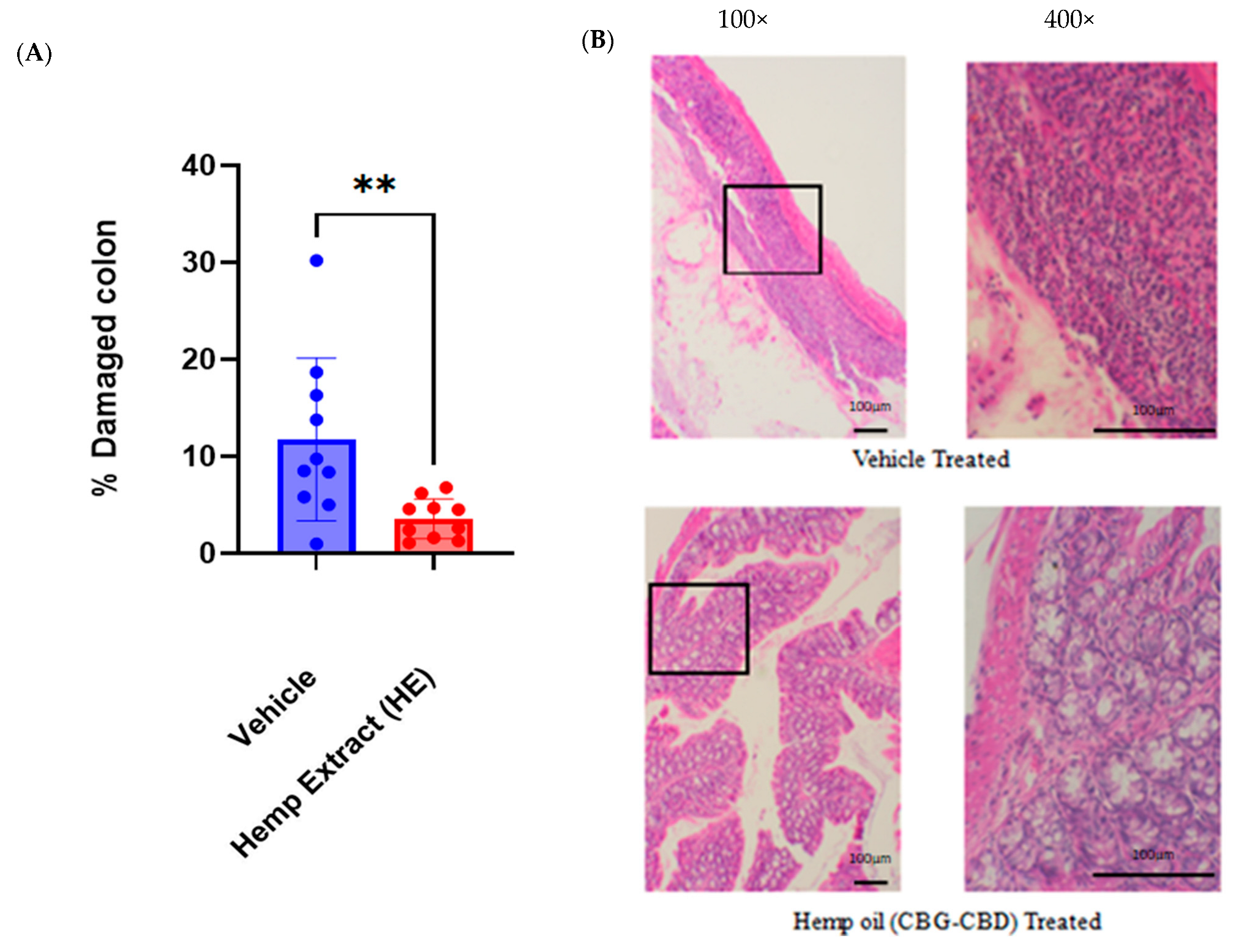
3.2. Oral Administration of CBD/CBG Hemp Extract Reduces Colonic Ulceration
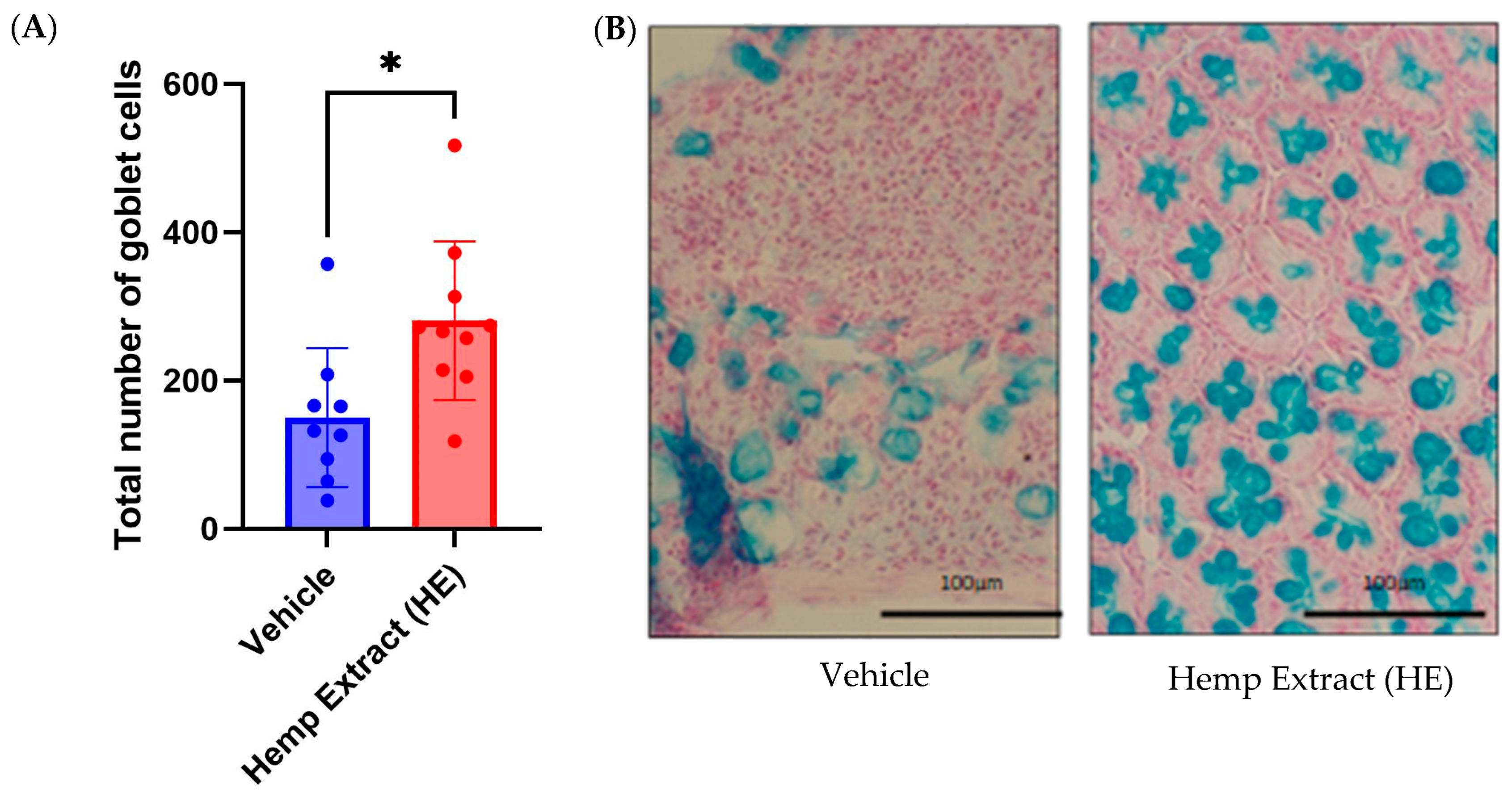
3.3. Hemp Extract Treatment Helps in Restoration of Goblet Cells
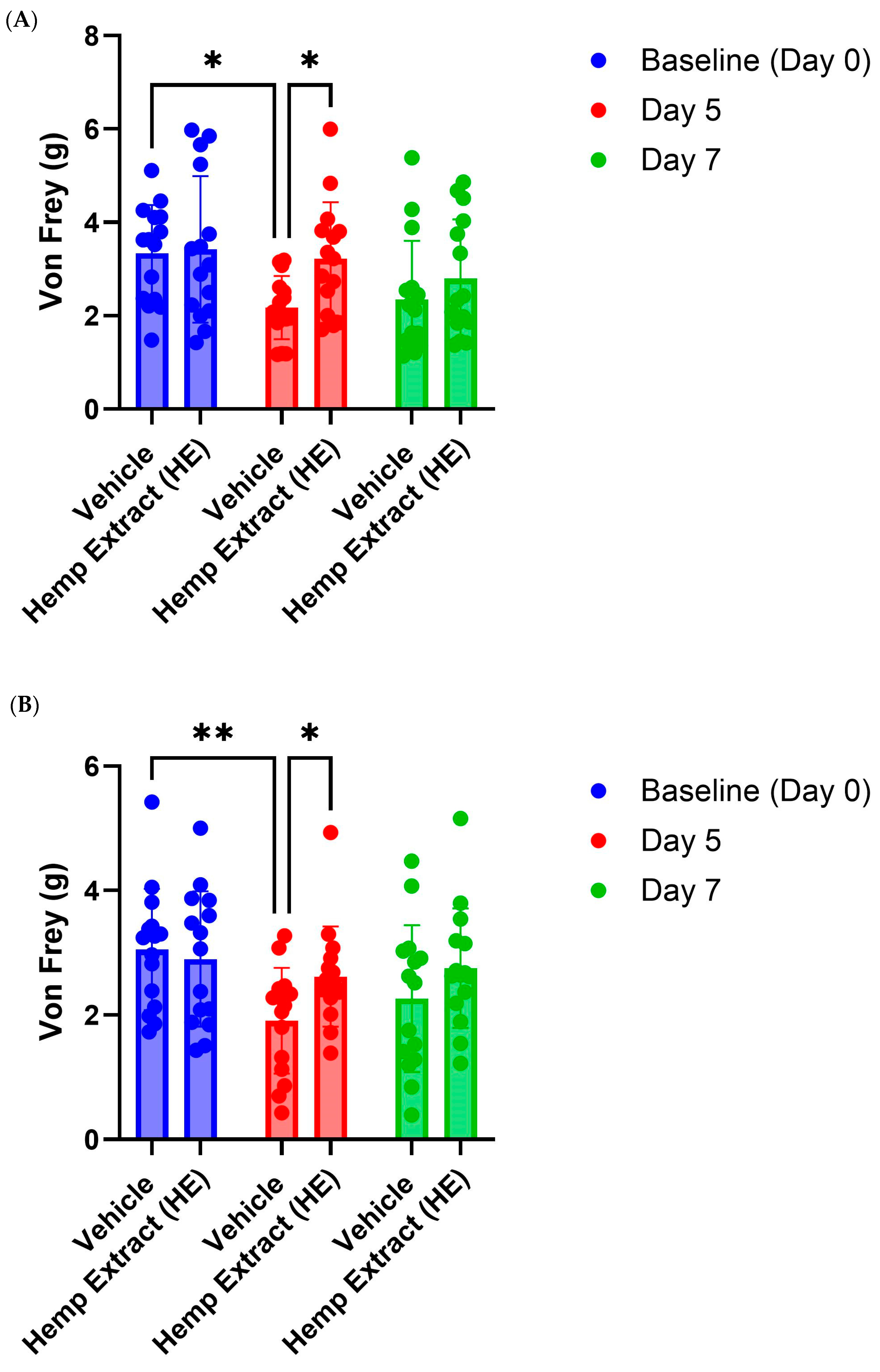
3.4. Hemp Extract Administration Reduces Ulcerative Colitis Associated Proxy Measures of Abdominal Pain
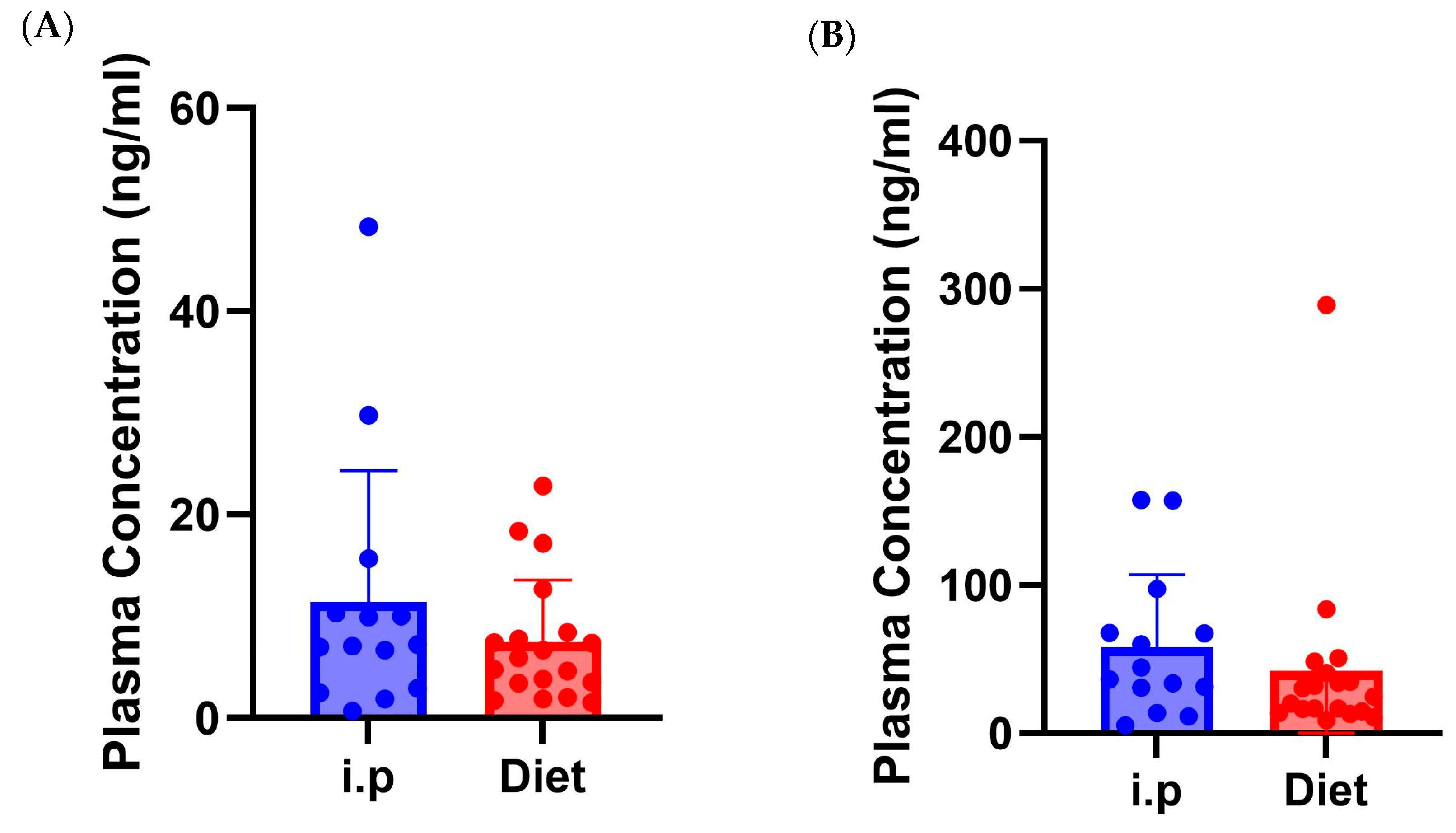
3.5. Route of Administration Impact on Plasma Levels of CBG and CBD
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| IBDI | inflammatory bowel disease |
| UC | ulcerative colitis |
| CBD | cannabidiol |
| CBG | cannabigerol |
| HE | hemp extract |
| H&E | hematoxylin and eosin |
References
- Gubatan, J.; Kulkarni, C.V.; Talamantes, S.M.; Temby, M.; Fardeen, T.; Sinha, S.R. Dietary Exposures and Interventions in Inflammatory Bowel Disease: Current Evidence and Emerging Concepts. Nutrients 2023, 15, 579. [Google Scholar] [CrossRef]
- Mendieta-Escalante, E.A.; Faber, K.N.; Dijkstra, G. Current and Emerging Autoantibodies in Ulcerative Colitis. Eur. J. Immunol. 2025, 55, e51721. [Google Scholar] [CrossRef]
- Omer, M.; Omer, O.I.; Ali, M.; Mohamed, I.O. Therapeutic Potentials of Phytochemicals in the Treatment of Ulcerative Colitis: Results from Animal Model Interventional Studies. J. Am. Nutr. Assoc. 2025, 1–13. [Google Scholar] [CrossRef]
- Huang, J.; Zhu, X.; Ma, Y.; Zhang, Z.; Zhang, J.; Hao, Z.; Wu, L.; Liu, H.; Wu, H.; Bao, C. Machine Learning in the Differential Diagnosis of Ulcerative Colitis and Crohn’s Disease: A Systematic Review. Transl. Gastroenterol. Hepatol. 2025, 10, 56. [Google Scholar] [CrossRef]
- Faggiani, I.; Fanizza, J.; Massironi, S.; D’Amico, F.; Allocca, M.; Furfaro, F.; Parigi, T.L.; Fiorino, G.; Danese, S.; Zilli, A. The Role of Diet in Inflammatory Bowel Disease: A Comprehensive Review of the Literature. Best Pract. Res. Clin. Gastroenterol. 2025, 77, 101995. [Google Scholar] [CrossRef]
- Silverberg, M.S.; Satsangi, J.; Ahmad, T.; Arnott, I.D.; Bernstein, C.N.; Brant, S.R.; Caprilli, R.; Colombel, J.-F.; Gasche, C.; Geboes, K.; et al. Toward an Integrated Clinical, Molecular and Serological Classification of Inflammatory Bowel Disease: Report of a Working Party of the 2005 Montreal World Congress of Gastroenterology. Can. J. Gastroenterol. 2005, 19, 5A–36A. [Google Scholar] [CrossRef] [PubMed]
- Gajendran, M.; Loganathan, P.; Jimenez, G.; Catinella, A.P.; Ng, N.; Umapathy, C.; Ziade, N.; Hashash, J.G. A Comprehensive Review and Update on Ulcerative Colitis. Dis. Mon. 2019, 65, 100851. [Google Scholar] [CrossRef]
- Melhem, H.; Regan-Komito, D.; Niess, J.H. Mucins Dynamics in Physiological and Pathological Conditions. Int. J. Mol. Sci. 2021, 22, 13642. [Google Scholar] [CrossRef]
- Mansouri, P.; Mansouri, P.; Behmard, E.; Najafipour, S.; Kouhpayeh, A.; Farjadfar, A. Novel Targets for Mucosal Healing in Inflammatory Bowel Disease Therapy. Int. Immunopharmacol. 2025, 144, 113544. [Google Scholar] [CrossRef] [PubMed]
- Le Berre, C.; Honap, S.; Peyrin-Biroulet, L. Ulcerative Colitis. Lancet 2023, 402, 571–584. [Google Scholar] [CrossRef] [PubMed]
- Fanizzi, F.; D’Amico, F.; Peyrin-Biroulet, L.; Danese, S.; Dignass, A. Treatment Targets in IBD: Is It Time for New Strategies? Best Pract. Res. Clin. Gastroenterol. 2025, 77, 101990. [Google Scholar] [CrossRef]
- Coates, M.D.; Clarke, K.; Williams, E.; Jeganathan, N.; Yadav, S.; Giampetro, D.; Gordin, V.; Smith, S.; Vrana, K.; Bobb, A.; et al. Abdominal Pain in Inflammatory Bowel Disease: An Evidence-Based, Multidisciplinary Review. Crohn’s Colitis 360 2023, 5, otad055. [Google Scholar] [CrossRef]
- Coates, M.D.; Johri, A.; Gorrepati, V.S.; Maheshwari, P.; Dalessio, S.; Walter, V.; Stuart, A.; Koltun, W.; Bernasko, N.; Tinsley, A.; et al. Abdominal Pain in Quiescent Inflammatory Bowel Disease. Int. J. Color. Dis. 2021, 36, 93–102. [Google Scholar] [CrossRef]
- Coates, M.D.; Dalessio, S.; Walter, V.; Stuart, A.; Bernasko, N.; Tinsley, A.; Razeghi, S.; Williams, E.D.; Clarke, K.; Vrana, K. Symptoms and Extraintestinal Manifestations in Active Cannabis Users with Inflammatory Bowel Disease. Cannabis Cannabinoid Res. 2022, 7, 445–450. [Google Scholar] [CrossRef]
- Bogale, K.; Maheshwari, P.; Kang, M.; Gorrepati, V.S.; Dalessio, S.; Walter, V.; Stuart, A.; Koltun, W.; Bernasko, N.; Tinsley, A.; et al. Symptoms Associated with Healthcare Resource Utilization in the Setting of Inflammatory Bowel Disease. Sci. Rep. 2022, 12, 10577. [Google Scholar] [CrossRef] [PubMed]
- Gemayel, N.C.; Rizzello, E.; Atanasov, P.; Wirth, D.; Borsi, A. Dose Escalation and Switching of Biologics in Ulcerative Colitis: A Systematic Literature Review in Real-World Evidence. Curr. Med. Res. Opin. 2019, 35, 1911–1923. [Google Scholar] [CrossRef] [PubMed]
- Velikova, T.; Sekulovski, M.; Peshevska-Sekulovska, M. Immunogenicity and Loss of Effectiveness of Biologic Therapy for Inflammatory Bowel Disease Patients Due to Anti-Drug Antibody Development. Antibodies 2024, 13, 16. [Google Scholar] [CrossRef]
- Anil, S.M.; Peeri, H.; Koltai, H. Medical Cannabis Activity Against Inflammation: Active Compounds and Modes of Action. Front. Pharmacol. 2022, 13, 908198. [Google Scholar] [CrossRef]
- Nagarkatti, P.; Pandey, R.; Rieder, S.A.; Hegde, V.L.; Nagarkatti, M. Cannabinoids as Novel Anti-Inflammatory Drugs. Future Med. Chem. 2009, 1, 1333–1349. [Google Scholar] [CrossRef]
- Leonard, B.E.; Aricioglu, F. Cannabinoids and Neuroinflammation: Therapeutic Implications. J. Affect. Disord. Rep. 2023, 12, 100463. [Google Scholar] [CrossRef]
- Kinnucan, J. Use of Medical Cannabis in Patients with Inflammatory Bowel Disease. Gastroenterol. Hepatol. 2018, 14, 598–601. [Google Scholar]
- Glickman, D.; Dalessio, S.; Raup-Konsavage, W.M.; Vrana, K.E.; Coates, M.D. The Impact of Cannabis Use on Clinical Outcomes in Inflammatory Bowel Disease: A Population-Based Longitudinal Cohort Study. Inflamm. Bowel Dis. 2024, 30, 1055–1061. [Google Scholar] [CrossRef] [PubMed]
- Coates, M.D.; Raup-Konsavage, W.M.; Vrana, K.E. Better Together? Individual and Synergistic Effects of Noneuphorigenic Cannabinoids on Visceral Sensation. J. Pharmacol. Exp. Ther. 2025, 392, 103593. [Google Scholar] [CrossRef] [PubMed]
- Richardson, J.D. Cannabinoids Modulate Pain by Multiple Mechanisms of Action. J. Pain 2000, 1, 2–14. [Google Scholar] [CrossRef]
- Rodriguez, C.E.B.; Ouyang, L.; Kandasamy, R. Antinociceptive Effects of Minor Cannabinoids, Terpenes and Flavonoids in Cannabis. Behav. Pharmacol. 2022, 33, 130–157. [Google Scholar] [CrossRef] [PubMed]
- Busquets-Garcia, A.; Bains, J.; Marsicano, G. CB1 Receptor Signaling in the Brain: Extracting Specificity from Ubiquity. Neuropsychopharmacology 2018, 43, 4–20. [Google Scholar] [CrossRef]
- Howlett, A.C. Pharmacology of Cannabinoid Receptors. Annu. Rev. Pharmacol. Toxicol. 1995, 35, 607–634. [Google Scholar] [CrossRef]
- Cabral, G.A.; Griffin-Thomas, L. Emerging Role of the Cannabinoid Receptor CB2 in Immune Regulation: Therapeutic Prospects for Neuroinflammation. Expert Rev. Mol. Med. 2009, 11, e3. [Google Scholar] [CrossRef]
- Correa, F.; Docagne, F.; Mestre, L.; Clemente, D.; Hernangómez, M.; Loría, F.; Guaza, C. A Role for CB2 Receptors in Anandamide Signalling Pathways Involved in the Regulation of IL-12 and IL-23 in Microglial Cells. Biochem. Pharmacol. 2009, 77, 86–100. [Google Scholar] [CrossRef]
- Hryhorowicz, S.; Kaczmarek-Ryś, M.; Zielińska, A.; Scott, R.J.; Słomski, R.; Pławski, A. Endocannabinoid System as a Promising Therapeutic Target in Inflammatory Bowel Disease—A Systematic Review. Front. Immunol. 2021, 12, 790803. [Google Scholar] [CrossRef]
- Tartakover Matalon, S.; Azar, S.; Meiri, D.; Hadar, R.; Nemirovski, A.; Abu Jabal, N.; Konikoff, F.M.; Drucker, L.; Tam, J.; Naftali, T. Endocannabinoid Levels in Ulcerative Colitis Patients Correlate with Clinical Parameters and Are Affected by Cannabis Consumption. Front. Endocrinol. 2021, 12, 685289. [Google Scholar] [CrossRef]
- Engel, M.A.; Kellermann, C.A.; Burnat, G.; Hahn, E.G.; Rau, T.; Konturek, P.C. Mice Lacking Cannabinoid CB1-, CB2-Receptors or Both Receptors Show Increased Susceptibility to Trinitrobenzene Sulfonic Acid (TNBS)-Induced Colitis. J. Physiol. Pharmacol. Off. J. Pol. Physiol. Soc. 2010, 61, 89–97. [Google Scholar]
- Anderson, B.D.; Sepulveda, D.E.; Nachnani, R.; Cortez-Resendiz, A.; Coates, M.D.; Beckett, A.; Bisanz, J.E.; Kellogg, J.J.; Raup-Konsavage, W.M. High Cannabigerol Hemp Extract Moderates Colitis and Modulates the Microbiome in an Inflammatory Bowel Disease Model. J. Pharmacol. Exp. Ther. 2024, 390, 331–341. [Google Scholar] [CrossRef]
- Couch, D.G.; Maudslay, H.; Doleman, B.; Lund, J.N.; O’Sullivan, S.E. The Use of Cannabinoids in Colitis: A Systematic Review and Meta-Analysis. Inflamm. Bowel Dis. 2018, 24, 680–697. [Google Scholar] [CrossRef]
- Pagano, E.; Capasso, R.; Piscitelli, F.; Romano, B.; Parisi, O.A.; Finizio, S.; Lauritano, A.; Marzo, V.D.; Izzo, A.A.; Borrelli, F. An Orally Active Cannabis Extract with High Content in Cannabidiol Attenuates Chemically-Induced Intestinal Inflammation and Hypermotility in the Mouse. Front. Pharmacol. 2016, 7, 341. [Google Scholar] [CrossRef]
- Deiana, S.; Watanabe, A.; Yamasaki, Y.; Amada, N.; Arthur, M.; Fleming, S.; Woodcock, H.; Dorward, P.; Pigliacampo, B.; Close, S.; et al. Plasma and Brain Pharmacokinetic Profile of Cannabidiol (CBD), Cannabidivarine (CBDV), Δ9-Tetrahydrocannabivarin (THCV) and Cannabigerol (CBG) in Rats and Mice Following Oral and Intraperitoneal Administration and CBD Action on Obsessive–Compulsive Behaviour. Psychopharmacology 2012, 219, 859–873. [Google Scholar] [CrossRef] [PubMed]
- Chassaing, B.; Aitken, J.D.; Malleshappa, M.; Vijay-Kumar, M. Dextran Sulfate Sodium (DSS)-Induced Colitis in Mice. Curr. Protoc. Immunol. 2014, 104, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Konsavage, W.M.; Jin, G.; Yochum, G.S. The Myc 3′ Wnt-Responsive Element Regulates Homeostasis and Regeneration in the Mouse Intestinal Tract. Mol. Cell. Biol. 2012, 32, 3891–3902. [Google Scholar] [CrossRef]
- Konsavage, W.M.; Roper, J.N.; Ishmael, F.T.; Yochum, G.S. The Myc 3′ Wnt Responsive Element Regulates Neutrophil Recruitment after Acute Colonic Injury in Mice. Dig. Dis. Sci. 2013, 58, 2858–2867. [Google Scholar] [CrossRef]
- Raup-Konsavage, W.M.; Cooper, T.K.; Yochum, G.S. A Role for MYC in Lithium-Stimulated Repair of the Colonic Epithelium After DSS-Induced Damage in Mice. Dig. Dis. Sci. 2016, 61, 410–422. [Google Scholar] [CrossRef] [PubMed]
- Sepulveda, D.E.; Morris, D.P.; Raup-Konsavage, W.M.; Sun, D.; Vrana, K.E.; Graziane, N.M. Cannabigerol (CBG) Attenuates Mechanical Hypersensitivity Elicited by Chemotherapy-induced Peripheral Neuropathy. Eur. J. Pain 2022, 26, 1950–1966. [Google Scholar] [CrossRef]
- Laird, J.M.A.; Martinez-Caro, L.; Garcia-Nicas, E.; Cervero, F. A New Model of Visceral Pain and Referred Hyperalgesia in the Mouse. Pain 2001, 92, 335–342. [Google Scholar] [CrossRef] [PubMed]
- López-Estévez, S.; Gris, G.; De La Puente, B.; Carceller, A.; Martínez, V. Intestinal Inflammation-Associated Hypersensitivity Is Attenuated in a DSS Model of Colitis in Sigma-1 Knockout C57BL/6 Mice. Biomed. Pharmacother. 2021, 143, 112126. [Google Scholar] [CrossRef]
- Barry, A.R. Patients’ Perceptions and Use of Natural Health Products. Can. Pharm. J. CPJ Rev. Pharm. Can. RPC 2018, 151, 254–262. [Google Scholar] [CrossRef]
- Klepser, T.B.; Doucette, W.R.; Horton, M.R.; Buys, L.M.; Ernst, M.E.; Ford, J.K.; Hoehns, J.D.; Kautzman, H.A.; Logemann, C.D.; Swegle, J.M.; et al. Assessment of Patients’ Perceptions and Beliefs Regarding Herbal Therapies. Pharmacotherapy 2000, 20, 83–87. [Google Scholar] [CrossRef]
- Snyder, F.J.; Dundas, M.L.; Kirkpatrick, C.; Neill, K.S. Use and Safety Perceptions Regarding Herbal Supplements: A Study of Older Persons in Southeast Idaho. J. Nutr. Elder. 2009, 28, 81–95. [Google Scholar] [CrossRef]
- Lynch, N.; Berry, D. Differences in Perceived Risks and Benefits of Herbal, over-the-Counter Conventional, and Prescribed Conventional, Medicines, and the Implications of This for the Safe and Effective Use of Herbal Products. Complement. Ther. Med. 2007, 15, 84–91. [Google Scholar] [CrossRef]
- Chiba, T.; Tanemura, N. Differences in the Perception of Dietary Supplements between Dietary Supplement/Medicine Users and Non-Users. Nutrients 2022, 14, 4114. [Google Scholar] [CrossRef] [PubMed]
- Tsokeva, Z.; Ganeva, M.; Gancheva, T.; Hristakieva, E. Dietary Supplements—Use and Safety Perception: A Study among Hospitalized Dermatology Patients. Int. J. Clin. Pharmacol. Ther. 2021, 59, 116–126. [Google Scholar] [CrossRef]
- Ibrahim, I.R.; Hassali, M.A.; Saleem, F.; Al Tukmagi, H.F. A Qualitative Insight on Complementary and Alternative Medicines Used by Hypertensive Patients. J. Pharm. Bioallied Sci. 2016, 8, 284–288. [Google Scholar] [CrossRef] [PubMed]
- Bogale, K.; Raup-Konsavage, W.; Dalessio, S.; Vrana, K.; Coates, M.D. Cannabis and Cannabis Derivatives for Abdominal Pain Management in Inflammatory Bowel Disease. Med. Cannabis Cannabinoids 2021, 4, 97–106. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, W.; Katz, S. Therapeutic Use of Cannabis in Inflammatory Bowel Disease. Gastroenterol. Hepatol. 2016, 12, 668–679. [Google Scholar]
- Ambrose, T.; Simmons, A. Cannabis, Cannabinoids, and the Endocannabinoid System-Is There Therapeutic Potential for Inflammatory Bowel Disease? J. Crohn’s Colitis 2019, 13, 525–535. [Google Scholar] [CrossRef]
- Nduma, B.N.; Mofor, K.A.; Tatang, J.; Ekhator, C.; Ambe, S.; Fonkem, E. The Use of Cannabinoids in the Treatment of Inflammatory Bowel Disease (IBD): A Review of the Literature. Cureus 2023, 15, e36148. [Google Scholar] [CrossRef]
- Naftali, T. Is Cannabis of Potential Value as a Therapeutic for Inflammatory Bowel Disease? Dig. Dis. Sci. 2019, 64, 2696–2698. [Google Scholar] [CrossRef]
- Singh, V.; Johnson, K.; Yin, J.; Lee, S.; Lin, R.; Yu, H.; In, J.; Foulke-Abel, J.; Zachos, N.C.; Donowitz, M.; et al. Chronic Inflammation in Ulcerative Colitis Causes Long-Term Changes in Goblet Cell Function. Cell. Mol. Gastroenterol. Hepatol. 2022, 13, 219–232. [Google Scholar] [CrossRef] [PubMed]
- Leoncini, G.; Cari, L.; Ronchetti, S.; Donato, F.; Caruso, L.; Calafà, C.; Villanacci, V. Mucin Expression Profiles in Ulcerative Colitis: New Insights on the Histological Mucosal Healing. Int. J. Mol. Sci. 2024, 25, 1858. [Google Scholar] [CrossRef]
- Gitter, A.H.; Wullstein, F.; Fromm, M.; Schulzke, J.D. Epithelial Barrier Defects in Ulcerative Colitis: Characterization and Quantification by Electrophysiological Imaging. Gastroenterology 2001, 121, 1320–1328. [Google Scholar] [CrossRef]
- Yeisley, D.J.; Arabiyat, A.S.; Hahn, M.S. Cannabidiol-Driven Alterations to Inflammatory Protein Landscape of Lipopolysaccharide-Activated Macrophages In Vitro May Be Mediated by Autophagy and Oxidative Stress. Cannabis Cannabinoid Res. 2021, 6, 253–263. [Google Scholar] [CrossRef]
- Silva, R.L.; Silveira, G.T.; Wanderlei, C.W.; Cecilio, N.T.; Maganin, A.G.M.; Franchin, M.; Marques, L.M.M.; Lopes, N.P.; Crippa, J.A.; Guimarães, F.S.; et al. DMH-CBD, a Cannabidiol Analog with Reduced Cytotoxicity, Inhibits TNF Production by Targeting NF-kB Activity Dependent on A2A Receptor. Toxicol. Appl. Pharmacol. 2019, 368, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Muthumalage, T.; Rahman, I. Cannabidiol Differentially Regulates Basal and LPS-Induced Inflammatory Responses in Macrophages, Lung Epithelial Cells, and Fibroblasts. Toxicol. Appl. Pharmacol. 2019, 382, 114713. [Google Scholar] [CrossRef] [PubMed]
- Borrelli, F.; Fasolino, I.; Romano, B.; Capasso, R.; Maiello, F.; Coppola, D.; Orlando, P.; Battista, G.; Pagano, E.; Di Marzo, V.; et al. Beneficial Effect of the Non-Psychotropic Plant Cannabinoid Cannabigerol on Experimental Inflammatory Bowel Disease. Biochem. Pharmacol. 2013, 85, 1306–1316. [Google Scholar] [CrossRef]
- Li, S.; Li, W.; Malhi, N.K.; Huang, J.; Li, Q.; Zhou, Z.; Wang, R.; Peng, J.; Yin, T.; Wang, H. Cannabigerol (CBG): A Comprehensive Review of Its Molecular Mechanisms and Therapeutic Potential. Molecules 2024, 29, 5471. [Google Scholar] [CrossRef]
- Farrell, K.E.; Callister, R.J.; Keely, S. Understanding and Targeting Centrally Mediated Visceral Pain in Inflammatory Bowel Disease. Front. Pharmacol. 2014, 5, 27. [Google Scholar] [CrossRef]
- Falling, C.L.; Stebbings, S.; David Baxter, G.; Siegel, C.A.; Gearry, R.B.; Mani, R. Somatosensory Assessments in Patients with Inflammatory Bowel Disease: A Cross-Sectional Study Examining Pain Processing Pathways and the Role of Multiple Patient Factors. Eur. J. Gastroenterol. Hepatol. 2022, 34, 503–511. [Google Scholar] [CrossRef]
- Casslén, B.; Andersson, A.; Nilsson, I.M.; Astedt, B. Hormonal Regulation of the Release of Plasminogen Activators and of a Specific Activator Inhibitor from Endometrial Tissue in Culture. Proc. Soc. Exp. Biol. Med. 1986, 182, 419–424. [Google Scholar] [CrossRef]
- Lashgari, N.-A.; Roudsari, N.M.; Momtaz, S.; Niazi Shahraki, F.; Zandi, N.; Pazoki, B.; Farzaei, M.H.; Ghasemi, M.; Abdollahi, M.; Abdolghaffari, A.H. Systematic Review on Herbal Preparations for Controlling Visceral Hypersensitivity in Functional Gastrointestinal Disorders. Curr. Pharm. Biotechnol. 2024, 25, 1632–1650. [Google Scholar] [CrossRef] [PubMed]
- Robaina Cabrera, C.L.; Keir-Rudman, S.; Horniman, N.; Clarkson, N.; Page, C. The Anti-Inflammatory Effects of Cannabidiol and Cannabigerol Alone, and in Combination. Pulm. Pharmacol. Ther. 2021, 69, 102047. [Google Scholar] [CrossRef] [PubMed]
- Khajuria, D.K.; Karuppagounder, V.; Nowak, I.; Sepulveda, D.E.; Lewis, G.S.; Norbury, C.C.; Raup-Konsavage, W.M.; Vrana, K.E.; Kamal, F.; Elbarbary, R.A. Cannabidiol and Cannabigerol, Nonpsychotropic Cannabinoids, as Analgesics That Effectively Manage Bone Fracture Pain and Promote Healing in Mice. J. Bone Miner. Res. 2023, 38, 1560–1576. [Google Scholar] [CrossRef]
- Pramhas, S.; Thalhammer, T.; Terner, S.; Pickelsberger, D.; Gleiss, A.; Sator, S.; Kress, H.G. Oral Cannabidiol (CBD) as Add-on to Paracetamol for Painful Chronic Osteoarthritis of the Knee: A Randomized, Double-Blind, Placebo-Controlled Clinical Trial. Lancet Reg. Health—Eur. 2023, 35, 100777. [Google Scholar] [CrossRef]
- Hameed, M.; Prasad, S.; Jain, E.; Dogrul, B.N.; Al-Oleimat, A.; Pokhrel, B.; Chowdhury, S.; Co, E.L.; Mitra, S.; Quinonez, J.; et al. Medical Cannabis for Chronic Nonmalignant Pain Management. Curr. Pain Headache Rep. 2023, 27, 57–63. [Google Scholar] [CrossRef]
- Mücke, M.; Phillips, T.; Radbruch, L.; Petzke, F.; Häuser, W. Cannabis-Based Medicines for Chronic Neuropathic Pain in Adults. Cochrane Database Syst. Rev. 2018, 3, CD012182. [Google Scholar] [CrossRef] [PubMed]
- Gamble, L.-J.; Boesch, J.M.; Frye, C.W.; Schwark, W.S.; Mann, S.; Wolfe, L.; Brown, H.; Berthelsen, E.S.; Wakshlag, J.J. Pharmacokinetics, Safety, and Clinical Efficacy of Cannabidiol Treatment in Osteoarthritic Dogs. Front. Vet. Sci. 2018, 5, 165. [Google Scholar] [CrossRef] [PubMed]
- Cásedas, G.; de Yarza-Sancho, M.; López, V. Cannabidiol (CBD): A Systematic Review of Clinical and Preclinical Evidence in the Treatment of Pain. Pharmaceuticals 2024, 17, 1438. [Google Scholar] [CrossRef]
- Izzo, A.A.; Capasso, R.; Aviello, G.; Borrelli, F.; Romano, B.; Piscitelli, F.; Gallo, L.; Capasso, F.; Orlando, P.; Di Marzo, V. Inhibitory Effect of Cannabichromene, a Major Non-Psychotropic Cannabinoid Extracted from Cannabis Sativa, on Inflammation-Induced Hypermotility in Mice. Br. J. Pharmacol. 2012, 166, 1444–1460. [Google Scholar] [CrossRef] [PubMed]
- Wen, Y.; Wang, Z.; Zhang, R.; Zhu, Y.; Lin, G.; Li, R.; Zhang, J. The Antinociceptive Activity and Mechanism of Action of Cannabigerol. Biomed. Pharmacother. 2023, 158, 114163. [Google Scholar] [CrossRef] [PubMed]
- Russo, E.B.; Burnett, A.; Hall, B.; Parker, K.K. Agonistic Properties of Cannabidiol at 5-HT1a Receptors. Neurochem. Res. 2005, 30, 1037–1043. [Google Scholar] [CrossRef]
- Tham, M.; Yilmaz, O.; Alaverdashvili, M.; Kelly, M.E.M.; Denovan-Wright, E.M.; Laprairie, R.B. Allosteric and Orthosteric Pharmacology of Cannabidiol and Cannabidiol-dimethylheptyl at the Type 1 and Type 2 Cannabinoid Receptors. Br. J. Pharmacol. 2019, 176, 1455–1469. [Google Scholar] [CrossRef]
- Raup-Konsavage, W.M.; Sepulveda, D.E.; Wang, J.; Dokholyan, N.V.; Vrana, K.E.; Graziane, N.M. Antinociceptive Effects of Cannabichromene (CBC) in Mice: Insights from von Frey, Tail-Flick, Formalin, and Acetone Tests. Biomedicines 2023, 12, 83. [Google Scholar] [CrossRef]
- Udoh, M.; Santiago, M.; Haneef, S.; Rodger, A.; Marlowe, C.K.; Barr, P.J.; Connor, M. Activation of CB2 Receptors by (−)-Cannabichromene but Not (+)-Cannabichromene. Cannabis Cannabinoid Res. 2024, 10, e94–e101. [Google Scholar] [CrossRef]
- Udoh, M.; Santiago, M.; Devenish, S.; McGregor, I.S.; Connor, M. Cannabichromene Is a Cannabinoid CB2 Receptor Agonist. Br. J. Pharmacol. 2019, 176, 4537–4547. [Google Scholar] [CrossRef]
- Romano, B.; Borrelli, F.; Fasolino, I.; Capasso, R.; Piscitelli, F.; Cascio, M.; Pertwee, R.; Coppola, D.; Vassallo, L.; Orlando, P.; et al. The Cannabinoid TRPA1 Agonist Cannabichromene Inhibits Nitric Oxide Production in Macrophages and Ameliorates Murine Colitis. Br. J. Pharmacol. 2013, 169, 213–229. [Google Scholar] [CrossRef]
- Trevaskis, N.L.; Shackleford, D.M.; Charman, W.N.; Edwards, G.A.; Gardin, A.; Appel-Dingemanse, S.; Kretz, O.; Galli, B.; Porter, C.J.H. Intestinal Lymphatic Transport Enhances the Post-Prandial Oral Bioavailability of a Novel Cannabinoid Receptor Agonist via Avoidance of First-Pass Metabolism. Pharm. Res. 2009, 26, 1486–1495. [Google Scholar] [CrossRef]
- Bar-Hai, A.; Domb, A.J.; Hoffman, A. Strategies for Enhancing the Oral Bioavailability of Cannabinoids. Expert Opin. Drug Metab. Toxicol. 2022, 18, 313–322. [Google Scholar] [CrossRef]
- Lacerda, M.; Carona, A.; Castanheira, S.; Falcão, A.; Bicker, J.; Fortuna, A. Pharmacokinetics of Non-Psychotropic Phytocannabinoids. Pharmaceutics 2025, 17, 236. [Google Scholar] [CrossRef]
- Ujváry, I.; Hanuš, L. Human Metabolites of Cannabidiol: A Review on Their Formation, Biological Activity, and Relevance in Therapy. Cannabis Cannabinoid Res. 2016, 1, 90–101. [Google Scholar] [CrossRef]
- Beers, J.L.; Fu, D.; Jackson, K.D. Cytochrome P450-Catalyzed Metabolism of Cannabidiol to the Active Metabolite 7-Hydroxy-Cannabidiol. Drug Metab. Dispos. Biol. Fate Chem. 2021, 49, 882–891. [Google Scholar] [CrossRef] [PubMed]
- Chayasirisobhon, S. Mechanisms of Action and Pharmacokinetics of Cannabis. Perm. J. 2020, 25, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Bansal, S.; Zamarripa, C.A.; Spindle, T.R.; Weerts, E.M.; Thummel, K.E.; Vandrey, R.; Paine, M.F.; Unadkat, J.D. Evaluation of Cytochrome P450-Mediated Cannabinoid-Drug Interactions in Healthy Adult Participants. Clin. Pharmacol. Ther. 2023, 114, 693–703. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Q.; Du, Z.; Qu, K.; Shen, Y.; Jiang, Y.; Zhu, H.; Zhang, X. Adverse Events of Epidiolex: A Real-World Drug Safety Surveillance Study Based on the FDA Adverse Event Reporting System (FAERS) Database. Asian J. Psychiatry 2023, 90, 103828. [Google Scholar] [CrossRef]
- Naftali, T.; Dor, M. Cannabis for the Treatment of Inflammatory Bowel Disease: A True Medicine or a False Promise? Rambam Maimonides Med. J. 2020, 11, e0008. [Google Scholar] [CrossRef] [PubMed]
- Naftali, T.; Bar-Lev Schleider, L.; Sklerovsky Benjaminov, F.; Lish, I.; Konikoff, F.M.; Ringel, Y. Medical Cannabis for Inflammatory Bowel Disease: Real-Life Experience of Mode of Consumption and Assessment of Side-Effects. Eur. J. Gastroenterol. Hepatol. 2019, 31, 1376–1381. [Google Scholar] [CrossRef] [PubMed]

| % Weight Loss | Stool Consistency | Hemoccult | |||
|---|---|---|---|---|---|
| 1–5% | 1 | Normal | 0 | Absent | 0 |
| 6–10% | 2 | Soft/Loose | 2 | Hemoccult positive | 2 |
| 11–20% | 3 | Diarrhea | 4 | Visible/Gross | 4 |
| >20% | 4 | ||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Godbole, S.S.; Sun, D.; Coates, M.D.; Himmelberger, V.J.; Roopchand, D.E.; Raup-Konsavage, W.M. Orally Administered CBD/CBG Hemp Extract Reduces Severity of Ulcerative Colitis and Pain in a Murine Model. J. Clin. Med. 2025, 14, 6095. https://doi.org/10.3390/jcm14176095
Godbole SS, Sun D, Coates MD, Himmelberger VJ, Roopchand DE, Raup-Konsavage WM. Orally Administered CBD/CBG Hemp Extract Reduces Severity of Ulcerative Colitis and Pain in a Murine Model. Journal of Clinical Medicine. 2025; 14(17):6095. https://doi.org/10.3390/jcm14176095
Chicago/Turabian StyleGodbole, Shivani S., Dongxiao Sun, Matthew D. Coates, Victoria J. Himmelberger, Diana E. Roopchand, and Wesley M. Raup-Konsavage. 2025. "Orally Administered CBD/CBG Hemp Extract Reduces Severity of Ulcerative Colitis and Pain in a Murine Model" Journal of Clinical Medicine 14, no. 17: 6095. https://doi.org/10.3390/jcm14176095
APA StyleGodbole, S. S., Sun, D., Coates, M. D., Himmelberger, V. J., Roopchand, D. E., & Raup-Konsavage, W. M. (2025). Orally Administered CBD/CBG Hemp Extract Reduces Severity of Ulcerative Colitis and Pain in a Murine Model. Journal of Clinical Medicine, 14(17), 6095. https://doi.org/10.3390/jcm14176095






